Abstract
The aims of the present study were 1) to evaluate the interlobular distribution of copper (Cu) in the liver of beef calves on a high-Cu diet, 2) to determine whether this distribution differs between Galician Blonds and Holstein Friesians, and 3) to determine whether in vivo needle biopsy provides an appropriate measure of overall hepatic Cu status. Liver biopsies were performed before slaughter on twenty-nine 10-month-old beef calves fed growing and finishing diets supplemented with 35 mg/kg of Cu sulfate (10 Galician Blonds, 9 Holstein Friesians, and 10 Galician Blond × Holstein Friesian crosses). At slaughter, samples taken from 6 regions of the liver (the internal and external faces of the right lobe; the left, caudate, and quadrate lobes; and the processus papillaris) were acid digested, and their Cu contents were determined by inductively coupled plasma atomic emission spectrometry. The highest Cu concentrations were found in the left lobe, followed by the processus papillaris, and the lowest Cu concentrations were found in the caudate and quadrate lobes. Different breeds differ in absolute hepatic Cu levels, but interlobular Cu distribution does not appear to depend on breed, at least when Galician Blonds are compared with Holstein Friesians. In vivo needle biopsy afforded accurate estimates of overall hepatic Cu status.
Several experimental studies found nonuniform distributions of certain metals within animal organs. For example, it is well established that cadmium accumulates more in renal cortex than in renal medulla. 7 By contrast, results on the intrahepatic heterogeneity of copper (Cu) concentration in horses 6,9 and sheep 3,4 are less conclusive. For bovine liver, interlobular differences in Cu accumulation were reported, 1,10 but relevant studies were few and appear to have involved few animals. 1,10 Hepatic Cu concentration is the most reliable indicator of animal's overall Cu status, 1,10,11 which is based on the fact that 50–60% of the body's Cu content is located in the liver. Although in live animals liver biopsy is recommended to evaluate chronic Cu accumulation in ruminants, 1,8,10 there are no studies to prove the effectiveness of this technique to evaluate the Cu burden in the whole organ. A nonuniform intrahepatic Cu distribution would throw further doubts on the validity of evaluating overall hepatic Cu status by means of liver biopsy. 1,8,10 Furthermore, the precision of the biopsy method is in any case limited by variation in tissue quantity and extraction site, and by contamination with blood, fat, and connective tissue. 1,9 The aims of the present study were 1) to evaluate the interlobular distribution of Cu in the liver of beef calves on a high-Cu diet, 2) to determine whether this distribution differs between Galician Blonds (GB) and Holstein Friesians (HF), and 3) to determine whether in vivo needle biopsy provides an appropriate measure of overall hepatic Cu status.
The study involved 29 beef calves (10 GB, 9 HF, and 10 GB × HF crosses), which were cared for and handled in compliance with Spanish Government guidelines (RD 1201/2005). On arrival to the feedlot, at age 8 weeks, the animals were weighed; dewormed with albendazole
a
and ivermectin
b
; vaccinated for clostridial infection
c
and for
Immediately before slaughter of the animals at age 10 months, needle biopsy of the liver was performed as follows. The biopsy incision site, located between the 11th and 12th right ribs on a line from the tuber coxae to the shoulder, was shaved, scrubbed 3 times with povidone-iodine, e scrubbed once more with 70% alcohol, and infiltrated with lidocaine. f A liver core was extracted with a biopsy needle, g rinsed with 0.01 M physiological saline solution (pH 7.4), drained of blood, placed in a polypropylene tube, transported on ice to the laboratory, and frozen at −20°C pending analysis. After slaughter, the liver was excised, and samples sized approximately 5 cm × 5 cm × 5 cm were taken from 6 regions: the internal and external (biopsy point) faces of the right lobe, the left, caudate and quadrate lobes, and the processus papillaris.
At the laboratory, postmortem liver samples were freed of connective tissue and major blood vessels and then homogenized. Subsamples of approximately 2 g were digested with 5 ml of concentrated nitric acid and 2 ml of 30% w/v hydrogen peroxide in a microwave digestion system, h and the resulting solution was transferred to polypropylene sample tubes and made up to 25 ml with ultrapure water for Cu determination.
Biopsy samples were weighed in glass digestion tubes and, over approximately 24 hr, were dried to constant weight in an oven at 85°C, after which the samples were digested for 24 hr at room temperature with 1 ml of concentrated nitric acid, placed for 60 min in a thermostatic block at 120°C ± 2°C (a thermostatic block was used to improve the limit of quantitation), treated with 1 ml of 30% w/v hydrogen peroxide, and made up to 5 ml with ultrapure water. The Cu concentration in the digested samples was determined by inductively coupled plasma atomic emission spectrometry, i in all cases with subtraction of absorbance values for blanks. Results of Cu concentration were given in wet weight by transformation of results of biopsy sample (dry weight) multiplying by 27.5% of DM.
An analytical quality control program was applied throughout the study. Analytical recovery from a certified reference material j was 92.2%. Analytical recoveries from 10 samples spiked at levels that afforded absorbance values 2–10 times higher than those of the unspiked samples ranged from 91% to 97%.
Non-normal data distribution was tested by using the Kolmogorov-Smirnov test. Two-way analysis of variance (ANOVA), with post hoc least significant difference tests, was used to test for interlobular and between-breed differences in postmortem Cu concentration. Association between Cu concentrations in postmortem and biopsy samples was evaluated by Pearson correlation analysis, and differences between these variables were evaluated by Student's
The Cu concentration data for each postmortem sampling site in each breed is summarized in Figure 1. An ANOVA showed a significant breed effect (F2,173 = 4.594,
The strong between-lobe correlations in Cu concentration, with
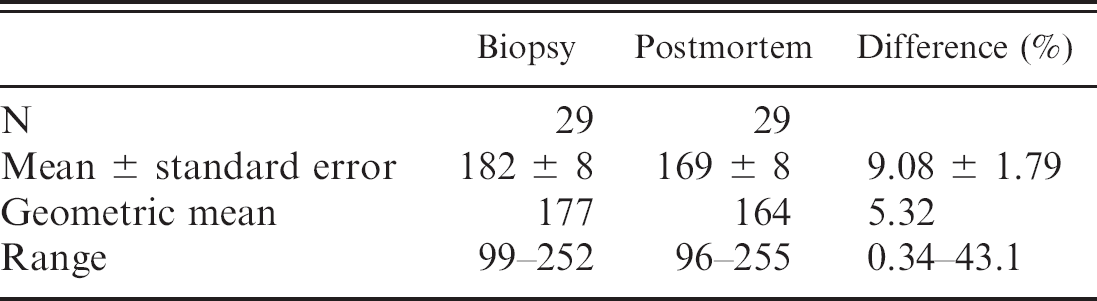
The Cu concentrations measured in biopsy samples and postmortem samples of the external right lobe (mean values of 182 and 169 mg/kg wet weight, respectively; Table 1) were not significantly different (
The results of the current study confirmed earlier reports 1,10 that Cu concentration in the liver is not homogeneous. However, whereas the otherwise uncharac-terized necropsied cow in a previous study 1 had its highest Cu concentration in the caudate lobe (which is also identified in another study 10 as the lobe with highest Cu levels), in the present study, Cu concentrations were highest in the left lobe and lowest in the caudate or quadrate lobes. This difference may be partly because of genetic factors, 5,11 partly because of differences in other dietary components, sex, and age, 5 and partly because of the difference in Cu levels and hence in dietary Cu content. In fact, in all lobes, Cu content was approximately 5 times higher in the present study than in a previous study. 1 It is known that, in sheep liver, Cu is concentrated in centrilobular zones, particularly around the central veins, when total hepatic Cu content is low, and only extends through the midlobular zones toward the portal triads as total Cu content rises. 4 Although there appear to be no significant differences in Cu concentration among left lobe, right lobe, and regions close to the portal vein after subclinical chronic Cu poisoning of sheep, 3 it seems possible that the pattern of accumulation may be somewhat different in larger animals, in which there are greater differences between lobes as regards the distance of central vessels from the superficial hepatic regions that were sampled in this study and others. More specifically, although deposition of Cu at low exposure levels may be greater in caudate lobe samples than in left lobe samples because the surface of the caudate lobe is closer than that of the left lobe to central vessels, at higher exposure levels, the critical factor may be the extent to which oxygen perfusion supports the metabolic activity involved in elimination of Cu in the bile, 2 elimination from regions farther from central vessels being less intense because of lesser perfusion. Further studies that compare the effects of diets with different levels of Cu supplementation would clarify this issue. It is also interesting in this respect that the pattern of Cu accumulation in the left lobe changed as Cu storage increased, tending to be slower at about 200 mg/kg wet weight (twice the generally accepted safe concentration 10 ), which suggests that Cu loading of greater intensity or longer duration might result in a different interlobular distribution.
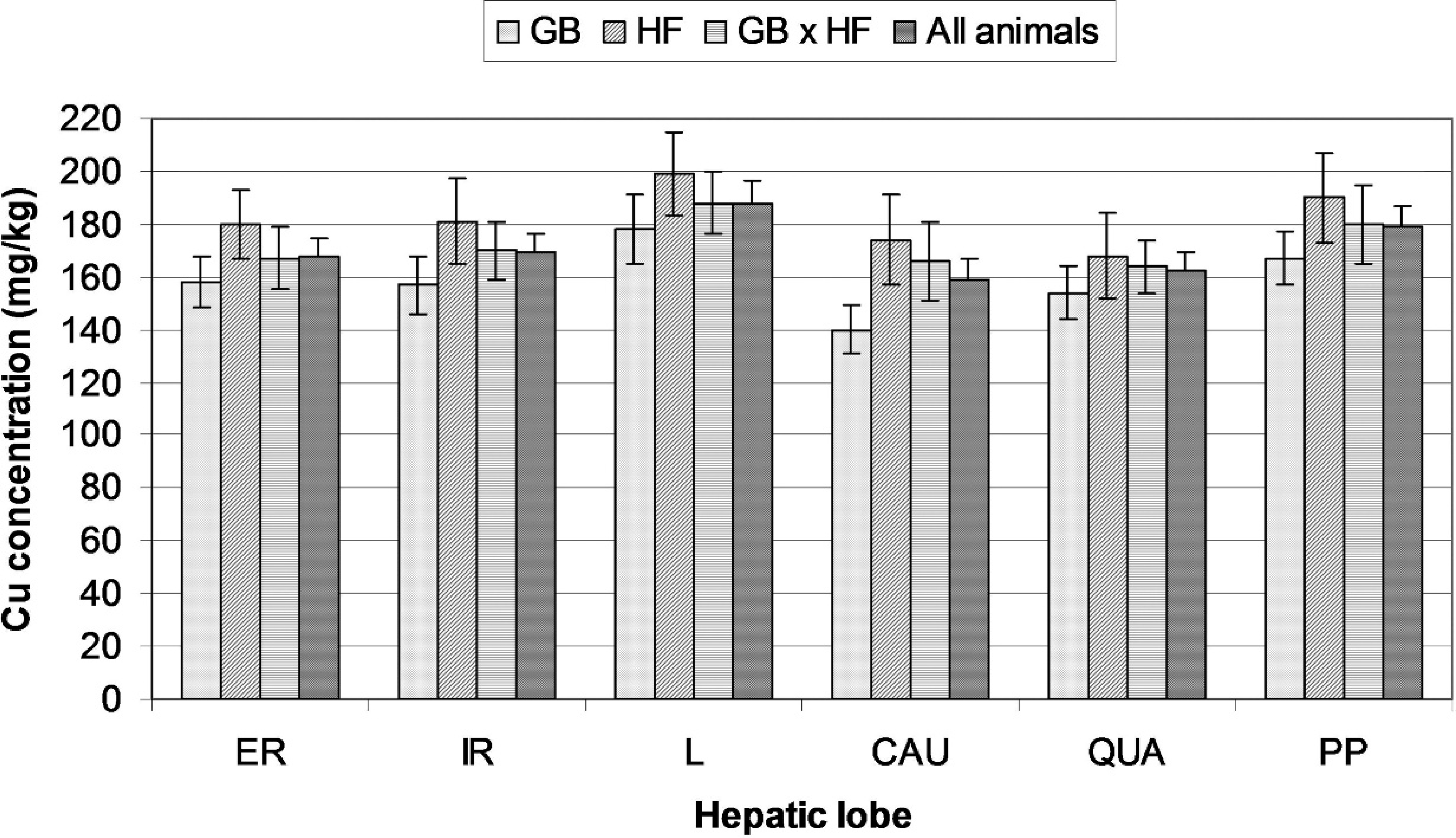
Mean copper (Cu) concentrations (mg/kg wet weight) in the hepatic lobes of each breed. Bars show standard errors. Analysis of variance showed a significant effect of breed (
In a previous study, biopsy-sized postmortem samples afforded significantly higher Cu concentrations than wedge samples when all lobes were considered jointly, but the difference was not clinically relevant. 1 In the present study, biopsy measurements of Cu concentration in the external right lobe did not differ significantly from postmortem measurements, and the 2 measurements correlated extremely well. In both studies, right lobe Cu concentrations were intermediate between and did not differ significantly from those of the other lobes. In view of this, and of the observed close between-lobe correlations in Cu concentration, it may be concluded that there is no evidence that needle biopsy of the right lobe does not adequately reflect overall hepatic Cu status.
In conclusion, the hepatic lobes of beef calves fed a high-Cu diet tend to develop different Cu concentrations, with highest concentrations in the left lobe and lowest in the caudate and quadrate lobes. However, these differences, which may derive from differences in blood supply and bile-excretion capacity, may depend on the absolute level of Cu accumulation, an issue that requires further research. Results in the current study seem to show a change in the interlobular Cu accumulation up to 200 mg/kg wet weight. Different breeds differ in absolute hepatic Cu levels, but interlobular Cu distribution does not appear to depend on breed, at least when GB are compared with HF. In the range of concentrations measured in this study, the Cu concentration of right lobe biopsy samples appears to be an adequate measurement of overall hepatic Cu status.
Copper concentrations (mg/kg wet weight) in biopsy and postmortem samples of the external right hepatic lobe.
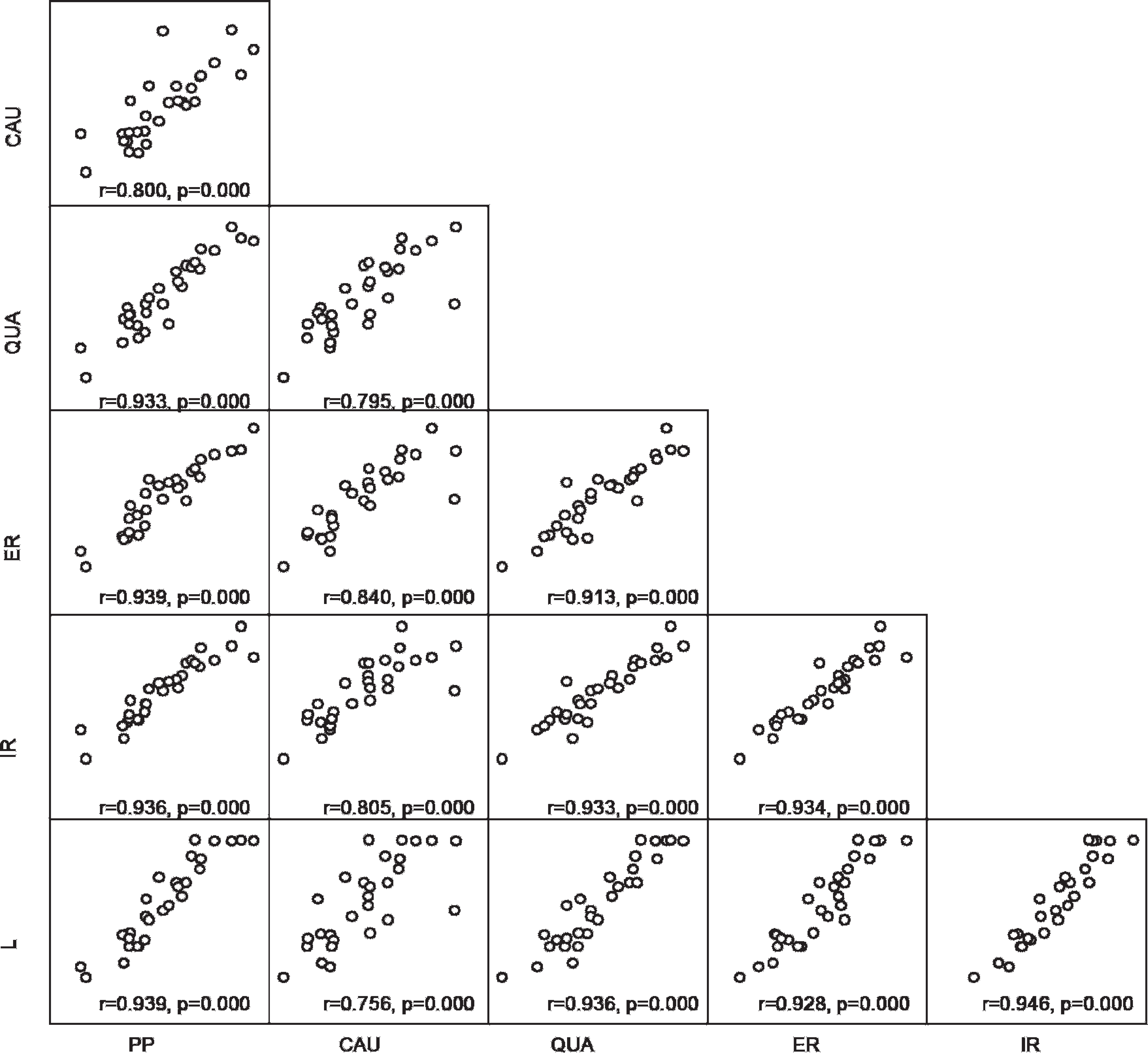
Scatter plots showing between-lobe correlation of copper concentrations. ER = external right lobe; IR = internal right lobe; L = left lobe; CAU = caudate lobe; QUA = quadrate lobe; PP = processus papillaris.
Footnotes
a.
Albensol-10%®, Fort Dodge Veterinaria, Girona, Spain.
b.
Ivomec®, Merial Laboratorios S.A., Barcelona, Spain.
c.
Toxipra-S7®, Hipra, Girona, Spain.
d.
Cattlemaster-4®, Pfizer, Madrid, Spain.
e.
Betadine®, Meda Pharma SA, Madrid, Spain.
f.
Xilocaina®, Laboratorios Ovejero, Leön, Spain.
g.
Super-CoreTM II Biopsy Instrument 14 G × 15 cm, Medical Device Technologies Inc., Gainesville, FL.
h.
Ethos Plus, Milestone Inc., Shelton, CT.
i.
Optima 4300 DV, Perkin Elmer Inc., Fremont, CA.
j.
Pig Kidney CRM (BCR 186), Institute for Reference Materials and Measurements, Geel, Belgium.
k.
SPSS Inc., Chicago, IL.
