Abstract
Objectives
Chronic kidney disease (CKD) is associated with accumulation of uremic toxins such as indoxyl sulfate (IS) in blood and tubular epithelial cells, which contribute to the progression of CKD. The aim of this prospective, randomised, two-centre clinical pilot study was to evaluate the effect of oral spherical carbonaceous adsorbent Renaltec (Porus One) on IS plasma concentration, progression of CKD, parameters of calcium/phosphate homeostasis (total calcium [tCa], phosphorus, fibroblast growth factor 23 [FGF-23]), urine protein:creatinine ratio (UPC) and faecal dysbiosis index (FDI) in cats with stable CKD International Renal Interest Society (IRIS) stage 2 and 3 over a 6-month period.
Methods
A total of 19 cats with CKD were randomly assigned to the Renaltec group (n = 10, eight stage 2, two stage 3) or control group (n = 9, eight stage 2, one stage 3). All cats received standard of care treatment according to IRIS guidelines. Cats in the Renaltec group received an additional 500 mg q24h of Renaltec administered mixed with food or a liquid cat treat. All cats were examined on day 0 (t0), after 3 months (t1) and after 6 months (t2). Statistical analysis was performed using generalised linear mixed models.
Results
At t0, the Renaltec group cats had significantly lower tCa (P = 0.026) than the control group cats. At t1, there were no differences between the groups, while at t2, the Renaltec group cats had significantly lower IS (P = 0.045) and UPC (P = 0.001) than cats in the control group. Progression of CKD was noted in two cats from each group. The Renaltec group cats had a significantly lower FDI at t2 compared with t1 (P = 0.023) and t0 (P = 0.009).
Conclusions and relevance
Administration of Renaltec over 6 months was associated with the mitigation of parameters linked to CKD progression. Given the pilot nature of the study, these findings should be interpreted with caution and warrant further investigation.
Plain language summary
Chronic kidney disease (CKD) is a common condition in older cats. It causes waste products such as indoxyl sulfate (IS) to rise in the blood. These substances can further damage the kidneys and accelerate the progression of the disease. The aim of this study was to find out whether the dietary supplement Renaltec (Porus One) can lower IS levels and whether it has other positive effects on the health of cats with CKD. A total of 19 cats with stable stage 2 or 3 CKD were randomly divided into two groups. All cats received the usual treatment in accordance with international guidelines. Ten cats were also given Renaltec once a day, while the other nine were not. The animals were examined at the start of the study, after 3 months and after 6 months. Various blood and urine values were measured, and the state of gut bacteria was examined. After 6 months, the cats that received Renaltec had lower levels of IS in their blood and less protein in their urine than the cats that did not receive Renaltec. Both are important factors that can influence the progression of the disease. The state of the intestinal bacteria also improved in the Renaltec group. In two cats in each group, the kidney disease worsened despite treatment. The results show that Renaltec could help to slow the progression of CKD in cats. However, as this was only a small pilot study, further research with more cats is needed to confirm the results.
Introduction
In recent years, there has been increasing interest in the role of uremic toxins, particularly of indoxyl sulfate (IS), in the progression of chronic kidney disease (CKD). IS belongs to the group of protein-bound indole compounds that are produced by intestinal microbes during the degradation of dietary L-tryptophan to indole, which is absorbed by the gut,1,2 metabolised to IS by the liver and excreted in the urine.3 –5
IS accumulates with increasing severity of CKD in cats.6 –9 The accumulation of IS in renal tubular cells leads to inflammation and damage to these cells in vitro. 10 In rats, IS accumulation disrupts the functionality of antioxidative defence mechanisms 11 and leads to suppressed Klotho expression through the production of reactive oxygen species and activation of nuclear factor-kappa B. 12 It accelerates glomerular sclerosis13,14 and is associated with renal fibrosis.14,15 IS induces intestinal epithelial cell damage in mice by disrupting mitophagy flow, leading to intestinal barrier injury. 16
In humans with CKD, increased serum IS concentrations activate immune cells, raise inflammatory markers and indicate oxidative stress. 17 In these patients, elevated IS blood levels are associated with heart disease, including aortic calcification, vascular stiffness, 18 endothelial and vascular smooth muscle cell dysfunction,18,19 coronary atherosclerosis 20 and left ventricular diastolic dysfunction, 21 all leading to a higher mortality risk.
Knowledge of these negative effects of IS led to experimental and clinical studies on the carbon-based intestinal adsorber AST-120 and its impact on plasma IS concentration and progression of CKD. In rats, oral administration of AST-120 for 10 weeks decreased IS plasma concentration, which was associated with a reduction in signs of oxidative stress. 22 In mice, AST-120 reduced IS-induced renal fibrosis. 23 In rats, a decrease in urine protein:creatinine ratio (UPC) was observed after treatment with AST-120 over 12–19 weeks, suggesting improved renal function.14,15 In humans with CKD, AST-120 significantly reduced IS blood concentrations after 12 weeks 24 and lowered creatinine levels in a 1-year study compared with a control group. 25
Renaltec (Porus One; Dechra Veterinary Products) is a selective intestinal adsorber for precursors of uremic toxins developed for cats with CKD. Similar to AST-120, it is carbon based and consists of small spherical particles (0.1–0.3 mm) with micropores that enhance the molecule’s adsorption capacity. A preliminary study showed that Renaltec reduces IS blood concentrations by 50% in geriatric cats. 26 A recent study also demonstrated that Renaltac lowered IS in cats with experimentally induced CKD. 27
Therefore, the aims of this prospective, randomised pilot study were to evaluate the effect of Renaltec in cats with International Renal Interest Society (IRIS) stage 2 and 3 CKD on plasma IS concentrations and disease progression. Furthermore, the effect on clinical parameters, serum electrolytes, fibroblast growth factor 23 (FGF-23) concentrations and the faecal dysbiosis index (FDI) was evaluated. It was hypothesised that Renaltec would lower IS plasma concentrations and positively influence parameters associated with CKD progression.
Materials and methods
Study design
The prospective, randomised, two-centre clinical pilot study included 19 cats with IRIS stage 2 and 3 CKD. All cats received standard-of-care treatment according to IRIS guidelines. Before inclusion, cats were randomly allocated to the Renaltec group or the control group. Cats in the Renaltec group received an additional 500 mg q24h of Renaltec with Add One (a liquid treat, Porus One; Dechra Veterinary Products) directly before or with their food. If further medication was indicated, an interval of 2 h was maintained between the administration of the drug and Renaltec. Between March 2019 and January 2023, 17 cats were treated at the LMU Small Animal Clinic, Ludwig Maximilian University Munich and two cats were treated at Tierklinik Oberhaching.
The study was approved by the ethical committee of the Centre for Clinical Veterinary Medicine, Ludwig Maximilian University Munich (reference number 154-14-01-2019). The study was conducted in accordance with the German Animal Welfare Act. All owners gave their written consent to participate before enrolling the cats in the study.
Patients
CKD was diagnosed based on history, clinical signs and persistent elevations of creatinine and symmetric dimethylarginine (SDMA) combined with a urine specific gravity (USG) <1035 at two time points at least 3 weeks apart. Sonographic assessment of the urinary tract was performed to exclude postrenal azotaemia due to upper urinary tract obstruction. CKD staging was performed according to IRIS guidelines. Exclusion criteria included proteinuria (UPC >0.4), hypercalcaemia, steroid treatment or uncontrolled systemic extrarenal diseases (chronic intestinal diseases, hepatic disease, pancreatic disease, diabetes mellitus or cancer). Screening for these comorbidities was based on detailed medical history, physical examination and laboratory investigations (complete blood count [CBC], serum chemistry, total thyroxine in cats aged >7 years). A complete abdominal ultrasound was performed in all cats with gastrointestinal signs or laboratory abnormalities that could not be explained with CKD. Cats receiving treatment for CKD-associated hypertension (amlodipine) were also included as well as cats with well-controlled hyperthyroidism. All cats received a commercial renal diet or a renal diet calculated by a veterinary nutritionist throughout the study period.
The following investigations were performed at the time of inclusion into the study (t0), after 3 months (t1) and after 6 months (t2): physical examination; systolic blood pressure measurement; CBC; serum chemistry profile; SDMA; urinalysis including USG, dipstick, urine sediment and UPC; and, if indicated, an aerobic bacterial culture (performed in cases of active sediment). EDTA plasma samples were collected at each time point for the measurement of plasma IS concentrations, and faecal samples were collected to determine the FDI. At t0, cats were randomly allocated to the Renaltec group or the control group.
Diet
Cats included in the study had been on a kidney diet for at least 3 weeks before t0. Cats were fed commercial renal diets (Hill’s Prescription Diet k/d wet and dry; Royal Canin early renal dry and renal wet and dry) or home-cooked meals with receipts calculated by Diet Check Munich or by nutritional specialists from the nutritional consulting service at the LMU Small Animal Clinic, Munich. At the beginning of the study, owners reported the types of food their cats were eating. A combination of dry and wet food was permitted according to owner preference, provided that all diets met renal diet criteria. Nutrient intake based on each cat’s body weight was calculated at the start of the study. On average, cats consumed 60 mg/kg body weight phosphorus (244.36 mg/MJ metabolisable energy [ME], 102 mg/100 kcal ME) with an average crude protein content of 3.99 g/kg body weight (16.14 g/MJ ME, 6.75 g/100 kcal ME) per day.
Assessment of treatment effect
Parameters to evaluate the treatment effect were the plasma IS concentration and the development of renal function assessed by serum creatinine and SDMA as well as phosphorus, total calcium (tCa) and potassium concentrations after 3 and 6 months. CKD progression was defined as a change to a higher IRIS stage and/or an increase in creatinine of more than 25% from baseline.
CBC and routine serum chemistry
Blood samples were collected from fasted cats (⩾12 h) by puncture of a peripheral vein. K-EDTA tubes were used for blood count analysis, processed immediately and analysed using fluorescence flow cytometry (Sysmex XT-2001; Sysmex). The remaining EDTA blood was processed for IS analysis.
Whole-blood samples were collected in serum tubes, centrifuged (5 mins at 2540 g) and analysed-in house. Sodium, potassium and chloride were measured with the ion-selective electrode module, while the remaining serum parameters were analysed using absorbance and photometry (Cobas Integra 400 Plus; Roche). SDMA was measured by an external commercial laboratory (IDEXX Kornwestheim); serum was sent by courier on the day of sampling.
Urinalysis
At t0, urine was collected by ultrasound-guided cystocentesis. At t1 and t2, urine samples were collected either by cystocentesis or by the owners at home using non-absorbent litter. USG was measured using refractometry. A dipstick test and a sediment examination were performed (SediVue DX; IDEXX). Samples with questionable or abnormal findings were also evaluated using light microscopy. If infection was suspected (in the presence of active sediment), an aerobic bacterial culture was obtained. UPC was measured by Cobas Integra 400 Plus (Roche). UPC was only performed in samples with an inactive sediment.
Plasma sample preparation for IS
Whole-blood samples were collected in K2-EDTA tubes and processed within 2 h. Samples were centrifuged for 5 mins at 630 g, and plasma was transferred to Eppendorf tubes and stored at –80 °C until measurement. IS was measured using an API5500 liquid chromatography with tandem mass spectrometry system as described in previous studies. 9
Serum preparation for FGF-23
Serum samples for FGF-23 analysis were stored at –80 °C until measurement. The measurement was performed by a commercial laboratory (LABOKLIN, Bad Kissingen) using a previously validated quantitative ELISA assay kit. 28
Faecal sample preparation for FDI
Owners collected fresh faecal samples at all time points and kept them refrigerated for up to 12 h before storage at –80 °C in the hospital. Samples were shipped frozen on dry ice to the Gastrointestinal Laboratory, Department of Small Animal Clinical Sciences, College of Veterinary Medicine and Biomedical Sciences (Texas A&M University, College Station, TX, USA), as described in previous studies. 29 One cat that had received antibiotics before enrolment was excluded from the FDI analysis. In addition, two cats that received antibiotics from t1 onwards and one cat from t2 onwards were excluded from FDI evaluation from the respective time point and all subsequent sampling time points. None of the cats included in the FDI analysis at any time point received medications known to affect the intestinal microbiota, such as antibiotics, proton pump inhibitors or H2 receptor antagonists, during the study period.
Statistical analysis
A power analysis was conducted based on the previously published mean (±SD) plasma IS concentration of 15 ± 5 mg/l in cats with IRIS stage 2 and 3 CKD 6 and the assumption of achieving a 50% reduction of plasma IS within 6 months of Renaltec administration, as demonstrated in an earlier study of geriatric cats. 26 For a power of 80% and a P value of 0.05, at least eight cats per group were required. Consequently, 10 cats were included in the Renaltec group and nine cats in the control group. Based on these assumptions, the minimum detectable effect for the study was an absolute difference of 7.5 mg/l in mean plasma IS concentration between groups, corresponding to a large effect size (Cohen’s d ≈ 1.5). The study was therefore powered to detect substantial treatment-related reductions in IS concentration.
Collected data were investigated using the statistical software R version 2023.06.1. Descriptive statistics included the calculation of the continuous variables as mean, SD and median with interquartile range and the categorical variables as counts and percentages. The normal distribution of continuous variables was assessed using the Shapiro–Wilk test. The difference between body weight in the Renaltec and control groups was evaluated using the Mann–Whitney U-test, while the difference in age was evaluated using the Student’s t-test. As a result of the presence of repeated measures among the time points, generalised linear mixed-effects models with individual animal ID as a random effect were chosen for analysis. The following model assumptions were always checked: (1) the normality of residuals was checked using the Shapiro–Wilk normality test; (2) the homogeneity of variances between groups was checked using the Bartlett test; and (3) the heteroscedasticity (constancy of error variance) was checked using the Breusch–Pagan test. In case assumptions were satisfied, generalised linear mixed-effects models were used (R package – lmer). In case assumptions were violated, robust linear mixed-effects models were applied (R package – robustlmm). The robust model was used for IS, creatinine, urea, total protein, phosphate, calcium, leukocytes, haematocrit, USG, UPC, FDI, FGF-23 and body weight. The generalised linear mixed model was used for SDMA, albumin, potassium, sodium chloride, sodium and blood pressure. P <0.05 was considered significant.
Results
Of the 65 cats with clinical signs compatible with CKD and assessed for enrolment, 19 with stable CKD were eligible for inclusion and randomly assigned to the Renaltec group or control group (Figure 1). The Renaltec group included 10 cats (eight at IRIS stage 2 and two at stage 3), while the control group included nine cats (eight at IRIS stage 2 and one at stage 3).
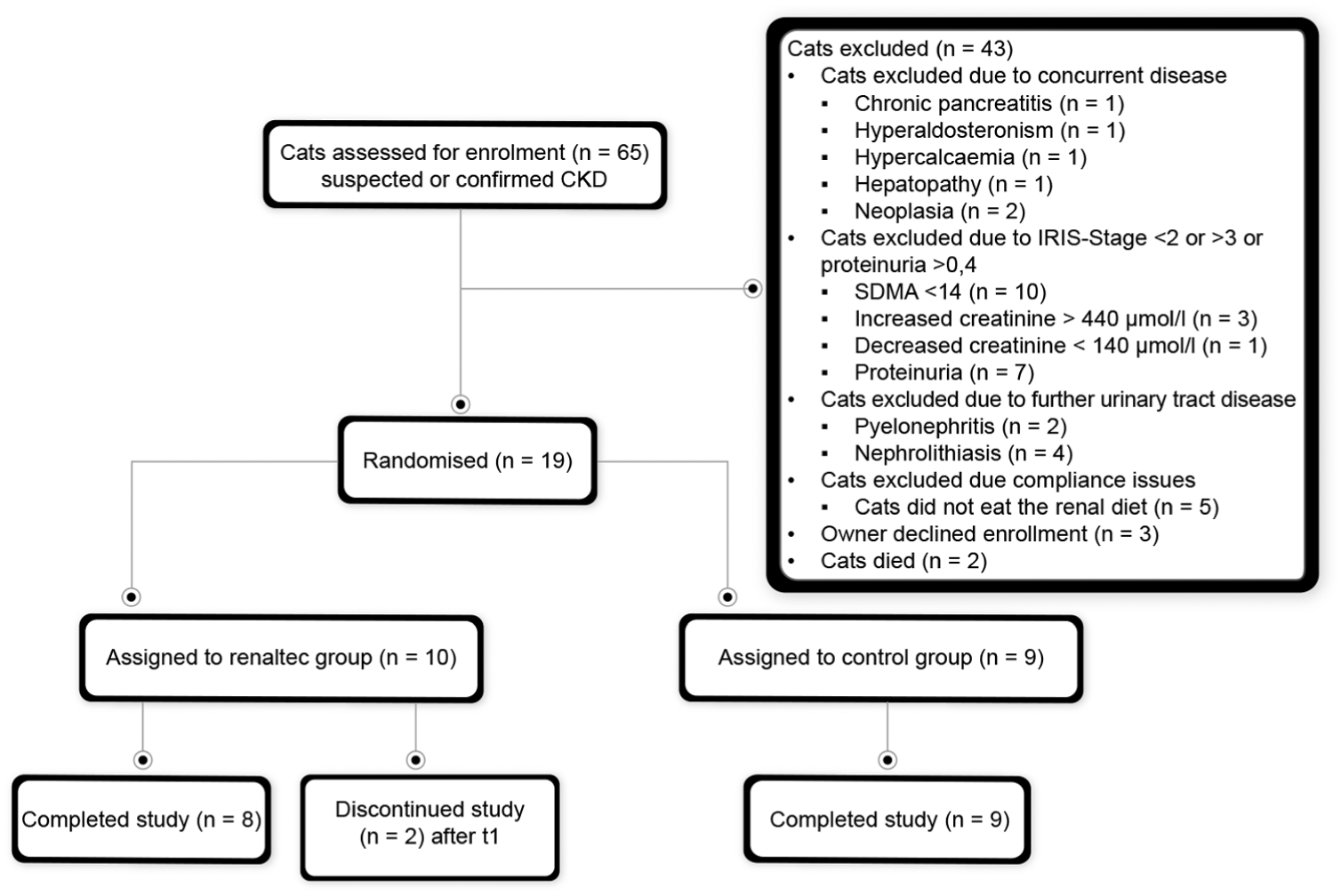
Flowchart showing the inclusion and exclusion of cats in the study. CKD = chronic kidney disease; IRIS = International Renal Interest Society; SDMA = symmetric dimethylarginine
The signalment of the cats is illustrated in Table 1. The Renaltec group included significantly more castrated male cats than the control group (P = 0.013). There were no other significant differences between the groups.
Signalment of the study cohort (age, breed, sex, body weight)

Data are n, mean ± SD or median (interquartile range). Differences in age between groups were evaluated using Student’s t-test, as the assumptions of normal distribution and homogeneity of variances were met. Differences in body weight were assessed using the Mann–Whitney U-test, as the assumption of normality was not met. P values for group comparisons are reported. Values in bold indicate statistically significant differences (P <0.05)
One Chartreux
One Chartreux, one Tonkinese, one Burmese, one Ragdoll, one Thai-mixed breed
At t0, laboratory results showed tCa concentrations within the reference interval for all cats; however, the cats in the Renaltec group had significantly lower tCa than those in the control group (P = 0.026) (Table 2). USG was significantly higher in the Renaltec group (P = 0.012). None of the cats in either group had an active urine sediment and none were proteinuric. One cat in the Renaltec group and two cats in the control group were in the borderline proteinuric range. No other significant differences were observed in CBC, serum chemistry, FGF-23, FDI or IS at t0.
Results of serum chemistry, haematology, urine analysis and faecal dysbiosis index (FDI) of the cats examined at t0 (baseline), t1 (after 3 months) and t2 (after 6 months), analysed using mixed effects models with group, time point and their interaction as fixed effects and individual cat ID as random effect

Data are estimates (95% confidence interval) for comparisons between groups within each time point. P values for between-group comparisons within each time point were obtained from estimated marginal means, with Tukey adjustment for multiple comparisons. P values <0.05 were considered statistically significant and are shown in bold
USG was determined using a hand refractometer
BP was measured using high-definition oscillometry
BP = blood pressure; FGF-23 = fibroblast growth factor 23; SDMA = symmetric dimethylarginine; UPC = urine protein:creatinine ratio; USG = urine specific gravity
Concurrent medication
In total, seven cats required additional long-term medication. In the Renaltec group, one cat received amlodipine for CKD-associated hypertension and another cat was treated with inhaled fluticasone for feline asthma. In the control group, four cats received amlodipine for hypertension, and one cat with well-controlled hyperthyroidism was treated with thiamazole throughout the study.
Administration of Renaltec
Questionnaires revealed that all cats in the Renaltec group reliably received the supplement once daily, either mixed with Add One or with their regular food. Cats that initially refused Renaltec mixed with Add One readily accepted it when offered with their regular food. Owners reported no refusals after this adjustment and no side effects or changes in acceptance throughout the study.
Treatment outcome
At t1, data from 19 cats were analysed, showing no statistically significant differences between the groups for any evaluated parameters. At t2, data from eight cats in the Renaltec group and nine in the control group were available. Two cats in the Renaltec group were excluded before t2: one on day 174 because of suppurative triaditis and another was euthanased shortly before t2 because of a forelimb neoplasm (exact date unknown).
At t2, cats in the Renaltec group had significantly lower plasma IS concentrations (P = 0.045) than cats in the control group. No significant differences were observed in CBC parameters, creatinine, SDMA, FGF-23, FDI, body weight or blood pressure between the groups. UPC values of eight cats of each group were available for analysis. UPC was not performed in one cat from the control group because of an active urine sediment. UPC was significantly lower in cats in the Renaltec group than in cats in the control group (P = 0.001) although values in both groups remained within the non-proteinuric range.
There were no statistically significant differences within the groups between t0 and t1. At t2, cats in the control group had a significantly lower haematocrit (P = 0.026) compared with t0, and a significantly lower body weight than at t0 (P = 0.015) and t1 (P = 0.017) (Table 3). Cats in the Renaltec group showed a significantly lower FDI at t2 compared with t1 (P = 0.023) and t0 (P = 0.009).
Results of the statistical analysis for serum chemistry, haematology, urinalysis, faecal dysbiosis index (FDI), blood pressure and body weight at the time intervals t0–t1, t0–t2 and t1–t2, analysed using mixed-effects models with group, time point and their interaction as fixed effects and cat ID as random effect

Values in parentheses are 95% confidence interval (CI). Reported are the pairwise estimates of difference (estimate [95% CI of difference]) between time points for each group, with Tukey-adjusted P values for multiple comparisons. P values <0.05 were considered statistically significant and are shown in bold
USG was determined using a hand refractometer
BP was measured using high-definition oscillometry
BP = blood pressure; FGF-23 = fibroblast growth factor 23; SDMA = symmetric dimethylarginine; UPC = urine protein:creatinine ratio; USG = urine specific gravity
One cat in each group progressed from IRIS stage 2 to stage 3, and one cat in each group showed an increase of creatinine of more than 25% from baseline (32% in the control group and 35% in the Renaltec group) without a change in IRIS stage (Figures 2 and 3).

Changes in creatinine, symmetric dimethylarginine (SDMA), haematocrit and indoxyl sulfate over a period of 6 months (t0 = baseline, t1 = after 3 months, t2 = after 6 months) in the Renaltec and control groups. Estimated marginal means, 95% confidence intervals and P values originate from mixed-effects regressions

Changes in phosphate, fibroblast growth factor 23 (FGF-23), urine protein:creatinine ratio (UPC) and faecal dysbiosis index over a period of 6 months (t0 = baseline, t1 = after 3 months, t2 = after 6 months) in the Renaltec and control groups. Estimated marginal means, 95% confidence intervals and P values originate from mixed-effects regressions
Discussion
This pilot study investigated the effects of oral administration of Renaltec in a population of client-owned cats with naturally occurring IRIS stage 2 and 3 CKD over a period of 6 months. The main findings were that cats in the Renaltec group had significantly lower plasma IS concentrations and a significantly lower UPC compared with those in the control group after 6 months. This was attributed to an increase in plasma IS concentrations in cats from the control group and a decrease in IS levels in the Renaltec group, with neither change being statistically significant on its own.
Marked reductions in IS concentrations have been reported in geriatric cats in a previous short-term study. 26 Given that these animals did not suffer from CKD and were consequently expected to have a lower initial level of uremic toxins, such findings are not directly comparable with cats with naturally occurring renal disease. Moreover, biological variability in uremic toxin concentrations has been demonstrated, 30 indicating that more moderate reductions in IS may also be meaningful, particularly when they are achieved under routine clinical conditions in cats with naturally occurring CKD and maintained over an extended period.
Previous studies on AST-120 have demonstrated its IS-lowering effect across various species under experimental conditions. In rats, significant reductions in IS levels were observed after 1022 and 1914 weeks at dosages of 0.40–0.45 g/100 g body weight, which are substantially higher than the dose used in the present study (0.5 g/cat/day, mean weight 5.12 ± 1.82 kg). In mice, findings have been less conclusive. A 12-week study using standard food containing 5% AST-120 in mice with experimentally induced renal dysfunction showed a non-significant trend towards lower IS levels, although body weight conversion was inconclusive. 31 Similarly, a study using a Western diet (diet with high amount of saturated fatty acids, carbohydrates and low fibre content) mixed with 8% AST-120 over 3 months in nephrectomised mice demonstrated a non-significant trend towards decreased IS levels. 31
In dogs with experimentally induced heart failure and healthy kidneys, a 50% reduction in IS levels was observed after 4 weeks of treatment with 1 g/kg body weight AST-120 per day. 32 Similarly, a multicentre, randomised, double-blind, placebo-controlled, dose-escalating study in humans with moderate to severe CKD demonstrated a dose-dependent reduction in serum IS levels after 12 weeks, with concentrations progressively decreasing as AST-120 doses increased (0.9–3 g/patient q8h). 24 In contrast, another study reported a significant IS decrease in CKD patients only after a minimum of 6 months of daily therapy with 6 g of AST-120. 33 In cats, a study involving 13 cats with experimentally induced CKD (IRIS stage 2 and 3), fed a standardised renal diet, reported significant serum IS reductions after 28 days of Renaltec dosing regimens (0.5 g q24h and 0.5 mg q12h). However, by day 56, significant reductions were maintained only with twice-daily dosing. The authors attributed this finding to the significant biological variability of uremic toxins and the small sample size, which may have limited the study’s statistical power. 27 In summary, these studies highlight the variability in IS-lowering effects across different species, dosing regimens and study durations. Notably, longer treatment durations and higher doses in both humans and other animals have demonstrated more consistent reductions in IS levels. Further research is required to optimise Renaltec dosing strategies and to clarify its long-term effects on IS levels, particularly in cats with elevated baseline IS levels and advanced stages of CKD.
Numerous studies have demonstrated that proteinuria serves as a critical prognostic factor for CKD in cats.34 –37 In the present study, cats with proteinuria (UPC >0.4) were excluded. At t2, cats in the Renaltec group showed significantly lower UPC values compared with those in the control group. Similar findings have been reported in multiple studies involving rats and humans. Studies in subtotal nephrectomised rats treated with AST-120 showed significantly lower urinary protein excretion compared with the control groups.14,15,38 Histological evaluations from these studies suggest that AST-120 may prevent glomerular sclerosis and tubulointerstitial fibrosis, which are conditions linked to urinary protein excretion.14,15 In addition, a clinical study in humans demonstrated that daily administration of AST-120 over 12 months significantly reduced proteinuria levels in non-diabetic patients with CKD compared with those receiving control treatment. The authors proposed that AST-120 treatment has a beneficial effect on reducing tubular damage. 39 Although the present study demonstrated a statistically significant reduction in UPC, this change occurred within the non-proteinuric range and is therefore unlikely to be clinically relevant in this cohort. However, the finding may still suggest a potential effect of Renaltec on urinary protein excretion that warrants further investigation in proteinuric CKD cats. In addition, future investigations incorporating histopathological analyses are necessary to explore potential effects on glomerular sclerosis and tubulointerstitial fibrosis.
No significant differences in serum creatinine levels or CKD progression were observed between the groups at any time point during this study. In human CKD studies, a reduction in serum creatinine was observed after more than 30 weeks of AST-120 treatment 25 but not after 12 weeks. 24 In contrast, rats treated with high doses of AST-120 showed reduced creatinine levels after only 12 weeks. 22 Given the limited number of studies on the effects of Renaltec in cats with CKD, it remains uncertain whether a longer treatment duration could result in significantly reduced creatinine levels in this species.
Multiple studies have shown that human patients with CKD exhibit an altered composition of gut microbiota compared with healthy controls.40 –43 A study investigating the potential of dietary probiotics in rats found a significant reduction in serum levels of IS, among other uremic toxins, suggesting a beneficial effect in reducing the accumulation of certain uremic toxins through modulation of the intestinal microbiome in CKD patients. 44 Studies in mice and rats treated with AST-120 demonstrated decreased tissue accumulation of IS and p-cresyl sulfate (pCS) in multiple organs, 45 a change in the composition of gut microbiota involved in the production of uremic toxin precursors 46 as well as an increase in faecal excretion of indole, p-cresol and phenol and the reduction of serum IS and pCS levels. 15 A similar effect in intestinal microbiota composition was observed in humans after 3 months of AST-120 treatment; although the trend towards reduced pCS levels was noted, these results did not reach statistical significance. 47 A study examining the faecal microbiome and serum IS concentrations in cats with CKD revealed decreased faecal bacterial diversity and richness, alongside higher serum IS concentrations, compared with control cats. However, the study was unable to establish a correlation between the concentration of the measured uremic toxins and the types of bacteria known to be involved in their production in humans. 9 In the present study, a significant decrease in the FDI within the Renaltec group was demonstrated at both 3 and 6 months after treatment. This improvement may be attributed to positive changes in gut microbiota composition after administration of Renaltec. Although Renaltec is a physical adsorber of indole in the intestinal lumen, the significant reduction in FDI observed in this study may suggest indirect effects on gut microbial composition, potentially mediated by altered substrate availability. These observations are in line with findings from studies on AST-120 and raise the hypothesis that intestinal toxin adsorbers could influence both systemic toxin burden and the intestinal microbiota in feline CKD. Further studies investigating the FDI in healthy cats, cats with CKD and those treated with Renaltec are warranted to better understand these effects.
In the present study, a significant decrease in haematocrit was observed in the control group between t0 and t2, although none of the cats was anaemic. The decrease, although statistically significant, was within the reference interval and therefore not considered clinically relevant in this small cohort of cats. Increased severity of CKD is associated with the development of anaemia: of cats with CKD, 5% (IRIS stage 2), 18% (IRIS stage 3) and 53% (IRIS stage 4) develop anaemia.48,49 A primary cause of anaemia is erythropoietin (EPO) deficiency due to impaired oxygen-sensing mechanisms and reduced EPO production by peritubular renal EPO-producing cells. 50 Studies in rats showed that exposure to increased IS levels increases oxygen consumption in renal tubular cells 51 and impairs the oxygen-sensing capacity of EPO-producing cells through a hypoxia-inducible transcription factor dependent mechanism. 52 Longitudinal studies are needed to determine whether lowering IS plasma levels could delay the progression of anaemia in CKD cats.
The present study has some limitations. Randomisation resulted in some baseline imbalances (sex, tCa, USG, concomitant medications) that could have influenced the observed treatment effects. In addition, a few cats did not complete the 6-month study period, which further reduced the effective sample size and the overall statistical power of the study.
There are also statistical considerations. To contextualise the observed 22% reduction (3.3 mg/l absolute difference in mean IS concentration), a post hoc sensitivity analysis was performed using the same assumptions as the a priori power analysis. Detecting this smaller effect at 80% power would require 37 cats per group (total of 74 cats). With the actual sample size (mean of 9.5 per group), the achieved power for the observed effect was approximately 27%, calculated via the pwr package in R. 53 This indicates that the study was underpowered for detecting subtler changes but aligns with its exploratory design, which was based on detecting a large hypothesised effect derived from prior data.
The FDI has not been validated in cats with CKD, and its interpretation should therefore be approached with caution. Moreover, individual differences in the administration of Renaltec (with or without Add One) and in the composition of the renal diets were not standardised. Although all cats were fed exclusively renal diets, the relative proportions of dry and wet food varied between individuals and over time. In addition, cats were fed renal diets from two different brands or a calculated ration, which did not have identical protein and phosphate contents (crude protein 13.1–18.2 g/MJ ME [5.48–7.61 g/100 kcal ME]; phosphate 124.6–315.8 mg/MJ ME [52.1–132.1 mg/100 kcal ME]). Furthermore, only cats in the Renaltec group received Add One as a liquid treat vehicle, which may have influenced the intestinal microbiota and should be considered a potential confounding factor.
Conclusions
Treatment with Renaltec was associated with significantly lower IS concentrations and significantly lower UPC after 6 months compared with a control group, although UPC values remained within the non-proteinuric range. Furthermore, cats in the Renaltec group had a significant improvement of FDI over time, whereas cats in the control group showed a significant decrease of haematocrit and body weight over time. Although these findings are encouraging and suggest a potential benefit of Renaltec in addition to treatment according to IRIS guidelines, they should be interpreted with consideration of the small sample size and other study limitations. As this was a pilot study, further controlled trials with larger populations are needed to confirm these results and to explore the long-term effects of Renaltec in feline CKD.
Footnotes
Author note
The data sets analysed during the current study are available from the corresponding author on reasonable request.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The manufacturer provided the products Porus One and Add One for the study participants, covered the costs of the laboratory investigations and funded the calculation of the home-cooked diets for two cats. The manufacturer had no role in the collection, analyses or interpretation of the data, in the writing of the manuscript or in the decision to publish the results.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognised high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from committee was therefore not specifically required for publication in JFMS. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers, tissues and samples) for all procedure(s) undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
