Abstract
Objectives:
Although angiotensin II receptor blockers are effective for patients with chronic kidney disease, dose-dependent renoprotective effects of angiotensin II receptor blockers in patients with moderate to severe chronic kidney disease with non-nephrotic proteinuria are not known. Our aim was to elucidate the dose-dependent renoprotective effects of angiotensin II receptor blockers on such patients.
Methods:
A multicenter, prospective, randomized trial was conducted from 2009 to 2014. Patients with non-nephrotic stage 3–4 chronic kidney disease were randomized for treatment with either 40 or 80 mg telmisartan and were observed for up to 104 weeks. Overall, 32 and 29 patients were allocated to the 40 and 80 mg telmisartan groups, respectively. The composite primary outcome was renal death, doubling of serum creatinine level, transition to stage 5 chronic kidney disease, and death from any cause. Secondary outcomes included the level of urinary proteins and changes in the estimated glomerular filtration rate.
Results:
There was no difference in the primary outcome (p = 0.78) and eGFR (p = 0.53) between the two groups; however, after 24 weeks, urinary protein level was significantly lower in the 80 mg group than in the 40 mg group (p < 0.05). No severe adverse events occurred in either group, and the occurrence of adverse events did not significantly differ between them (p = 0.56).
Conclusion:
Our findings do not demonstrate a direct dose-dependent renoprotective effect of telmisartan. The higher telmisartan dose resulted in a decrease in the amount of urinary protein. Even though high-dose angiotensin II receptor blockers may be preferable for patients with stage 3–4 chronic kidney disease, the clinical importance of the study results may be limited. The study was registered in the UMIN-CTR (https://www.umin.ac.jp/ctr) with the registration number UMIN000040875.
Introduction
Renoprotective therapy, including inhibition of the renin angiotensin system (RAS), in patients with chronic kidney disease (CKD) is important to support renal function, especially in patients with overt proteinuria.1,2 Several systematic reviews have evaluated the risk of combined renal outcomes of treatment with angiotensin II receptor blockers (ARBs) in CKD patients and indicated that ARBs have a favorable effect on renal outcomes.3–6
Although diabetes is still the predominant cause of renal failure in patients undergoing hemodialysis in Japan, the proportion of patients with nephrosclerosis has increased to nearly that of patients with chronic glomerulonephritis. 7 According to the African American Study of Kidney Disease and Hypertension (AASK) trial, nephrosclerosis tends to be associated with a lower degree of proteinuria than diabetic nephropathy or glomerulonephritis, and the administration of RAS inhibitors to patients with nephrosclerosis has little effect on renal outcomes compared with beta-blockers or calcium channel blockers. 8 In addition, there is a concern that lower glomerular blood pressure (BP) may be associated with reduced glomerular filtration rates in patients with nephrosclerosis, 9 especially in elderly patients with moderate to severe CKD. 10 Therefore, safety and efficacy should be considered when administering RAS inhibitors to CKD patients with non-nephrotic proteinuria.
It has been demonstrated that RAS inhibition by ARBs may exert renoprotective effects in a dose-dependent manner.11–16 For example, 300 mg irbesartan exerts stronger effects than 150 mg irbesartan in terms of urinary albumin excretion and overt diabetic nephropathy prevention. 15 In these studies, none of the participants had moderate to severe renal insufficiency, such as CKD stage 3–4; only one study investigated dose-dependent renoprotection in patients with moderate to severe CKD and found a higher degree of proteinuria. 16 Consequently, we attempted to elucidate whether ARBs exert dose-dependent renoprotective effects in patients with moderate to severe CKD and non-nephrotic proteinuria. We conducted a prospective multicenter open-label analysis to clarify the dose-dependent renoprotective effect and safety of the long-term administration of the ARB telmisartan on patients with stage 3 or 4 CKD without nephrotic level proteinuria (e.g. 3.5 g/day).
Methods
Study design
This was a multicenter, prospective, open-label, randomized control study with an inclusion period from 1 March 2009 to 29 February 2012. Participants were included if they were at least 20 years old, had stage 3–4 CKD with an estimated glomerular filtration rate (eGFR) of 15–60 mL/min/1.73 m2, were receiving antihypertensive therapy, and had sustained BP control. The exclusion criteria were as follows: heavy liver dysfunction like Child-Pugh class C cirrhosis and/or poor bile secretion, possibility of pregnancy or lactation, history of hypersensitivity to telmisartan or its analogous drugs, nephrotic proteinuria (>3.5 g/gCr), corticosteroid or immunosuppressant use within the past 6 months, acute myocardial infarction within the past 6 months, congestive heart failure, cerebral infarction, transient ischemic attack, and renal artery stenosis. After considering the eligible patients in our facilities during the period of this trial, the sample size was settled to 100. Written informed consent was obtained from all participants. All procedures involving human participants were performed in accordance with the ethical standards of the institutional and/or national research ethics committee (Nagasaki University Hospital IRB approval number 08090558-6) and the 1964 Helsinki Declaration and its amendments or comparable ethical standards. The study was registered in the UMIN-CTR (https://www.umin.ac.jp/ctr) under registration number UMIN000040875.
Patients were observed for 104 weeks, and the target BP level was less than 130/80 mm Hg. Patients with a urinary protein level of >1 g/gCr have a target BP level of <125/75 mm Hg, in accordance with the Japanese Society of Nephrology guidelines. 17 Throughout the follow-up period, antihypertensive drugs were allowed as long as they were not RAS inhibitors, such as angiotensin-converting enzyme inhibitors (ACEi) or anti-aldosterone drugs.
Run-in and screening periods (4 weeks)
Four weeks before randomization, ACEi, ARBs, and direct renin inhibitors were discontinued, and the patients were prescribed 20 mg telmisartan. In fact, telmisartan was prescribed within 4 weeks before the randomization. During this period, patients with a sustained potassium level of 5.5 mEq/L under appropriate anti-hyperkalemia therapies, including diet guidance, ion exchange resin use, and metabolic acidosis adjustment, and those who experienced a 30% increase in the serum creatinine level were excluded from further analyses.
Observational period (8 weeks)
Participants were randomized 1:1 into pre-allocated block sizes (block size 4) and stratified by age (60 years and over or less), sex (male or female), and CKD stage (3 or 4) using the UMIN-INDICE system. Participants in Group A (low-dose group) received telmisartan at an up-titrated dose of 40 mg at 4 weeks, and this dose was continued for an additional 4 weeks. Participants in Group B (high-dose group) received telmisartan at an up-titrated dose of 40 mg over the first 4 weeks; thereafter, the dose was increased to 80 mg for the next 4 weeks. All participants were expected to receive nutritional guidance (salt restriction and low-protein diet) at least one time during the observational period. The study protocol is illustrated in Figure 1.

Study protocol. The study protocol consisted of a screening period (4 weeks), observational period (8 weeks), and treatment period.
Data collection
BP, blood and urinary chemistry data (serum creatinine, eGFR, and urinary protein), history of adverse events, and patients’ backgrounds were collected at the entry of this study and again at week 0 (initiation of telmisartan) and weeks 12, 24, 52, 76, and 104. Regarding the baseline data, we divided the patients into three categories based on the amount of proteinuria, which was proposed as a substitute for the microalbuminuria categories of the CKD heat map. Specifically, a protein–creatinine ratio (PCR) < 142 mg/gCr corresponds to the albumin–creatinine ratio (ACR) stage A1, ACR < 30 mg/g, and PCR of 142–660 mg/gCr corresponds to the ACR stage 2, ACR of 30–299 mg/gCr, and PCR > 660 mg/g corresponds to the ACR stage 3, and ACR > 300 mg/g. 18 The eGFR was calculated according to the Japanese Society of Nephrology as 194 × Cr−1.094 × age−0.287 for men and the same value × 0.739 for women. 19
Outcomes
The primary outcome was a composite of a doubling of the serum creatinine level, transition to CKD stage 5, end-stage renal disease onset, or death from any cause. Secondary outcomes included proteinuria and eGFR decline. Regarding the follow-up schedule, participants visited the facilities every 1–3 months.
Statistical analysis
All analyses were conducted in accordance with the intention-to-treat principle. Continuous values are shown as mean ± standard deviations and/or median ± interquartile ranges. Categorical variables are expressed as percentages. Statistical analyses, including t tests, Wilcoxon rank sum tests, and Fisher’s exact tests, were used to evaluate differences between the groups. BP, eGFR, and urinary protein excretion were analyzed by a repeated measures analysis of variance (ANOVA), and multiple comparisons were conducted when the ANOVA was significant. The event occurrence rate was evaluated using the Kaplan–Meier method and compared using a log rank test. All statistical analyses were performed using JMP 13 software (SAS Institute Inc., Cary, NC, USA). Differences with p < 0.05 (two-tailed) were considered statistically significant.
Results
Clinical characteristics of patients
Seventy-three patients were screened during the run-in period. Of these, 12 patients were excluded due to sustained hyperkalemia (n = 1), high-degree proteinuria (n = 6), and lost consent or dropout (n = 5). The remaining 61 eligible participants were randomly allocated to Group A (telmisartan 40 mg; 32 patients) and Group B (telmisartan 80 mg; 29 patients). The baseline characteristics of patients are shown in Table 1. There were no significant differences in the backgrounds of the patients between the two groups, except with regard to the serum sodium level; Group A had a higher serum sodium level than Group B (p = 0.02). In Group A, 27 participants completed the 104-week analysis or reached the primary outcome; in Group B, 23 participants completed the analysis or reached the primary outcome. The flow chart of patients is shown in Figure 2.
Patient characteristics at week 0.

ADPKD: autosomal dominant polycystic kidney disease; Hb: hemoglobin; BUN: blood urea nitrogen; eGFR: estimated glomerular filtration rate; Na: serum sodium; K: serum potassium; Cl: serum chloride; LDL: low-density lipoprotein.
Statistical analyses were done using t tests and Wilcoxon rank sum tests.
Median (interquartile range).

Flow chart of patient inclusion. Flow chart was based on the CONSORT guidelines.
Primary outcome
This study was conducted from 2009 to 2014, and the average follow-up period was 88.6 weeks. In total, five patients in Group A reached the primary outcome: three patients reached CKD stage 5 (eGFR < 15 mL/min/1.73 m2), one patient died from acute myocardial infarction, and one patient suddenly died from cardiovascular disease (etiology unknown). Furthermore, five patients reached the primary outcome in Group B: four patients reached stage 5 CKD, and one patient began hemodialysis. Overall, there was no significant difference in the primary outcomes between these two groups (renal events free rate (p = 0.33) and renal and death free events (p = 0.78)). The Kaplan–Meier curves are shown in Figure 3.

Survival analysis of patients reaching the primary outcome. Kaplan–Meier curves of patients segregated by (a) composite renal outcome and (b) renal outcome and all causes of death. Straight line: Group A; telmisartan 40 mg. Dotted line: Group B; telmisartan 80 mg.
Secondary outcomes and BP
Throughout the follow-up period, there were no significant differences in mean systolic or diastolic BP between the groups (p = 0.30; Figure 4(a)). Furthermore, there was no significant difference between the groups in mean eGFR change (p = 0.53; Figure 4(b)). In contrast, Group B showed a significantly lower degree of proteinuria than Group A (p = 0.02), and the post hoc analyses showed that there were significant differences after 24 weeks (p < 0.05). When limited to patients whose proteinuria exceeded 142 mg/gCr at the baseline (Group A (n = 24) and Group B (n = 18)), there were no significant differences between the two groups (p = 0.11).
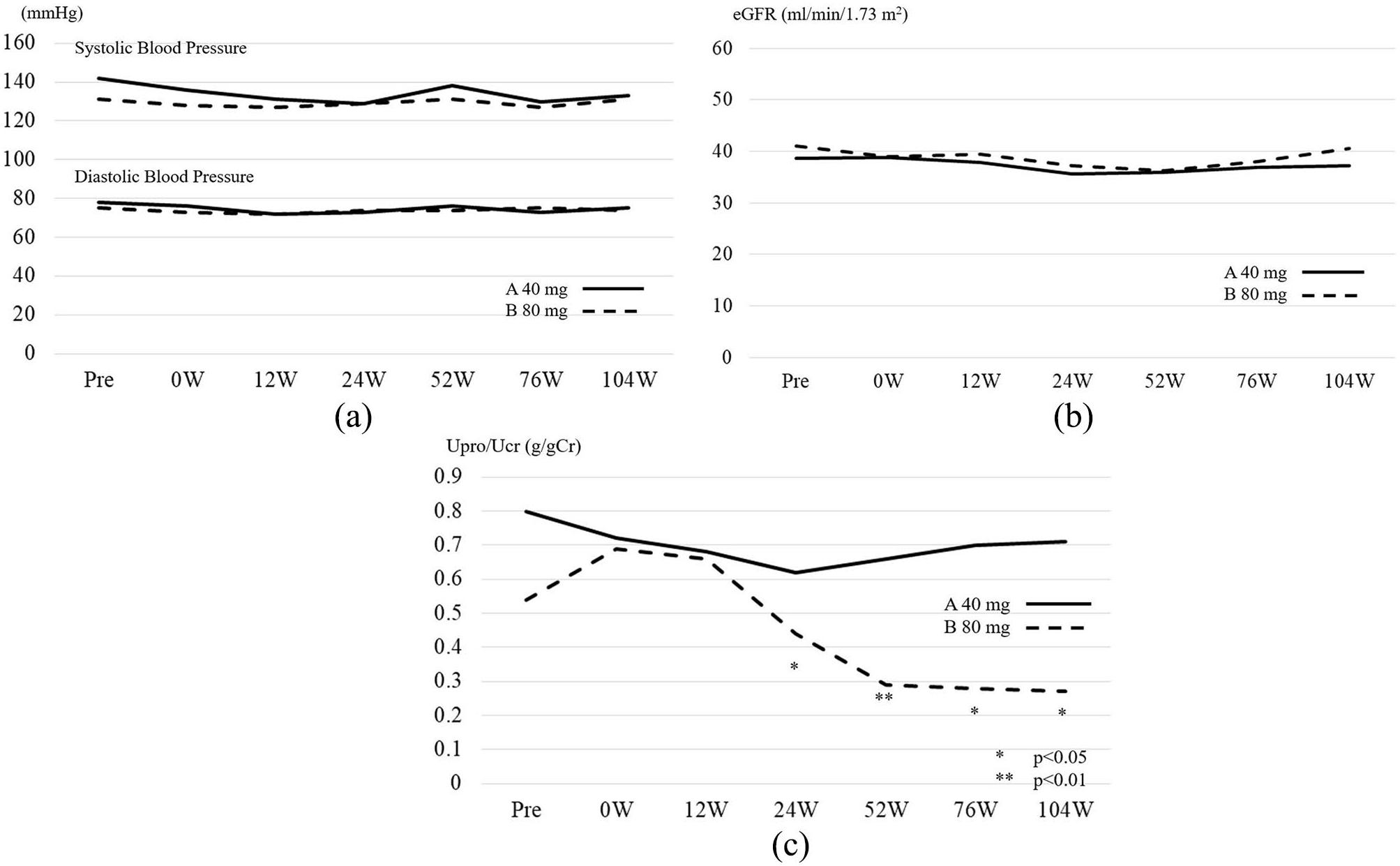
Blood pressure and secondary outcome analysis between low- and high-dose telmisartan groups: (a) mean changes in blood pressure, (b) estimated glomerular filtration rate, and (c) proteinuria. Straight line: Group A; telmisartan 40 mg. Dotted line: Group B; telmisartan 80 mg.
The changes in the urinary protein levels in both groups are shown in Figure 4(c) and Table 2.
Changes in urinary protein (urinary protein/urinary creatinine) over time.

Values are expressed as mean ± standard deviation and median (interquartile range).
p < 0.05; **p < 0.01. Post hoc analyses were performed using Wilcoxon rank sum test.
Adverse events
In Group A, two patients died during the follow-up period as described above, but this was not thought to be directly associated with the administration of telmisartan. Two patients in Group B experienced hyperkalemia, and one suffered from taste disturbance. All three patients recovered after stopping telmisartan administration. No severe adverse events were observed throughout the follow-up period, and there was no significant difference in the occurrence of adverse events between the two groups. A summary of adverse events is shown in Table 3.
Adverse events.

Statistical analysis was performed using Fisher’s exact tests.
Discussion
We conducted a multicenter, prospective, open-label, randomized controlled trial to elucidate the dose-dependent renoprotective effect of telmisartan in non-nephrotic proteinuria patients with moderate to severe CKD. We did not find a significant difference in the primary outcome between the low- and high-dose telmisartan groups. Among the secondary outcomes, there was no significant difference in the eGFR, but the proteinuria in the high-dose group after 24 weeks was significantly lower than that in the low-dose group. Overall, telmisartan did not improve the hard renal outcomes, such as doubling the serum creatinine level or end-stage renal disease, in this study.
There is a concern that ARBs or ACEis for CKD patients with non-nephrotic proteinuria are of no clinical benefit. 20 The AASK trial of antihypertensive therapy showed that ramipril had a beneficial effect in patients with a urine PCR of ⩽0.22 but not in patients with low-degree proteinuria, suggesting that the renoprotective effect of RAS inhibitors was only applicable to patients with some degree of proteinuria (e.g. 0.22 g/gCr). 8
An angiotensin II receptor blockade by ARBs depends on their dose, 21 and there have been several clinical trials describing the efficacy of supramaximal doses.11–14 Clinical dosing of ARBs was determined according to their antihypertensive effect, as they were initially approved for this purpose, 22 with their anti-proteinuric effects as secondary outcomes. A previous study showed that the administration of benazepril or losartan had a dose-dependent renoprotective effect in patients with advanced proteinuric CKD (inclusion criteria: serum creatinine level of 1.5–5.0 mg/dL and urinary protein level > 1.0 g/day lasting more than 3 months) and resulted in reduced risk of hard renal outcomes. 16 In contrast, in this study, we targeted CKD patients with non-nephrotic proteinuria. On the basis of our findings, the renoprotective effect of ARBs may be limited in this patient population.
In this study, there was no rapid decrease in the urinary protein level in the high-dose telmisartan group, but after 24 weeks, there was a significant difference in the proteinuria between the low- and high-dose groups. Reducing intraglomerular pressure by ARBs undoubtedly plays a key role in alleviating proteinuria and glomerular hypertension shortly after the administration of RAS inhibitors, 23 but the proteinuria continuously decreased over a period of weeks to months. The precise mechanisms of lowering urinary proteins independent of intraglomerular pressure are unknown although RAS inhibition has other positive effects on the kidneys; for example, the administration of ACEi increases nephrin expression in podocytes. 24 In addition, the overexpression of angiotensin II receptors on podocytes is associated with proteinuria through foot process effacement or the local detachment of podocytes. 25 Furthermore, RAS inhibitors may be further developed to include reduced levels of transforming growth factor β and connective tissue growth factor and improved permselective properties of the glomerular membrane.26,27 Unless RAS inhibitors cause a rapid decline in the eGFR or unmodifiable hyperkalemia, long-term RAS inhibition would be advantageous for patients with CKD.
RAS inhibition includes some safety considerations, particularly because ARBs or ACEi for advanced CKD may increase the risk of hyperkalemia or elevated serum creatinine. 28 In such patients, angiotensin II contracts efferent arterioles to maintain intraglomerular pressure; however, under RAS inhibition, the GFR would be reduced. Nevertheless, the renoprotective effects conferred could be tolerable and advantageous for such patients. For example, benazepril decreased the risk of renal failure by 43% in patients with serum creatinine levels of 3.1–5.0 mg/dL, without severe adverse events. 29 A meta-analysis found that ARBs were well tolerated, and the proportion of adverse events was lower than that of the BP-lowering effect when the administration dose was increased. 22 In this study, there were no significant differences in the occurrence of adverse events when telmisartan was administered at high or low doses, and hyperkalemia was observed only in the high-dose group. Because hyperkalemia is a life-threatening adverse event of RAS inhibition, it should not be overlooked in patients with moderate to severe CKD, 30 especially in elderly patients.10,31
There was no significant difference in primary outcome between the high-dose (80 mg) and the low-dose (40 mg) groups; this might have been due to the lack of statistical power in this study. We attempted to estimate a sample size for this study, but we could not perform a pilot study because of the limited number of eligible patients. In addition, when we initiated the study design, there was no appropriate study that could be used as a reference to estimate the sample size. However, the Renoprotection of Optimal Antiproteinuric Doses (ROAD) trial elucidated the dose-dependent renoprotective effect of ARB in moderate to severe CKD patients and showed that the proportion of patients in the low-dose losartan group (fixed 50 mg) who reached the primary end point was approximately 30%, whereas in the high-dose losartan group (titrated from 50 to 200 mg), the value was approximately 15% of patients in 3 years. 16 Consequently, the risk reduction rate was approximately 50%, and 77 participants would have been needed in one arm in this study for a statistical power of 80% and a two-sided significance level of 0.05. There were several patient characteristic differences between the ROAD study and our study, such as age and etiologies of renal failure. Specifically, the patients’ backgrounds of ROAD study were as follows: the mean age was approximately 50 years old, the mean eGFR was approximately 30 mL/min/1.73 m2, and the median of proteinuria was 1.4 g/day; furthermore, >50% of participants contracted glomerular disease including diabetic nephropathy. Despite the large differences between the ROAD study and ours, the results of the ROAD study suggest that our study does not seem to have enough statistical power and could not provide the anticipated results.
This study had several critical limitations. As we described above, there might have been a type II error in this study due to the small number of participants. Even though we intended to include as many participants as possible by not limiting etiologies of renal function, such as diabetes, the total number of participants was 61. This study was conducted in an open-label manner, and thus many biases may have been present. In addition, complications, etiologies for renal failure, and the level of proteinuria were not taken into consideration in the randomization. For example, the proportion of diabetes was higher in Group B (p = 0.10), and this might have affected the results. In addition, the diagnosis of nephrosclerosis was not based on renal biopsies, and we could not exclude the possibility of other etiologies. The dropout rate in both groups was approximately 10%, which may have unintentionally biased the results. Moreover, the lack of data in this study may confound the interpretation of our observations. Furthermore, there was a significant difference in urinary protein levels between the high- and low-dose groups after 24 weeks, but the decrease rates from baseline were not significantly different. This is because some participants had almost no proteinuria at baseline, and therefore, worsening of proteinuria could not be observed. Finally, since this study was conducted in daily clinical practice, we did not standardize instruments or the way of measuring BP. In addition, the methods used for blood chemistry and urinary examinations were not standardized among the facilities; this might have affected the results.
Conclusion
Our results do not clearly indicate a dose-dependent renoprotective effect of telmisartan. On the other hand, the amount of proteinuria was significantly lower in the high-dose group, without significant adverse effects, such as reduced eGFR, in either group. Since renal hard outcomes were not improved in this study, the clinical relevance of the study results may be limited.
Footnotes
Acknowledgements
The authors thank the participants and facilities where this study was conducted, including the Department of Nephrology Nagasaki University Hospital, Nagasaki Medical Center, Omura Municipal Hospital, Nagasaki Harbor Medical Center, Shinzato Clinic, Nagasaki Renal Center, Isahaya General Hospital, Sasebo General Hospital, Hokusho Central Hospital, St. Francisco Hospital, and Miyazaki Naika Clinic.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Nagasaki University Hospital Institutional Review Board approval number 08090558-6.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Written informed consent was obtained from all subjects before the study.
Trial registration
UMIN-CTR UMIN000040875. This trial was retrospectively registered. When the study design was inspected by our institutional review board (IRB) in 2008, the registration in systems was not mandatory as far as our facility, at least, and our trials were approved without UMIN-CTR registration. Our facility IRB do not approve any prospective trial without registration today. Participants data were collected through UMIN-INDICE; anonymous participants data were registered online. Both UMIN-INDICE system and UMIN-CTR are administered by University Hospital Medical Information Network (UMIN) Center, Japan.
