Abstract
Case summary
Primary portal vein hypoplasia (PVH) is a rare congenital vascular disorder characterised by underdevelopment of the portal veins, leading to hepatic hypoperfusion and hypoplasia of the affected liver lobes. Although PVH is occasionally diagnosed in dogs, its occurrence in cats is exceptionally rare, with only a few cases reported. This case report describes the early detection of PVH in a 1-year-old female Russian Blue cat that exhibited persistently elevated liver enzyme activity without any clinical signs. CT imaging revealed hypoplasia of the left portal branch and a reduction in the volume of specific liver lobes. Histopathology confirmed PVH, revealing hypoplastic portal veins, mild bile duct and arteriolar hyperplasia, and hepatocellular vacuolar degeneration. The cat remained asymptomatic during the 18-month follow-up period.
Relevance and novel information
This case highlights the critical role of advanced imaging modalities, particularly CT, in the early diagnosis of PVH in cats and emphasises the potential for early intervention in asymptomatic cases. These findings contribute to the limited body of knowledge on feline PVH, expanding the understanding and paving the way for further research into its prevalence, pathophysiology and optimal diagnostic approaches in veterinary practice.
Keywords
Introduction
Primary portal vein hypoplasia (PVH) is a congenital vascular disorder characterised by the underdevelopment of microscopic portal veins, leading to hepatic hypoperfusion and subsequent hypoplasia of the affected liver lobes.1 –5 Differentiating PVH from secondary portal vein hypoplasia caused by macroscopic conditions – such as portosystemic shunt (PSS), arterioportal fistula and portal vein thrombosis – is crucial because these conditions also contribute to chronic hepatic hypoperfusion. Therefore, a definitive diagnosis of PVH necessitates both histopathological examination and advanced imaging techniques, particularly CT angiog-raphy (CTA).1,2,4,6
In dogs, PVH has been documented, occasionally presenting with clinical features similar to those of congenital PSS and often coexisting with congenital PSS. When PVH occurs as an isolated condition, the clinical signs are typically milder than those associated with PSS, with less severe neurological and gastrointestinal signs.1,3,4,7,8 In contrast, PVH is extremely rare in cats, and only a few reports describe its early detection before the onset of clinical signs or secondary complications associated with portal hypertension.5,7,9 This case report presents the early imaging and histopathological findings of PVH in a cat, contributing valuable information to the limited literature on this rare condition.
Case description
A 1-year-old spayed female Russian Blue cat with persist-ently elevated liver enzymes for more than 6 months was referred for further evaluation. The cat remained asymptomatic, and initial treatments, including fluid therapy, S-adenosylmethionine, antioxidants and ursodeoxycholic acid, failed to normalise the elevated liver enzyme activity.
On physical examination, the cat had a normal body condition score and a body weight of 4.5 kg. At the time of referral, serum biochemistry revealed elevated alanine aminotransferase (ALT) activity of 686 U/l (reference interval [RI] 12–130) and aspartate aminotransferase (AST) of 119 U/l (RI 0–48). Cholesterol, triglyceride, glucose, blood urea nitrogen, creatinine, albumin and globulin levels were all within normal limits. Additional hepatic function tests, including measurement of blood ammonia concentration, fasting serum bile acids, 2-h postprandial serum bile acids, bilirubin and coagulation times, were all within normal RIs. Urinalysis revealed no presence of ammonium urate crystals or other abnormalities. Thoracic and abdominal radiographs showed no remarkable findings, and liver size was normal. No signs of renal calculi or cystoliths were observed (Figure 1). On abdominal ultrasonography, the visualised liver parenchyma generally revealed no specific abnormalities; however, the left-sided liver lobes were not clearly visualised. Instead, what appeared to be abdominal fat was observed in the areas where these lobes would typ-ically be located (Figure 2).

(a) Right lateral and (b) ventrodorsal abdominal radiographs of the cat, demonstrating normal liver size without any remarkable findings in the abdominal organs. No evidence of other pathological changes were observed

(a,b) Ultrasonographic images obtained in the longitudinal plane of the liver demonstrate generally normal hepatic echogenicity and homogeneous parenchymal texture in the visualised portions. Although no distinct abnormalities were observed within the clearly visualised hepatic parenchyma, the left-sided liver lobes were not clearly visualised. GB = gallbladder
CT was performed using a 32-slice multidetector CT scanner (Revolution ACT; GE Healthcare) to assess the hepatic lobes and the portal vascular system. The scan parameters included the following: slice thickness 1.3 mm; pitch 0.9; tube current 70 mA; and tube voltage 120 kV. Contrast-enhanced CT imaging revealed that the left portal branch was markedly hypoplastic compared with the right portal branch; it primarily supplied only the right medial lobe, while its branches to the quadrate lobe, left medial lobe, left lateral lobe and papillary process of the caudate lobe were not clearly identified. Correspondingly, the left medial lobe, left lateral lobe and papillary process of the caudate lobe were observed to be small, whereas the quadrate lobe maintained a normal size despite the altered portal supply (Figure 3). Overall, liver attenuation appeared normal, with homogeneous contrast enhancement. No abnormal intra- or extrahepatic tortuous vessels were detected, and no evidence of arteriovenous connections was found. In addition, no portal vein thrombosis or indirect signs of portal hypertension (such as abdominal congestion, ascites or engorged abdominal lymphatics) were observed. Based on these imaging findings, a tentative diagnosis of PVH was made; however, congenital liver lobe hypoplasia, cholangiohepatitis and chronic hepatitis could not be definitively excluded.
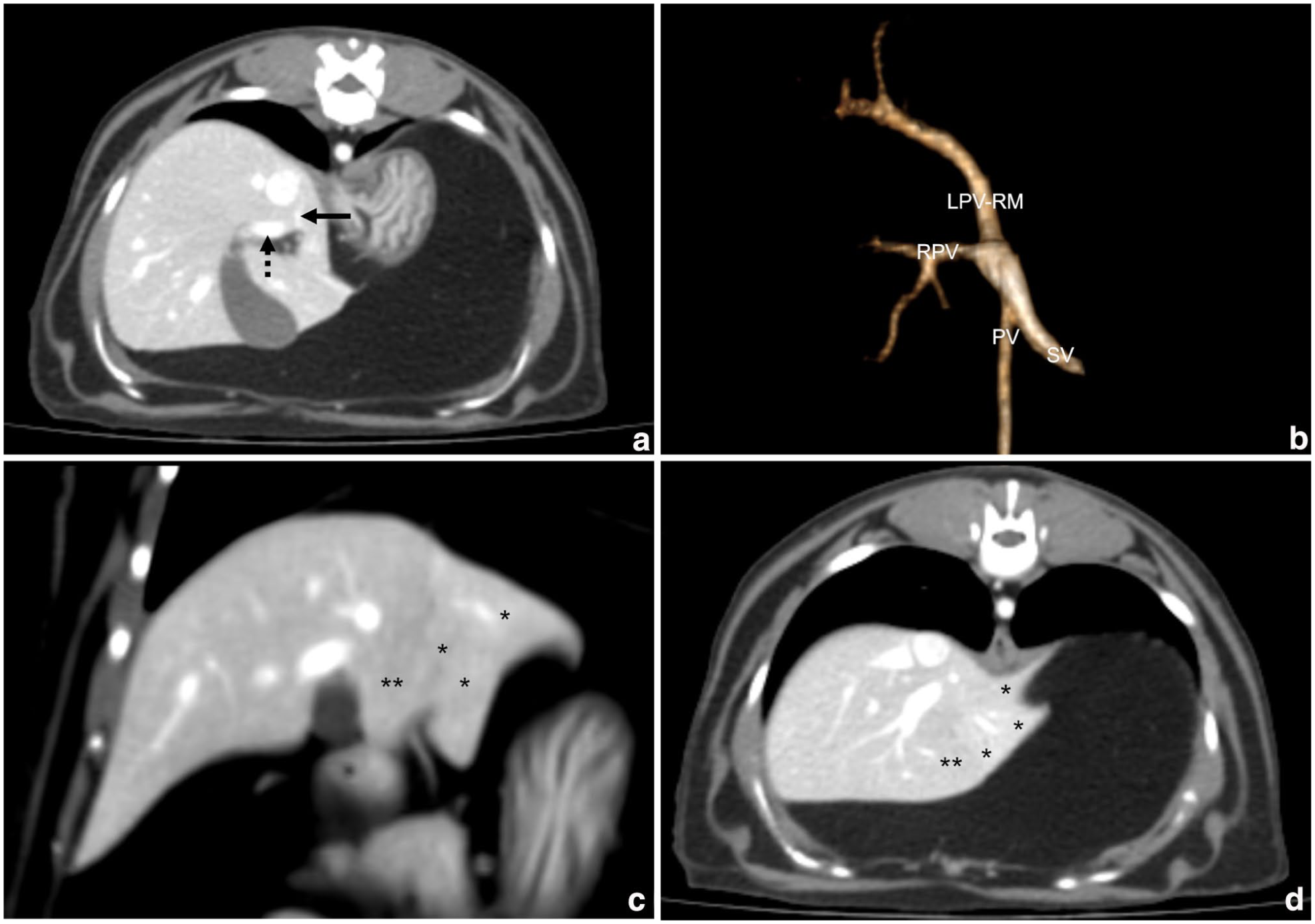
Contrast-enhanced CT images of the liver and three-dimensional reconstruction of the intrahepatic portal venous system of the cat. (a,d) Transverse and (c) dorsal contrast-enhanced CT images of the liver, and (b) a three-dimensional reconstructed image of the portal venous system. (a) The dashed arrow indicates the normally branching right portal vein (RPV), which supplies the body of the caudate process and the right lateral lobe. The solid black arrow points to the hypoplastic left portal vein (LPV); the LPV primarily supplies only the right medial lobe (RM), with its branches to the quadrate lobe, left medial lobe, left lateral lobe and papillary process of the caudate lobe not clearly identified. (c,d) Asterisks (*) denote the reduced volume of the left medial lobe, left lateral lobe and the papillary process of the caudate lobe compared with the right-sided liver lobes. Double asterisks (**) indicate the quadrate lobe, which, although appearing normal in size, also showed no clear evidence of vascular supply from the hypoplastic LPV. No evidence of a portosystemic shunt, arterioportal fistula or portal vein thrombosis was observed. PV = portal vein; SV = splenic vein
An exploratory laparotomy was performed. Intra-operatively, the right lateral, right medial, quadrate and main body of the caudate lobes appeared grossly unremarkable in size, colour and texture. In contrast, the left medial lobe, left lateral lobe and the papillary process of the caudate lobe were observed to be markedly diminutive, each estimated to be less than 1 cm in its largest dimension. To obtain adequate samples for histopath-ology, punch biopsies were performed on the accessible lobes, specifically the right lateral, right medial, quadrate and main body of the caudate lobe. Tissue samples were fixed in 10% neutral-buffered formalin and evaluated by a board-certified veterinary pathologist. Histopathological examination of all the sampled lobes revealed mild lobular atrophy, hypoplastic portal veins accompanied by both mild arteriolar and biliary ductular hyperplasia, and mild hepatocellular vacuolar degeneration (Figure 4). These findings supported the diagnosis of PVH.

Histopathological findings in feline primary portal vein hypoplasia (PVH). (a) A relatively well-formed portal area is shown (20× objective). The largest vascular channel in the centre of the image, the portal vein (*), appears relatively normal. An adjacent bile duct (solid arrow) is present, and mild arterial hyperplasia (dashed arrows) is also confirmed in this portal tract. H&E stain. Scale bar = 50 µm. (b) This portal area (40× objective) demonstrates an irregularly collapsed and small portal vein (*), characteristic of primary PVH. This is accompanied by biliary hyperplasia (indicated by dashed arrows) and arterial hyperplasia (indicated by solid arrows). Surrounding hepatocytes exhibit mild vacuolar degeneration. H&E stain. Scale bar = 50 µm. H&E = haematoxylin and eosin
Over the 18-month follow-up period, the cat received continuous symptomatic treatment. Throughout this period, the cat exhibited no clinical signs related to PVH, physical examinations consistently revealed no distinct abnormalities and periodic radiographic examinations also consistently revealed no findings suggestive of generalised hepatomegaly. However, mild elevations in liver enzyme activity (ALT and AST) persisted. Long-term follow-up was not possible because of loss of contact with the owner.
Discussion
PVH is more frequently diagnosed in dogs than in cats, and its clinical presentation in dogs is well characterised. In dogs, PVH may present with neurological and gastrointestinal signs similar to those seen in congenital PSS; however, isolated PVH typically exhibits a milder clin-ical course and a more favourable prognosis.1 –4,8 Recent studies indicate that up to 57.1% of dogs with PVH remain asymptomatic, with elevated liver enzyme activity as the only detectable abnormality. 8 In contrast, previously reported feline cases of PVH have often been associated with clinical signs.5,9 However, the present case uniquely demonstrated persistently elevated liver enzyme activity without any apparent clinical signs, and no clinical signs developed over the 18-month follow-up period.
Diagnosing PVH solely by histopathology can be challenging, necessitating the use of advanced imaging modalities such as ultrasonography, scintigraphy and CTA to rule out macroscopic vascular anomalies contributing to hepatic hypoperfusion.6,7,9 Notably, CTA provides high-resolution vascular mapping, enabling clinicians to differentiate between isolated PVH and other conditions, such as congenital PSSs. 6 In this case, CTA not only identified left portal vein hypoplasia but also provided detailed visualisation of subtle changes in the liver lobe size or perfusion that may be missed with other imaging modalities. These findings emphasise the importance of CTA in early detection of focal or localised PVH.
PVH remains an exceedingly rare condition in cats, and the limited reported cases suggest that its presentation differs from that observed in dogs. To the best of our knowledge, only three reports have described PVH in cats without concurrent congenital PSS. In one such case, a 9-month-old Birman cat diagnosed with PVH via trans-splenic portal scintigraphy and liver biopsy exhibited clinical signs, including periodic ptyalism, apathy and circling, and laboratory findings indicative of liver failure. 9 Notably, D’Anjou et al 7 suggested that, unlike in dogs, microhepatica in feline PVH may not always be detectable on ultrasonography. Consistent with this case, where macroscopic hypoplasia was limited to specific liver lobes rather than affecting the entire liver, radiography or ultrasonography may not clearly identify such subtle or focal microhepatica.
This study further underscores the critical role of CT imaging in the early diagnosis of PVH cases where gross pathological changes are observed in only some lobes. Unlike ultrasonography, CT imaging facilitates easier differentiation of liver lobes through the visualisation of vascular branching. In normal feline anatomy, the main portal vein divides into right and left branches, with the larger left portal vein typically supplying the right medial lobe, quadrate lobe, papillary process and both left lobes. 10 The CT findings in this case, which detailed a hypoplastic left portal branch with a restricted distribution primarily to the right medial lobe, highlighted a marked deviation from this expected normal anatomy. This vascular anomaly correlated directly with the observed selective lobar hypoplasia (specifically, the left medial, left lateral and papillary process of the caudate lobes), while the quadrate lobe, despite also lacking a clear LPV supply, remained normal in size. These findings emphasise the importance of considering PVH as a differential diagnosis in cats with persistently elevated liver enzyme activity, even in the absence of distinct clinical signs or generalised microhepatica detectable by conventional radiography or ultrasonography. Consequently, advanced imaging techniques such as CT are warranted to accurately evaluate individual liver lobes and their vasculature when PVH is suspected. Furthermore, previous reports have described cases of feline PVH complicated by secondary acquired PSS, neurological signs and signs of liver failure. 5 This highlights the critical role of detailed imaging in the early detection of PVH in asymptomatic cats, particularly those without concurrent PSS.
This study has several limitations. First, direct measurement of portal vein pressure was not performed, potentially overlooking a key parameter for evaluating hepatic haemodynamics. Second, the absence of dedicated portal-phase CT examinations may have limited the detection of subtle vascular abnormalities affecting hepatic perfusion. Furthermore, the markedly diminutive liver lobes identified on imaging and during surgery could not be biopsied, precluding a complete understanding of the spectrum of microscopic changes across all affected parenchyma. Finally, the lack of a long-term follow-up prevented conclusions regarding the progression or clinical significance of subclinical PVH.
Conclusions
This case demonstrates that feline PVH can be detected early, before the onset of clinical signs or secondary complications, when advanced imaging modalities such as CT are utilised. Consequently, PVH should be considered in the differential diagnosis of cats with persistently elevated liver enzymes, even in the absence of liver lobe microhepatica.
Footnotes
Acknowledgements
The authors thank colleagues at the Department of Veterinary Medical Imaging, Jeonbuk National University, and the staff at Nel Animal Medical Foundation for their support in preparing this case report.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognised high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS Open Reports. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers, tissues or samples) for all procedure(s) undertaken (prospective or retrospective studies). For any animals or people individually identifiable within this publication, informed consent (verbal or written) for their use in the publication was obtained from the people involved.
