Abstract
Background:
Kidney transplantation is the ideal treatment for patients with end-stage renal disease. However, this population has a higher risk of cancer. This highlights the importance of early screening to improve outcomes in the detection of neoplasms, associated risk factors, and prompt treatment in this population.
Objectives:
The objective of this study is to determine the incidence and risk factors associated with post-transplant cancer.
Methods:
This was a historical analytical cohort observational study that evaluated the incidence of cancer and associated risk factors in kidney transplant recipients in the Clínica Universitaria Colombia program between 2007 and 2022.
Result:
A total of 555 patients were included, with a predominance of males (61.1%). An incidence of posttransplant cancer was documented in n = 79 (14%) patients at a follow-up of 84.7 months, with skin neoplasms in n = 33 (41.7%) of cases, followed by solid organ neoplasms n = 31 (39.2%). Associated risk factors identified were age at the time of transplantation being statistically significant (Hazard ratio (HR): 1.04; 95% confidence interval (CI): 1.02–1.06), a history of cancer prior to kidney transplantation (HR: 3.62; 95% CI: 1.46–8.99), and diabetes mellitus (HR: 2.08; 95% CI: 1.24–3.49). A statistically significant relationship was documented as a protective factor for mammalian Target of Rapamicyn (mTOR) inhibitors and solid organ neoplasms with relative risk (RR) 0.414 (95% CI: 0.19–0.88). No increase of posttransplant lymphoproliferative disorder (PTLD) was found with co-stimulation signal inhibitor-based therapy.
Conclusions:
In a Latin American kidney transplant cohort the incidence of post-transplant malignancy was found to be 14% in a median follow-up of 7 years. This study found that risk factors include age, history of cancer prior to transplantation, and diabetes mellitus. Use of mTOR pathway inhibitors was associated with a reduced risk of solid organ neoplasms. No increase in the frequency of PTLD was found in patients receiving costimulation signal inhibitors.
Introduction
Kidney transplantation provides a solution to a number of complications associated with chronic kidney disease. However, being a kidney transplant recipient increases the risk of infections, cardiovascular events, and neoplasms. The latter being the third leading cause of mortality, following infectious and cardiovascular death, with an incidence of 12%–14.8% respectively.1,2 Mortality is up to three times higher in patients who develop cancer after a kidney transplant than in those who do not, and there is also a higher risk of graft loss given the need to reduce the intensity of immunosuppression.2,3 Postkidney transplant neoplasms can develop as de novo neoplasms, recurrence of a previous neoplasm or transmission from the donor. Among the most frequently occurring neoplasms developed after a solid organ transplant, specifically a kidney transplant, skin neoplasms have been documented. These can be either melanoma or nonmelanoma. Other solid organs affected include the pulmonary, gastrointestinal, urinary, and gynecologic systems, as well as the hematologic system, which can present as posttransplant lymphoproliferative disorder (PTLD).4,5
The risk factors associated with the development of posttransplant cancer vary according to ethnicity, geographical location, and lifestyle. The condition is more prevalent in females, elderly patients, and individuals with a history of previous transplantation. 6 It has also been linked to posttransplant neoplasms, the etiology of chronic kidney disease, time on renal replacement therapy, smoking, transfusions, high body mass index (BMI), and the development of acute or chronic rejection. 7 There is also an association with some viral infections such as human papillomavirus, Epstein–Barr virus, herpes virus 8, and cytomegalovirus as additional risk factors. 8
At the national and regional levels in Latin America, there are few studies evaluating the incidence of neoplasms in patients who have received a kidney transplant with identification of risk factors, making it difficult to determine prevention and early detection strategies. The objective of this study is to identify the incidence of posttransplant cancer and describe the presence of associated risk factors in kidney transplant recipients.
Methods
We did a historical analytical cohort observational study where we checked out the medical records from patients who got a kidney transplant between 2007 and 2022 in the kidney transplant program at the Clínica Universitaria Colombia and the Clínica del Country in Bogotá, Colombia. The inclusion criteria were patients older than 18 years of age with a first or second kidney transplant from a deceased or living donor, with a histological diagnosis of post-transplant cancer at any time during follow-up.
Patients who were lost to follow-up due to transfer to another center, or with medical histories without cancer diagnosis data or without a report of the diagnostic pathology were excluded from the analysis.
To calculate the sample size, a precision of 3%, a confidence level of 95%, and a design effect (DEFF) of 1 were used, and it was compared with different studies to determine the sample size, leaving 200 as the highest sample of patients to be collected. The size was calculated using Epidat software version 4.1 (xunta de galicia conselleria de sanidade); data collection was performed by reviewing medical records using simple random sampling. Data collection was carried out by reviewing medical records from the institution’s different software programs (AVICENA and SOPHIA) by keralty. The researchers entered the data in a previously established format, which was then entered into a database created in Microsoft Excel. In our group, we have a database with all the patients in active follow-up in the program, this database is fed directly from the patients’ medical records with the exact date of the diagnoses, in addition the criteria to include the cancer diagnosis had to include the pathology report and the exact date of the diagnosis
For the analysis, categorical variables were described as percentages and absolute frequencies. Continuous variables were described as median and interquartile range (IQR), given that they do not have a normal distribution, which was explored using the Shapiro–Wilk normality test. A bivariate analysis was performed using the chi-square test or Fisher’s exact test for categorical variables. In the case of quantitative variables, the Mann–Whitney U test was performed. The cumulative incidence was calculated based on patients who developed their first cancer diagnosis, relative to the total at-risk population (total number of transplant patients during the period) at the start of the follow-up cohort.
The survival analysis was based on time zero, the date of transplant, with total follow-up in months until the date of death or the last follow-up available in clinical records.
Survival until the presentation of the first neoplasm was estimated using a Kaplan–Meier analysis.
Subsequently, a Cox regression model was performed to calculate the univariate Hazard ratio (HR) with a 95% confidence level, and finally, a multivariate model was performed with a variable selection process using the stepwise regression strategy, seeking the most accurate model with the Akaike criterion by choosing the clinical variables statistically related to posttransplant neoplasms. The information was analyzed using R software version 4.3.2. (R Core Team R Foundation for statistical computing).
The study was conducted in accordance with the ethical principles for medical research involving human subjects set forth in the Declaration of Helsinki. In this study, the participants’ medical records were reviewed. Therefore, according to Colombian regulations issued by the Ministry of Health through Resolution 8430 of 1993, it was considered a risk-free investigation and did not require the signing of an informed consent form. The study was approved by the research ethics committees of Clínica Universitaria Colombia, with protocol number 042-24, and by the research committee of the Fundación Universitaria de Ciencias de la Salud, with protocol number 743.
Results
Demographic and clinical characteristics of the population
A total of 649 patients who underwent kidney transplantation during the study period were included, 94/649 patients (14.4%) were lost to follow-up, most of them due to transfer to other centers for follow-up. 555 met the selection criteria (Figure 1), with a median follow-up of 84.7 months and a median age of 44.8 years with an IQR (2.8–56.3); males were predominant n = 339 (61.1%).

Flow chart.
In terms of the cause of chronic kidney disease, an unknown etiology was found in n = 207 patients (37.3%), followed by glomerular disease in n = 164 (29.5%). Out of the general population, 460 patients (82.9%) received a transplant from a cadaveric donor, and 68.5% (n = 380 patients) received induction with thymoglobulin. Some differences were found in the clinical and demographic variables of the population with posttransplant cancer compared to the population without cancer. These differences included the type of previous dialysis therapy, BMI, history of cancer prior to transplant and use of induction therapy with thymoglobulin or basiliximab. Table 1 shows the demographic, transplant, and immunosuppressive therapy characteristics in the total cohort and among patients who developed cancer versus those who did not.
Demographic variables.
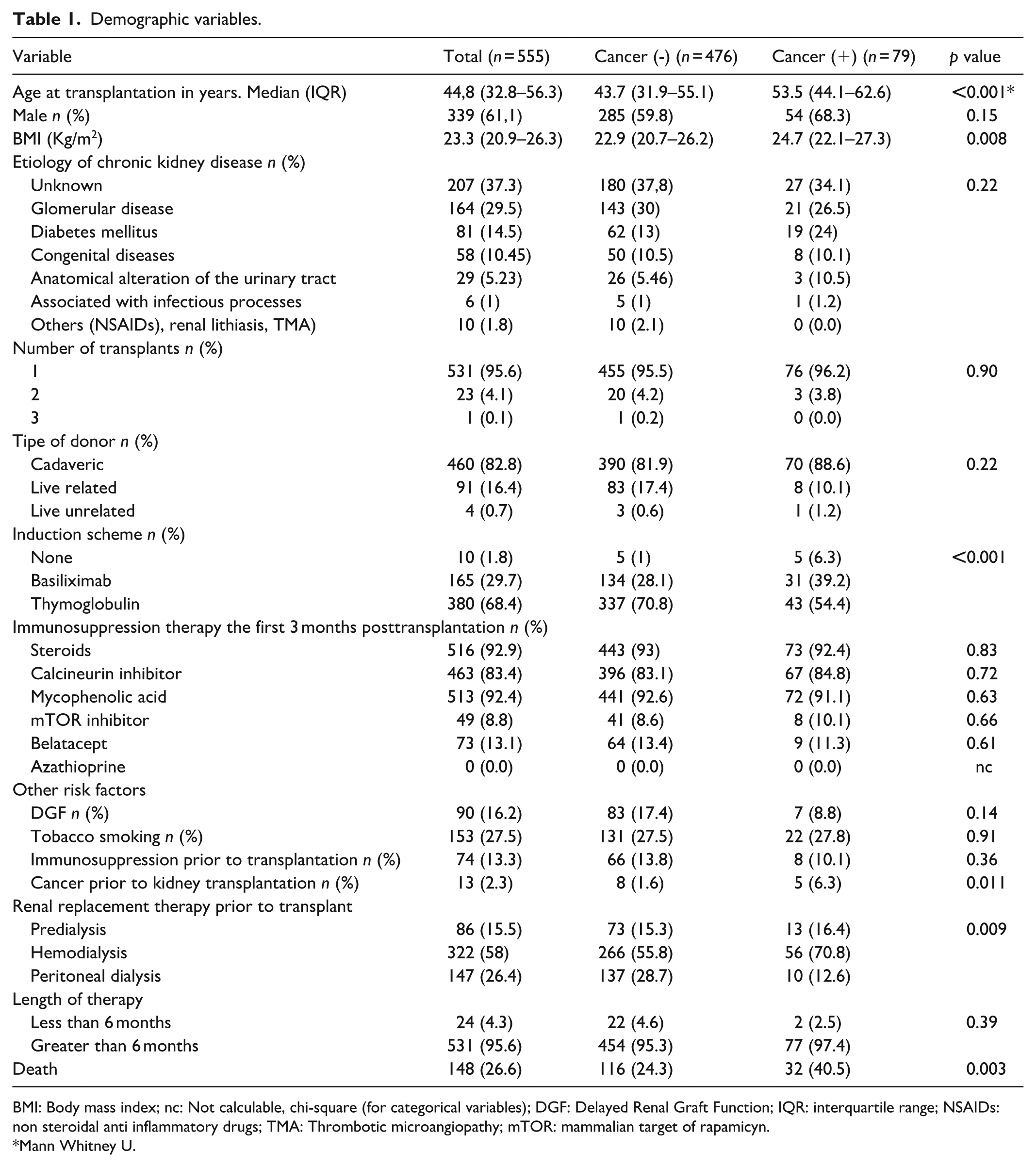
BMI: Body mass index; nc: Not calculable, chi-square (for categorical variables); DGF: Delayed Renal Graft Function; IQR: interquartile range; NSAIDs: non steroidal anti inflammatory drugs; TMA: Thrombotic microangiopathy; mTOR: mammalian target of rapamicyn.
Mann Whitney U.
Incidence and risk factors in the post-transplant cancer population
Our population shows an incidence of 14.2% (n = 79) of cancer after receiving a kidney transplant at a median of 84.7 months. Skin neoplasms were the most frequent, accounting for 41.7% (n = 33), followed by solid organ neoplasms, accounting for 39.2% (n = 31), and, to a lesser extent, hematological neoplasms, accounting for 18.9% (n = 15). Of the population with cancer, 68.4% (n = 54) were men. In 10.13% (n = 8) of patients, cytomegalovirus disease was documented, and in 2.53% (n = 2), Epstein–Barr virus was documented prior to the presentation of the neoplasm. Additionally, in 39.24% (n = 31), rejection was present before the first neoplasm was documented. Regarding the immunosuppression regimen in the cancer population, 31.6% (n = 25) used Calcineurin inhibitor (CNI), 41.8% (n = 33) used belatacept, 88.6% (n = 70) used mycophenolic acid, and 84.8% (n = 67) used steroids 1 year prior to the diagnosis of the first cancer. The immunosuppression regimen of the cancer population is described in Table 2. Use of mTOR inhibitor was found to be protective for development of solid organ neoplasm with relative risk (RR), 0.414 (95% Confidence interval (CI): 0.19–0.88).
Immunosuppression 1 year prior to diagnosis of each cancer.

No association was found in our series between the use of belatacept and an increase in the incidence of skin neoplasms, solid organ neoplasms, or PTLD (Table 3).
Relationship between immunosuppressants and type of neoplasms.
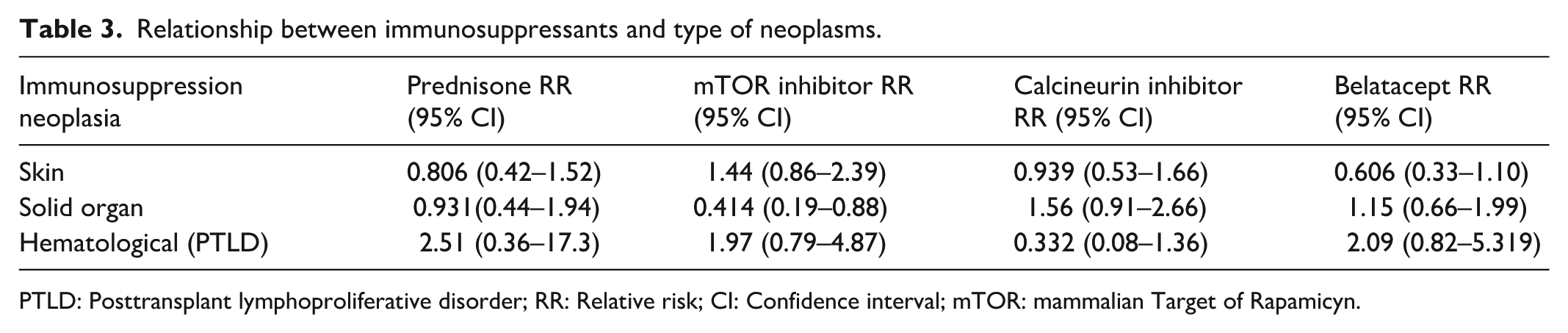
PTLD: Posttransplant lymphoproliferative disorder; RR: Relative risk; CI: Confidence interval; mTOR: mammalian Target of Rapamicyn.
Univariate and multivariate analysis
HR was calculated using a Cox regression model, demonstrating that age at the time of transplantation and a history of cancer are statistically significant risk factors for posttransplant neoplasia, with HR, 1.04 (95% CI: 1.02–1.06) and HR, 3.62 (95% CI: 1.46–8.99), respectively. Other clinically significant variables are presented in Table 4. In the multivariate analysis, age at transplantation was found to be a statistically significant risk factor with an HR of 1.04 (95% CI: 1.03–1.06, p = 0.001), and a history of cancer with an HR of 2.14 (95% CI: 0.85–5.4, p = 0.108), although the latter was not statistically significant but was included in the final model.
Univariate and multivariate analysis by Cox regression.

BMI: Body mass index; Inf: Infinite; HR: Hazard ratio; CI: Confidence interval; NSAIDs: non steroidal anti inflammatory drugs; TMA: Thrombotic microangiopathy.
Overall and graft survival
Among the whole transplant cohort, 148 deaths were documented during follow-up, with a median survival (time at which 50% of the cohort died) of 167 months. Of the 148 deaths during follow-up, 17 (11.4%) patients died from causes associated with neoplasia; hematological cancer n = 7 (4.7%) and gastrointestinal tract cancer n = 3 (2.02%) were the most frequent. Other causes of death in the whole transplant cohort were infection in 93 (62.8%) cases (22 due to COVID-19), cardiovascular disease in 26 (17.5%) cases, and other causes in 12 (8.1%) cases. Death-censored graft loss was observed in 44 patients (7.9%). Of these patients, four had posttransplant cancer, one of which was cancer-related.
Regarding survival within the study cohort, no statistically significant differences were observed between patients who developed cancer and those who did not, according to the log-rank test (p = 0.53) (Figure 2).

Kaplan–Meier analysis of survival in patients with cancer versus noncancer.
Regarding renal function and proteinuria in patients with cancer, an increase in proteinuria of 0.2 g/24 h (p 0.19) was documented and a decrease in the glomerular filtration rate (GFR) measured by CKD-EPI of 2 ml/min/1.73 m2 (p 0.31) in a 2-year follow-up after diagnosis of posttransplant cancer, without being statistically significant (Figure 3(a), (b))
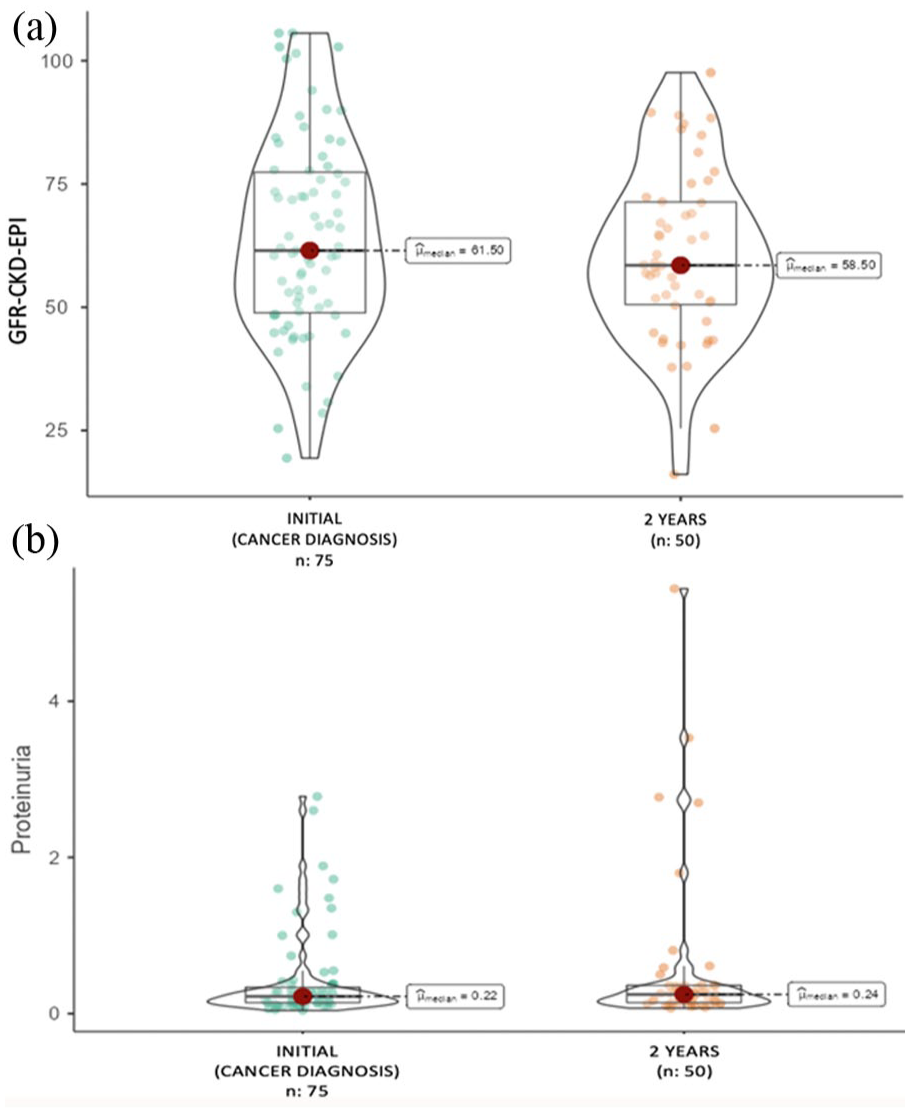
Changes in (GFR) and 24 h proteinuria: (a) Changes in GFR: (b) Changes in proteinuria in 24 h.
Discussion
In general, kidney transplants increase the risk of posttransplant cancer by 1.5–3 times compared to the rest of the population. 8 There are few reports in Latin America describing the incidence of postkidney transplant cancer. Pinto Filho V et al., describe a cancer frequency of 4.2% in a cohort of patients in Brazil over a 10-year period out of a total of 599 kidney transplant patients, with nonmelanoma skin cancer being the most common, accounting for around 60% of cases, followed by other types of cancer such as lung, breast, and colon cancer. This highlights the need for regional studies evaluating incidence and risk factors. 9 Our study documents the most relevant epidemiological findings in patients with posttransplant cancer in the Colombian population, identifying an incidence of posttransplant cancer of 14.2%. This is similar to what has been documented in other studies and is within the expected range according to demographic distribution, which is between 1.9% and 18%.1,8,10,11
In reviews such as that conducted by Al-Adra et al, skin neoplasms are reported to have a cumulative incidence of more than 60% in patients with posttransplant cancer. In our study, nonmelanoma skin cancer was found to be the most frequent (41.7%), with a predominance of basal cell carcinoma, followed by solid organ cancer (39.2%), of which gastrointestinal tract involvement was the most frequent, similar to that reported by Re Sarto et al., who found that in 177 patients with at least 1 posttransplant cancer, nonmelanoma skin cancer was predominant (55.1%), followed by solid organ cancer (38.6%), with differences in the type of neoplasms and organ involvement, respectively. These findings differ from those of Krishnan et al. who documented in a follow-up of 21,844 patients with kidney transplants between 1980 and 2016 from the Australian and New Zealand Dialysis and Transplant Registry (ANZDATA) PTLD as the most frequent cancer (25%), followed by urogenital tract neoplasms (18%) and melanoma (12%), confirming the geographical variability in the incidence of posttransplant neoplasms, which depends on the different lifestyles of the population.6,12,13
In the analysis of demographic, pathological, and transplant-related risk factors, a higher incidence was observed in males, consistent with the findings reported in 2019 by Giocoa et al. In another study, a mean follow-up of 7.8 years was conducted on 535 patients with kidney transplants, in whom 39 cases of posttransplant cancer were documented, with a predominance of males (n = 17); this was also confirmed in 2021 by Fröhlich et al., in Berlin with a follow-up of 1417 patients, of whom 154 presented with posttransplant cancer, also predominantly in males. 14
In terms of the etiology of chronic kidney disease, an unknown etiology predominated in 37% of cases. This differs from the findings reported by Resarto et al., in which there was greater precision in the etiological diagnosis of chronic kidney disease. In their study, a maximum of 12% of cases were indeterminate or unknown. They also reported an association between posttransplant neoplasms and the cause of renal involvement. This association was mainly observed in cases of pathology requiring immunosuppressive treatment, such as vasculitis. 10 Additionally, our study showed a relationship between posttransplant cancer and a history of diabetes mellitus HR, 2.08 (CI: 1.24–3.49), differing from the report by Kasiske et al, who, in a cohort of 35,765 kidney transplants, found that having diabetes mellitus as the etiology of chronic kidney disease was a protective factor for posttransplant nonskin neoplasms RR, 0.82 (95% CI: 0.71–0.95) and for nonmelanoma skin neoplasms RR, 0.63 (95% CI: 0.54–0.73), 15 highlighting the need for further research into the post-transplant cancer population and the importance of understanding the etiology of kidney damage through multicenter studies.
Our study found a statistically significant increase in risk between age at the time of transplantation and the presence of posttransplant cancer in univariate and multivariate analyses, showing that for each year of increase in the recipient’s age at the time of receiving a kidney transplant, the risk of posttransplant neoplasia increases 1.04 times, a finding that aligns with that reported by Webstern et al. in 2007 using a cohort of 15,183 transplant patients from the Australian and New Zealand Dialysis and Transplant Registry (ANZDATA), finding a statistically significant relationship in patients over 55 years of age who received a kidney transplant and posttransplant cancer, HR, 2.6 (95% CI: 2.3–2.9) for women and HR, 1.9 (95% CI: 1.7–2.1) for men 16 ; confirmed by Liu et al. in 2022, who also determined an association similar to our study (HR, 1.057; 95% CI: 1.004–1.112). 6
Only a few studies have followed up transplant patients with a history of neoplasia, and a significant increase in the risk of recurrence or de novo neoplasms has been identified in this type of patient. Our study documents a statistically significant relationship between a history of neoplasia and the incidence of posttransplant cancer HR, 3.62 (95% CI: 1.46–8.99), confirming the importance of strict follow-up of patients after solid organ transplantation regardless of the type of neoplasm previously presented, even though the different remission times are met according to the type of tumor and histological variety previously presented. 17
Multiple risk factors associated with posttransplant cancer have been identified, such as immediate graft function, the use of thymoglobulin as induction therapy, maintenance medications, exposure to certain viral pathogens with oncogenic capacity, genetic factors, history of smoking, type of renal replacement therapy and its duration, as well as the use of immunosuppressants.1,3,5,14 In our study, an association was documented between the development of cancer and a BMI in the overweight range, with no association found with any of the other variables mentioned above.
The prolonged use and cumulative doses of maintenance immunosuppressive drugs have been linked to the presence of posttransplant neoplasms, as these drugs promote cellular changes. This effect is greater with calcineurin inhibitors than with mTOR inhibitors.8,11 mTOR inhibitors have been reported to decrease the incidence of posttransplant neoplasms, as reported by Campistol et al. In a multicentre, randomized, open-label study of 430 European patients who were switched to sirolimus after 3 months and were free of calcineurin inhibitors, a reduction in the incidence of posttransplant skin and solid organ neoplasms was observed at 5 years (RR, 0.343; 95% CI: 0.205–0.574 and 9.6% versus 4.0%, p = 0.032, respectively). These findings are consistent with those of our study, in which mTOR inhibitors were found to act as a protective factor against posttransplant neoplasms, particularly solid organ neoplasms (RR, 0.414; 95% CI: 0.19–0.88), which was statistically significant.18,19
With the availability of new medications such as co-stimulation inhibitors (Belatacept), a higher incidence of PTLD was documented in patients with negative Epstein–Barr serology compared to those with positive serology. 20 These findings were documented in real-life studies such as those conducted by Larsen et al. using the ENLiST registry with 933 Epstein–Barr seropositive patients treated with Belatacept, showing a cumulative incidence of 0.08 per 100 person-years for PTLD without central nervous system (CNS) involvement and 0.03 per 100 person-years without CNS involvement, 21 less than the incidence established for seronegative patients. In our study, no statistically significant relationship was documented between the use of immunosuppression and posttransplant neoplasms, especially PTLDs.
Of the 148 reported deaths, 32 patients had posttransplant cancer. Of these patients, 17 (11%) died as a direct result of the neoplasm as described by Rosales et al. who identified a cohort taken from the dialysis and transplant registry of New Zealand and Australia between 1980 and 2013, which included 15,183 patients, in which 5284 deaths were documented, of which 1061 (20%) were due to cancer, 22 confirming posttransplant cancer as the third leading cause of mortality in kidney transplant patients after infections and cardiovascular events.1,2
When it comes to changes in GFR and 24 h proteinuria in a 2 years follow-up in our cohort, we didn’t find any statistically significant variations, unlike what Imamura et al. reported using a cohort of 1973 kidney transplant recipients, where 50% of patients with posttransplant cancer lost the graft at 5 years of follow-up after cancer diagnosis. 23
Limitations
This is a retrospective cohort study, and the mean follow-up time is short for assessing an outcome such as cancer and its impact on mortality. There is a significant loss of patients during follow-up.
There was difficulty in assessing the association with immunosuppression prior to cancer diagnosis; given that drug titration is a frequent practice preventing full description of its use; this highlights the need for research on cancer detection, diagnosis and therapy to enable more timely and accurate diagnoses and to improve curative treatments in kidney transplant recipients.
Another limitation of our study is that the sample size was calculated based on the expected incidence of cancer in the population, using precision parameters for proportion estimates, rather than on the statistical power required to detect a specific hazard ratio in the Cox regression analysis. Consequently, while the sample was adequate for estimating incidence with the desired precision, it may have been underpowered to detect certain associations, particularly for exposures with low prevalence or small effect sizes.
Conclusion
There is evidence of a high incidence of posttransplant cancer, being nonmelanoma skin cancer the most frequent. Hematologic and gastrointestinal tract cancer were the leading causes of cancer-related death. We identified statistically significant risk factors for the presentation of cancer such as age at the time of transplantation, history of cancer prior to transplantation and diabetes mellitus as etiology of chronic kidney disease and protective factors such as the use of mTOR inhibitors. It is considered necessary to implement screening, prevention and early detection programs for patients with renal transplants, particularly those with risk factors identified in our population. This data also provides a basis for the implementation of machine learning models to predict risk in our population.
Footnotes
Acknowledgements
To the teachers and fellow residents, who contributed to the development of the research work through scientific advice and data collection.
Author contributions
Torres R, Montero C, Mendoza E, and Ruano C participated in the original research design, data collection, analysis of results, and preparation of the main document. Andrade D performed the methodological design and statistical analyses. Reina M, Roselli C, Flechas JA, and Olivares O participated in the final review of the main document.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Presentations
The abstract of this original work was presented as a poster at the 2025 World Transplant Congress, San Francisco, USA
