Abstract
Objectives
The study aimed to assess the clinical efficacy, safety and outcomes of legally prescribed and compounded GS-441524, with or without prior remdesivir, in cats with confirmed or highly suspected feline infectious peritonitis (FIP) in the Netherlands. A standardised diagnostic and treatment protocol was used to evaluate clinical response, adverse effects and prognostic factors.
Methods
This prospective, multicentre study included 127 client-owned cats with FIP, enrolled between June and November 2023 from three Dutch veterinary clinics. Diagnosis followed American Association of Feline Practitioners (AAFP)/EveryCat guidelines, incorporating clinical findings, laboratory data, imaging, cytology and RT-qPCR performed on either fine-needle aspirates or effusion. Cats were treated for a minimum of 12 weeks with oral GS-441524, with some initially receiving intravenous remdesivir before transitioning to oral therapy. Dosing was based on disease form (effusive, non-effusive, ocular, neurological). Follow-up included physical examinations and clinicopathological monitoring. Descriptive statistics, Kaplan–Meier survival analysis and Cox regression were used for data analysis.
Results
Of the 127 cats, 55.9% had effusive, 19.7% non-effusive, 16.5% ocular and 7.9% neurological FIP; 21.3% showed mixed forms. Most cats were aged under 2 years (87.4%), male (77.2%) and purebred (60.6%). Common clinical signs included lethargy (93%) and inappetence (83%). Overall survival was 78%. Ocular FIP had the highest survival (100%), while neurological FIP had the lowest (60%). Elevated serum bilirubin and creatinine concentrations at presentation were associated with reduced survival. Adverse effects were generally mild and transient, with vomiting (33%) and diarrhoea (29%) most commonly reported.
Conclusions and relevance
This study confirms that GS-441524, with or without prior remdesivir, is an effective and well-tolerated treatment for naturally occurring FIP. Clinical improvement was typically rapid; resolution of laboratory abnormalities took longer. These findings support the legal use of GS-441524 in feline practice and highlight the need for further research to refine treatment protocols.
Plain language summary
Feline infectious peritonitis (FIP) was long considered a fatal disease in cats. It is caused by a mutated form of feline coronavirus and occurs in two main forms. Effusive (wet) FIP leads to the accumulation of fluid in the abdomen or chest. Non-effusive (dry) FIP does not cause fluid build-up but can affect internal organs such as the liver, kidneys, lymph nodes, eyes (ocular FIP) or brain, and spinal cord (neurological FIP). With the development of antiviral drugs such as GS-441524, many cats can now recover. GS-441524 can be given orally and blocks replication of the virus that causes FIP. Its injectable prodrug, remdesivir, is converted in the body to GS-441524 and is sometimes used at the start of therapy before switching to oral medication. In the Netherlands, GS-441524 is now legally available for veterinarians, but real-world data have been limited. This prospective, multicentre study evaluated 127 cats with confirmed or highly suspected FIP, treated in three Dutch veterinary clinics. Diagnosis followed international guidelines and was based on clinical signs, blood tests, imaging, cytology and RT-qPCR testing of effusion or fine-needle aspirate samples, which detects genetic material of the virus causing FIP. All cats received oral GS-441524 for at least 12 weeks. Some were initially treated with intravenous remdesivir before switching to the oral form. Most cats were young (under 2 years), male and purebred. Overall survival was 78%. Cats with ocular FIP had the best outcome (100% survival), while those with neurological disease had the lowest (60%). Reported side effects were mild and short-lived, mainly vomiting and diarrhoea. This study shows that GS-441524, with or without prior remdesivir, is an effective and well-tolerated treatment for naturally occurring FIP.
Keywords
Introduction
Feline infectious peritonitis (FIP) is a common systemic viral disease of domestic and wild felids, caused by feline coronavirus (FCoV).1,2 FCoV occurs as two different biotypes. The first, feline enteric coronavirus (FECV), is ubiquitous in cats worldwide, especially in multi-cat households, and is not in itself an important pathogen. Cats with a FECV infection are either asymptomatic or show mild self-limiting gastrointestinal signs. The other biotype is the mutated virulent biotype, also called feline infectious peritonitis virus (FIPV). Initial infection almost always involves the non-mutated biotype. In a small portion of cats with FCoV (5–14% in multi-cat environments), a spontaneous mutation will occur causing a change in cell tropism from enterocytes to monocytes/macrophages. This biotype switch is a key event in the pathogenesis of FIP. This allows for systemic spread of FCoV and causes a fatal immune-mediated multisystemic infection.3 –7
The incidence of FIP is related to a number of host, virus and environmental co-factors, such as population density, immune status and age of the cat, breed predisposition, virulence and viral load.6 –9 Most cats with FIP show non-specific clinical signs, such as lethargy, loss of appetite, fluctuating fever and weight loss or failure to gain weight. However, the clinical presentation of FIP is complex and variable. In general, an effusive form with a relatively rapid course and widespread vasculitis, which manifests as fluid accumulation in the body cavities, is distinguished from a non-effusive form where (pyo)granulomatous lesions are present. The non-effusive form is often more chronically progressing over weeks to months. Clinical signs depend on which organs are affected. The kidneys, liver, intestines (including local lymph nodes), central nervous system and eyes are most commonly involved; however, there is considerable overlap between effusive and non-effusive disease and clinical signs can also change over time.4,7,10 –12
The definitive diagnosis of FIP can be challenging because of the lack of pathognomonic clinical signs and laboratory changes, especially in the non-effusive form of the disease. A definitive diagnosis usually relies on demonstration of FCoV antigen or RNA on effusions or fine-needle aspirates (FNAs) in association with typical FIP histopathological changes. However, a range of supporting diagnostic evidence can be obtained from haematology (non-regenerative anaemia, lymphopenia), certain biochemical parameters (eg, hypergammaglobulinaemia, hyperbilirubinaemia, albumin:globulin (A:G) ratio <0.4), investigation of effusion and cytology of liver, spleen and/or mesenteric lymph nodes (pyogranulomatous inflammation).2 –4,9,10,13,14
FIP is fatal when left untreated. Ground breaking studies published in 2018 and 2019 showed that the antiviral nucleoside analogue GS-441524 was efficacious in the treatment of experimentally induced and spontaneously occurring FIP.15 –17 Lack of veterinary licensing has led to the emergence of an unlicensed drug market, both for GS-441524 and remdesivir (GS-5734), a prodrug of GS-441524. 18 Reputedly, many thousands of cats have undergone treatment globally, with remission achieved successfully in many cats. However, the importation of unlicensed medicinal products for veterinary use remains illegal in many parts of the world.10,19 –21 In the early 2020s, veterinarians in Australia and the UK gained legal access to a compounded, injectable formulation of remdesivir and later also a tablet formulation of GS-441524 through a veterinary compounding pharmacy (BOVA).13,22,23 As the UK is no longer a member of the EU, it is still impossible for Dutch veterinarians to obtain these products legally. In early 2023, the Veterinary Pharmacy of the Faculty of Veterinary Medicine in Utrecht obtained the human medicinal product remdesivir (Veklury; Gilead Sciences) for intravenous (IV) use, as well as raw GS-441524 from a Good Manufacturing Practice (GMP)-certified facility, which was compounded into capsules. In June 2023, a multicentric, prospective study was started with the aim to document the use of legally prescribed and sourced oral GS-441524, with or without prior IV remdesivir using a standardised diagnostic, treatment and monitoring protocol to obtain descriptive information on the efficacy of the medication, description of the clinical course, any adverse effects and prognostic factors.
Materials and methods
Study cohort, inclusion and exclusion criteria, and case recruitment
This multicentre, prospective study was conducted at the University Clinic for Companian Animals of the Faculty of Veterinary Medicine in Utrecht, AniCura Specialist Referral Centre Haaglanden and IVC Evidensia Referral Hospital Arnhem. Client-owned cats suspected of having FIP were enrolled after referral by primary veterinarians or through direct owner initiative. The study was actively promoted through veterinary magazines, websites, social media, webinars, podcasts and information letters to general practitioners. A central email address was established to facilitate enrolment and response to inquiries.
Cases were recruited between June and November 2023. Written informed consent was obtained from all clients. All reviewed cat data were pseudonymised and contained no identifiable personal details of the owner/client. No untreated control group was included, as withholding effective treatment was considered unethical given the fatal prognosis of untreated FIP. Cats remained under owner care, with owners responsible for treatment costs and veterinary services. This study was approved by the Utrecht Animal Welfare Authority (16205-3-03).
Exclusion criteria for enrolment in the study included: (1) treatment at any point with a nucleoside analogue from a source other than the GS-441524 or remdesivir provided by the Veterinary Pharmacy of Utrecht University; or (2) absence of a confirmed or highly suspected FIP diagnosis. Cases were excluded from data analysis if treatment or patient data were incomplete or inadequate for evaluation.
Diagnosis of FIP
The following diagnostics were performed for all cats before inclusion:
(1) Signalment and history with use of a standardised questionnaire (see S1 and S2 in the supplementary material).
(2) Physical examination, including body weight, body condition score (BCS) and muscle condition score (MCS), 24 neurological and ophthalmological examination (see S3 in the supplementary material).
(3) Laboratory tests. All tests were primarily conducted at the University Veterinary Diagnostic Laboratory (UVDL). The minimum database included complete blood counts performed on an ADVIA 2120i (Siemens Healthcare) and biochemistry analyses (serum creatinine, alanine aminotransferase [ALT], bile acids, bilirubin) performed on an Olympus AU 680 (Beckman Coulter). Protein profiles were determined by protein electrophoresis (Hydragel β1-β2, Hydrasis; Sebia). Previously established reference intervals (RIs) were applied. Haematology results from external laboratories obtained up to 2 days before inclusion were accepted. Biochemistry results performed up to 5 days before inclusion, either in-house or externally, were also accepted.
(4) Abdominal and/or thoracic ultrasound performed by a board-certified radiologist, resident radiologist under supervision of a board-certified radiologist or an experienced veterinary ultrasonographer.
(5) If effusion was present: effusion was aspirated and protein concentration was determined. Cytology was used to assess the likelihood of FIP and exclude other diseases. FCoV RT-qPCR targeting the 7b gene was performed at the Veterinary Microbiological Diagnostic Center (VMDC) at Utrecht University. 25 RNA was extracted using the QiAamp Viral RNA Mini Kit (Qiagen), and amplification used TaqMan Fast Virus 1-Step Master Mix on a LightCycler 96 system (Roche). Quality controls included positive and negative PCR controls, an internal amplification control (Phocine distemper virus), a feline housekeeping gene (GAPDH) and extraction blanks. 26 Results were reported as positive or negative; if positive, the crossing point (CP) value was included.
(6) If no effusion was present: FNAs were taken from the liver, spleen and/or mesenteric lymph nodes for cytology to exclude other diseases and to obtain material for FCoV RT-qPCR. For the PCR test, samples were deposited in Eppendorf tubes filled with RNA shield to conserve RNA. The extraction of RNA and RT-qPCR were performed as described above.
(7) Externally performed FCoV RT-qPCR or immunohistochemistry for FCoV antigen were accepted.
The diagnosis of FIP was based on the AAFP/EveryCat FIP diagnosis tool algorithms. 4
The categories of FIP diagnosis used were:
Confirmed: compatible signalment, history, clinical examination findings, haematology/serum biochemistry/imaging findings and consistent histopathology or cytology (on body cavity fluid, such as effusion, or FNA samples of affected tissues) with a positive FCoV RT-qPCR and/or positive immunohistochemistry for FCoV antigen.
Highly suspicious: compatible signalment, history, clinical examination finding, haematology/serum biochemistry/imaging findings and consistent histopathology or cytology (on body cavity fluid, such as effusion or FNA samples of affected tissues) without a positive FCoV RT-qPCR.
Cats that did not meet the specified criteria – based on their signalment, medical history, clinical examination, results from haematology, biochemistry, imaging or other diagnostic tests inconsistent with FIP – were considered unlikely to have FIP and were therefore excluded from the study.
Cat data were used to classify the predominant type of FIP that was present. The types of FIP were effusive, non-effusive, ocular signs dominant or neurological signs dominant. If a cat had more than one type of FIP, the cat was categorised based on the main type of FIP, but the other type(s) of FIP present were also recorded (secondary FIP, tertiary FIP).
Treatment protocol
GS-441524 capsules of 10 mg, 20 mg and 40 mg used in treatment were compounded and dispensed by the Veterinary Pharmacy of the Faculty of Veterinary Medicine of Utrecht University. Remdesivir was obtained via leftover stock at several human hospitals in the Netherlands.
All cats with confirmed or highly suspicious FIP were included regardless of their form of FIP or severity of disease. At the clinician’s discretion, treatment could begin before all diagnostic results were available. Oral GS-441524 capsules were the first-line treatment. In cats too obtunded to swallow, diluted IV remdesivir was used initially, with a switch to oral GS-441524 once feasible. Remdesivir use was closely monitored for adverse effects such as thrombophlebitis or anaphylaxis. Subcutaneous administration was not permitted because of injection-site pain. Choice of initial treatment route (IV remdesivir or oral GS-441524) was left to the discretion of the treating clinician and was guided by the cat’s clinical presentation and overall condition.
The dose of both GS-441524 and remdesivir was as follows:
Uncomplicated effusive or non-effusive FIP: 10–12 mg/kg q24h PO or IV
Ocular involvement: 15 mg/kg q24 PO or IV
Neurological involvement: 10 mg/kg q12h PO or 20 mg/kg q24h IV
The calculated GS-441524 dose was rounded up to the nearest available capsule size. Capsules were administered directly or mixed with a small amount of wet food or treat. Doses were adjusted during the study based on body weight, and treatment duration was at least 12 weeks. Supportive medications, such as antiemetics, non-steroidal anti-inflammatory drugs and appetite stimulants, were permitted. Gabapentin or pregabalin could be given before veterinary visits to reduce stress. Systemic glucocorticoids were only used when strictly necessary, such as for posterior uveitis.
Monitoring protocol
Physical evaluations were conducted at 2, 4, 8 and 12 weeks after treatment initiation, and 4 weeks after treatment (Figure 1). If treatment was extended beyond 12 weeks, an additional checkup was performed at 20 weeks. At each visit, a standardised questionnaire was used to record side effects, and a physical, neurological and ophthalmological examination was performed. Blood samples (3–4 ml) were collected into EDTA and serum tubes for complete blood count and biochemistry, including creatinine, ALT, bile acids, bilirubin, albumin, gamma globulins and A:G ratio. Additional testing was performed at the clinician’s discretion. All laboratory analyses were conducted at the UVDL.

Timeline of monitoring
If at week 6 the clinical or clinicopathological abnormalities persisted or worsened, the dose of GS-441524 was increased by 3–5 mg/kg/day. Treatment was stopped for the following reasons: (1) if the cat had been treated for at least 12 weeks (84 days); (2) if all clinical signs of FIP had resolved; (3) serum bilirubin concentration was within the RI; and (4) gamma globulin levels were 20 g/l or below (RI 4–8).
At weeks 1, 3, 5, 6, 7, 9, 10 and 11, the cat owners were contacted by telephone or email to complete a questionnaire assessing the cat’s clinical condition (Figure 1, see also S4 in the supplementary material). Owners were also asked about the ease of administering GS-441524, with the following response options: (1) ‘Easily administered’; (2) ‘I am able to give the medication, but it is not always easy’; (3) ‘Some days I am not able to give the medication’; or (4) ‘Unable to administer at all’.
Statistical analysis
Data were collected in Excel (version 2501, build 16.0.18429.20132) and exported into SPSS Statistics (version 29.0.2.0; IBM) for statistical analysis. Assessment of normality of distribution of continuous data was performed using the Shapiro–Wilk test. Collected data on haematology and biochemistry values were further analysed using descriptive statistics and linear mixed models with a fixed effect of time and individual animals as random effect. Data on population, clinical signs, physical signs, survival and relapse were analysed using descriptive statistics. The χ2 test was used to compare the proportions of categorical variables. Survival curves were calculated using the Kaplan–Meier product limit analysis. Differences in survival between groups were determined with the log-rank test. Cats were additionally censored for survival when they were alive at the time of data accrual closure, distinguishing death due to other reasons or lost to follow-up (the last day the cat was known to be alive was used). Univariate Cox regression analysis, followed by multivariate Cox forward regression analysis including all parameters with P <0.20 in univariate analysis, was performed to evaluate influence of different clinical and laboratory parameters on survival. Results were considered statistically significant if they had a P value <0.05.
Results
Study cohort
A total of 127 cats were enrolled. One cat, diagnosed with concurrent lymphoma during the study, received 12 weeks of treatment; as FIP was confirmed, data up to week 12 were included in the analysis.
Purebred cats made up 60.6% (77/127) of the study population. The most common breeds were British Shorthairs (24, 18.9%), Ragdolls (10, 7.9%) and Siberian Forest Cats (10, 7.9%). Other purebreeds included Maine Coons (n = 7), Norwegian Forest Cats (n = 5), Bengals (n = 4), Siamese (n = 4), British Longhairs (n = 3), Persians (n = 2) and one cat each of Birman, Devon Rex, Egyptian Mau, Exotic Shorthair, Kurilian Bobtail Longhair, Russian Blue, Scottish Straight and Sphynx. The remaining 39.4% (50/127) were non-purebred cats, comprising 42 (33.1%) domestic shorthairs and eight (6.3%) purebred crosses.
Most cats (87.4%) were aged under 2 years at intake (median age 13 months, range 3–150). Of the 127 cats, 98 (77.2%) were male (55.1% castrated, 22.0% intact) and 29 (22.8%) were female (17.3% spayed, 5.5% intact).
Cat origins were as follows: 46 (36.2%) were acquired from breeders, 27 (21.3%) from private sellers, 18 (14.2%) from shelters/rescues and 14 (11.0%) from other sources. The origin was unknown in 22 (17.3%) cats.
In 35 (27.6%) cases, the FIP-diagnosed cat was the only cat in the household. In 53 (41.7%) cases, one other cat was present; in 20 (15.7%) cases, two or more other cats were present; and in 19 (15.0%) cases, it was unknown.
Type of FIP
The type of FIP was documented for all 127 cats: 71 (55.9%) had effusive FIP, 25 (19.7%) had non-effusive FIP, 21 (16.5%) had ocular FIP and 10 (7.9%) had neurological FIP. Secondary FIP types were identified in 27 (21.3%) cats, most commonly secondary effusive FIP (16 cats, 12.6%). Four (3.1%) cats each had secondary neurological or non-effusive FIP. Three (2.4%) cats had secondary ocular FIP. Two cats developed a third FIP type: one neurological and one ocular.
Concurrent diseases
Concurrent disorders were identified in 14 (11%) cats. The most common comorbidities were immune-mediated haemolytic anaemia (IMHA) and pneumonia, each diagnosed in six (5%) cats. Two cats had both IMHA and pneumonia, and one cat with acute kidney injury (AKI) was also affected by pneumonia. Additional concurrent conditions included exudative polyarthritis, feline leukaemia virus (FeLV) infection, feline immunodeficiency virus (FIV), fibroadenomatous hyperplasia, malignant lymphoma and AKI, each observed in one cat.
Clinical signs
Clinical signs at intake were based on owner questionnaires, with complete data available for 123/127 cats. Most frequently reported signs were lethargy (114/123, 93%) and inappetence (102/123, 83%). Other common signs are listed in Table 1. Uncommon clinical signs included weight gain and polyuria (n = 11 each), abnormal gait (n = 5), increased vocalisation (n = 4), licking inanimate surfaces (n = 3), abnormal behaviour (n = 3), polyphagia (n = 2), excessive grooming/scratching (n = 2), increased salivation/swallowing (n = 1) and pica (n = 1).
Clinical signs observed in 123 cats with feline infectious peritonitis at intake, in order of prevalence

Data are n (%)
Physical examination findings
Findings from physical examinations conducted at intake by veterinarians at 1/3 veterinary hospitals were documented for all 127 cats. Mean body weight at the time of presentation was 3.25 kg (range 0.86–7.55). The most common physical abnormalities were pyrexia (76/127, 60%), tachycardia (61/127, 48%) and tachypnoea (39/127, 31%). A full list of physical findings is provided in Table 2.
Findings from physical examination of 127 cats with feline infectious peritonitis at intake, in order of prevalence

Data are n (%)
Clinicopathological findings
At intake, all 121 cats in which serum gamma globulin levels were measured exhibited hypergammaglobulinaemia. Other notable clinicopathological findings included hyperbilirubinaemia in 104/122 (85%) cats, lymphocytopenia in 97/123 (79%) cats and hypoalbuminaemia in 92/124 (74%) cats. These and other clinicopathological findings are summarised in Table 3.
Clinicopathological findings observed in 127 cats with feline infectious peritonitis at intake, in order of prevalence

Data are n (%)
A:G = albumin:globulin; ALT = alanine aminotransferase
FCoV RT-qPCR
RT-qPCR for FCoV was performed on effusion samples (thoracic or abdominal) from 76 cats at the VMDC and in an external laboratory for an additional three cats. Of the samples analysed at the VMDC, 72/76 (95%) tested positive and 4/76 (5%) tested negative. The mean (±SD) CP value for positive effusion samples was 26.7 ± 4.4.
RT-qPCR was also performed on FNAs in 43 cats at the VMDC and in one additional cat at an external laboratory. Of the VMDC-tested samples, 31/43 (72%) were positive and 12/43 (28%) were negative. The mean CP value for positive aspirate samples was 28.5 ± 5.8.
Immunohistochemistry for FCoV was performed in one cat, yielding a positive result. Consequently, no additional molecular testing was undertaken for this case. There was no RT-qPCR result for five cats, either because the sample was lost or because the fluid could not be aspirated because of uncooperative behaviour. Two cats had a positive RT-qPCR result of both effusion and FNAs.
Treatment
A total of 84 (66%) cats started with oral GS-441524 and 43 (34%) with IV remdesivir. The mean dose of oral GS-441524 was 12.9 mg/kg (range 10–22.8). The mean dose of IV remdesivir was 12.8 mg/kg (range 10–20). Of the 127 cats included in the study, 99 (78%) completed at least 12 weeks of treatment. Extended treatment was given in 31/99 cats (31%) because of insufficient reduction in gamma globulin levels (most cases) or inadequate clinical improvement. Treatment was prolonged by 1 week in one cat, 2 weeks in 12 cats, 4 weeks in 15 cats and 8 weeks in one cat. Two additional cats also received extended treatment, but the exact duration was not documented.
Outcome
Clinical signs
Lethargy and inappetence resolved within 2 weeks in most cats, except for five and seven cases, respectively. Concurrent improvements were noted in urinary, respiratory and ocular signs, as well as hypo- and polydipsia. Although 21% of cats still lost weight after 2 weeks, 66% had gained weight. Between week 4 and the final follow-up, 81–93% of cats continued to gain weight, depending on the time point. No differences in weight were observed between cats with effusive and non-effusive forms of FIP at any time. Abdominal distension, initially present in 58 cats, persisted in 10% at 2 weeks, three cats at 4 weeks and had resolved completely by week 16.
Vomiting and diarrhoea increased slightly at 2 weeks (vomiting: 20% to 22%; diarrhoea: 20% to 30%) before gradually declining. By week 8, vomiting occurred in 14% and diarrhoea in 13% of cats. These signs persisted in 7–14% (vomiting) and 5–11% (diarrhoea) at various time points and had resolved in all cats by their last follow-up. Respiratory and ocular signs were reported in 21% and 26% of cats, respectively, up to their final evaluations.
Clinicopathological findings
Median haematocrit at intake was 0.25 l/l (interquartile range [IQR] 0.21–0.3), increasing after treatment initiation and normalising within 2–4 weeks (Figure 2a).
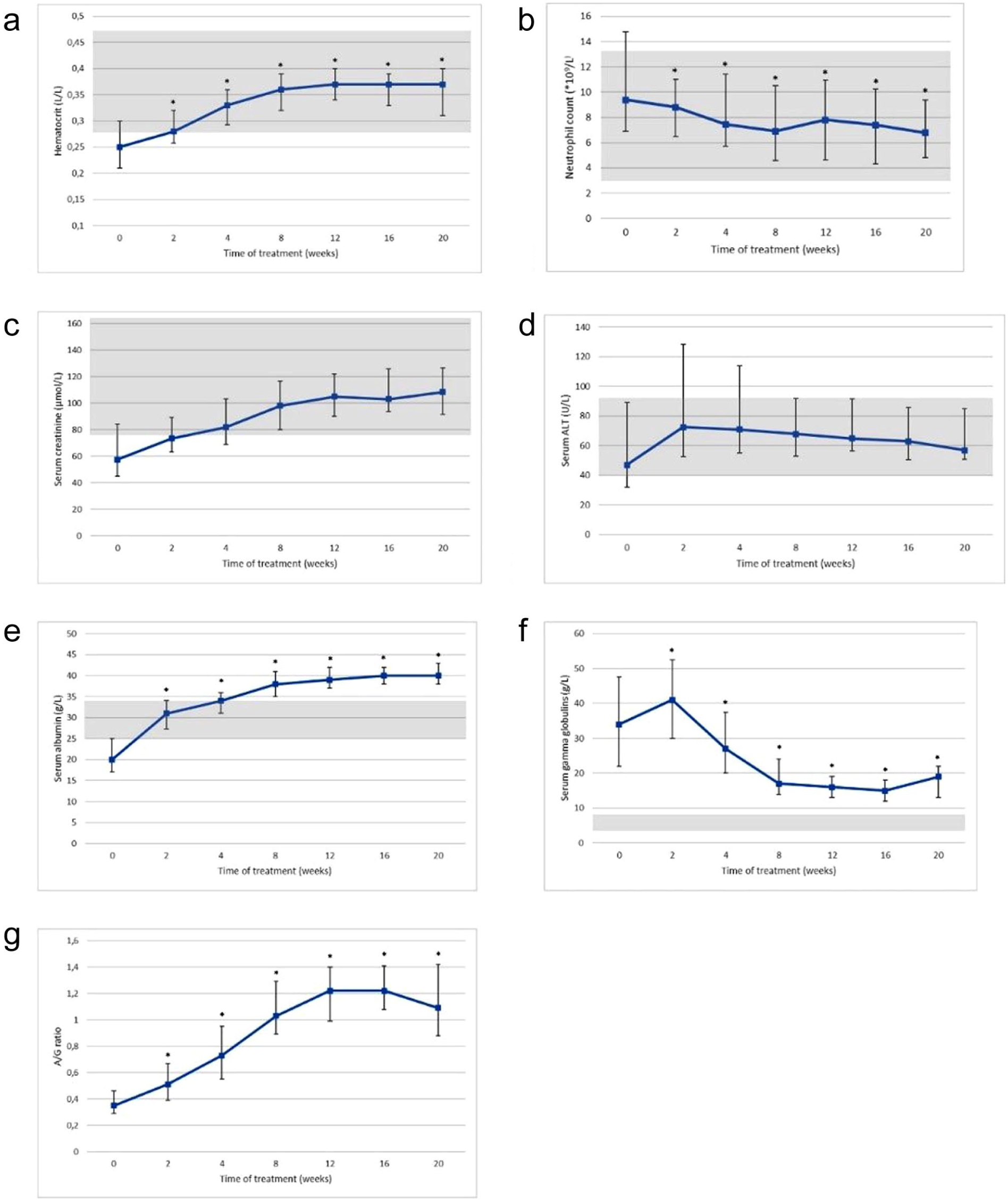
Median laboratory values and interquartile range (the lower error bar depicts Q1 and the upper error bar depicts Q3), for cats with feline infectious peritonitis during treatment with GS-441524 for (a) haematocrit, (b) lymphocyte count, (c) serum creatinine level, (d) serum alanine aminotransferase (ALT) level, (e) serum albumin level, (f) serum gamma globulin level and (g) serum albumin:globulin ratio. Significant differences (P ⩽0.05) between intake and respective checkups are depicted by asterisks (*). Grey areas indicate reference interval
Median lymphocyte count at intake was 1.3 × 109/l (IQR 0.6–1.9), rising to within the RI from week 2 onwards (Figure 2b). Median serum creatinine at intake was low at 58 µmol/l (IQR 45–84), increasing to within the RI by week 4, though 34% of cats still had low levels at that time (Figure 2c).
At intake, 46/123 (37%) cats had low and 25/124 (20%) had elevated ALT concentrations. After 2 weeks of treatment, low ALT concentrations decreased to 7%, while elevated ALT concentrations rose to 37%. Both abnormalities declined over time, reaching 2% and 16%, respectively, by week 16 (Figure 2d).
Serum albumin concentrations were low in 92/124 (74%) cats at baseline but increased rapidly after treatment initiation and normalised by week 8 (Figure 2e). Elevated albumin was rare at intake (2/124, 2%) but increased in 90/97 (93%) cats by week 16.
Median serum gamma globulin concentration at intake was 34 g/l (IQR 22–48), peaking at 41 g/l (IQR 30–53) after 2 weeks of treatment before declining again below the 20 g/l threshold by week 8 and remained low thereafter (Figure 2f).
The A:G ratio rose from a baseline median of 0.35 (IQR 0.29–0.46) to stabilise by week 16, with a slight decline noted at week 20 (Figure 2g). Gamma globulin concentrations were significantly higher in cats with effusive FIP compared with those with non-effusive forms at multiple time points. At baseline (T0), median levels were 33.2 g/l in effusive cases and 39.4 g/l in non-effusive cases, but the difference did not reach statistical significance (P = 0.060). From week 2 onwards, cats with effusive FIP consistently showed significantly higher gamma globulin concentrations: at week 2, median levels were 48.7 g/l vs 36.7 g/l (P <0.001), and at week 4, 34.2 g/l vs 26.0 g/l (P <0.001). The difference remained statistically significant at week 8 (22.2 g/l vs 18.1 g/l; P = 0.008) and week 12 (18.6 g/l vs 15.7 g/l; P = 0.037), but not at week 16 (16.8 g/l vs 15.1 g/l; P = 0.188), suggesting convergence of gamma globulin levels over time.
Survival
By the end of the study, 99/127 (78%) cats were alive. Most deaths (26/28, 93%) occurred within the first 8 days of treatment and were primarily attributed to advanced disease or severe comorbidities such as pneumonia or IMHA. One additional cat was euthanased on day 17 because of FeLV coinfection and progressive clinical deterioration, while another died on day 29 from an accidental injury unrelated to FIP. Excluding these two cases, the FIP-related survival rate rose to 80%. Notably, for cats surviving beyond the first 8 days of treatment, the calculated 1-year survival was 98% (Figure 3).

Kaplan–Meier survival curve for 127 cats with feline infectious peritonitis treated with nucleoside analogues GS-441524 with or without remdesivir. The vertical bars demonstrate censored observations
Cats with effusive FIP had a 70% survival rate, those with non-effusive FIP 84% and those with neurological FIP 60% at the end of the study. All cats with ocular FIP survived. Notably, ocular FIP was associated with a significantly higher chance of survival compared with the effusive form (hazard ratio [HR] 0.001, 95% confidence interval [CI] 0.000–0.010; P <0.001) (Figure 4).

Kaplan–Meier curves for 127 cats with FIP treated with nucleoside analogues GS-441524 with or without remdesivir for the different types of feline infectious peritonitis. The vertical bars demonstrate censored observations
Although British Shorthairs had a shorter mean survival time (180 days) compared with Ragdolls (267 days), domestic shorthairs (357 days) and Siberian Forest Cats (381 days) (Figure 5), the difference was not statistically significant (P = 0.19).

Kaplan–Meier curves for 127 cats with feline infectious peritonitis treated with nucleoside analogues GS-441524 with or without remdesivir for different breeds of cats. The vertical bars demonstrate censored observations
Univariate Cox regression analysis (Table 4) found no significant association between survival and age, sex, BCS or MCS, antiviral dose or treatment centre. Among categorical variables, ocular FIP was associated with an improved prognosis compared with the effusive form. Lower body temperature at presentation and lower thrombocyte counts, as well as elevated serum bile acids and bilirubin, were associated with reduced survival. Variables with a P value <0.20 were included in the multivariate regression analysis, which also accounted for potential confounding.
Results of univariate Cox proportional hazards regression analysis of clinical and laboratory variables predicting death in 127 cats treated with nucleoside analogues GS-441524 with or without remdesivir

A:G = albumin:globulin; ALT = alanine aminotransferase; BCS = body condition score; CI = confidence interval; FIP = feline infectious peritonitis; HR = hazard ratio; MCS = muscle condition score; T0 = baseline (presentation)
After multivariate analysis (Table 5), only bilirubin and creatinine remained significant predictors. Each 1 µmol/l increase in bilirubin raised the hazard of death by 2.9% (P <0.001), and each 1 µmol/l increase in creatinine by 0.3% (P = 0.022).
Results of multivariate Cox proportional hazards regression analysis of clinical and laboratory variables predicting death in 127 cats treated with nucleoside analogues GS-441524 with or without remdesivir

CI = confidence interval; HR = hazard ratio; T0 = baseline (presentation)
Post-treatment clinical concern
Post-treatment clinical concerns were reported in five (4%) cats between 21 and 112 days after completing GS-441524 treatment. A strict protocol for defining relapse was not established, as this was not a primary aim of the study. Relapse was virologically confirmed in only one cat, while in the remaining four, FCoV RT-qPCR was either negative or not performed, and alternative causes were considered more likely. One cat developed a decline in haematocrit and rising gamma globulin levels 47 days after treatment despite a stable clinical condition; no FCoV PCR was performed, and the cat was retreated for nearly 30 weeks, remaining in remission at last follow-up. Another cat presented with fever and nasal discharge 112 days after treatment completion; FCoV PCR was negative and no clinical improvement was observed during 11 days of GS retreatment. These signs were attributed to other causes, and the cat recovered without further therapy. A third cat showed pyrexia, recurrent anterior uveitis and increased gamma globulin levels 44 days after treatment; effusive FIP was confirmed by positive FCoV PCR on abdominal fluid, and GS-441524 treatment was restarted. After 4 weeks, the cat was lost to follow-up because of relocation. One cat failed to fully respond to the initial treatment course and developed thoracic effusion; blood FCoV PCR was negative but increasing the GS-441524 dose to 10 mg/kg q8h led to clinical improvement and resolution of effusion after a second 12-week course, with remission maintained at follow-up. The fifth cat developed lethargy and mildly increased gamma globulin levels 21 days after stopping GS; no FCoV PCR was performed. GS retreatment was initiated but discontinued after 4 weeks because of lack of clinical response. The cat recovered without further intervention and remained well at the time of writing.
Administration of medication
A total of 84 owners reported that administering GS-441524 was easy; eight reported managing daily administration, though it was not always easy. In 35 cases, cats died early or owners did not respond.
Adverse effects
Diarrhoea was reported in 28/98 (29%) cats during treatment, mostly within the first 2 weeks, and resolved in most by weeks 2–4; only five cats had diarrhoea at week 16. Vomiting occurred in 32/98 (33%) cats and followed a similar pattern, with only seven cats affected at week 16. Increased appetite was noted in 67/98 (68%) cats at the 2-week check-up. Other reported possible adverse effects included pruritus (n = 4), increased grooming or scratching (n = 9), lip-licking, salivation or swallowing (n = 3), inappropriate urination (n = 3), post-medication deep sleep (n = 2), alopecia (n = 2), flaky skin (n = 1), coat rippling (n = 1), ammonia-like urine odour (n = 1) and increased oestrus behaviour (n = 1).
No serious adverse effects, such as thrombophlebitis or anaphylaxis, were observed with remdesivir.
Discussion
This prospective multicentre clinical trial demonstrates that oral GS-441524 effectively improves clinical and clinicopathological parameters in cats with naturally occurring FIP, is well tolerated and can result in sustained clinical remission. The overall survival rate was 78%, which is consistent with or somewhat lower than other studies. Treatment success extended across diverse FIP presentations and included cats with comorbidities such as pneumonia or IMHA. The possible difference in survival rate likely reflects our inclusion of all cats regardless of disease severity, including neurological cases and those requiring IV treatment, which are often excluded from previous reports.2,3,16,21,22,27 Neurological FIP is associated with poorer outcomes and may have contributed to the lower survival rate. This is likely due to limited penetration of antiviral drugs across the blood-brain barrier, resulting in subtherapeutic concentrations in the central nervous system (CNS) and incomplete viral clearance. In addition, CNS inflammation and damage caused by the disease can be severe and often irreversible, contributing to poorer outcomes. Delayed diagnosis and treatment initiation, owing to the variable and sometimes subtle neurological signs, may also negatively impact prognosis.2,3,16 Practical factors such as travel requirements for treatment may have further impacted outcomes. Previous studies often included smaller cohorts and used subcutaneous GS-441524, which is linked to injection-site reactions and increased treatment-related stress.13,16,22,27
The study population reflected typical FIP demographics, with most cats being young, purebred males.4,28 –32 British Shorthairs were the most common purebred, aligning with a 2024 Dutch owner survey that shows this breed as the most popular purebred in the Netherlands, potentially explaining their over-representation. 33
Clinical signs such as lethargy and inappetence predominated, with improvement typically within 2–4 weeks.4,13,22 Pyrexia was present in 60% of cats at initial presentation, consistent with prior studies.10,13,14,29 Tachycardia (48%) and tachypnoea (31%) were more prevalent, possibly influenced by stress during handling. 34
All cats exhibited hypergammaglobulinaemia at diagnosis, differing from previous reports, possibly due to differences in case selection or diagnostic criteria.10,14,22,27,29 Cats with effusive FIP had significantly higher gamma globulin levels than those with non-effusive forms at multiple time points, suggesting a more pronounced inflammatory response in the effusive form. Gamma globulin concentrations increased further after 2 weeks of treatment before declining rapidly. This transient rise may reflect reabsorption and subsequent excretion of effusion fluid rich in gamma globulins, along with associated weight loss (22%), as previously described.16,22 Once effusions resolve, serum levels decrease as recovery progresses.
Serum ALT elevation occurred in 37% of cats after treatment initiation, consistent with feline and human remdesivir studies13,21,22,35 –37 Although the mechanism remains unclear, virus-induced inflammation, hypoxic injury or drug toxicity may contribute. ALT generally declined after treatment cessation but did not fully normalise in all cats by week 16.
Interestingly, 66% of cats presented with low serum creatinine concentrations. In dogs, this is typically associated with loss of muscle mass. 38 This may also explain the findings in this cohort, given that 76% of cats experienced weight loss likely accompanied by muscle wasting. Furthermore, creatinine RIs are determined for healthy, adult cats, while an important subset of cats in this study were kittens. 39 Although pairwise comparisons of creatinine concentrations between time points were not statistically significant, a significant overall time effect was observed. This may reflect gradual increases from a variable baseline, with high variability potentially reducing statistical power. The observed rise may indicate muscle mass recovery, though mild renal impairment – reported in some human remdesivir recipients – cannot be excluded.35 –37,40
Unexpectedly, 96% developed hyperalbuminaemia during treatment, a novel finding not previously reported. Hyperalbuminaemia is typically associated with dehydration or fluid loss, but this explanation seems unlikely for all affected cats. Further research is needed to understand the underlying mechanisms causing this side effect during treatment with GS-441524. One hypothesis is that albumin, a negative acute-phase protein, increases as acute inflammation resolves; a transient rebound effect may even result in hyperalbuminaemia.
Elevated bilirubin and creatinine concentrations were significant predictors of reduced survival in cats with FIP, in line with other studies. Hyperbilirubinaemia likely indicates hepatic involvement or cholestasis due to systemic inflammation, while increased creatinine reflects renal impairment or dehydration, both associated with poorer prognosis.16,19,21,41 Regular monitoring of these parameters during treatment may aid in identifying high-risk cats and optimising supportive care. Further studies are needed to clarify the underlying pathophysiology and improve outcome prediction.
The oral GS-441524 formulation was well tolerated and easier to administer compared to subcutaneous injections, facilitating at-home care – a key advantage as nearly 40% of owners find completing oral treatments challenging.42,43 No severe systemic adverse events were reported. Gastrointestinal signs, including diarrhoea (29%) and vomiting (33%), were common after initiation of GS-441524 treatment in our cohort, typically mild and resolving within 2–4 weeks. These findings are consistent with previous reports where gastrointestinal adverse effects occurred in approximately 2–20% of cats treated with GS-441524 or remdesivir.13,19,20 Pruritus was also reported in some cats (4–9%), which was similar to other studies where pruritus was found in less than 5% of cats.21,27 Distinguishing FIP-related signs from treatment-related adverse effects was challenging, as these signs were frequently present at intake. Persistent signs likely reflect underlying disease, whereas new or worsening signs during clinical improvement may represent treatment effects or other factors such as dietary changes. Polyphagia was frequently reported by owners during treatment, likely reflecting clinical recovery rather than a direct drug effect. This aligns with previous reports where increased appetite was commonly noted as cats improved clinically, although specific data on polyphagia are limited.13,19,27 Adjunctive prednisolone use in some cats may have further contributed to increased appetite. Hyperactivity was occasionally observed but generally considered a manifestation of improved health rather than a treatment-related side effect. Separate from behavioural changes, the adverse events described in this cohort overlapped with those seen in humans, including diarrhoea and a skin rash; however, acute hypersensitivity reactions commonly reported in human patients were not observed in these cats.35 –37,40
Possible relapse occurred in five (4%) cats, consistent with previous reports.13,16,22,27 Only one case was virologically confirmed and one cat recovered without retreatment, suggesting true relapse was unlikely in some instances. In strict clinical terms, relapse of FIP should ideally be defined by recurrence of clinical signs combined with virological or pathological confirmation of active FCoV infection (eg, positive FCoV PCR, immunostaining or consistent effusion/cytology) and/or a clear response to retreatment with GS441524. Therefore, if FCoV PCR is negative, and the cat either does not respond to GS441524 treatment or improves without retreatment, labelling the event as a confirmed relapse is questionable. In such cases, it is more appropriate to describe the situation as a suspected relapse, relapse-like signs or a non-FIP illness mimicking relapse, especially when other causes such as stress, coinfections or immune-mediated diseases are plausible. The underlying cause of these suspected relapses remains unclear but may involve incomplete viral clearance due to suboptimal dosing, high viral load or antiviral resistance.43,44 In addition, some cats were lost to follow-up after their final check, so we cannot exclude the possibility that further relapse occurred but was not reported by the owners. Further studies are warranted to clarify the nature of suspected relapses and to optimise treatment pharmacokinetics/pharmacodynamics and monitoring protocols.
Our study has several limitations. Not all cats had FCoV RNA detected in samples. The lack of a definitive (or very likely) diagnosis is a common challenge in FIP studies. This is because the ability to perform further diagnostics in a case can be difficult or not desirable because of financial constraints or the cat’s condition. A positive response to treatment in cases with suspected FIP is sometimes used as strong diagnostic support for FIP. Nonetheless, initiating antiviral therapy in the absence of a confirmed or very likely FIP diagnosis entails certain risks. These include the possibility of missing an alternative, potentially treatable condition, as well as the inappropriate use of antiviral agents, which may contribute to the emergence of antiviral resistance with broader One Health implications. Therefore, thorough discussions with owners are vital to ensure informed consent and appropriate case management.
Second, the study involved multiple treatment centres and clinicians, which introduced some variability in treatment decisions such as IV vs oral therapy initiation, hospitalisation and concurrent medications, despite a clear protocol.
No untreated control group was included, as withholding treatment from cats with FIP is considered unethical given the fatal prognosis without intervention.
Blood sample collection occasionally proved difficult, leading to missing data at certain time points. In addition, acute phase proteins such as alpha-1 acid glycoprotein, now commonly used to aid FIP diagnosis and monitoring, were not measured as these assays were unavailable at our laboratory during the study period.4,45–47
Conclusions
This prospective, multicentre study confirms that legally sourced, orally administered GS-441524, sometimes proceeded by IV remdesivir, is an effective and well-tolerated treatment for naturally occurring FIP in cats, achieving an overall survival rate of 78%. Clinical improvement typically occurs rapidly, with most signs resolving within 2–4 weeks, although some respiratory and ocular signs may persist for longer. Clinicopathological abnormalities often require a longer period to normalise. The treatment demonstrated efficacy across various FIP presentations, including effusive, non-effusive, ocular and neurological forms, and was successful even in cats with concurrent illnesses. Adverse effects were generally mild and transient, predominantly involving gastrointestinal signs, which resolved without interruption of therapy. Higher serum bilirubin and creatinine concentrations at presentation were associated with poorer prognosis, while ocular FIP was linked to a significantly better outcome. Post-treatment suspected relapses were uncommon, often lacked virological confirmation and some cases responded to retreatment while others resolved without further therapy. These findings support the broader clinical use of legally prescribed GS-441524 in managing FIP and highlight the need for continued research into treatment optimisation and long-term outcomes.
Supplemental Material
S1
Owner questionnaire intake.
Supplemental Material
S2
Consultation questionnaire.
Supplemental Material
S3
Physical examination.
Supplemental Material
S4
Email or phone questionnaire.
Footnotes
Acknowledgements
The authors would like to thank all the cat owners, veterinary nurses (especially Kimberley Sneepels, Leonie Habermehl, Cheryl Jansen, Leonie Smit and Anouk Arntzen), all the staff of the UVDL, VMDC and the Veterinary Pharmacy, and all the radiologists participating at this study.
Supplementary material
The following files are available as supplementary material:
S1: Owner questionnaire intake.
S2: Consultation questionnaire.
S3: Physical examination.
S4: Email or phone questionnaire.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognised high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers, tissues and samples) for all procedure(s) undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
