Abstract
Objectives
The aim of the present study was to evaluate the efficacy of oral molnupiravir (MPV; EIDD-2801) in cats with naturally occurring feline infectious peritonitis (FIP), with a subset of cats being administered a known immune stimulant (liposome-toll-like receptor agonist complex [LTC] orally.
Methods
A prospective, open-label longitudinal single-center clinical trial was conducted. Cats with FIP were enrolled and treated with oral MPV (10–21 mg/kg PO q12h) for 84 days. A subset of cats (41 cats with effusive FIP) was randomized to concurrently be administered the oral immune stimulant. Cats were evaluated at 0, 4, 8 and 12 weeks followed by a 12-week observation period.
Results
A total of 73 cats were included in the study and 77% of the cats survived to 6 months. The median total bilirubin concentrations were significantly different (P = 0.0007) between the survivors vs non-survivors. Relapses occurred in 12% of the cats (at 9–99 days after discontinuing treatment), and all achieved remission during a second course of treatment. Clinicopathologic features associated with FIP normalized during the study period; however, some cats showed decreased cholesterol levels and lymphocytosis during treatment. No adverse effects necessitated discontinuation of either treatment. No effects of the LTC were apparent in this study.
Conclusions and relevance
MPV administered at 10–21 mg/kg PO q12h for 12 weeks is well tolerated and an effective treatment (77% success) for all forms of naturally occurring FIP, with a relapse rate of 12%. These results support those of other studies showing that MPV is an effective treatment for cats diagnosed with FIP. Additional studies will be required to determine if any benefits might be derived from the LTC treatment.
Introduction
Feline infectious peritonitis (FIP) is a disease caused by feline coronaviruses (FCoVS) and, despite marked efforts and many theories, the pathogenesis of FIP is still not fully understood. 1 Left untreated, FIP is almost always fatal, with most cats succumbing within weeks to months of diagnosis.2,3 Recent studies have reported the use of drugs such as the nucleoside analog GS-441524 and molnupiravir (MPV; EIDD-2801) as successful treatments for FIP.4 –9 However, more treatment options for cats with FIP, especially those that relapse, are needed.
Recently, a study showing pharmacokinetics/pharmacodynamics data from the antiviral drug EIDD-2801 (which has US Food and Drug Administration Emergency Use Authorization for treatment of SARS-COV-2 in humans) showed promise for its use and safety in cats. 10 This recent study of pharmacokinetic properties of MPV in healthy specific pathogen-free cats established that orally administered MPV at 10 mg/kg achieves plasma levels greater than the established corresponding half-maximal effective concentration (EC50) values. In that study, the MPV prodrug was detected in the serum of all three cats at low levels in the first 12 h after administration, while the beta (β)-d-N4-hydroxycytidine (EIDD-1931) metabolite was detected at much higher levels at 12–24 h after administration. This study reported no evidence of acute organ toxicity in any of the cats based on the pre- and post-treatment complete blood count (CBC) and serum biochemistry panels; however, all three of the MPV-treated cats demonstrated variable signs of nausea, including hypersalivation and/or vomiting, after oral administration of MPV (10 mg/kg).
One study of 18 cats with FIP being treated with MPV showed that MPV might be an effective and safe treatment for domestic cats with FIP at a dose of 10–20 mg/kg twice daily. 7 In that study, increased serum alanine transaminase (ALT) activity was found in 3/18 cats, all at days 7–9, and all cats recovered without any medications or need to stop the treatment. 7 There are also uncontrolled data from client surveys that suggest efficacy of this product as rescue treatment after failure of GS-441524 therapy as well as a first-line treatment option for cats with FIP. 9
Another study evaluating MPV in cats with FIP showed that it is an effective antiviral treatment for effusive FIP, with 8/10 cats achieving remission at 16 weeks while the two non-survivors died within the first 24 h of treatment. 8 Survival of cats treated with oral MPV was non-inferior to historic control cats treated orally with GS-441524. 8 Survival analysis revealed no difference in outcomes between cats treated with MPV monotherapy and those treated with remdesivir/GS-441524. Adverse events associated with MPV therapy included neutropenia and transient elevations in hepatic enzyme activities. 11
Cell-mediated immunity is believed to play an important role in controlling or eliminating the mutated FIP virus. Cats in the terminal stages of FIP have severe depletion of the CD4+ and CD8+ T lymphocytes necessary for mounting cell-mediated immunity.12,13 With immune dysregulation being a major component of the pathophysiology of FIP, treatment with an antiviral immune stimulant that triggers strong endogenous interferon (IFN) production would be a rational approach. It has been suggested that antiviral drugs can act synergistically with immunomodulatory treatments to improve patient outcome and survival in different human viral diseases, such as influenza virus, hepatitis C virus and human immunodeficiency virus.14 –16 This synergism has been proposed to improve long-term treatment outcomes for cats with FIP as well.
Recently, studies evaluating polyprenyl immune stimulant (PI) and a new liposome-toll-like receptor (TLR) agonist (TLR3 and TLR9) complex (LTC) have been published, providing some evidence that the compounds are immune stimulants. The commercially available PI is believed to upregulate innate immunity via TLR2 and TLR6, 17 but peer-reviewed data supporting that claim in cats are not available. This product is licensed by the US Department of Agriculture for the reduction in the severity of signs of feline herpesvirus 1 (FHV-1) in cats over the age of 8 weeks and has been evaluated in cats with non-effusive FIP, with some positive results.18 –21 The LTC immune stimulant has been extensively studied in multiple different veterinary species, including cats.22,23 The compound has been patented and is currently in pre clinical development as a feline immune stimulant. The effects of the LTC on the innate immune system have been well documented in healthy cats. 22 In addition, we showed effects against FHV-1 in two different models in a follow-up study.23,24 In another study, we demonstrated that a similar compound could be given safely by parenteral inoculation to cats, with a positive effect on chronic rhinitis. 25 A new study comparing two immune stimulants (PI and LTC) for antiviral activity in cats, using peripheral blood mononuclear cells from healthy animals, showed that both compounds upregulated IFN-alpha (α), IFN-gamma (γ) and interleukin-1β gene expression. 26 Treatment with LTC induced significantly greater expression of IFN-α and IFN-γ, suggesting that activation of the TLR3 and TLR9 pathways produces broader IFN responses in vitro than activation of the TLR2 and TLR6 pathways. This study also demonstrated significant protection against FIP virus-induced cytopathic effects when Fcwf-4 cells were treated with conditioned medium from LTC-activated leukocytes. 26
The objectives of the current study were to determine the efficacy of MPV as a first-line treatment for naturally occurring FIP in cats and to determine the efficiency of oral LTC as an adjunct treatment of cats with the effusive form of FIP. It was hypothesized that MPV would be effective and safe as a first-line treatment for FIP and that the LTC would help cats with FIP achieve or maintain remission.
Materials and methods
Study design
The study was a prospective, open-label longitudinal single-center clinical trial. MPV was sourced from Natural Micron Pharm Tech (NM PharmTech; see supplementary material) with a purity greater than 99% also confirmed by liquid chromatography-mass spectrometry after purchase. Orally administered MPV was formulated as excipient-less powder in size 3 gelatin capsules in different strengths (20–70 mg) and the amount of MPV in the capsules was verified by a licensed pharmacy (Monument Pharmacy, Monument, CO, USA). Cats with FIP were enrolled and all were administered oral MPV (10–21 mg/kg PO q12h) for 84 days. The doses followed the previous pharmacokinetics study in healthy and FIP cats and currently recommended dosing.10,27 Higher doses were used for cats with ocular and neurological involvement. Owners were allowed to give medications with small amounts of food or treats. Cats with effusive FIP were randomized into groups to be administered the LTC (0.1 ml/kg PO once weekly) or placebo (0.1 ml/kg PO once weekly; 0.1 mM Tris buffer with 10% sucrose sterile filtered). The LTC was prepared as described previously. 22
Cats were evaluated at 0, 4, 8 and 12 weeks followed by a 12-week observation period. The study followed ethical guidelines and was approved by the Institutional Animal Care and Use Committee (protocol number 4484, approval date 8 September 2023, and 5961, approval date 30 July 2024). Cats were recruited through the urgent care service at the referral hospital as well as by referral from referring veterinarians directly to the clinical trial when the patient was suspected to have FIP.
Inclusion criteria
Cats suspected of having FIP by an internal medicine specialist were considered for enrollment. For a cat to be included, a diagnosis of FIP had to be (a) confirmed: positive for FCoV RNA using quantitative RT-PCR as previously reported; 28 or (b) presumptive: have clinical signs (eg, fever, abdominal fluid, uveitis, neurological signs) and clinicopathological findings consistent with FIP (eg, hyperglobulinemia, hypoalbuminemia, low albumin:globulin raio [A:G], high protein [>3.5 g/dl effusion], cytology findings assessed by a veterinary clinical pathologist to be consistent with FIP) and other differential diagnoses ruled out (eg, negative Toxoplasma gondii serology, no evidence of neoplasia, bacterial or fungal disease on cytology). Cats were excluded from the clinical trial if immunosuppressive medications (eg, immunosuppressive doses of glucocorticoids) or antiviral medications had been administered before enrollment. Cats were also excluded if hospitalization for intravenous fluid therapy or blood transfusion for severe anemia was required. Cats with chronic kidney disease of International Renal Interest Society (IRIS) stages 3 and 4, 29 congestive heart failure, diabetes mellitus and/or uncontrolled hyperthyroidism were also excluded from the study. Cats were characterized based on the form of FIP as effusive (pleural, peritoneal or pericardial) or non-effusive and whether there was neurological or ocular involvement.
Data collected
Patient data collected included signalment, clinical signs, physical examination findings (pyrexia defined as a rectal temperature of >103.1°F [>39.2°C]), 30 diagnostic tests including hematology, serum biochemistry, urinalysis (if performed), infectious disease testing such as feline leukemia virus (FeLV) antigen and feline immunodeficiency virus (FIV) antibodies within the preceding month (performed by the referring veterinarian or at the initial visit using the SNAP FeLV/FIV test; IDEXX), diagnostic imaging when performed, cytology and/or biochemistry of fluid samples, fine-needle aspiration and testing for FCoV RNA by RT-PCR. Treatment dosage, outcome and any adverse effects of MPV were also recorded.
Statistical analysis
Data were recorded in Excel (Microsoft). Variables were assessed for normality by visual assessment of histograms and use of the Shapiro–Wilk test. Descriptive statistics were used to report the data. Data were reported as median (range) and interquartile range (IQR). Categorical data of the number of cats alive or dead at the end of the initial treatment period were grouped according to whether they had achieved remission or not at the end of the 12-week treatment period. Friedman analysis was used to compare the medians of the clinicopathological parameters in the follow-up blood work. The χ2 test was used to compare the number of relapses between the LTC vs placebo groups.
Results
Study cohort
A total of 73 cats with a confirmed or presumptive diagnosis of FIP were recruited between September 2023 and November 2024. Of these, the majority were domestic shorthairs (n = 54, 74%) or domestic longhairs (n = 8, 11%) and only 11 (15%) cats were pedigree breeds: Maine Coon (n = 4), Sphynx (n = 1), British Shorthair (n = 1), Ragdoll (1), Bengal (n = 1), Siamese (n = 1), Snowshoe (n = 1) and Siberian (n = 1). There were 38 (53%) castrated males, 27 (37%) spayed females, four (5%) intact males and four (5%) entire females. The majority (54/73, 74%) of cats were aged under 1 year at diagnosis (median age 0.6 years, IQR 0.5–1.0). The oldest cat enrolled in the study was aged 13 years.
Clinical findings
Of the 73 cats, most (n = 47, 64%) were diagnosed with effusive FIP: peritoneal effusion (n = 39, 53%), pleural effusion (n = 7, 10%), and both pericardial and pleural effusion (n = 1, 1%) were detected. Non-effusive FIP was diagnosed in 26/73 (36%) cats. Between both effusive and non-effusive cases, 10/73 (14%) cats had ocular involvement, 4/73 (5%) cats had neurological involvement and 2/73 (3%) cats had both ocular and neurological involvement. Of these cats, 43 (59%) had confirmed FIP by RT-PCR, while 30 (41%) cats had presumptive FIP. Out of the 30 presumptive cats, 22 (73%) were PCR negative and no PCR was performed in eight (27%) cats. In 43 cats, FCoV RNA was amplified by RT-PCR, from peritoneal effusion from 30 (70%), pleural effusion from five (11%), jejunal lymph node aspirates from three (7%), splenic aspirates from three (7%), aqueous humor from one (2%) and an ileum mass aspirate from one (2%). In addition, in 4/43 (5%) cats, RT-PCR was positive on more than one sample. The most common clinical signs and findings are shown in Table 1.
The most common clinical signs and findings at the time of feline infectious peritonitis diagnosis in 73 cats

Data are n (%)
Laboratory results
Table 2 shows CBC results at the initial visit for 71 cats; in two cats, the EDTA blood clotted and CBC could not be performed. A total of 51/71 (72%) cats were anemic, with a median hematocrit of 28.0% (IQR 24.0–32.0). Neutrophilia was seen in 38/71 (54%) cats and one cat presented with mild neutropenia (1.7 × 103/µl). A total of 46/71 (65%) cats had lymphopenia on presentation and one cat presented with mild lymphocytosis (6.7 × 103/µl). Serum biochemistry results for all 73 cats are shown in Table 3. An A:G of 0.4 or below was detected in 25 (34%) cats, with only 10 (14%) cats having a serum A:G of 0.6 or greater. Hyperglobulinemia occurred in 54 (74%) cats and hypoalbuminemia occurred in 66 (90%) cats. A total of 48 (66%) cats presented with hyperbilirubinemia.
Complete blood cell count results at the time of feline infectious peritonitis diagnosis (week 0)

IQR = interquartile range; MCHC = mean corpuscular hemoglobin concentration; MCV = mean corpuscular volume; RI = reference interval
Serum biochemistry results at the time of feline infectious peritonitis diagnosis (week 0)
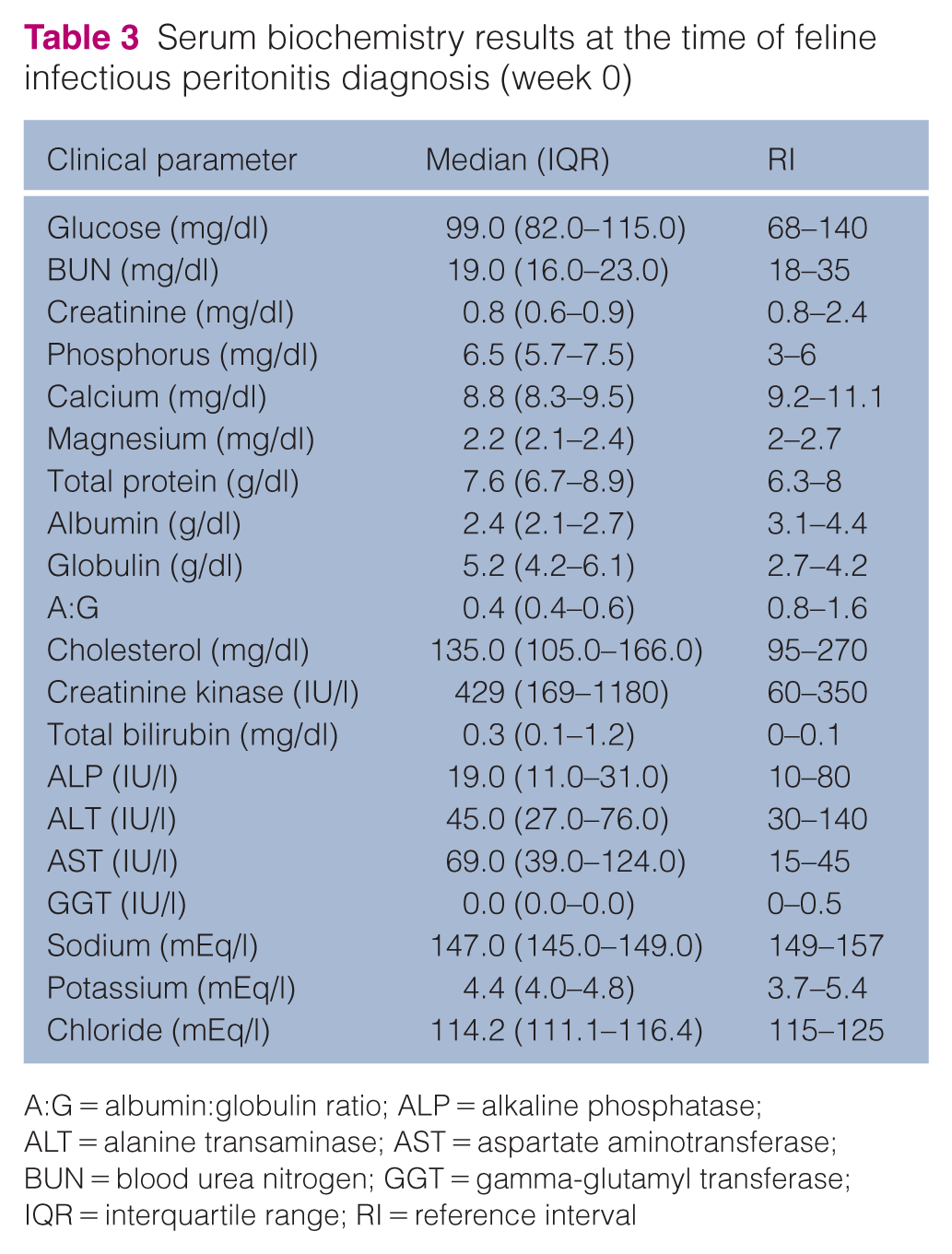
A:G = albumin:globulin ratio; ALP = alkaline phosphatase; ALT = alanine transaminase; AST = aspartate aminotransferase; BUN = blood urea nitrogen; GGT = gamma-glutamyl transferase; IQR = interquartile range; RI = reference interval
Urinalysis was performed in 29/73 (40%) cats and showed median urine specific gravity (USG) of 1.053 (IQR 1.041–1.059), median pH of 6.5 (IQR 6.0–7.0) and median urine protein:creatinine ratio (UPC) of 0.36 (IQR 0.25–0.57).
All but one cat tested negative for FeLV and FIV. One cat was FeLV/FIV negative in the referring veterinarian’s records before inclusion but was then diagnosed with FeLV antigen and confirmed with FeLV infection by PCR after enrolment.
Abdominal ultrasonography
Abdominal ultrasonography was performed in 23/73 (32%) cats. All 23 (100%) cats had more than one abnormality on abdominal ultrasonography, with a median of four abnormalities in one cat (range 2–6). The most common findings were lymphadenopathy (21/23, 91%), peritoneal effusion (21/23, 91%), mottled or heterogenous spleen (7/23, 31%), intestinal wall thickening (7/23, 31%), splenomegaly (5/23, 22%), medullary rim sign (5/23, 22%), heterogenous kidneys (5/23, 22%), gallbladder thickening or edema (5/23, 22%), renomegaly (4/23, 17%), heterogeneous or hypoechoic liver (4/23, 17%), nodular liver (3/23, 13%), renal nodules (2/23, 9%), hepatomegaly (1/23, 4%), hyperechoic mesentery (1/23, 4%), splenic nodules (1/23, 4%), pancreatic nodules (1/23, 4%) and ileal mass (1/23, 4%).
Cytology findings
Fluid analysis and cytology were performed on effusions from 46/47 (97%) cats. The median total protein in the effusion was 6.0 g/dl (IQR 5.4–7.1), and the median nucleated cell count was 3200/µl (IQR 1900–5700).
Treatment and follow-up
Antiviral treatment
All cats were treated with MPV for 12 weeks. Of the 73 cats, four (5%) had therapy prolonged to 16 weeks when clinicopathological abnormalities or clinical signs had not resolved at week 12. The median dosage of MPV was 13.8 mg/kg PO q12h (IQR 12.5–15.4) and the maximum dosage was 21.4 mg/kg PO q12h. Of the 73 cats, 56 (77%) survived to 6 months (3 months of therapy and 3 months of observation) and 16 (22%) died or were euthanized. One cat had to be excluded from the study because of disease progression and its owners elected to add GS-441524 to the treatment. There was a median of 4 days to death or euthanasia (range 1–11). The median total bilirubin concentrations were significantly different (P = 0.0007) between the survivors vs non-survivors (Figure 1). The median total bilirubin in alive cats was 0.2 mg/dl (IQR 0.1–0.8) compared with 1.9 mg/dl (IQR 0.5–3.0) in the cats that died or were euthanized. Of the 56 cats that lived, seven (12%) relapsed and required a secondary course of treatment when signs referable to FIP (pyrexia, lethargy and inappetence) recurred at 9–99 days after discontinuing treatment. All seven cats were administered a second course of treatment using a median dose of 22.4 mg/kg (IQR 21.8–24.8), which was higher than for their primary course, and all achieved remission. It should be noted that none of the cats relapsed with the same form of FIP that was initially diagnosed. All cats at relapse had a non-effusive form, with 3/7 cats also having neurological involvement. At the initial FIP diagnosis, these cats had effusive (4/7 cats) or non-effusive (3/7) FIP, with two of those having ocular involvement and no neurological signs. The two cats with ocular involvement both presented with neurological signs at the time of relapse. Cats were prescribed maropitant (1 mg/kg PO q24h) and transdermal mirtazapine if they were presented with decreased appetite and/or nausea. Cats with neurological and ocular involvement were prescribed a tapering dose of prednisolone starting at 1 mg/kg/day. All were tapered off before the end of the 12 weeks of treatment.
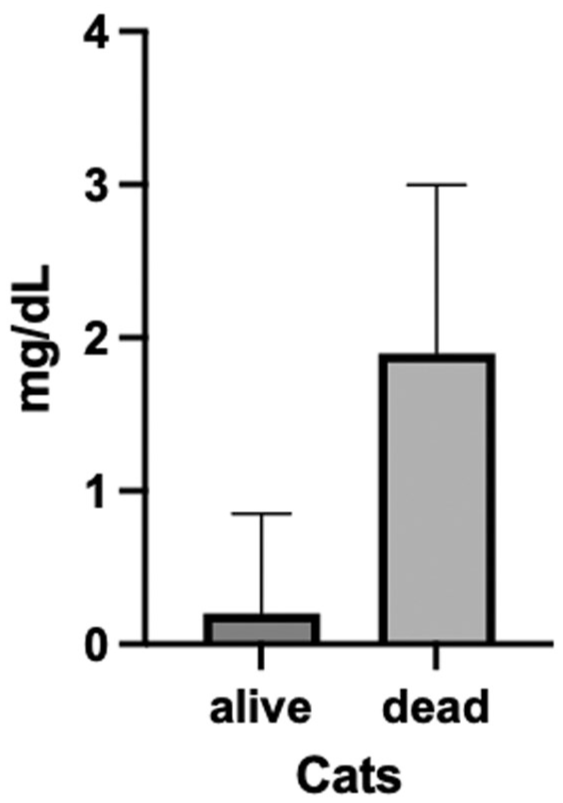
Total bilirubin concentrations at diagnosis of cats that survived vs died. The box represents the median and the vertical line is the interquartile range
Side effects from MPV were minimal. Most common side effects were gastrointestinal: nausea (n = 4), hypersalivation (n = 4), constipation (n = 3), diarrhea (n = 2), hyporexia (n = 1) and vomiting (n = 1). All of the side effects observed resolved within the first week of therapy, except in one cat that remained intermittently nauseous and had to be treated with anti-nausea medication throughout the treatment period. Pruritus was reported in three cats during the study. Folded ears developed in a cat that relapsed and was treated for the second time with a high dose of MPV (26.4 mg/kg PO q12h). Mild neutropenia developed in one cat at week 8 and persisted until week 12, and one cat developed mild neutropenia at week 12. At week 4, 8/56 (14%) cats developed a mild to moderate increase in ALT activity; of them, two cats had high ALT activity at the time of the primary diagnosis. All of these cats had normal ALT activity by week 8. At week 8, two cats had a mild increase in ALT activity (these were different from the cats with high ALT activity at week 4); at week 12, ALT activity was mildly increased in two cats (one new cat and one that had an ALT activity increase at week 8). No other side effects were noticed. No adverse effects necessitated drug discontinuation.
Clinicopathologic findings during the first 12 weeks of treatment
Marked and rapid clinical improvements were observed in all 56 cats that completed 12 weeks of treatment. Follow-up blood work was available for 56 cats that were alive at 4, 8 and 12 weeks of treatment. Results are shown in Tables 4 and 5. On CBC, there was a significant improvement in hematocrit (P ⩽0.0001) (Figure 2) and neutrophilia (P ⩽0.0001) (Figure 3) by the end of the treatment at week 12. Mild eosinophilia developed in three cats in week 4 of treatment and resolved in all three (two cats by week 8 and one cat by week 12). One cat developed mild eosinophilia in week 8 of treatment that persisted until week 12, and an additional three cats developed mild eosinophilia in week 12. On serum biochemistry, there was a significant improvement in A:G (P ⩽0.0001) (Figure 4), albumin (P ⩽0.0001) (Figure 5) and globulin (P ⩽0.0001) (Figure 6) concentrations, as well as total bilirubin concentrations (P ⩽0.0001) (Figure 7), by the end of the treatment, and total bilirubin normalized by week 4. There was also a significant increase in lymphocytes (P ⩽0.0001) (Figure 8) and a decrease in cholesterol (P = 0.0311) (Figure 9) while on antiviral treatment.
Complete blood cell count results at weeks 4, 8 and 12 of treatment

Data are median (interquartile range)
MCHC = mean corpuscular hemoglobin concentration; MCV = mean corpuscular volume; RI = reference interval
Serum biochemistry results at weeks 4, 8 and 12 of treatment

Data are median (interquartile range)
A:G = albumin:globulin ratio; ALP = alkaline phosphatase; ALT = alanine transaminase; AST = aspartate aminotransferase; BUN = blood urea nitrogen; GGT = gamma-glutamyl transferase; RI = reference interval

Hematocrit (%) over the 12 weeks of treatment. Each dot represents the median and the vertical line is the interquartile range

Neutrophil count over the 12 weeks of treatment. Each dot represents the median and the vertical line is the interquartile range

Albumin: globulin ratio over the 12 weeks of treatment. Each dot represents the median and the vertical line is the interquartile range

Albumin concentrations over the 12 weeks of treatment. Each dot represents the median and the vertical line is the interquartile range
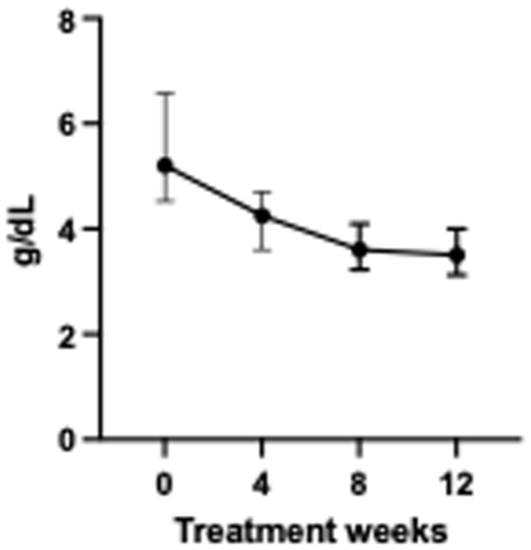
Globulin concentrations over the 12 weeks of treatment. Each dot represents the median and the vertical line is the interquartile range

Total bilirubin concentrations over the 12 weeks of treatment. Each dot represents the median and the vertical line is the interquartile range

Lymphocyte count over the 12 weeks of treatment. Each dot represents the median and the vertical line is the interquartile range

Cholesterol concentrations over the 12 weeks of treatment. Each dot represents the median and the vertical line is the interquartile range
Repeated urinalysis was performed in 18/29 (62%) cats and showed a median USG of 1.051 (IQR 1.030–1.058), median pH of 6.5 (IQR 6.0–7.0) and median UPC of 0.10 (IQR 0.08–0.12). One cat developed worsening dyspnea on day 4 of treatment and needed repeated thoracocentesis; the fluid was negative for FCoV on RT-PCR. Another cat had repeated abdominocentesis in week 4 due to persistent effusion; the fluid was negative for FCoV on RT-PCR.
Treatment with immune stimulant vs placebo
The 41 cats with effusive FIP were randomized into groups and administered the LTC (0.1 ml/kg PO once weekly, n = 21 cats) vs placebo (0.1 ml/kg PO once weekly, n = 20 cats). In total, 31/41 (76%) cats survived (17/21 [81%] in the LTC group and 14/20 [70%] in the placebo group) to 6 months (3 months of therapy and 3 months of observation) and 10/41 (24%) cats died or were euthanized. The median number of days in which the cats died or were euthanized was 4 (range 1–10). Of the 31 cats that survived, 17 (55%) were treated with LTC and 14 (45%) were in the placebo group. There was no statistical difference between the LTC and placebo groups in A:G during the treatment (Figure 10). Moreover, of the 31 cats that lived, four (13%) relapsed and required a secondary course of treatment when signs referable to FIP (pyrexia, lethargy and inappetence) recurred. All four cats commenced a second course of treatment using a median dose of 22.4 mg/kg (IQR 21.9–23.1). Of the four cats that relapsed, three were treated with LTC and one was in the placebo group; however, the difference between the groups was not statistically significant (P = 0.443).

Albumin: globulin ratio over the 12 weeks of treatment in cats with effusive feline infectious peritonitis in the liposome-toll-like receptor agonist complex (LTC) and placebo groups.
Discussion
In this prospective clinical trial with MPV as a first-line treatment for 73 cats with naturally occurring FIP, MPV treatment resulted in clinical remission in 77% of the cats enrolled by the end of 6 months. Most cats that died or were euthanized did not survive longer than 4 days. The treatment with MPV was safe, with no adverse events noted that necessitated discontinuation of the medication, but gastrointestinal side effects, mild and mostly transient increases in ALT activity, mild neutropenia and folded ear tips were observed in a small number of cats in this and in other studies.7,9,11
Survival rates observed here were similar to those in the other studies using MPV as a first-line drug for FIP.7,8,11 The median dosage of MPV was 13.8 mg/kg PO q12h and the maximum dosage was 21.4 mg/kg PO q12h for 12 weeks, similar to what has been reported previously as a first-line drug for FIP.7,8,11 However, a previous study with the nucleoside analogue GS-441524 reported that 6-week treatment with antivirals may be sufficient for FIP, and additional studies evaluating shorter courses of treatment are needed to better define the optimal treatment protocol. 31 The survival rate with MPV in this study and other studies is very similar to the approximately 80–85% success in treatment with GS-441524; studies have reported that GS-441524 and MPV have similar effects and safety in cats with FIP.8,32 However, the mechanism of action of MPV includes the introduction of mutations into the viral genome during replication, which raises concern for genetic selection of antiviral-resistant strains of FIPV. 33 In our study, 12% of the cats relapsed and required a secondary course of treatment when signs referable to FIP (pyrexia, lethargy and inappetence) recurred at 9–99 days after discontinuing treatment. All seven cats commenced a second course of treatment using a higher dose of MPV (median dose 22.4 mg/kg, IQR 21.8–24.8) and all achieved remission during this second course of treatment, which makes resistance less likely as a cause of relapse in these cats, but further studies are needed to evaluate the risk of mutations and possible antiviral resistance with MPV in cats with FIP. Host mutagenicity is also a concern with MPV treatment, and long-term safety studies monitoring for adverse outcomes and teratogenicity are needed to further assess the safety of MPV treatment in cats. 34
The signalment of the cats in the present study was similar to that in previous reports as most (74%) cats were young, aged under 1 year, at diagnosis. The majority were domestic shorthairs and domestic longhairs (85%), and only 15% of cats were pedigree cats, which is a much lower proportion than previously reported. This could be explained by the lower number of pedigree cats in the USA.4,35–39 There was one cat that initially tested negative for FeLV at the referring veterinary clinic; however, on repeated testing after enrolment, this cat was confirmed to be FeLV positive on both antigen and PCR tests. Since this was discovered after the patient was already enrolled into the study, and a recent study showed that FeLV-positive cats have the same prognosis when treated for FIP as retroviral-negative cats, 40 the cat was allowed to remain in the study. The presenting clinical signs and physical findings were also consistent with other studies,4, 41–42 with lethargy and hyporexia frequently reported as non-specific clinical signs and pyrexia occurring in approximately 58% of the cats. More cats (64%) were diagnosed with effusive FIP than non-effusive FIP (36%). Of the cats, 14% had ocular involvement, 5% presented with neurological involvement, and 3% had both ocular and neurological involvement.
Rapid resolution of clinical signs associated with FIP was achieved in all cats that survived to 6 months, again consistent with previous studies.4,42 All presenting clinicopathological abnormalities (eg, anemia, hyperbilirubinemia, hyperglobulinemia, hypoalbuminemia and low A:G) normalized in the cats on MPV treatment. Cholesterol was the only parameter that decreased while on treatment and the reason for this is unclear. It is possible that some of these cats might have developed gastrointestinal disease after recovery from FIP, given that several cats had abnormal gastrointestinal tract findings on ultrasonography, which could lead to malabsorption and cause low cholesterol levels. The total bilirubin concentrations were higher in cats that died or were euthanized, and high bilirubin concentrations have been previously reported in cats as a poor prognostic indicator. 43 There was a statistically significant increase in lymphocytes in the cats during MPV treatment, a finding also reported in cats treated with GS-441524.4,44 This likely reflects an immune or antiviral response. A previous study noted lymphocytosis in cats receiving GS-441524, with PCR for antigen receptor rearrangements indicating a reactive lymphoid cell population; 44 however, further studies are needed to understand why cats on antiviral therapy develop lymphocytosis. Mild eosinophilia developed in a few cats, as similarly reported in previous studies,4,42 but the underlying cause is unclear. Because fecal examinations were not performed, parasitic coinfections cannot be ruled out, and dermatologic conditions such as allergies remain possible. In humans with COVID-19 caused by SARS-CoV-2, increased eosinophil counts are thought to hold positive prognostic value. 45 Additional studies are needed to determine the significance of eosinophilia in cats with FIP receiving antiviral therapy.
Of the 31 cats with effusive FIP that survived, 55% were treated with LTC and 45% were in the placebo group, possibly suggesting some benefit in the response to treatment. However, among the cats with effusive FIP, more cats in the LTC group relapsed vs the placebo group (four vs one, respectively). However, the difference between the groups in response rates or number of relapses was not statistically significant; therefore, larger numbers of treated cats are needed to see if the addition of an immune stimulant may be beneficial. It is also possible that the LTC dose used in this study was not high enough. Further studies are needed to evaluate the effects of LTC and other immune stimulants in both effusive and non-effusive FIP, as previous studies have suggested benefit from immune stimulants in FIP treatment.18,21 There was no statistical difference in A:G between the LTC and placebo groups during treatment; A:G was assessed because normalization of this ratio is considered a positive response to treatment.
One limitation of this study is that it was an open trial with one drug and no untreated control group. However, because FIP is a lethal disease, withholding treatment would be unethical. The results of MPV treatment described here can still be compared with historical data and with outcomes from GS-441524 treatment. Our criteria for a presumed diagnosis of FIP – compatible history, signalment, clinical signs, blood work abnormalities and positive response to antiviral medications – are consistent with recommendations from the Advisory Board for Cat Diseases (ABCD), 46 and over half (59%) of the cats also had positive RT-PCR results. Future studies comparing MPV with other antiviral treatments should be randomized and blinded to better evaluate efficacy. Another limitation is that the first recheck was at 4 weeks, making it impossible to assess early resolution of laboratory abnormalities, as all cats showed marked improvement by that time. Finally, there were numbers of cats with different presentations, and cats with effusive disease may respond differently from those with neurological involvement.
Conclusions
MPV administered at 10–20 mg/kg orally q12h for 12 weeks is an effective treatment for all forms of naturally occurring FIP, with 77% of cats achieving remission. The risk of relapse is low (approximately 12%), and cats that do relapse seem to respond well to a second course of treatment using a higher MPV dose. The medication is well tolerated, with no adverse effects observed that required discontinuation. Further studies are needed to determine whether shorter MPV treatment protocols would be appropriate for different forms of FIP, and to evaluate whether LTC can aid in FIP treatment.
Supplemental Material
Supplementary file 1
Certificate of analysis
Footnotes
Supplementary material
The following file is available as supplementary material:
Supplementary file 1: Certificate of analysis
Conflict of interest
SD and MRL both hold equity and officeholder positions in Laporte Immunotherapeutics, a Fort Collins startup company that is developing LTC as a veterinary immunotherapeutic. The remaining authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The study was funded by the EveryCat Health Foundation (grant number W21-017-2).
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals and procedures that differed from established internationally recognized high standards (‘best practice’) of veterinary clinical care for the individual patient. The study therefore had prior ethical approval from an established (or ad hoc) committee as stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers, tissues and samples) for all procedure(s) undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
