Abstract
Approximately 10% of cats with diabetes mellitus (diabetes) are euthanased at diagnosis and a further 10% euthanased within the first year of treatment, despite diabetes being a treatable disease. This review presents a spectrum of veterinary care aimed at providing practitioners with a range of treatment options to discuss with owners, potentially helping to prevent euthanasia at the time of diagnosis. Barriers such as owner concerns about lifestyle impact, pet welfare and presence of comorbidities contribute to decisions to euthanase, while financial limitations can significantly restrict access to veterinary care for cats with diabetes. This highlights the urgent need to implement a spectrum of care approach for feline diabetes with the aim of reducing euthanasia rates and improving owner and cat outcomes.
Plain language summary
Diabetes mellitus is a common and increasingly diagnosed disease in cats. Unfortunately, about one in 10 cats are euthanased at the time of diagnosis, even though diabetes is a treatable condition that can be managed. Decisions about euthanasia are often influenced by both cat-related factors (such as age, temperament or concurrent diseases) and owner-related factors (including time and financial demands, lifestyle impacts, pet welfare, fear of hypoglycaemia and giving injections, and challenges with boarding or travel). These factors reflect the significant caregiver burden that can accompany management of a diabetic cat. To support both cats and their owners, veterinarians should focus on clear and thorough owner discussions and developing individualised and flexible treatment and monitoring plans tailored to each cat’s needs and the owner’s circumstances. By applying a ‘spectrum of care’ approach – offering a range of practical and affordable options – veterinarians can make diabetes care more accessible and sustainable for all clients. The ultimate goals in managing feline diabetes are to reduce euthanasia rates, empower owners through education and shared decision-making, and improve quality of life for both cats and their caregivers without compromising care. The primary costs of diabetes management arise from treatment, monitoring, diet and veterinary care. Substituting a more affordable option in any of these areas can reduce overall expenses generally without impacting care. By being aware of the available options and their associated costs, veterinarians can offer flexible, individualised and informed treatment plans that address financial and practical barriers, ultimately reducing treatment costs and enhancing prognosis, owner satisfaction and feline wellbeing.
Keywords
Introduction
Prevalence and causes of feline diabetes mellitus
Diabetes mellitus (diabetes) is the second most common endocrinopathy reported in cats.1 –5 Diabetes is not a single disease with the same pathogenesis in all cats; rather, diabetes is heterogenous, and pathogenesis can vary between individual cats. The majority of cats have clinical signs and islet histology analogous to advanced type 2 diabetes mellitus in humans.6,7 In some cats, diabetes is attributed to other specific types of diabetes causing loss of beta cells secondary to pancreatitis, or secondary to endocrinopathies that result in marked insulin resistance, such as hypersomatotropism (acromegaly) and hypercortisolism.8,9 Diabetes is characterised by persistent hyperglycaemia; this requires some degree of failure of beta cells to secrete sufficient insulin to maintain normoglycaemia. 10
Euthanasia risk
There is a substantial risk of euthanasia when a cat is diagnosed with diabetes, with 10% reported to be euthanased at the time of diagnosis and a further 10% euthanased within the first 12 months of treatment. 11 Although the most common reason for euthanasia is presence of a concurrent disease,11,12 there are many other owner-related reasons which, alone or together with the presence of concurrent disease, lead to a decision to euthanase. These include the cost of treatment and monitoring, concerns about pet welfare, worry about hypoglycaemia, impact on owner’s lifestyle, giving injections, and time and cost demands for regular veterinary visits. 13 Pet-related factors associated with an increased risk of euthanasia are the presence of concurrent disease and older age of the cat. 11 After treatment implementation, emergence of other comorbidities, such as neoplasia or chronic kidney disease, is the most common reason for euthanasia and reflects the senior age of diabetic cats at diagnosis.11,12 Other reported factors influencing the decision to euthanase include lack of response or compliance, clinical hypoglycaemia, expectations from others to euthanase the cat, emergency costs for diabetic ketoacidosis (DKA), poor veterinary support and impacts on the owner’s quality of life.11,14
Spectrum of care in feline diabetes mellitus
Spectrum of care is a flexible approach to diabetes where veterinarians can offer a range of treatment, monitoring, diet and veterinary care options that are practical and achievable for the client without compromising care. There is no single ‘gold standard’ or best approach to diabetes but rather a continuum of evidence-based options allowing broader access to veterinary care. Costs of managing diabetes primarily arise from drug therapy (insulin or sodium-glucose cotransporter-2 [SGLT2] inhibitor drugs), monitoring tools, diet and veterinary care. Selecting a more affordable option in one or more aspects of care can dramatically reduce overall costs with minimal impact on quality of care.
To optimise quality of life and survival, the veterinarian should engage in sympathetic discussions regarding the client’s budget and commitment to treatment of their cat. Cost estimates should be provided to the client as well as options for monitoring and drug administration that are aligned with the client’s circumstances, considering the demand on the client’s time and availability. It is crucial to allow the client to ultimately make the choice on a level of care, provided the welfare of the cat is acceptable. Ideally, information on the impact of these choices on remission, survival and quality of life for the owner and cat should also be provided. However, robust data are not yet available to provide this with confidence. Clinicians should be prepared to offer flexible, practical and individualised options. In many circumstances, treatment and monitoring protocols can be tailored to benefit the owners with little-to-no negative impact on health outcomes for the cat. There is no single, uniform and ‘correct’ approach to managing feline diabetes.
Diagnosis of diabetes and prediabetes and implications for treatment
In humans, prediabetes is considered an intermediate stage between normal glycaemia and diabetes, in terms of diagnostic criteria, and as a precursor to diabetes in terms of metabolic risk. Although some measures of dysglycaemia have been identified in non-diabetic cats,15 –17 there are no established criteria for diagnosis of prediabetes as a precursor to diabetes. Although there are no clinical data yet, considering the similarities between human and feline diabetes, it is likely that in cats, as in people, interventions that reverse mild dysglycaemia (even before it meets the criteria for diagnosis of diabetes) might delay or prevent the onset of clinical diabetes. This is especially relevant to addressing predisposing lifestyle factors such as obesity. Minimising load on beta cells to secrete insulin by reducing dietary carbohydrate is also likely advantageous, although long-term studies are needed to establish this recommendation.18 –21
The upper limit of the 95% reference interval (RI) for blood glucose concentration in fasted, non-stressed cats is approximately 6.3–6.5 mmol/l (113–117 mg/dl).15,22,23 However, these cutpoints are affected by methodology, including type of glucose meter, method of blood collection, time interval from collection to measurement and anticoagulant used. 22 Assuming stress hyperglycaemia has been ruled out, persistent, fasting blood glucose concentrations above this should be considered prediabetic, subclinical diabetic or diabetic, and each has different management considerations.15,23,24 The Agreeing Language in Veterinary Endocrinology (ALIVE) project has defined the diagnosis of diabetes as a random fasted or unfasted blood glucose concentration of 15 mmol/l (270 mg/dl) or higher associated with a hyperglycaemic crisis (either DKA or hyperosmolar hyperglycaemic state) or classic clinical signs together with at least one additional criteria of increased glycated proteins or repeatable urinary glucose in a non-stressed home environment. 25 If persistent blood glucose concentrations are greater than 7 mmol/l (>126 mg/dl) but less than 15 mmol/l (<270 mg/dl), at least two of the three criteria are required: classic clinical signs, increased glycated proteins or repeatable glucosuria in a non-stressed home environment (Figure 1). 25 Subclinical diabetes is defined as meeting the ALIVE criteria for diabetes without the classic clinical signs of diabetes. Based on the ALIVE criteria, a diabetic cat with subclinical diabetes would, at a minimum, need to exhibit glycosuria on more than one occasion, unrelated to stress, if no classical clinical signs were present. However, because the renal threshold for glucose is approximately 14–16 mmol/l (250–290 mg/dl),26,27 some cats with persistently elevated glucose concentrations below 15 mmol/l (270 mg/dl) may not meet the ALIVE criterial for a diabetes diagnosis, although many would likely still exhibit at least subtle clinical signs and glucosuria.

Criteria for diagnosing feline diabetes mellitus according to Agreeing Language in Veterinary Endocrinology (ALIVE) consensus. BG = blood glucose
The precautionary principle suggests that if there is a possibility of significant harm, and scientific evidence is not definitive, then action should be taken to prevent or minimise that harm rather than waiting for conclusive proof. 28 Although data in cats are limited regarding clinically relevant cutpoints, assuming stress hyperglycaemia has been ruled out, the authors recommend, based on the veterinary literature and human medicine cutpoints, that a blood glucose concentration of approximately 9–10 mmol/l (160–180 mg/dl) be considered the cutpoint for feline diabetes,22,28 –30 while concentrations exceeding 15 mmol/l (270 mg/dl) and meeting the ALIVE criteria are consistent with clinical diabetes. 25 The authors propose that cats with a persistent fasting glucose concentration of 7–<10 mmol/l (126–<180 mg/dl) be considered prediabetic15,22,23 and 10–15 mmol/l (180–<270 mg/dl) be considered subclinical diabetic. 25 Highlighting the importance of persistent dysglycaemia as a risk factor for clinical diabetes, cats in diabetic remission with moderately impaired fasting glucose (⩾7.5–9 mmol/l [⩾135–162 mg/dl]) and/or moderately impaired intravenous (IV) glucose tolerance (>14 mmol/l [>252 mg/dl] 3 h after glucose 1 g/kg IV) had a high risk of relapse within 9 months of testing. 23 In contrast, no cat in remission with normal fasting glucose and glucose tolerance relapsed within the 18 months of follow-up. Hence, dysglycaemic cats in diabetic remission should be managed similarly to prediabetic cats. 23
In cats, stress hyperglycaemia is a major confounding factor when attempting to diagnose prediabetes, subclinical diabetes and diabetes using a random blood glucose concentration (measured at any time of the day and in relation to eating). Stress cannot be ruled out as a cause of hyperglycaemia based on the magnitude of the elevation.24,31 Blood glucose concentrations exceeding the renal threshold (15 mmol/l [270 mg/dl]) and resulting in glycosuria24,32 are not uncommon in stressed cats, 15 with blood glucose concentrations exceeding 55 mmol/l (1000 mg/dl) reported. 24 However, mild elevation in blood glucose cannot be discounted as stress-related just because it is mild without further diagnostic testing, as it could also represent prediabetes or subclinical diabetes, especially if other risk factors are present – for example, age older than 8 years, obesity or concurrent glucocorticoid administration.
Cats meeting the ALIVE criteria for a diagnosis of diabetes should be treated without delay. In the authors’ opinion, provided appropriate monitoring is in place, the risk of life-threatening complications after implementation of therapy is lower than the risk of life-threatening complications associated with not treating a diabetic cat. If the ALIVE criteria are not met, either persistent hyperglycaemia should be documented in the home environment using continuous glucose monitoring (CGM) or glucometer-measured blood glucose, glycosuria documented using urine dipsticks (or other methods for identifying glucosuria in cat litter) or at least two additional criteria to demonstrate reasonable confidence that the hyperglycaemia is not solely stress-induced. The authors recommend using increased glycated haemoglobin (Hb) A1c (>3.6%, RI 1.9–3.1, Baycom assay), 33 increased beta hydroxybutyrate (BHB) (>0.18 mmol/l [>1.9 mg/dl], Precision Xtra Ketone Meter; Abbott) 34 or increased fructosamine. Notably, if fructosamine is within the RI, it does not rule out prediabetes or subclinical diabetes.33,35 It is also important to take into consideration that serum BHB can be elevated in sick cats with diseases other than diabetes, and RIs and cutpoint concentrations for diabetes are dependent on the methodology used for measurement.36,37
The earlier treatment is implemented in the course of the disease process – for example, when blood glucose is between 7 mmol/l (126 mg/dl) and less than 15 mmol/l (<270 mg/dl)) – the less intensive the treatment required to control blood glucose, and potentially the better the outcome in terms of remission and therefore survival. The degree of hyperglycaemia at diagnosis is not predictive of remission; 12 however, after 17 days of treatment, cats with lower mean daily blood glucose concentrations (<16 mmol/l [<288 mg/dl]) were more likely to achieve remission. 38 At higher blood glucose concentrations – for example, 20 mmol/l (360 mg/dl) or higher – there is likely minimal insulin secretion, due either to suppression of insulin secretion and/or loss of beta cells, which cannot be differentiated at diagnosis. 39
Goals of treatment
There is no single, uniform and ‘correct’ approach to managing feline diabetes. As veterinarians, setting realistic expectations for treatment goals for clients is important for a successful outcome. Keep in mind that pet owners’ expectations are often set by the veterinarian’s expectations, but also by their prior lay knowledge of the disease and its impact. Often, owners’ prior knowledge is relevant to diabetes in humans and not to their pets. The long-term consequences of diabetes in humans and pets are very different. The primary goals of therapy in diabetic cats are as follows: (1) improvement in pet’s and owner’s quality of life; (2) improvement in clinical signs; (3) prevention of DKA and clinical hypoglycaemia and minimisation of other complications; (4) normalisation of body condition score; and (5) diabetic remission, when appropriate for the cat and client (Figure 2). Whether remission is a therapeutic goal for an individual cat will require consideration of all the factors and needs to be discussed with the owner.

Realistic expectations for therapy goals in feline diabetes mellitus. Adaptation from Agreeing Language in Veterinary Endocrinology (ALIVE) consensus on treatment goals of feline diabetes mellitus.25 DKA = diabetic ketoacidosis
Remission
Achieving remission might be a reasonable goal for many newly diagnosed cats, considering that it is associated with increased survival,12,40 decreased overall expenses and better quality of life; however, this should be discussed with the owner and considered in the context of multiple factors. 14 Factors associated with a higher probability of remission include early implementation of tight glycaemic control,41,42 onset of diabetes after steroid administration,12,40 breed (Norwegian Forest Cat and European-type Burmese),12,14 low carbohydrate diet 21 and effective treatment of underlying cause (eg, cats treated surgically for hypersomatotropism). 43 Cats that have been diagnosed longer than 6 months and/or have other comorbidities, such as peripheral neuropathy or hypercholesterolemia, have a significantly lower probability of achieving remission.44,45 Before the advent of SGLT2 inhibitors, tight glycaemic control required intensive insulin treatment and monitoring. However, the intensity of treatment and monitoring that might be required to achieve remission might not be an appropriate choice for some owners. Interestingly, although the evidence is scarce, the incidence of clinical hypoglycaemia is low in cats treated with long-acting insulin such as glargine U-100 using intensive, euglycaemia or remission-oriented protocols and may not differ from incidence when using less intensive protocols.41,44 However, these protocols are more labour intensive, associated with more biochemical hypoglycaemia and not practical for all clients. Remission can occur in cats treated with less intensive protocols;12,40 it is therefore important that the client makes that choice in an informed manner, aided by scientific evidence provided by the veterinarian. In rats, the rapid glucose-lowering effect of SGLT2 inhibitors reduces glucotoxicity. 46 Given their similar effectiveness in lowering blood glucose in cats, SGLT2 inhibitors may likewise reduce glucotoxicity, supporting beta cell recovery and possibly increasing the chance of remission. However, there are currently no data on the probability of achieving remission using SGLT2 inhibitors.
Spectrum of care and factors that influence therapeutic approach
Spectrum of diabetes in cats influences therapeutic approach
Diabetes is a heterogenous disease, with variation in both duration and severity among cats, the latter largely determined by the relative contribution of beta-cell dysfunction and insulin resistance. The pathogenesis of diabetes exists along a spectrum ranging from predominant insulin resistance to predominantly beta-cell dysfunction and anywhere in between. Individual cats may fall anywhere along this continuum, and their position can shift with time as diabetes advances or contributing factors resolve. Cats with diabetes and hypersomatotropism represent one end of the spectrum of severe insulin resistance, with minimal or reversible beta-cell dysfunction, as shown by high remission rates after hypophysectomy. In other cats, beta-cell dysfunction – resulting in inadequate insulin secretion due to the suppressive effects of high glucose concentrations, combined with beta-cell death (both influenced by timing of diagnosis) – may be the predominant feature.7,39 Specific types of diabetes associated with diseases such as hypersomatotropism, diabetogenic drugs, hyperadrenocorticism or pancreatitis affect a subset of cats and influence treatment recommendations, owner choices and prognosis. 23
Spectrum of clinical presentation influences therapeutic options and goals for glycaemic control
Although there is no prospective high-level evidence that setting a specific glycaemic goal is correlated with a specific treatment outcome, 47 it is well documented that persistently high glucose concentrations supress insulin secretion to the point of marked ketonaemia, and protocols aiming for near-normal blood glucose concentrations are associated with higher remission rates, 42 particularly in newly diagnosed diabetic cats. Glycaemic control must always be assessed in light of clinical signs. However, several factors can influence decisions around the appropriate level of glycaemic control for diabetic cats, particularly when using insulin (Figure 3). In newly diagnosed cases, where the likelihood of remission is higher, tighter control (70–150 mg/dl) may be the goal. In contrast, cats with comorbidities causing inappetence and vomiting may be at greater risk for hypoglycaemia, making moderate control (150–400 mg/dl) more appropriate. For cats with limited life expectancy or a history of recurrent hypoglycaemia, moderate control may offer a balance between minimising clinical signs and avoiding risks. Similarly, moderate control is recommended for cats with hypersomatotropism, as they are at risk of hypoglycaemia and have little to no chance of achieving remission without surgery or radiation. Clinically well cats without ketonuria may be candidates for an SGLT2 inhibitor (Table 1).48 –50 All these factors need to be weighed and used as guidelines to determine a suitable individualised therapeutic regimen that will achieve an appropriate level of glycaemic control tailored for the client’s circumstances and their cat.

Glycaemic targets with therapy based on clinical presentation. For cats with diabetes treated with insulin, glycaemic targets should be based on disease features and patient characteristics. Therapy goals may vary between cats, with a focus of excellent glycaemic control in some cats vs moderate glycaemic control in others. DKA = diabetic ketoacidosis
Factors guiding the initial choice of monotherapy for feline diabetes; no single factor should be used in isolation to decide on the most appropriate therapy

IGF = insulin-like growth factor; N = no; RI = reference interval; SGLT2 = sodium-glucose cotransporter 2; Y = yes
Spectrum of owner characteristics influences therapeutic options for cats with diabetes mellitus
A crucial determinant of appropriate therapy for diabetes in an individual cat is the owner. A thorough discussion with the owner is critical to identify their goals and capabilities, and to clarify to the owners the veterinary perspective on diabetes care (which often differs from the perspective on human diabetes). The owner’s main objectives, financial capacity, tolerance for risk and complications, expectations and technical abilities, and time limitations should be considered in tailoring treatment for that particular owner and cat (Figure 4). Some owners choose a less intensive treatment approach (in terms of glycaemic control) because that requires less monitoring. The monitoring protocol for SGLT2 inhibitors is generally less demanding in the long term than insulin-monitoring protocols and may be a more suitable option for some owners. Other owners wishing to increase the probability of remission with insulin therapy may opt for normal or near-normal blood glucose concentrations and more intensive monitoring.
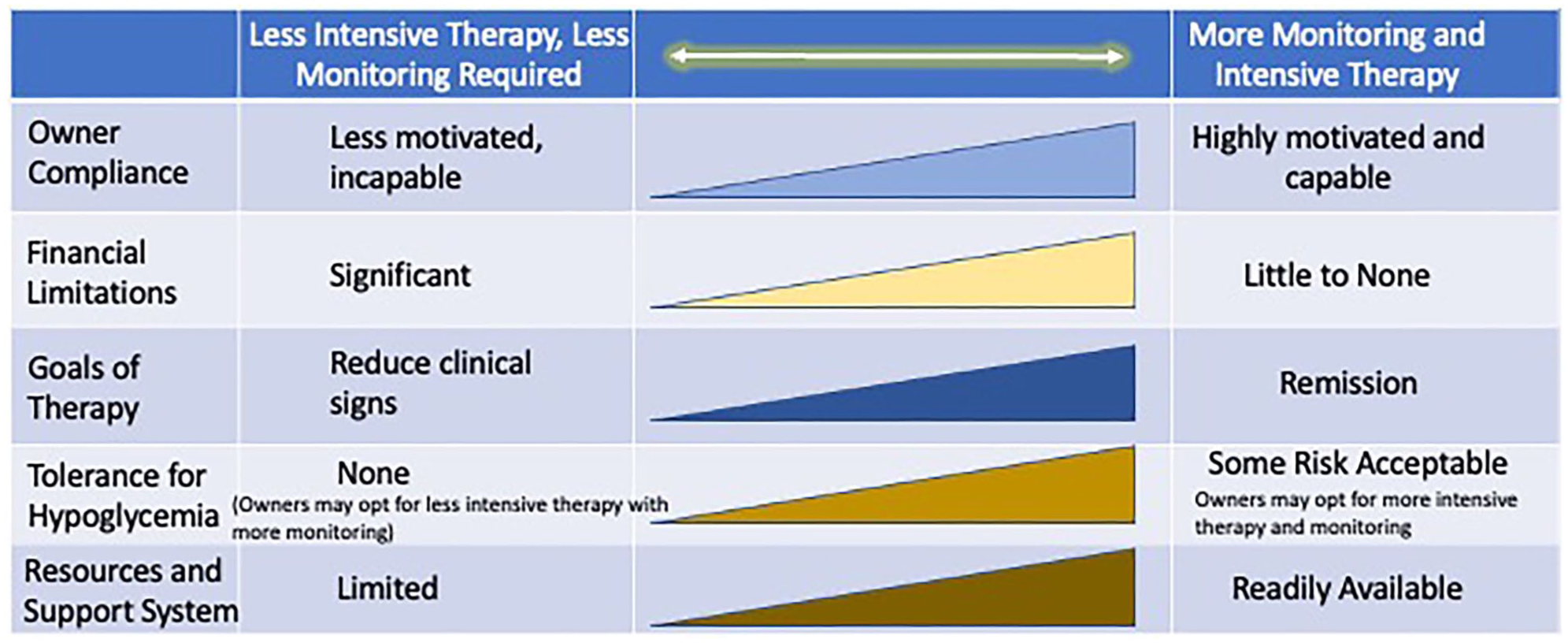
Spectrum of owner characteristics for treating cats with diabetes mellitus. In developing a treatment protocol for ‘best’ compliance in cats with diabetes mellitus, it is critical to involve the owner and design the protocol to match their goals, capabilities and limitations
The owner’s technical ability to handle subcutaneous injections, and their willingness and availability to give those injections in the short and long term, might affect the choice of insulin formulation and delivery, eg, injection pens vs a syringe and needle, or whether insulin is used at all (eg, alternatively SGLT2 inhibitors). Injection pens are more accurate in the units of insulin delivered and are often easier for the client to use; however, they typically restrict dosing changes to 1 unit when using formulations that are labelled for human use. Smaller adjustments (0.5 units) are possible with pens designed for insulin formulations registered for veterinary use. Taking all these factors into consideration, a personalised therapeutic and monitoring approach can be created to ‘best fit’ an individual cat and owner.
Approaches to therapy
There are three main treatment strategies that may or may not be appropriate for the cat, depending on whether sufficient endogenous insulin concentrations are present to prevent ketoacidosis and owner preference (Table 1):
Low carbohydrate diet (<2 g/100 kcal carbohydrate [CHO]; 12 canned) and normalisation of body condition for cats with prediabetes or subclinical diabetes. If dysglycaemia does not respond to a low carbohydrate diet, consider trialling a glucagon-like peptide 1 (GLP-1) receptor agonist (currently off-label use).
SGLT2 inhibitors ± low carbohydrate diet (<2 g/100 kcal CHO; canned), and normalisation of body condition (Table 1) for cats with subclinical and clinical diabetes.
Low carbohydrate diet (<2 g/100 kcal CHO; canned) and normalisation of body condition plus insulin (glargine 300 U/l, glargine 100 U/ml, protamine zinc insulin [PZI] or porcine lente) (Table 1) for cats with clinical diabetes.
As experience with SGLT2 inhibitors grows, if closely monitored, combined insulin and SGLT2 inhibitor protocols may be appropriate for some cats, although this currently remains off-label. 51
Drug therapy
Many factors should be considered to guide therapy in cats with diabetes, such as clinical presentation, concurrent illnesses and laboratory abnormalities (Table 1). The cost of therapy may also dictate available therapies for each individual owner and cat.
In prediabetic cats (suggested definition glucose 7–<10 mmol/l [126–<180 mg/dl]), where dysglycaemia does not respond to a low carbohydrate diet, trialling once-weekly GLP-1 receptor agonists can be considered. Currently, there is no evidence supporting the use of GLP-1 agonists as monotherapy in cats with prediabetes, although they have been used off-label in obese and diabetic cats.52 –56 In cats, GLP-1 agonists increase insulin secretion and may assist with weight loss, effects that are also well established in humans.47,57
Therapy for cats with diabetes involves insulin or an SGLT2 inhibitor. Clinically well diabetic cats without ketonuria may be candidates for SGLT2 inhibitors (Table 1). 48 In most cats, these drugs rapidly normalise blood glucose over 1–7 days, with low glycaemic variability and minimal risk of hypoglycaemia. A recent study compared cats, including previously insulin-treated and naive diabetic cats, that were randomised to receive treatment with porcine lente insulin or velagliflozin. Velagliflozin was not inferior to insulin, with an overall treatment success rate of 54% compared with 42% in insulin-treated cats. 50 There was no difference in improvement of polyuria/polydipsia and polyphagia, but mean and minimum blood glucose concentrations were lower for the velaglifloxin group at all time points (7, 21, 45 and 91 days), but were only statistically different under 91 days, 50 whereas fructosamine concentration was significantly lower at all time points for cats treated with velaglifloxin. Of the cats treated with porcine lente insulin, 7.6% developed clinical hypoglycaemia and 7% of velagliflozin-treated cats developed DKA. 50 In the pivotal bexagliflozin study, 5% of cats developed DKA, but no cases of clinical hypoglycaemia were reported. 48 Although uncommon, SGLT2 inhibitors can cause DKA, which is typically characterised by glucose concentrations below 13.9 mmol/l (250 mg/dl) along with acidosis and ketonaemia, with or without ketonuria, termed euglycemic ketoacidosis (eDKA). This may develop rapidly and occurs most frequently within the first 2 weeks of initiating therapy. Development of eDKA with an SGLT2 inhibitor indicates that the cat has insufficient endogenous insulin to suppress ketone formation and needs exogenous insulin. Therefore, monitoring is required, including at-home monitoring for ketonaemia or ketonuria, especially in the first 14 days, to facilitate early institution of insulin (and glucose) therapy and prevent severe ketoacidosis. Where insulin or an SGLT2 inhibitor alone is insufficient to control hyperglycaemia, a carefully monitored combination of both may be considered, although this remains off-label use. SGLT2 inhibitors have also been used successfully in poorly controlled insulin-treated diabetic cats. 51
In diabetic cats with hypersomatotropism due to profound insulin resistance, achieving glycaemic control with insulin alone is often challenging. When surgery or radiation is not feasible, treatment with an SGLT2 inhibitor may be considered as an alternative to further increasing the insulin dose to improve glycaemic control, although this remains off-label use. A recent clinical trial in eight diabetic cats with hypersomatotropism showed improved glycaemic control with velagliflozin, either as monotherapy or in combination with insulin. 58
Insulin therapy is indicated for cats with DKA, reduced appetite, lethargy or other signs of systemic illness. The presence of ketonuria or moderate to marked ketonaemia may indicate inadequate endogenous insulin to suppress ketone production and the need for exogenous insulin; however, these findings should always be interpreted in the context of the overall clinical picture. In contrast, for clinically stable diabetic cats with a good appetite, SGLT2 inhibitors may be a reasonable option when combined with appropriate monitoring of response to therapy, particularly in the first 2 weeks.
Considerations to reduce costs and drug availabilities
Multiple options are available for reducing the cost of treatment (Table 2). There is currently no strong evidence to suggest that one insulin formulation is superior to any other in terms of long-term outcomes because many additional factors influence outcome. However, some pharmacological characteristics of specific formulations might help the veterinarian prioritise insulin choices. Insulin solutions (eg, glargine U100 and even more so, glargine U300) are associated with decreased inter- and intra-day glucose variability, and therefore could ease the burden of monitoring and decrease the risk of clinical hypoglycaemia. 59 For some clients whose cats require insulin, price and convenience may be more important than insulin type when choosing between different formulations. Furthermore, specific insulin types might be available at different price ranges. For example, insulin glargine U100 as Lantus may be more expensive than the generic version or one of the biosimilars (Semglee, Basaglar). The same insulin type can be prescribed in a pen or a syringe/needle form, with some pens being more expensive than their syringe counterparts and vice versa.
Comparison of approximate costs of a similar insulin dose of different insulins for feline diabetes mellitus in US (US$) and Australian (AU$) dollars (cost available online as of 23 August 2025)

P = pen; PZI = protamine zinc insulin; U = unit; V = vial
Geographic location affects costs of drugs and monitoring, as well as availability and regulatory approval of some therapies (Table 2). For example, insulin formulations other than those approved for veterinary use are substantially cheaper in Australia compared with the USA (Table 2). In some countries, as in the UK, the first choice of insulin for cats with diabetes must be a formulation approved for veterinary use. In these countries, depending on cost and availability, PZI (Prozinc) would be the first choice and lente (Vetsulin/Caninsulin) the second choice. 38 However, in most countries, any insulin can be chosen at the veterinarian’s discretion. At the time of publication, detemir is no longer available, and inhibitors labelled for veterinary use (Bexacat and Senvelgo) are approved in the USA and Senvelgo is approved in parts of Europe, Canada, Australia and the UK. In countries where a veterinary SGLT2 inhibitor is not yet approved, other SGLT2 inhibitors approved for human use are widely available. For example, at least one SGLT2 inhibitor (dapaglifozin 10 mg tablets) has been tested in healthy and diabetic cats, with efficacy reported for doses of 2.5–10 mg per day in individual diabetic cats.60,61 Veterinarians should be aware of costs and availability of different therapies for cats with diabetes in their country, and these should be discussed with the client. For example, depending on the geographical location, biosimilar or generic glargine may be a cheaper option than an SGLT2 inhibitor, but the ease of oral administration and less intense long-term monitoring using an SGLT2 inhibitor compared with twice daily subcutaneous insulin injections may be more important than cost for some clients.
Diet
Low carbohydrate diets are important in reducing the glycaemic load on the beta cells to produce insulin, therefore increasing the probability of remission. 21 Weight loss reduces insulin resistance, and therefore body condition should be normalised. 17 However, depending on the level of overweight or obesity, normalisation of body weight can take 6–12 months to achieve at a safe weight loss rate of 1–2% of body weight per week, 62 but has a high failure rate. Based on the authors’ experience, frequent monitoring of body weight – for example, every 2 weeks – and appropriate adjustment of energy fed is successful. The addition of a GLP-1 receptor agonist (off-label) could also be considered.
Prescription diets for cats with diabetes may be too expensive or not palatable to some cats and may not have the lowest carbohydrate content (ie, <2 g/100 kcal). An alternative is to use an over-the-counter canned diet with low carbohydrate content, resulting in substantial savings. In one study, diabetic cats fed commercial wet food (low carbohydrate) diets were three times more likely to achieve diabetic remission than those fed veterinary prescription diets (wet and/or dry). 14 Homemade diets formulated by a board-certified veterinary nutritionist may be preferred by some owners who do not want to feed a commercial diet. (Table 3). In humans, low carbohydrate diets are known to increase the risk of eDKA when used with SGLT2 inhibitors. 63 However, studies are needed to determine whether the same risk applies to cats, as their carbohydrate metabolism differs fundamentally from that of humans. As obligate carnivores, cats have a limited capacity to utilise dietary carbohydrates and rely heavily on gluconeogenesis to maintain glucose homeostasis. Despite the theoretical risk, some veterinarians do recommend the combination of low carbohydrate diets and SGLT2 inhibitors in cats, based on the known glycaemic benefits of carbohydrate restriction and the species-specific differences in metabolism. If combining a low-carbohydrate diet with SGLT2 inhibitor therapy, the SGLT2 inhibitor should be started first for at least 2 weeks, then the diet introduced. This stepwise strategy helps determine whether any gastrointestinal signs are drug- or diet-related.
Comparisons of approximate costs of prescription and over-the-counter low carbohydrate (CHO) diets to feed a 3.6 kg cat in US (US$) and Australian (AU$) dollars (cost available online as of 23 August 2025)

Costs and approaches to monitoring
There are many options for monitoring cats with diabetes (Table 4). The most appropriate approach depends on multiple factors, including the treatment selected, stage of disease (eg, DKA, clinical or subclinical diabetes), the individual cat’s characteristics, cost considerations and owner preferences. One simple and free tool is the ALIVE Diabetic Clinical Score (DCS), a standardised scoring system that assesses glycaemic control based on owner-reported signs: polyuria/polydipsia, weight loss, activity level and appetite. 25 A higher DCS may indicate the need to increase insulin, while a low score may suggest a dose reduction is appropriate.
Comparison of costs of different approaches to monitoring feline diabetes in US (US$) and Australian (AU$) dollars

ALIVE = Agreeing Language in Veterinary Endocrinology; CGM = continuous glucose monitoring; CHO = carbohydrate; POC = point of care; U = unit
Baby scales are inexpensive (typically US$40–$60), provide accurate assessment of body weight, and eliminate the cost and stress of a veterinary visit just to accurately weigh the cat. In addition, the development of clinical signs such as weight loss and polyuria/polydipsia might potentially be recognised through at-home devices, such as automatic water dispensers or smart litter box systems that weigh the cat and/or determine frequency of urination. Options for simplifying and reducing the cost of monitoring include measuring water intake (use a specific jug of water) or urine output (weigh the litter tray), although these might be most useful in single-cat households.
Additional commonly used monitoring tools include CGM, fructosamine, HbA1c, BHB, urine ketones or blood glucose concentration. Costs and availability can vary depending on geographic location. Regardless of the monitoring strategy used, findings should always be interpreted in conjunction with the cat’s clinical signs.
Serial blood glucose measurements (blood glucose curves), either performed at home or in hospital, have been used for monitoring glycaemic response to insulin therapy but have significant limitations associated with large day-to-day variability in response, as well as missed glucose nadirs or hypoglycaemic and hyperglycaemic episodes, which make it difficult to make appropriate decisions about insulin dose adjustments. 64 Surprisingly, because of the considerable day-to-day variability in curves generated at home, the agreement between blood glucose curves performed at home is not superior to agreement between home and hospital blood glucose curves. 64 In addition, most glucometers for human use underestimate the actual feline glucose concentration, which needs to be considered when interpreting glucose concentrations around the upper and lower limits of normal. Challenges of home blood glucose monitoring for owners include technical difficulties, time involved, mild to moderate life restrictions and concern for hurting the cat, although with experience most owners can perform this and may experience positive impact for both owner and cat. 65 Despite these limitations, measuring blood glucose at home can be cost effective and, if performed, should be interpreted alongside DCS and other clinical and physical examination findings.
Glycated proteins like fructosamine and HbA1c reflect glycaemic control over the preceding 3–7 days or 68–77 days, respectively.35,66,67 Fructosamine is increased by persistent hyperglycaemia and influenced by dysproteinaemia,35,68 hyperthyroidism, 69 sex, 70 diet 71 and other diseases, 72 whereas HbA1c is increased by persistent hyperglycaemia and affected by anaemia. 33 Glycaemic changes in fructosamine precede HbA1c, thus allowing more frequent insulin adjustments every 1–3 weeks, whereas HbA1c is useful for long-term monitoring every 2–2.5 months. Glycated proteins can be used as an assessment of glycaemic control in cats on insulin or SGLT2 inhibitor therapy. Because assay results can vary, fructosamine and HbA1c concentrations should be measured using the same validated assay for each cat. Interpretation is best when based on trends rather than a single value, alongside DCS, history and physical examination. Both glycated proteins require a blood sample, necessitating a visit to a veterinarian or technician, which adds to the cost, but they remain cost-effective monitoring tools.
Urine glucose monitoring from the litter is inexpensive; however, it should not be used alone for insulin titration, as it reflects the average blood glucose over the bladder filling period rather than real-time concentrations. However, consistently negative urine glucose may indicate a need to reduce the insulin dose, but further evaluation using additional monitoring parameters is essential.
Interstitial glucose rather than blood glucose concentration is measured using CGM systems and has a lag time of approximately 5–30 mins, although this is dependent on the rapidity of blood glucose changes. 73 Therefore, an exact correlation with blood glucose concentration is not expected. Use of CGM has been validated in cats with diabetes and DKA, and commonly identifies nadirs and hypoglycaemic events.73 –76 An important caveat is that when blood glucose drops to below 2.8 mmol/l (<50 mg/dl), the FreeStyle Libre2 overestimates actual blood glucose in cats. 77 Although challenges have been reported by cat owners, including premature sensor detachment (57%), poor tolerance by cat (60%) and cost (50%), CGM is the authors’ preferred method of monitoring. 78 To mitigate expenses, the authors recommend allowing owners to apply sensors at home and using CGM intermittently – initially to guide insulin dose adjustments, and then once an appropriate dose is achieved, applying a sensor every 3–4 months to monitor glycaemic control. CGM is beneficial for monitoring glycaemic control when using insulin, but it is not necessary for cats only treated with an SGLT2 inhibitor, because clinical hypoglycaemia is not a risk and blood glucose concentration has minimal within- or between-day variation compared with insulin. However, to determine if the cat is in remission, CGM can be useful to monitor glycaemic response to a gradual reduction of insulin or SGLT2 inhibitor dose.
Cats treated with insulin should be monitored with a level of intensity that is commensurate with desired glycaemic control (Figure 3). Clinical signs might be the only monitoring tool used when remission is not a goal and target average blood glucose is around the renal threshold, although clients should be made aware that aiming for a higher average glucose concentration does not preclude the development of hypoglycaemia. Closer monitoring is required when aiming for lower blood glucose concentrations.
In cats receiving SGLT2 inhibitors, monitoring of ketones (blood or urine), glycaemic control and body weight is recommended, especially in the first 14 days. Glycaemic control can be assessed using spot blood glucose measurements or fructosamine concentrations. When appropriate, home blood glucose can be measured to reduce the owner’s costs and veterinary visits. Body weight loss exceeding 5% within 2–3 days or more than 8% within the first 2 weeks of therapy may indicate an increased risk for DKA, possibly indicating the need to implement insulin therapy. 49
Ketone monitoring during SGLT2 inhibitor treatment can be performed using blood or urine samples. The predominant ketone body in DKA is BHB, which increases approximately 5 days 39 earlier in the blood than acetoacetate appears in the urine. Blood BHB measurement is more sensitive for the early detection of ketosis than urine dipsticks. Blood BHB testing has been associated with reduced emergency assessments, reduced hospitalisations and time to recovery from DKA, and lowered healthcare costs when compared with urine acetoacetate testing in people with type 1 diabetes mellitus. 79 Ketone meters are inexpensive, and veterinarians prescribing SGLT2 inhibitors ideally should have one in their practice. Although ketonuria will occur approximately 5 days after the onset of ketonaemia, monitoring urine ketones remains a low cost and practical approach. 39 Monitoring urine ketones was successful in both velagliflozin clinical trials and, if performed at home, is inexpensive and easy. 50 Motivated owners can also periodically measure BHB or urine ketones at home, and should be instructed to also do this if any signs of illness or behavioural changes occur.
When monitoring cats receiving SGLT2 inhibitors, clinical assessment should take precedence over absolute BHB or urine ketone values. Modest BHB or urine ketone elevations in a clinically stable cat may be physiologically appropriate and may not require discontinuation of therapy. In contrast, any degree of BHB or urine ketone elevation in an ill cat is of greater clinical significance and SGLT2 inhibitor cessation is likely warranted. Because evidence-based BHB cut-offs for diabetic cats on SGLT2 inhibitors are lacking, decisions to continue or discontinue therapy should rely on the integration of clinical findings with biochemical data rather than adherence to fixed numerical thresholds.
Cats with persistent impaired fasting glucose concentrations or prediabetes (suggested definition of 7–<10 mmol/l [126–<180 mg/dl]) that are being managed using an ultra-low carbohydrate diet and no other drug therapy for glycaemic control, do not require close glycaemic monitoring, but would likely benefit from periodically monitoring blood glucose at home (depending on the degree of hyperglycaemia) to detect worsening dysglycaemia and facilitate early implementation of appropriate drug therapy. Changes in demeanour, especially lethargy, unintentional weight loss, or changes in water intake or urination, should be investigated immediately because clinical ketoacidosis can develop rapidly.
Conclusions
Diabetes mellitus in cats is treatable, and a spectrum of care can be offered to develop individualised treatment plans. The approach to clinical diabetes includes offering various monitoring and treatment options tailored to the owner’s capacity and the cat’s medical needs, such as adjusting insulin types and frequencies, monitoring, dietary changes and the potential use of newer drugs like SGLT2 inhibitors. The recent availability of SGLT2 inhibitors, which have little risk of clinical hypoglycaemia and allow drug delivery in food, are likely to reduce barriers to treatment. By providing flexible and tailored options, veterinary care becomes more accessible to a wider range of owners and their cats.
Footnotes
Acknowledgements
The authors acknowledge Ashleah Williams for her assistance with referencing and Andre D’Couto for his support with formatting.
Conflict of interest
The authors declare the following potential conflicts of interest related to this research. JSR reports no conflicts of interest, as she has not received funding for this project within the past 5 years. JM discloses employment at the University of Florida and consultancies with, and research support from, Elanco, Dechra and Merck. CG discloses employment at the University of Florida, as well as support and consultancy relationships with Animal Health, Dechra, Okava, Merck Animal Health, BiomEdit, Edge Animal Health, CEVA and BI.
Funding
The authors disclose the following sources of funding for this research. JSR’s salary is supported through donations to the Australian Pet Welfare Foundation. CG is employed by the University of Florida. JM is employed by the University of Florida.
Ethical approval
This work did not involve the use of animals and therefore ethical approval was not specifically required for publication in JFMS.
Informed consent
This work did not involve the use of animals (including cadavers) and therefore informed consent was not required. No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
