Abstract
Objectives
Feline infectious peritonitis (FIP) is a serious disease that arises due to feline coronavirus infection. The nucleoside analogues remdesivir and GS-441524 can be effective in its treatment, but most studies have used unregulated products of unknown composition. The aim of the present study was to describe the treatment of FIP using legally sourced veterinary-prescribed regulated veterinary compounded products containing known amounts of remdesivir (injectable) or GS-441524 (oral tablets).
Methods
Cats were recruited via email advice services, product sales contacts and study publicity. Cats were excluded if they were deemed unlikely to have FIP, were not treated exclusively with the veterinary compounded products, or if there was a lack of cat and/or treatment (including response) data. Extensive cat and treatment data were collected.
Results
Among the 307 cats recruited, the predominant type of FIP was most commonly abdominal effusive (49.5%) and then neurological (14.3%). Three treatment protocols were used; remdesivir alone (33.9%), remdesivir followed by GS-441524 (55.7%) and GS-441524 alone (10.4%). The median (range) initial treatment period duration and longest follow-up time point after starting treatment were 84 (1–330) days and 248 (1–814) days, respectively. The most common side effect was injection pain (in 47.8% of those given subcutaneous remdesivir). Of the 307 cats, 33 (10.8%) relapsed, 15 (45.5%) during and 18 (54.5%) after the initial treatment period. At the longest follow-up time point after completion of the initial treatment period, 84.4% of cats were alive. The cats achieving a complete response within 30 days of starting treatment were significantly more likely to be alive at the end of the initial treatment period than those cats that did not.
Conclusions and relevance
Legally sourced remdesivir and GS-441524 products, either alone or used sequentially, were very effective in the treatment of FIP in this group of cats. Variable protocols precluded statistical comparison of treatment regimens.
Introduction
Feline infectious peritonitis (FIP) is a systemic inflammatory disease caused by infection with feline coronavirus (FCoV), although only a small percentage of FCoV-infected cats develop FIP.1 –3 The cat’s immune response also plays a role in the pathogenesis of FIP.4,5 When FIP develops, FCoV infection induces (pyo)granulomatous vasculitis and/or serositis, 6 leading to disease that may or may not be accompanied by high-protein effusions. FIP associated with effusion development (especially in the abdominal cavity) is the most common type of FIP encountered.7–16 A range of other systemic signs can occur, such as pyrexia, inappetence, lethargy,9,12,13,17,18 abdominal lymphadenopathy,9,19,20 ocular signs21,22 and/or neurological signs.23,24 A definitive diagnosis usually relies on demonstration of the FCoV antigen or RNA in association with typical FIP histopathological changes, but a range of supporting diagnostic evidence can be obtained from haematology, serum and/or fluid sample (eg, effusion) biochemistry (eg, hyperglobulinaemia, increased alpha-1-acid glycoprotein [AGP], serum amyloid A, hyperbilirubinaemia, reduced albumin to globulin [AG] ratio), cytology (pyogranulomatous inflammation) and FCoV antigen or FCoV RNA detection (on samples such as effusions or fine-needle aspirates [FNAs]).1,8,17,25,26
Left untreated, FIP is almost always fatal, with most cats succumbing within weeks to months of diagnosis.15,27 Early ground-breaking studies showed that the nucleoside analogue GS-441524 was effective in the treatment of experimentally induced 28 and spontaneous 29 FIP, although the majority of cases were effusive FIP. Initial dosages of GS-441524 were 2–5 mg/kg once daily by subcutaneous (SC) injection for 14–84 days (and occasionally more than 84 days if serum protein concentrations remained elevated).28,29 The initial field study 29 excluded cats with neurological or ocular signs due to concerns about poor penetration of GS-441524 into the brain and/or eye. 28 However, subsequently, four cats were successfully treated for neurological FIP with higher doses (5–10 mg/kg SC) of GS-441524 for at least 84 days. 30 Since then, evidence has mounted for the efficacy of GS-441524, administered increasingly by the oral route, in large numbers of cats with FIP, although the preparations used have been unlicensed and unregulated, meaning that the content and purity of any GS-441524 within them and administered to cats was not known or determined.10,11,22,31–33 Indeed, in one report of the treatment of cats with GS-441524, independent analysis of the unregulated preparation used to treat the 18 cats showed it to contain more than double the dose of GS-441524 than the manufacturer implied on the label, 34 and similar findings were reported in another study, 35 showing that it is not possible to determine the actual doses used of unlicensed or unregulated products, making generalised treatment recommendations very difficult. Despite these limitations, it is clear that oral GS-441524 treatment of FIP is effective; all 18 cats (16 with effusions) recovered in a prospective study using 84 days of oral GS-441524 treatment. 11 Additionally, retrospective studies using 84 days of GS-441524 treatment (primarily oral, but SC treatment was used in some cats) showed that 116/141 (82.2%) cats with FIP and effusions, 32 137/161 (85.1%) cats with ‘mixed’ signs of both effusive and non-effusive FIP disease and 153/163 (93.9%) of cats with FIP without effusions 33 were treated successfully.
Remarkably, in one study on treatment using owner-reported data (via surveys), only 8.7% of owners had received help from their veterinarian in administering treatment to their cat with FIP, and most had obtained treatment information from online resources. 10 This is likely due to the difficulties in veterinary use of unregulated preparations because, in many countries, their use, including prescribing and administration, by veterinarians is illegal and/or can result in removal of a veterinarian’s licence to practise, depending on the country’s regulations. Thus, many veterinarians feel unable to help owners directly with the treatment of FIP in the absence of legally available regulated or licensed products, despite it being suggested that supportive veterinary care during the early days of treatment is critical to a successful outcome. 11
In 2021, oral GS-441524 became legally available as a regulated veterinary compounded ‘Specials’ product in Australia (since August 2021), the UK (since November 2021) and some other countries (dependent on their importation regulations), offering a route, in these countries, for veterinarians to treat cats with FIP using a legally sourced veterinary-prescribed GS-441524 product of known content and purity. The composition of this product is assayed by a validated analytical method and must comply with specifications before release. Physical and chemical properties are tested in each batch and ‘A Certificate of Analysis’ has to be produced.
Remdesivir (GS-5734) is another, more phosphorylated, nucleoside analogue that is rapidly converted to GS-441524 in mammals.36,37 Remdesivir has been suggested as a treatment for respiratory coronavirus diseases in humans, most notably COVID-19 due to SARS-CoV-2, although clear evidence for a beneficial effect in humans is lacking and the results of different studies are contradictory. 38 Remdesivir has also been considered for FIP treatment in cats, 39 but its safety and efficacy have not been established in controlled peer-reviewed publications. However, favourable reports of the use of remdesivir to treat FIP in small numbers of cats have emerged,40 –43 including the use of a regulated veterinary compounded ‘Specials’ product of injectable remdesivir (of known composition, as outlined above for GS-441524) legally available for cats in Australia (since November 2020), the UK (since August 2021) and some other countries (dependent on importation regulations, similar to GS-441524). As this is a drug that is registered for human use in almost all jurisdictions, its use in cats with FIP has been subject to fewer legal constraints than the unlicensed drug GS-441524, and various human formulations made under licence are widely available in many countries, including India, South Africa and Japan (R Malik, 2023, personal communication). Although a greater body of evidence has been published for the efficacy of GS-441524 compared with remdesivir, and comparative studies of GS-441524 and remdesivir do not exist, in some countries only remdesivir is legally available to prescribe for feline use. This is because, in the absence of being able to import veterinary compounded ‘Specials’ products, some countries’ rules allow veterinarians to prescribe human licensed products to animals (ie, remdesivir licensed for use in humans). Thus, more data on the use of remdesivir in cats with FIP are required to enable more cats to receive treatment safely when this is available. The importation regulations and documentation required for veterinary compounded ‘Specials’ products in some countries require that peer-reviewed evidence on the efficacy of specific products must be provided before importation is permitted (E Jones, 2023, personal communication).
The main aim of this retrospective study was to describe the use of legally prescribed and sourced veterinary compounded remdesivir and/or GS-441524, containing known amounts of active agents, in a large number of cats with FIP, to obtain descriptive information on their efficacy and any adverse effects. An additional aim was to publish treatment data that might help facilitate importation of such products into more countries, allowing a greater number of veterinarians to legally treat cats with FIP, with resulting advantageous welfare outcomes.
Materials and methods
Case recruitment
Cases were recruited to the study by enrolling cats treated with the legally sourced, veterinary-prescribed regulated veterinary compounded ‘Specials’ preparations (also known as extemporaneous preparations) of injectable remdesivir and/or GS-441524 tablets, manufactured by BOVA in the UK and Australia and imported to some additional countries.
Cats were sourced from: (i) records from a free FIP advice email service (via FIPadvice@gmail.com) run by five of the authors (SST, DGM, ENB, SS and ST) to help veterinarians with the diagnosis and treatment of FIP; (ii) records of the manufacturer (BOVA) of veterinary practices treating FIP cases that had given permission to be contacted by the authors; (iii) advice emails to another author (RM); and (iv) study publicity via social media and professional speaker engagements at conferences and veterinary continuing professional development events (SST, DGM, ENB, ST, SC and JMN). Cases were recruited between March 2022 and September 2022 and follow-up sought in December 2022; treatment for FIP had been given from 2020 to 2022. All reviewed cat data were anonymised and contained no identifying personal details of the owner/client, and permission was sought from all clients to allow entry of their pets’ anonymised data into the study. Where necessary, gatekeeper approval for access to social media pages was sought or authors themselves were the gatekeepers. A Self-Assessment for Governance and Ethics (Animal Research) was undertaken at the University of Surrey (Application ID: 638929-638920-91768170), which determined that this project did not require a full ethical review by the University of Surrey.
Exclusion criteria comprised: (i) absence of a confirmed, very likely or highly suspicious diagnosis of FIP (see below under ‘Diagnosis of FIP’); (ii) treatment, at any point, with a nucleoside analogue from a source other than the veterinary compounded BOVA remdesivir or GS-441524 products (at the time of the study, these were the only veterinary compounded products containing remdesivir and GS-441524 that were legally available in the UK); and (iii) inadequate cat and/or treatment data.
Cat data collected
Cat data collected included: country of origin, signalment, clinical signs and their duration, physical findings (pyrexia defined as a rectal temperature of >39.2°C 44 or the description ‘pyrexia’ recorded in cat data), diagnostic tests (including haematology, serum biochemistry, urine analysis, infectious disease testing, radiography, ultrasonography, advanced imaging [CT or MRI], cytology and/or biochemistry of fluid samples, FNAs, histopathology samples, testing for FCoV antigen by immunostaining and testing for FCoV RNA by RT-PCR) and treatment data (for further details, see below). Whether a sibling or housemate had confirmed or suspected FIP was also tabulated when recorded.
Diagnosis of FIP
Cat data were used to categorise the diagnosis of FIP by one of the board-certified internal medicine specialist authors (SST), based on the European Advisory Board for Cat Diseases’ FIP diagnostic tool algorithms.1,17 The categories of FIP diagnosis used were:
1. Confirmed: consistent signalment, clinical signs, physical findings, haematology/serum biochemistry/imaging findings AND consistent histopathology or cytology (on body cavity fluid, such as effusion, or cerebrospinal fluid [CSF], or FNA samples of affected tissues) WITH positive immunostaining for FCoV antigen
2. Very likely: consistent signalment, clinical signs, physical findings, haematology/serum biochemistry/imaging findings AND consistent histopathology or cytology WITH confirmed identification of FCoV RNA by RT-PCR on appropriate samples (eg, body cavity fluid, such as effusion, or CSF, or FNA samples of affected tissues)
3. Highly suspicious: consistent signalment, clinical signs, physical findings, haematology/biochemistry/imaging findings but WITHOUT confirmed presence of FCoV RNA by RT-PCR, or FCoV antigen by immunostaining on appropriate samples (either negative result or testing not performed).
Any cats that did not meet the above criteria, due to their signalment, clinical signs, physical findings, haematology/biochemistry/imaging findings and/or any other testing performed that was not consistent with FIP, were categorised as being unlikely to have FIP and were excluded from the study.
Type of FIP
Cat data were used to characterise the predominant (ie, main) type of FIP present by one of the board-certified internal medicine specialist authors (SST). Cats were characterised based on the predominant clinical signs and physical findings. When the predominant type of FIP could not be determined (due to limited diagnostic testing or reported data), the type of FIP was deemed ‘uncharacterised’. Presence of an effusion (at any location) at diagnosis was documented. The following categorisations were used for the predominant type of FIP:
Effusion – thoracic (this included pleural and/or pericardial effusions)
Effusion – abdominal (this included retroperitoneal effusions)
Effusion – both cavities (ie, thoracic and abdominal)
Neurological signs dominant
Ocular signs dominant
Abdominal pathology dominant (excluding signs and findings due to an abdominal effusion, in which case effusion – abdominal was used to categorise). This category included cases where the predominant signs or findings were mesenteric, splenic and/or renal abnormalities and/or abdominal lymphadenopathy
Uncharacterised.
Where a cat had more than one type of FIP (eg, a cat had an abdominal effusion plus ocular signs), the cat was categorised based on the predominant type of FIP but the other type, or types, of FIP present were also recorded as ‘additional’ FIP type(s).
Treatment data collected
Treatment data, including response data, were recorded as to whether either remdesivir or GS-441524 was used alone or both were used serially in the same cat (ie, remdesivir given initially, then a transition to GS-441524). It is important to note that the regulated remdesivir was available before GS-441524, and this influenced protocol use. Additionally, an 84-day period of treatment was usually given due to recommendations based on Pedersen’s seminal studies and further published studies using unregulated nucleoside analogues.11,32,33 The term ‘initial treatment period’ was used to describe the first continuous period of nucleoside analogue treatment (in days). Route of administration, frequency of daily administration, dose (in mg/kg of the cat’s current weight), any alterations in dose (to a new dose in mg/kg) and duration of treatment (in days) were recorded. Any other treatments given were also recorded. The number of veterinary monitoring reviews each cat underwent, as well as the timing of these (in days from the start of nucleoside analogue treatment), was also recorded, together with the results at each review (clinical signs, physical findings and diagnostic tests, such as blood tests and imaging results, if the cat was alive or dead, and if the cat had died or had been euthanased).
Each cat was assigned a response-to-treatment category at every veterinary review follow-up time point during treatment, at the end of their initial treatment period and at any further follow-up time points, by one of the authors (SST) based on available information (clinical signs, physical findings and diagnostic test results). Additionally, each cat was assigned a response-to-treatment category at the follow-up time point as close to (but within) 30 days of treatment as possible, if this information was available. The response-to-treatment categories were:
‘Complete’ if the cat appeared clinically healthy, based on all available information
‘Partial’ if only partial improvement was reported and/or some abnormalities were still present based on available information
‘No’ if there was no response (ie, no improvement based on available information or static or worsening signs).
The results of each follow-up for the cats with only a partial or no response to treatment were collated as descriptive data, including whether such cats were alive or dead at the end of the initial treatment period and at the longest follow-up time point (see below for further information).
The time to normalisation of temperature, clinical signs, serum bilirubin concentration, haematocrit or packed cell volume and serum globulin concentration, time to resolution of effusion and time to achieving a serum AG ratio of greater than 0.4 were also recorded.
Cats were also categorised according to whether or not they had any relapse of FIP; a ‘relapse’ was defined as a recurrence of clinical signs, physical findings and/or diagnostic test results consistent with FIP (as decided by one of the board-certified internal medicine specialist authors [SST]) after an initial response to nucleoside analogue treatment. Any cats with a relapse were subdivided as to whether the relapse occurred ‘during’ the initial treatment period or ‘after’ completion of the initial treatment period. Details of any management (eg, alteration in drug dose, repeat treatment) and/or outcome of the relapse (eg, euthanasia) were also recorded.
The longest follow-up time point (in days) after starting treatment (ie, day 1 being the first day of treatment) for each cat was recorded, as well as the longest follow-up time point (in days) after completion of the initial treatment period, if treatment had been completed (ie, day 1 being the first day after treatment had been completed). Whether the cat was alive or dead at these time points was also recorded.
Any perceived adverse effects seen in association with remdesivir or GS-441534 treatment were recorded and described. For inclusion as a clinicopathological side effect, the clinicopathological variable needed to have been normal pre-treatment but then measured and found to be abnormal during or after treatment.
Any neutering or vaccination elective procedures performed during or after the nucleoside analogue treatment were also recorded, as well as any adverse clinical response to these procedures.
Statistical analysis
Data were recorded in Excel, version 2212 (Microsoft Corp) and exported into SPSS, version 29.0.0.0 (IBM Corp) for analysis. Descriptive statistics were used to report the data. Data were reported as median (range). Categorical data of the number of cats alive or dead at the end of the initial treatment period grouped according to whether they had achieved a complete response within 30 days were compared with χ2 or Fisher’s exact tests. P <0.05 was considered statistically significant.
Results
In total, 318 cats were initially recruited to the study. Eleven of these cats were then excluded: six due to part treatment with sources of remdesivir or GS-441524 other than BOVA veterinary compounded preparations, three had inadequate treatment (including response) data available and two were not categorised as having a confirmed, very likely or highly suspicious diagnosis of FIP (both of these cats were thought ‘unlikely’ to have FIP). Thus, 307 cats were included in the main study cohort.
Cat data
The 307 cats comprised 174 from the UK, 115 from Australia, 11 from Sweden, five from South Africa and two from Japan. In total, 105 clinics provided data for the 307 cats.
Breeds were recorded for 306 cats and was unknown in one instance. Roughly two-thirds of cats (192/306; 62.8%) were purebred, with the three most prevalent breeds being British Shorthair (57/192; 29.7%), Ragdoll (32/192; 16.6%) and Maine Coon (15/192; 7.8%). Other purebreds included Oriental Shorthair (14), Bengal (10), Norwegian Forest Cat (seven), Scottish Fold (six), Burmilla and Russian Blue (six each), Burmese and Persian (five each), Exotic Shorthair, Siberian and Tonkinese (four each), Birman, Siamese and Sphynx (three each), Devon Rex (two), Abyssinian, Australian Mist, Cornish Rex, Minuet, Munchkin and Savannah (all one each). The remaining cats (113/306; 36.9%) were non-purebreds with 110 listed as domestic short-, medium- or long-haired cats and two recorded as purebred crosses.
Age was known for all 307 cats; the median (range) age at diagnosis was 11 (3–187) months, with just over half of the cats (161/307 cats; 52.4%) aged under 12 months.
Sex and neuter status were known for all 307 cats; almost two-thirds were male (194/307; 63.2%) of which just over three-quarters were neutered (147/194; 75.8%). Of the female cats, a similar proportion were neutered (79/113; 69.9%).
Of the 257 cats for which data were available for housemates or siblings, 12.1% (31/257) were reported to have or had a housemate or sibling affected by suspected FIP (although unconfirmed as a diagnosis in most cases). However, the 31 cats included six from the same multi-cat environment in the UK, most likely representing a rarely seen ‘outbreak’ of FIP.45 –49
Clinical signs and physical findings were reported for 306 of the 307 cats (Table 1). Lethargy (287/306; 93.8%), inappetence (231/306; 75.5%) and weight loss (131/306; 42.8%) were the most commonly described clinical signs. Neurological and ocular signs were seen in 20.3% (62/306) and 13.1% (40/306) of the cats, respectively.
Clinical signs and physical findings of cats with feline infectious peritonitis
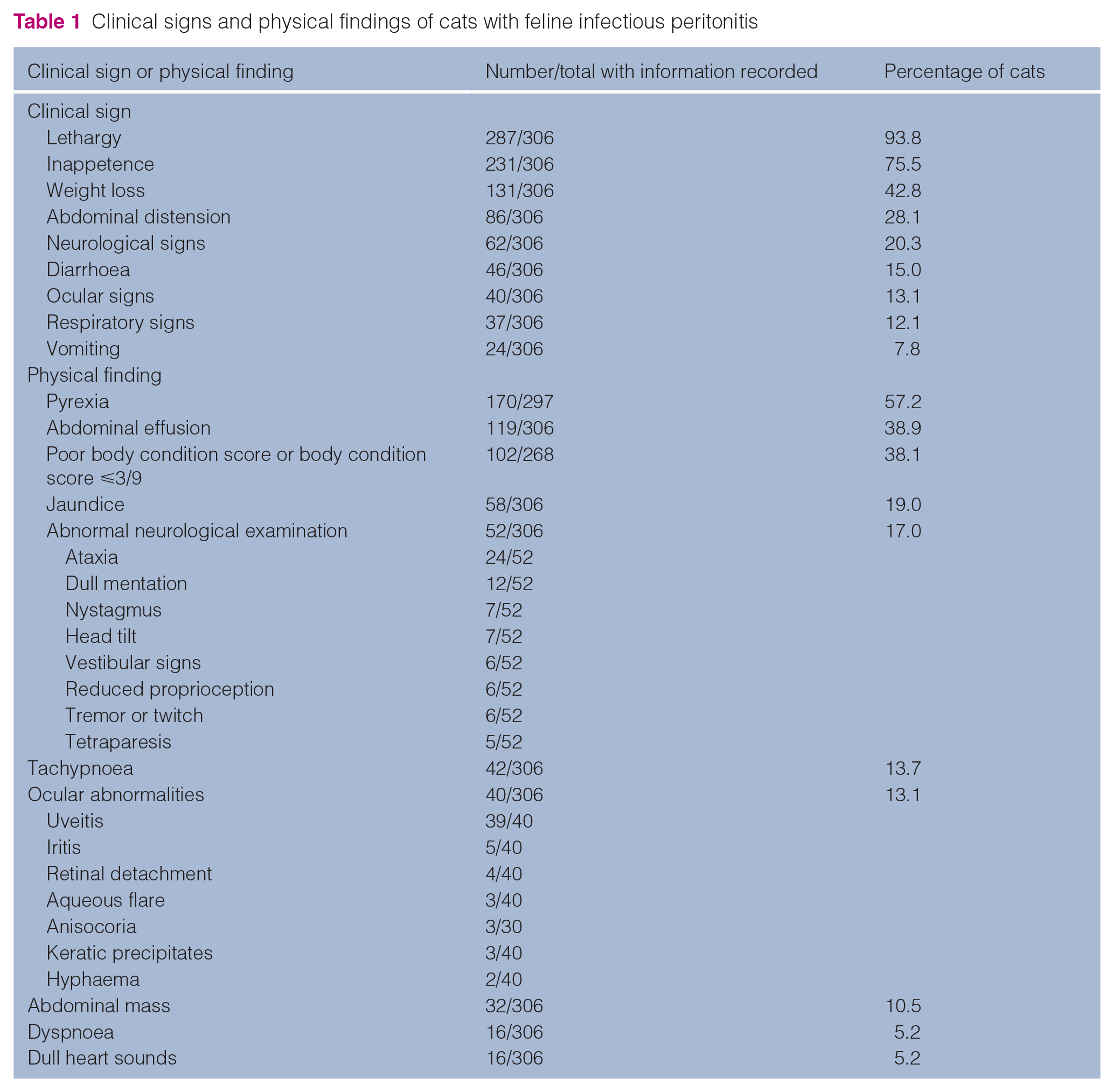
For the 297 cats with an adequate history recorded, pyrexia had frequently been present (170/297; 57.2%), with a median (range) temperature of 39.8 (39.3–41.0)°C. An abdominal effusion was the next most commonly reported physical finding with 119/306 (38.9%) cats so affected. The median (range) weight at diagnosis was 3.0 (0.96–7.7) kg and body condition was scored or described in the records of 268 cats, with 102/268 (38.1%) said to be in poor body condition or with a body condition score of ⩽3/9. An abdominal mass was found in 10.5% (32/306) of cats. Other clinical signs and physical findings and their frequency are shown in Table 1. The median (range) duration of clinical signs prior to diagnosis was 10 (1–210) days.
Diagnostic testing results were available for most cats (eg, haematology in 297/300 [99.0%] and serum biochemistry in 300/306 [98.0%]). Table 2 shows the clinicopathological results, as well as feline leukaemia virus antigen, feline immunodeficiency virus antibody, FCoV antibody, serum AGP, diagnostic imaging, cytology and histopathology results, and reports the number of cats evaluated with each diagnostic test. A low serum AG ratio of ⩽0.4 occurred in 74.8% (175/234) of cats, with only 8.6% (20/234) of cats having a serum AG ratio of ⩾0.6. Hyperglobulinaemia occurred in 74.2% (221/298) and hypoalbuminaemia occurred in 54.4% (149/274) of cats. Forty-two percent (120/286) of cats were hyperbilirubinaemic (compared with 19.0% [58/306] that were jaundiced on clinical examination, as shown in Table 1).
Diagnostic testing results for cats with feline infectious peritonitis

Text is as recorded in submitted data and relates to text of results obtained from laboratory †Calculated as proportion of cats having full abdominal ultrasound AG = albumin to globulin; AGP = alpha-1 acid glycoprotein; CSF = cerebrospinal fluid; FCoV = feline coronavirus; FeLV = feline leukaemia virus; FIV = feline immunodeficiency virus; HCT = haematocrit; ICC = immunocytochemistry; IHC = immunohistochemistry; LN = lymph node; PCV = packed cell volume; POCUS = point of care ultrasound; WBC = white blood cells
FCoV RNA detection by RT-PCR was performed in 143/303 (47.2%) cats; these included 46/143 (32.2%) by FCoV RT-quantitative PCR, 50/143 (35.0%) by IDEXX FIP Virus RealPCR and 41/143 (28.7%) by RT-(non-quantitative) PCR, and the type of PCR was not recorded for the remaining six cats. Test results were available for 140 of the 143 RT-PCRs performed; 111/140 (79.3%) were positive, with the remaining 29/140 (20.7%) negative. FCoV antigen immunostaining by direct immunocytochemistry was performed in 37/307 (12.1%) cats, and was positive in 26/37 (70.3%) cats. FCoV antigen immunostaining by immunohistochemistry (IHC) on tissue biopsy samples was performed in 6/307 (2.0%) of cats, with all six (100%) positive.
Two cats underwent necropsy after they died or were euthanased following deterioration during treatment; one that already had a confirmed diagnosis of FIP by FCoV antigen immunostaining of pleural fluid and the second that had been categorised as a diagnosis of FIP being ‘very likely’ due to a positive RT-PCR for FCoV RNA on an effusion sample. Samples collected at post-mortem examination in both cats showed histopathological changes, including pyogranulomatous inflammation, consistent with FIP, but FCoV antigen IHC was not performed.
Diagnosis of FIP
Only 9.5% (29/307) of the cats were categorised as having a ‘confirmed’ diagnosis of FIP. One-third (102/307; 33.2%) were categorised as ‘very likely’ to have FIP, while most (177/307; 57.7%) were categorised as being ‘highly suspicious’ for a diagnosis of FIP.
Type of FIP
The predominant type of FIP was characterised in 299/307 (97.4%) cats and uncharacterised in the remainder (8/307; 2.6%) (Figure 1). In total, 213/299 (71.2%) cats had an effusion at diagnosis and 86/299 (28.8%) did not. The numbers of each predominant type of FIP were: 152/307 (49.5%) effusion – abdominal, 44/307 (14.3%) neurological signs dominant, 35/307 (11.4%) abdominal disease dominant, 27/307 (8.8%) effusion – thoracic (pleural and/or pericardial), 23/307 (7.5%) ocular signs dominant and 18/307 (5.9%) effusion – both cavities. Additional types of FIP were identified concurrent to the predominant type in 46/299 (15.4%) cats (Table 3), including 12 cats with dominant neurological or ocular signs that also had effusions, and 10 cats with both ocular and neurological signs. Lastly, six cats had a third additional type of FIP present, as described in Table 3.

Pie chart illustrating the number and percentage of each type of predominant feline infectious peritonitis among the 307 cats in the study
Additional type of feline infectious peritonitis (FIP) seen alongside the predominant type of FIP in 46 of the 307 cats in the study

Six cats also had a third type of additional FIP described
Three cats with neurological signs dominant and ocular signs also had effusion – abdominal
One cat with neurological signs dominant and ocular signs also had effusion – both cavities
One cat with ocular signs dominant and neurological signs also had abdominal pathology
One cat with effusion – abdominal and ocular signs also had neurological signs
Treatment data
The nucleoside analogue treatments given are shown in Table 4; as described earlier, the availability of the treatments determined the specific protocols used. Around one-third of the cats (104/307; 33.9%) were treated with remdesivir alone. Over half the cats (171/307; 55.7%) were serially treated with remdesivir first and then GS-441524 afterwards. In these cases, remdesivir was transitioned to GS-441524 at a median (range) of 15 (2–150) days after commencing remdesivir (note that both drugs were not given together). Only 32/307 cats (10.4%) were treated with GS-441524 alone. Thus, overall, 275/307 cats (89.6%) received remdesivir and 203/307 cats (66.1%) received GS-441524.
Data on the dose and duration of treatment are presented in Table 4 and Table 5. Of the 275 cats that received remdesivir, the initial administration route was intravenous (IV) in around half of those cats (153/275; 55.6%) and SC in the remainder (122/275; 44.4%). For the 153 cats that initially received remdesivir IV, the administration route was changed to SC in 114/153 (74.5%) of cats after a median (range) of 3 (1–17) days of IV administration. No further qualitative data (eg, dilution, speed of injection) about the method of administration of remdesivir were consistently available in records/submitted data (although the recommendations for IV administration of remdesivir were to dilute the 10 mg/kg dose to 10 ml in saline and then administer it over 10–20 mins 43 ). All cats receiving remdesivir were treated once a day. Oral GS-441524 was administered once daily to most cats (164/203; 80.8%) but twice daily to around one-fifth of cats (39/203; 19.2%).
Nucleoside analogue treatment data for cats with feline infectious peritonitis, including response and follow-up, according to treatment group
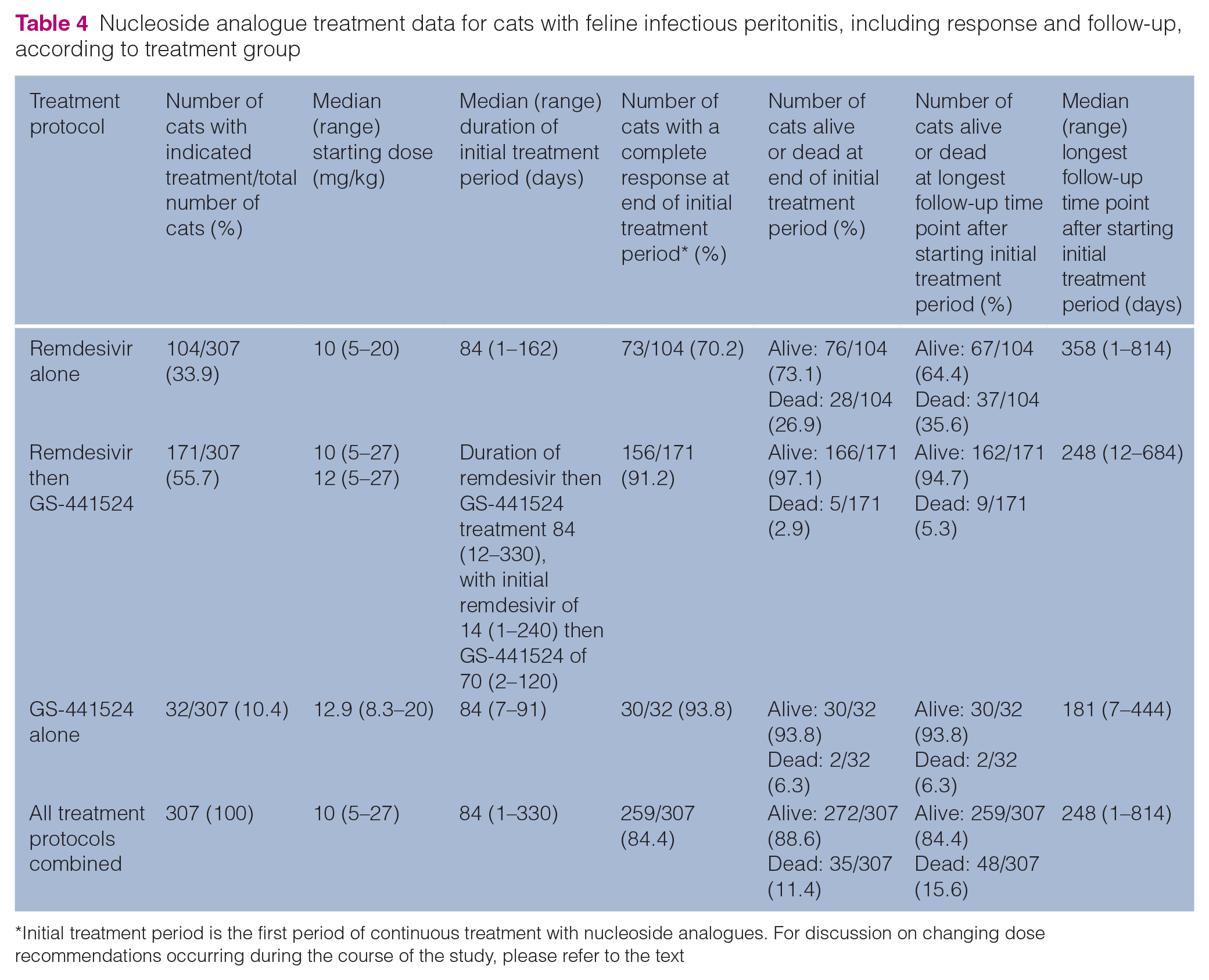
Initial treatment period is the first period of continuous treatment with nucleoside analogues. For discussion on changing dose recommendations occurring during the course of the study, please refer to the text
Nucleoside analogue treatment data for cats with feline infectious peritonitis (FIP), including response and follow-up, according to predominant type of FIP present

NA = not applicable
Starting doses of remdesivir and GS-441524 are illustrated in Figure 2 and Table 4. The median (range) starting dose of remdesivir in the group given remdesivir alone was 10 (5–20) mg/kg, while the starting dose of GS-441524 in the group given GS-441524 alone was 12.9 (8.3–20) mg/kg. The median (range) starting doses of the group given serial remdesivir and then GS-441524 were 10 (5–27) mg/kg for remdesivir and 12 (5–27) mg/kg for GS-441524. Around one-quarter (82/307; 26.7%) of cats had their nucleoside analogue dose increased during treatment, as shown in Table 5. At the time that the newly available veterinary compounded remdesivir became available in Australia, the preliminary advice was to start with a high dose of remdesivir IV and then reduce the dose when given SC; this was later altered in the light of evolving experience. Hence, 15/307 (4.9%) cats had a dose decrease when changed from IV to SC remdesivir.

Boxplot showing starting doses of remdesivir and GS-441524 as sole treatment and in the combination group treated with remdesivir and GS-441524 sequentially. The boxes are edged by the 25th and 75th percentiles of the data, with the median (50th percentile) shown by a line within the box. The crosses indicate the mean of the data. The whiskers mark the 5th and 95th percentiles and values beyond these upper and lower bounds are considered outliers, marked with coloured dots
The median (range) initial treatment period for the 307 cats was 84 (1–330) days. Fifty-one of 307 (16.6%) cats were treated for longer than 84 days, with the median (range) initial treatment period in these 51 cats being 105 (87–330) days. In total, 52/307 (16.9%) cats were treated for less than 84 days with a median (range) of 8 (1–83) days. Fourteen of these 52 cats were treated for between 70 and 83 days, and two of these 14 cats were euthanased during their initial treatment period; one with ongoing neurological signs and one with the development of haemothorax of unknown cause. Thirty-eight of 52 cats were treated for less than 70 days; five were alive at the end of the initial treatment period (treatment lasted a median [range] of 60 [30–69] days) and the remaining 33 were euthanased or died early in the initial treatment period, at a median (range) of 3 (1–55) days.
Corticosteroids were given to 61 of the 307 (19.9%) cats; 18 received parenteral dexamethasone (median [range] dose 0.13 [0.05–1.3] mg/kg) for a median (range) duration of 2 (1–30) days, and 33 cats received oral prednisolone at a median (range) dose of 1 (0.5–2.0) mg/kg for a median (range) duration of 7 (3–14) days. Twelve cats received topical ophthalmic preparations containing corticosteroids, although the dose and duration were not recorded.
In terms of other treatments given, these included antibiotics (such as cefovecin and doxycycline) to 148/307 (48.2%) cats, systemic non-steroidal anti-inflammatory drugs to 41/307 (13.4%) cats, mefloquine to 14/307 (4.6%) cats (dose not recorded) and feline recombinant interferon-omega to one cat (0.3%). No cats received polyprenyl immunostimulant.
More than half of the cats (174/307; 56.7%) received non-specific supportive therapy with drugs including mirtazapine (55/174; 31.6%), maropitant (45/174; 25.9%), gabapentin (35/174; 20.1%), opioid analgesia (24/174; 13.8%), IV fluid therapy (23/174; 13.2%), topical non-corticosteroid-containing ophthalmic preparations (21/174; 12.1%), ondansetron (8/174; 4.6%), blood transfusions (5/174; 2.9%) and anti-seizure drugs (4/174; 2.3%). Only two cats received ‘hepatoprotectant’ treatments (such as S-adenosylmethionine and silybin).
The median (range) number of veterinary monitoring reviews was 3 (1–10) with the first review occurring at a median (range) of 10 (1–114) days after commencing treatment.
At completion of their initial treatment period, 272/307 cats (88.6%) were alive. At the longest follow-up time point available (see data below), 259/307 cats (84.4%) were alive (Figure 3 and Tables 4 and 5). Of the 13 cats that were alive at the completion of their initial treatment period but not alive at the longest follow-up time point available after completion of the initial treatment period, 11 (84.6%) had relapsed with clinical signs consistent with FIP (for details, see below), one had died from a road traffic accident and one was euthanased for pituitary neoplasia (the attending veterinarian did not consider this to be related to FIP).

Bar chart showing the percentage of the 307 treated cats that were alive at the longest follow-up time point after starting treatment and also showed a complete response to the nucleoside analogue treatment indicated
At completion of their initial treatment period, most cats (259/307; 84.4%) cats had a ‘complete’ response to treatment and the remainder had either a ‘partial’ (18/307; 5.9%) or ‘no’ (30/307; 9.8%) response to treatment. More details on response to treatment are provided in Table 5.
Of the 18 cats with only a ‘partial’ response at completion of the initial treatment period, further evaluation of their data showed that 10 remained alive and eight were dead at the longest follow-up time point. Of the 10 cats with a partial response that were still alive, four had already had the initial treatment period extended beyond 84 days, with a median (range) initial treatment period of 119 (104–189) days, one had a repeat 84-day course of GS-441524 after stopping briefly, one was treated with oral mefloquine (25 mg once daily) and four cats were monitored only. Interestingly, 4/10 cats with a partial response that were alive at the longest follow-up time point had persistent, but static, neurological signs but were otherwise normal at 1–240 days after completing the initial treatment period, two had persistent hyperglobulinaemia but were otherwise clinically normal at 90 and 180 days after 84 days of treatment, two had static ocular signs (present at diagnosis) at 10 and 240 days after finishing the initial treatment period, one had a persistent small volume abdominal effusion and lymphadenopathy but remained otherwise normal at review 210 days after finishing the initial treatment period, and one had ongoing stable azotaemia and ultrasonographic changes in the kidneys 35 days after completion of 84 days of treatment, but was otherwise clinically normal. Of the eight cats with a partial response that were dead at the longest follow-up time point, the median (range) follow-up time point at which they had died or were euthanased was 55 (2–330) days following the start of treatment.
Of the 30 cats that were recorded with ‘no’ response to treatment, all died or were euthanased during the initial treatment period. These 30 cats had a median (range) time of death recorded at a follow-up time point of 3 (1–55) days following the start of treatment. Of these 30 cats, 12 died, 10 were euthanased due to deterioration of clinical signs, five were euthanased due to the development or worsening of neurological signs and three were euthanased due to a lack of improvement.
Within the first 30 days of treatment, 271/307 cats (88.3%) were re-examined and, of these 271 cats, 76 (28.0%) had a complete response, whereas 72.0% (195/271) did not. Of the 76 cats with a complete response within 30 days of treatment, 75 (98.7%) were alive at the end of the initial treatment period, significantly more than those that did not have a complete response within 30 days of treatment but were alive at the end of the initial treatment period (160/195; 82.1%) (P <0.001).
Figure 4 shows the time to normalisation (in days) for temperature, clinical signs, serum bilirubin concentration, haematocrit or packed cell volume and serum globulin concentration, as well as time to resolution of effusion and time to achieve a serum AG ratio >0.4. These data suggest that, during remdesivir and/or GS-441524 treatment, the serum globulin concentration takes the longest to normalise of the measurements tracked, as has been reported previously. 31 Over one-quarter of cats (47/176; 26.7%) that had serum biochemistry reassessed within 30 days of starting treatment had an initial worsening of hyperglobulinaemia before subsequent normalisation, as has been reported previously. 29 , 42

Boxplot showing time to normalisation in days of various clinical and clinicopathological parameters during treatment for feline infectious peritonitis. HCT = haematocrit; AG = albumin to globulin ratio. The blue boxes are edged by the 25th and 75th percentiles of the data, with the median (50th percentile) shown by a line within the box. The crosses indicate the mean of the data. The whiskers mark the 5th and 95th percentiles and values beyond these upper and lower bounds are considered outliers, marked with blue dots. HCT boxplots represent HCT or packed cell volume, depending on which was reported in the haematology profiles
Overall, 33/307 (10.8%) cats relapsed with a recurrence of clinical signs, physical findings and/or diagnostic test results consistent with FIP. Considering the 33 cats that relapsed, the percentages of each of the predominant types of FIP categories at initial diagnosis that relapsed were as follows: 5/23 (21.7%) cats with ocular signs dominant; 4/27 (14.8%) cats with effusion – thoracic dominant; 5/44 (11.4%) cats with neurological signs dominant; 14/152 (9.2%) of cats with effusion – abdominal dominant; 2/35 (5.7%) cats with abdominal pathology dominant: 2/18 (11.1%) cats with effusion – both cavities dominant; and 1/8 (12.5%) cats with the uncharacterised FIP category relapsed. The 33 cats that relapsed included 17 cats (51.5%) that relapsed with neurological signs, five (15.1%) with weight loss, inappetence and pyrexia, three (9.1%) with effusions, three (9.1%) with uveitis, two with jaundice and an abdominal mass, two with severe anaemia and one with both uveitis and neurological signs. Of the 33 cats that relapsed, 25/33 (75.8%) did so with clinical signs different from their initial predominant FIP subtype.
Fifteen of the 33 cats (45.5%) that relapsed did so during the initial treatment period at a median (range) of 40 (3–90) days into treatment, while the remaining 18 (54.5%) relapsed after completion of the initial treatment period, at a median (range) of 14 (7–450) days after stopping treatment; 15/18 (83.3%) were within 60 days of stopping treatment, but three cats suffered later relapses at 90, 390 and 450 days. Thus, 30/33 cats (90.9%) relapsed, either during their initial treatment period or within 60 days of stopping it.
Of the 15 cats that relapsed during the initial treatment period, eight were then treated with an increased dose of the same drug, of which 7/8 responded; these were treated for a median (range) of 148 (114–204) days in total during their initial treatment period, with a median (range) longest follow-up time point after completing their initial treatment period of 370 (7–510) days. The one cat that failed to respond to an increased dose, and the remaining seven cats that were not treated further, were euthanased at a median (range) of 84 (2–330) days into the initial treatment period.
Of the 18 cats that relapsed after completing the initial treatment period, 10/18 were treated by restarting nucleoside analogues and eight responded, 2/10 did not respond and 2/10 that responded relapsed again (one euthanased, one responded to a third, higher-dose treatment course with a longest follow-up time point after completing this third course of 520 days). The median (range) longest follow-up time point from completion of the initial treatment period for cats that relapsed but responded to repeat treatment was 90 (7–520) days. The remaining 8/18 were euthanased at a median (range) of 23 (7–450) days after completing the initial treatment period.
The median (range) of the longest follow-up time point after starting the initial treatment period was 248 (1–814) days. The median (range) of the longest follow-up time point after completion of the initial treatment period was 180 (0–730) days. Further information on follow-up time points according to treatment group is presented in Table 4.
Adverse effects of treatment
Adverse effects were recorded in 176/307 cats (57.3%), including clinical adverse effects in 122/307 (39.7%) and clinicopathological abnormalities in 107/245 cats (43.7%) that had blood samples taken during the initial treatment period. The most frequent adverse effects are shown in Figure 5. Localised pain or discomfort associated with SC injection of remdesivir was reported in almost half (122/255; 47.8%) of all the cats that received SC remdesivir. The clinicopathological adverse effects seen (in cats treated with remdesivir alone, GS-441524 alone or both drugs) were increased serum alanine transaminase (ALT) activity (in 71/250 of cats [28.4%] in which this was measured, with a median [range] value of 110 ([47–1260] U/l), eosinophilia (in 38/253 [15.0%] with a median [range] of 2.2 [0.9–4.9] × 109/l), lymphocytosis (in 26/244 [10.7%] cats with a median [range] of 8.2 [6.1–13.3] × 109/l), increased serum alkaline phosphatase activity (in 23/281 [8.2%] cats with a median [range] 81 [range 55–155] U/l) and increased serum creatinine concentration (in 8/243 [3.3%] cats, with a median [range] of 180 [148–383] µmol/l). The proportion of cats with elevated ALT was higher in cats treated with GS-441524 (either in combination with remdesivir or alone, being 37.5% and 30.8%, respectively) than cats treated with remdesivir alone (8.3%). Conversely, cats treated with GS-441524 alone had a lower proportion with eosinophilia (3.9%) compared with cats treated with remdesivir alone or in combination with GS-441524 (19.4% and 14.8%, respectively).

Bar chart showing the percentage of the 307 treated cats showing the most common adverse effects seen during injectable remdesivir and/or oral GS-441524 treatment. GS-441524 is an oral medication and so pain on injection relates to injectable remdesivir administration only. Figures within bars are the percentage of cats affected. ALT = alanine transaminase
Other less frequent adverse effects included sores and thickened skin at remdesivir SC injection sites, seen in 9/255 of cats (3.5%), change in the colour of the fur (from black to white) at the interscapular injection site (1/255 cats), subdued demeanour immediately after remdesivir injection (both when administered SC and IV; seen in 7/275 [2.5%] of cats), hypotension after IV remdesivir (seen in 5/255 [2.0%] of cats) and diarrhoea after starting oral GS-441524 (seen in 4/203 [2.0%] of cats). Three of the cats in the study (3/307; 1.0%) developed pruritus; one had generalised pruritus that resolved after changing from SC remdesivir to oral GS-441524, one had facial pruritus during the initial treatment period with remdesivir, and one had pruritus around the interscapular remdesivir SC injection site. One cat developed thrombocytopenia, without associated clinical signs, with GS-441524 treatment (platelet count prior to treatment was within the reference interval, whereas the platelet count was 6 × 109/l on day 31 of treatment [not confirmed by blood smear examination] and 40 × 109/l on day 80 [confirmed by blood smear examination]; this persisted after GS-441524 was stopped and the thrombocytopenia eventually responded to corticosteroid therapy).
Vaccination or neutering elective procedures
Twenty-three cats were vaccinated after finishing the initial treatment period at a median (range) of 60 (21–300) days, while one cat was vaccinated during the initial treatment period. Twenty-one cats were neutered (both males and females) after finishing the initial treatment period at a median (range) of 60 (1–240) days and one cat was neutered during treatment. None of these cats had evidence of FIP relapse or other adverse sequelae following vaccination or neutering.
Discussion
The present study describes a very substantial cohort of 307 cats with a presumed or confirmed diagnosis of FIP treated using specific regulated veterinary compounded nucleoside analogues of known composition in countries where these products can be legally prescribed by veterinary surgeons to cats under their care. The legality of the drugs has facilitated access to data relating to direct veterinary input into the care of these cats. In agreement with previous studies10,11,22,28 –33,40,42 reporting the use of varied (usually unregulated or illegal) nucleoside analogue preparations, in the present study, all three treatment protocols (ie, remdesivir alone, remdesivir then GS-441524 and GS-441524 alone) were highly effective in treating this previously fatal disease. An impressive 88.6% of cats were alive at the end of the initial treatment period, and 84.4% at the longest follow-up time point after completion of the initial treatment period (ie, at a median [range] of 180 [0–730] days). Interestingly, these favourable results were obtained without the use of additional immunomodulatory treatments (such as polyprenyl immunostimulant, and feline recombinant interferon-omega was given to only one cat) as reported by others.22,31
Importantly, we cannot directly compare the efficacy of treatment with remdesivir alone with the use of remdesivir followed by GS-441524, or GS-441524 given as monotherapy. This is because this was not a randomised controlled clinical trial and, as such, cannot assess efficacy. The protocols used to treat the cats with FIP evolved rapidly during the study,42,43,50 as more information was obtained through the experience of managing these cases. This led to changes, notably increases, in recommended doses and duration of treatment, aiming to optimise the response to treatment and try to prevent relapses. In addition, the type of FIP present influenced the protocol, with higher doses usually recommended for cats with ocular or neurological signs.11,30,43,50 The high cost of treatment is likely to be one reason for not giving a simpler higher dose to all cases. In terms of drug choice, for the cohort of cats described in the present study, remdesivir was the first, and initially only, nucleoside analogue to become legally available; thus, the initial treatment protocols comprised remdesivir alone. The regulated veterinary compounded GS-441524 became available several months after remdesivir, such that protocols evolved to transition from remdesivir (which could be given IV, if necessary, to critically ill cats, or SC for longer-term use) to oral GS-441524 treatment. Finally, oral GS-441524 monotherapy was used in the cats recruited later in the study, once experience had shown it to be of comparable efficacy to the treatment regimens including remdesivir (see below). Thus, the first cats recruited into the study were given remdesivir alone, and often at a lower dose, than those given treatment later in the study; this would likely have impacted the response to treatment. This can be seen in Figure 2 and Tables 4 and 5, which suggest a trend for lower doses of remdesivir being used when it was the only treatment given (to cats recruited early in the study) compared with the doses used when remdesivir was used principally as a prelude to oral GS-441524.
Remdesivir is more expensive (on a per-mg basis) than oral GS-441524, and has additional increased costs and challenges due to its injectable administration compared with an oral tablet that can be readily halved or quartered, and this could have influenced the likelihood of owners electing to euthanase a cat following a clinical setback or slow improvement while remdesivir was being administered as the only available treatment. The subsequent availability of compounded GS-441524 tablets provided a cheaper and less stressful form of treatment, which no doubt influenced owner and veterinary choice, especially once confidence in its efficacy evolved. For this cohort of cats, remdesivir, as the only injectable formulation, was the only agent used to treat cats that could not tolerate oral medications and, because such cats tend to be those that are sickest due to their FIP disease, this could have biased the population of cats given remdesivir towards those that were most sick, which may also have influenced the response to treatment; this further prohibits direct comparison of the treatment groups.
Based on our collective experience, it is the opinion of the authors that, given the ease of treatment with oral GS-441524, its lower cost and the excellent response rate seen in the present study, most cats with FIP should probably be treated with oral GS-441524, with the use of injectable remdesivir reserved for two situations. The first is the scenario where remdesivir is the only nucleoside analogue available, or the most affordable option, which is the case in some countries, including many developing nations, where it is made inexpensively as a human drug under licence, with such formulations being available for cat owners (R Malik, 2023, personal communication); in this situation, it is important to note that 73.1% of cats treated with remdesivir only were alive at the end of the initial treatment period. Further research, ideally comprising controlled clinical trials, may reveal protocols that can confer greater survival using this agent. Second, in some cats and kittens that present late with advanced disease, oral administration of GS-441524 might be impossible or unreliable, such that an injectable treatment (IV or SC) is required (eg, in cases with severe ileus or gastrointestinal malabsorption, neurological signs affecting ability to swallow, collapse, risk of aspiration of food after a seizure, etc). It has been suggested31,35 that oral, rather than injectable, administration of a nucleoside analogue might be more efficacious because it targets the major intestinal site of FCoV replication. Studies of successful responses to injectable GS-441524 certainly exist,10,28 –30 but comparative studies on the efficacy and bioavailability of the active moieties of oral GS-441524 and parenteral remdesivir (including its efficiency of conversion into GS-441524) are currently lacking, although they are emerging (S Coggins, 2023, personal communication). The original dose and treatment duration (84 days) protocols for these agents were based on the recommendations of veterinarians in Australia who had already used these formulations successfully in cats with FIP 42 (R Malik, S Coggins, D Hughes and JM Norris, 2023, personal communication).
The signalment of the cats in the present study was broadly similar to that of previously described populations of cats with FIP; most of the cats were purebred (63.4%),51 –56 male (63.2%)13,53–57 and under 12 months of age (52.4%).9,15,53,54,56 The presenting clinical signs and physical findings were also consistent with other studies;10,13,58,59 lethargy and inappetence were frequently reported as non-specific clinical signs, and pyrexia occurred in around two-thirds of cats. Cats presented more commonly (71.2%) with effusions than without,7 –15 and 20% of the cats had neurological signs, while 13% had ocular disease (Table 1). One recent paper, describing a large case series of cats treated for FIP, reported a lower percentage to have effusions (57%), with the remainder of cats having neurological or ocular manifestations; 10 that study was reliant on owner-reported clinical signs in cats that were largely not being monitored by veterinarians, compared with the veterinary assessment of all cats in the present study, which should have improved the accuracy of descriptions and possibly resulted in earlier presentation of the cats in their clinical course due to owners seeking veterinary care. Importantly for practitioners, 15.4% of cats had clinical signs of FIP of more than one type, suggesting that looking for effusions and examining the eyes (eg, of cats with neurological signs) may aid the diagnosis and full characterisation of FIP cases.
One study limitation and interesting discussion point is that only 9.5% of cats in the study met the stringent diagnostic criteria that confirms a diagnosis of FIP, with only another one-third of cats (33.2%) having FCoV RNA detected in samples, leading to a diagnosis of FIP being ‘very likely’, the remainder being highly suspicious. This lack of definitive diagnosis in many cats is not dissimilar to other studies on the outcome of cats treated for FIP10,11,22,29 –33,40 because, increasingly, financial resources for treatment are being prioritised over those to confirm the diagnosis, and the ability to perform further diagnostic testing in a case can be problematic due to access to testing or the condition of the cat. A positive response to treatment in suspected cases of FIP is sometimes used as strong diagnostic support for FIP. However, not confirming a diagnosis of FIP ahead of commencing antiviral treatment does create a risk that (i) an alternative treatable diagnosis is overlooked and (ii) these antivirals are administered without justification and may increase risks of antiviral resistance, with potential One Health impacts. We recommend that veterinarians discuss this risk with owners ahead of treatment, so that informed decisions are made so that the most appropriate approach is taken for each case.
It is noteworthy (Table 2) that 85.3% of the effusions, 75.0% of the tissue FNAs and 46.2% of the CSF samples (as well as both of the two aqueous humour samples) submitted for FCoV RNA RT-PCR gave positive results in the present study. This highlights the usefulness of FCoV RNA RT-PCR testing in cases in which FIP is a differential diagnosis, as has been suggested previously.1,8,20,26,60 FCoV antigen immunocytochemistry (ICC) staining is more specific for the diagnosis of FIP than FCoV RNA RT-PCR8,26 but none of the three FNA samples sent for ICC testing gave positive results. However, the ICC yield from effusion samples was far higher than for FNA, with 76.5% of 34 effusion samples positive for FCoV antigen ICC. Although treatment for suspected FIP in the absence of a definitive diagnosis can be effective and, when successful, is used by some to ‘confirm’ the diagnosis, as outlined above, it is still important for veterinarians to be as confident as possible in a diagnosis of FIP before starting antivirals. We still recommend, when finances permit, that veterinarians try to demonstrate FCoV antigen or FCoV RNA within FNA or fluid samples using ICC and RT-PCR, respectively, to make a diagnosis of FIP as complete as possible. However, we know that FCoV distribution and detection is variable in cats with FIP and a negative result for these tests does not exclude a diagnosis of FIP. 60 Additionally, delaying treatment while diagnostic tests are pending is suboptimal for the cat; in-house cytology and effusion biochemistry can expedite exclusion of other causes of clinical signs (eg, pyothorax) and facilitate prompt use of antivirals.
The proportion of cats responding to treatment and being alive at both the end of their initial treatment period and the longest follow-up time point available was similar between the predominant FIP types (Table 5). This may be, in part, because of the rapid evolution, adoption and availability of optimised protocols to treat different types of FIP.30,50,61 As mentioned earlier, initial protocols from when remdesivir first became legally available in the countries in which the authors and their collaborators practise included recommendations to administer higher doses of nucleoside analogues to cats with neurological or ocular involvement, and this dose recommendation was adopted in some of the cats in the present study, as shown in the median dose values in Table 5, although a range of doses was used. A study that used owner-reported dose data from surveys reported similar results, in that cats with neurological or ocular manifestations received higher doses of nucleoside analogues than those without these signs. 10 It is not possible, however, to directly compare doses administered in different studies due to the unknown purity and content of the unlicensed and/or unregulated products. The present study highlights the need for more treatment response data to be generated in cats treated with known and accurately quantified nucleoside analogues and veterinary care to guide treatment decisions, as well as data on any effect of route of administration and formulation.
As seen in previous studies,10,29 relapses of FIP did occur, and many were successfully managed with extended or repeated treatment courses, typically at increased doses. It is possible that some of these relapsing cats initially presented with occult ocular or neurological involvement, such that initial dose selection was inadequate. Indeed, previous studies62 –65 have shown the presence of ocular and neurological inflammation and FCoV at higher frequencies on extensive diagnostic testing than was suspected based on clinical signs alone. The vast majority (90.9%) of relapses occurred during the initial treatment period, or within 60 days of stopping. It is not possible to say whether more frequent veterinary monitoring reviews in these cats might have resulted in earlier detection of a lack of response to treatment and subsequent appropriate dose escalation. Given the lack of toxicity observed with remdesivir and GS-441524 treatment, the decision to use appropriate higher doses according to FIP type is appropriate. In addition, serum levels may vary between cats, necessitating higher doses for individual cats based on response (D Gunn-Moore, 2023, personal communication). Three-quarters of relapsing cats did so with clinical signs different from their initial predominant FIP type, and half were with neurological signs, as reported previously. 10 Hence, vigilance for novel clinical signs during and after antiviral treatment is recommended.
Treatment with nucleoside analogues in the present study was well tolerated, but potential side effects were observed in 57.3% of the cats, the most common reported being pain on injection in 47.8% of cats treated with SC remdesivir. Previously reported adverse effects when treating cats with parenteral nucleoside analogue products have included, similar to the present study, pain or discomfort at SC injection sites (with both remdesivir and GS-441524),10,28 -30 as well as transient exacerbation of pleural effusion (when given IV remdesivir), 42 which was not clearly seen in the present study (although cats with thoracic effusions can require repeat thoracocentesis early in the treatment course as fluid can re-accumulate before the cat responds to treatment). The cause of pain on injection is unclear and may be multifactorial (eg, volume of fluid injected, injection site used, temperature of the fluid injected); however, most clinicians suspect that the low pH of the solution is a significant factor. The pH of the veterinary compounded remdesivir used in the present study (pH 4.1) was considerably less acidic than the human-licensed remdesivir product available in the UK (pH 1.4; Veklury) (N Bova, 2023, personal communication) and it was hoped that any injection pain would thereby be minimised. Not all reports describe pain following SC remdesivir, with one case report of successful treatment in a cat with confirmed FIP using remdesivir IV for 3 days followed by remdesivir SC for an additional 77 days without problems; this cat remained in remission at the 7-month follow-up at the time of publication. 41 Injectable SC GS-441524 has similarly been associated with pain on injection; 82.0% of cats treated with owner-sourced GS-441524 were reported to have vocalised at the time of injection and 76.1% were reported to have exhibited signs of pain at the injection site, while around half (51.7%) reported scarring and scabbing. 10 Pain or discomfort on injection has also been reported during administration of the protease inhibitor antiviral agent GC376 for FIP. 66 These reactions are often a major reason for preferring oral medications, even though advice is available to try to help minimise the pain on injections (such as the use of gabapentin, which was commonly used and helpful in many cats treated in the present study).43,50
Previously reported clinicopathological changes associated with oral GS-441524 administration in cats have included a mild increase in liver enzyme activities for ALT, lymphocytosis or eosinophilia. 11 Eosinophilia was reported in the present study in around 20% of cats treated with remdesivir, although it did not result in premature drug withdrawal and resolved after completion of the course of therapy. By contrast, eosinophilia was not seen as frequently in cats treated with GS-441524 (3.9%). Increased serum ALT activity was seen in over 30% of cats treated with GS-441524; again, it did not lead to premature withdrawal of treatment and resolved on completion of treatment, without the administration of ‘hepatoprotectants’. Elevated ALT activity was seen less frequently in cats treated with remdesivir alone (8.3%). The causal pathomechanisms underlying these clinicopathological changes are unknown at this time. Development of, or worsening of, azotaemia was not reported in any cats treated with remdesivir in the present study. This is of note because acute kidney injury has been associated with remdesivir use in humans,67,68 although causality remains unproven. 69
When to stop nucleoside analogue treatment has been a topic of some debate in communications with several of the authors via the FIP advice line they contribute to (SS Taylor, S Tasker, EN Barker, D Gunn-Moore and S Sorrell, personal communications), particularly if abnormalities (especially certain clinicopathological changes in the absence of clinical signs) persist at 84 days, which is the typical time of completion of the primary course of therapy. One small retrospective study of 42 cats with confirmed or suspected FIP documented the successful use of attaining normal serum AGP measurements to differentiate cats that fully recovered from FIP (26 cats) from those did not (16 cats), 31 and it may be that AGP could be used as an indicator to stop antiviral therapy, including allowing for treatment courses of shorter durations. Further prospective studies are required to confirm this. Although further research is required, it is of interest that four cats in the present study had persistent static neurological signs yet showed long-term survival, two cats with hyperglobulinaemia that persisted at the end of the initial treatment period had not relapsed up to 180 days after stopping therapy, and one cat with a persistent small volume effusion and lymphadenopathy remained well at 210 days after completing the initial treatment period. Further research is required, but our current advice is to initially investigate and then closely monitor such cases for relapse; lack of a complete response may be because these signs are due to, for example, fibrosis or permanent loss of neurons following severe FIP pathology and inflammation, although this has not been confirmed. Lymphadenopathy has been reported in a cat following treatment with GS-441524 and recovery from FIP, without any apparent clinical effect and no associated FCoV immunostaining. 34
The limitations of the present study included its retrospective nature, with data derived from a collaboration of a large number of clinicians working in different environments and countries, during a period of flux when FIP treatments and availability, as well as specialist recommended treatment doses and duration, were changing and evolving. The present study does not provide answers to all of the questions we have regarding the treatment of FIP with regard to optimal treatments, doses, duration and routes of administration. Ultimately, in the present study, the diagnosis, treatment and monitoring employed by the veterinarians were at their individual discretion. As well as the science of therapeutics, decisions were impacted by discussion with owners regarding affordability of treatment and compliance preferences. Recruitment of cases via multiple routes could have introduced bias regarding case submission.
The strength of the present study, which sets it apart from most other studies, is the exact knowledge of the strength of diagnosis alongside what nucleoside analogue was administered and at what dose, which helped our understanding of the response to certain treatment decisions (Table 4). FIP is fatal without effective treatment. These data described herein may permit clinicians who have access to these products to predict the likelihood of response if administered at the doses described, pending description of prospective clinical trials upon which treatment decisions are ideally based.
By virtue of the treatment success in the present study, the majority of cats that had a complete response to treatment remain alive. This, alongside the variations in dosage and follow-up time points after treatment when data were collected (eg, 2020 compared with 2022), precludes survival data analysis. It is hoped that survival will continue over a far longer period than we were able to report at our longest follow-up time point. Of interest in this regard is that one of the 18 ‘cured’ cats in an oral GS-441524 treatment study in which all 18 cats with FIP recovered (the shortest follow-up time was 99 days after completion of the 84-day treatment course) 11 sadly died as a result of a road traffic accident 164 days after finishing treatment. 34 That cat underwent necropsy and no gross or microscopic evidence of FIP was found, nor were FCoV antigen or FCoV RNA found in any tissues (although lymphadenopathy was found, as described above). This supports the theory that there is elimination of FCoV from all tissues following successful treatment of FIP using oral GS-441524, again giving hope for longevity of life and permanent cure following treatment.
This retrospective study also reports the successful vaccination and neutering of cats that have completed treatment for FIP, which is encouraging due to previous concerns that possible stress associated with these procedures may lead to a relapse of FIP. However, we recommend that cat friendly practices are always adopted to minimise stress. 70
Conclusions
The nucleoside analogues remdesivir and GS-441524, both alone and used sequentially, are highly effective in the management of FIP when administered at the doses and duration described in the present study. These data have been used to make summary recommendations for veterinarians for whom these products are legally available, which sadly does not include all feline practitioners at this time. 50 The easier administration of GS-441524 tablets over injectable remdesivir, which is often associated with pain when given by SC injection, makes it more likely to be the first-line treatment in countries in which GS-441524 tablets are available. However, remdesivir is still appropriate to use where this is the only nucleoside analogue legally available or in rare cases in which cats do not tolerate oral medications, at least during initial treatment until clinical improvement results in tolerance of oral medication. Positive responses within the first 30 days of therapy may be associated with better outcomes. Randomised controlled clinical trials are required to further compare and optimise treatment and monitoring regimens.
Footnotes
Acknowledgements
We thank the veterinary clinics generously providing data for this project including in the UK: Animal Medical Centre, Ark Vets, Aura Veterinary, Baildon Veterinary Centre, Bay Veterinary Group, Cheshire Vets, Clare Veterinary Group, Companion Care (Weston Super Mare), Cotswold Vets, Cramar Vets, Davies Veterinary Specialists, Donaldson Vets, Dovecote Veterinary Specialists, Dragon Vets, East Neuk Veterinary Clinic, Eastcott Veterinary Referrals, University of Edinburgh R(D)SVS Small Animal Hospital, Goddard Veterinary Group, Henwick Vets, Highcroft Veterinary Referrals, Langford Vets, London Cat Clinic, Lumbry Park Veterinary Specialists, Maple Veterinary Clinic, Medivet (Battersea, Croxley Green, Evegate, Hinkley), Millcroft Vets, Montgomery Veterinary Clinic, Moray Coast Vet Group, North Downs Veterinary Referrals, Oakhill Vet Centre, Oaks Veterinary Centre, Paragon Veterinary Referrals, Pride Veterinary Centre, Queen’s Veterinary School Hospital, Cambridge University, Rutland Veterinary Centre, Severn Veterinary Centre, Southern Counties Veterinary Specialists, Swift Referrals, The Green Vets, The Ralph, Top Cat Veterinary Centre, Valley Vets, Vets For Pets (Corstophine, Walton Vale), Vets Now Manchester, Veterinary Referral Centre, Watkins and Tasker Vets, West Mount Vets, Whitstable Bay Veterinary Centre and Wylie Veterinary Centre; in Australia: Advanced Vetcare, Alphington and Fairfield Vets, Bombaderry Vets, Cat Specialist Services, Canberra Cat Clinic, Challenger Vet Hospital, Concord Veterinary Hospital, Greencross Vets, Hills Veterinary Clinic, Northside Veterinary Specialists, Pacific Vetcare, Paddington Cat Hospital, Perth Cat Hospital, Pet Hub Balaclava, Red Hill Vets, Reedy Creek Vets, Rolystone Animal Hospital, Small Animal Specialist Hospital, Sylvania Veterinary Clinic, The Cat Clinic (Mt Gravatt, Hobart), Veterinary Referral Hospital, Vets@Acacia Gardens, Vet HQ Double Bay, Vet HQ Darlinghurst, Walkerville Vet and Willoughby Veterinary Hospital; in South Africa: Brackenhurst Animal Hospital and King Edward Veterinary Hospital; in Sweden: Anicura Djursjukhuset Albano, Blue Star Veterinary Hospital and Vasby Djursjukhus; and in Japan: Morita Animal Hospital. Additional thanks go to Zoey Broughton for assistance with data collation and Fabienne Marie for support and providing information.
Author note
The authors wish to highlight to readers that, during peer review of this paper, two additional descriptive case series on the treatment of FIP with remdesivir and/or GS-441524 were published: Coggins SJ, Norris JM, Malik R, et al.
Conflict of interest
No external companies had any role in the writing of, or in the decision to publish, the research presented. All of the research was performed by the authors independently. ENB, DGM, SC and SST had all funds from BOVA for the provision of continuous professional development material on FIP (articles, webinars, conference presentations) donated to a fund for FIP research. ENB, DGM, SS, SST and ST comprise a group of five RCVS recognised specialists in feline and internal medicine who run a free-of-charge email advice line on FIP for veterinarians to aid with the diagnosis of FIP and its treatment. Individual authors have been consultants for, or received research funding and honoraria from, various commercial companies and other organisations; full details available on request.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognised high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS. Although not required, where ethical approval was still obtained, it is stated in the manuscript
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers) for all procedure(s) undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
