Abstract
Objectives
To evaluate the effect of transmucosal glucagon powder (Baqsimi; Amphastar Pharmaceuticals) on blood glucose (BG) concentrations in healthy cats and describe adverse reactions to its administration.
Methods
A randomized, controlled, crossover study was conducted on six healthy cats with a 7-day washout period between treatments. Transmucosal glucagon powder was administered intranasally and rectally and compared with intranasal placebo. Blood was collected at −15 and −1 mins before glucagon administration and 5, 15, 25, 35, 45 and 60 mins after to evaluate BG, plasma glucagon concentrations (pGlucagon) and plasma potassium concentrations (K+). Stress scores and adverse effects were recorded at all time points.
Results
Median pGlucagon in the nasal and rectal groups increased from baseline (nasal: 12.2 pmol/l, range 3.5–44.1; rectal: 6.9 pmol/l, range 2.9–21.1) to 218.5 pmol/l (range 7.9–349.8; P = 0.02) and 349.8 pmol/l (range 67.4–349.8; P = 0.01), respectively, 15 mins after administration. Median BG increased from baseline (101 mg/dl, range 91–110) 15 mins after nasal (137.5 mg/dl, range 104–251; P = 0.006) and rectal (229 mg/dl, range 99–285; P = 0.002) administration. Median K+ decreased from baseline (nasal: 3.8 mmol/l, range 3.6–4.1; rectal: 3.7 mmol/l, range 3.5–3.9) to 3.4 mmol/l (range 3.1–3.6; P = 0.04) at 15 mins with nasal administration, and to 3.2 mmol/l (range 3.1–3.6; P = 0.04) at 15 mins and 3.1 mmol/l (range 2.9–3.4; P = 0.01) at 25 mins with rectal administration. No significant changes were detected in the placebo group. No serious adverse effects were noted.
Conclusions and relevance
Transmucosal glucagon administration is effective in raising BG with minimal side effects in healthy cats. Future studies are needed to quantify the efficacy and safety of transmucosal glucagon in diabetic cats, especially during hypoglycemic crises.
Introduction
Hypoglycemia is a major limiting factor in the management of diabetes mellitus in patients receiving insulin therapy. Diabetic humans and animals have impaired counter-regulatory responses to insulin-induced hypoglycemia, which leads to a lack of, or insufficient, glucagon secretion in response to hypoglycemic events (but does not limit response to exogenous glucagon).1–3 In people and in dogs, the use of continuous glucose monitoring (CGM) allows for a more accurate identification of low glucose episodes compared with intermittent monitoring. 4 In cats, there are currently no reports on the frequency of hypoglycemia using CGM. When home blood glucose (BG) monitoring was used with an intensive insulin protocol, a high frequency (94%) of subclinical hypoglycemia (BG <50 mg/dl) was reported, with a low frequency (2%) of clinical hypoglycemia. 5 While the frequency of subclinical hypoglycemia is lower (6–31%) in studies utilizing in-clinic BG curves with non-intensive insulin protocols, the frequency of clinical hypoglycemia is similar or higher (2% and 7%).6,7 Clinical signs, when present, appear to be mostly neurologic in nature. 8
In surveys that investigated the quality of life of the owners of diabetic pets, as well as perceived quality of life of their diabetic pets, owners’ fears of hypoglycemia had one of the largest negative impacts on their quality of life.9,10 The American Diabetes Association recommends that glucagon is routinely prescribed to people who are at risk for severe hypoglycemic episodes. 9 Glucagon administration is recommended for use during severe hypoglycemic events, in which the patient is unable to consume glucose or other carbohydrates themselves or does not have access to such products. 11 Glucagon is not routinely prescribed to diabetic dogs and cats receiving insulin therapy, and its use in veterinary medicine is limited to the treatment of hypoglycemia refractory to intravenous (IV) dextrose administration. 12 It has already been shown that glucagon increases BG in cats; however, all available injectable glucagon formulations that are intended for emergency use (ie, ready-to-inject pens) deliver doses that consistently cause nausea and vomiting in cats. 13 As such, currently available injectable glucagon can be extremely dangerous to use in the hypoglycemic cat that is comatose and seizing.
Baqsimi (Amphastar Pharmaceuticals) is an intranasal glucagon powder medication recently approved for use in diabetic individuals for severe hypoglycemic events. Baqsimi delivers 3 mg of glucagon powder in a simple one-step dispensing device. The powder is formulated to be passively absorbed through the nasal mucosa in humans. 14 Pharmacokinetic studies of the drug in both pediatric and adult populations showed dose-responsive increases in plasma glucagon levels, with overall lower plasma glucagon levels compared with smaller intramuscular doses of glucagon without significant pharmacodynamic differences.15,16 Acutely, glucagon administration in people decreases serum potassium concentrations. 14 Toxicology studies have been carried out using rat, dog and rabbit models, and have shown no long-term adverse effects of subchronic administration of the intranasal glucagon. 17 Baqsimi has not been previously studied in cats.
As a transmucosal glucagon formulation, Baqsimi has the potential to be used emergently at home by owners to treat life-threatening hypoglycemia and with no need for technical expertise. Baqsimi might also improve the quality of life of owners of diabetic pets by reducing their fear of hypoglycemia and stress related to the potential hypoglycemia-induced death of their pets. Glucagon could be another tool for owners to have on hand, especially owners who do not have fast access to emergency medical care. While other glucagon formulations exist, the pricing of Baqsimi is comparable with other premixed syringes of glucagon and takes less expertise to administer given that injectable glucagon is intended to be delivered intramuscularly. 18 Baqsimi has a shelf-life of 2 years. 14 While dextrose solutions or corn syrup have been historically recommended for pet owners to utilize in these situations, in an obtunded or seizing patient, oral/buccal administration of liquids poses a risk for aspiration events. Transmucosal glucagon also holds a potential benefit for in-hospital use when vascular access is not easily obtained and a severe hypoglycemic event occurs.
The primary aim of this study was to evaluate if Baqsimi, administered intranasally and rectally, is effective in raising BG concentrations in healthy cats. A secondary aim was to describe acute adverse reactions to Baqsimi administration.
Materials and methods
Animals
Six neutered domestic shorthair purpose-bred cats (four female, two male), aged 7–8 years, were included in this study. The cats were sourced from a professional vendor 3 years before the study and had been living in the cat colony at the University of Florida. All cats were overweight or obese with a body condition score range of 6–8 on a 9-point scale. The median body weight was 5.0 kg (range 3.9–6.2). Cats were group-housed in facilities accredited by the Association for Assessment and Accreditation of Laboratory Animal Care International with a fixed 13–11-h light–dark cycle, ambient temperature of 22.2°C and 50% humidity. All cats were socialized and acclimatized to catheter bandages and routine handling and restraint for at least 12 months before the start of the study. Extensive environmental enrichment was provided, including 1–3 h of daily human interaction and 24-h access to various toys and climbing apparatus. Cats were fed commercial dry cat food (2060 Teklad Global Cat Diet; Envigo) ad libitum in sufficient amounts to maintain body weight. Water was available for cats at all times. Cats were deemed healthy based on routine weekly physical examinations, systemic blood work (complete blood count and serum biochemistry panels performed approximately 2 years before the start of the study) and the absence of clinical signs of disease. Experiments were performed in ambient temperatures between 20°C and 24°C in their routine environment. All animal use was approved by the University of Florida Institutional Animal Care and Use Committee (protocol number 202300000345).
Blood glucose, glucagon and potassium measurements
All blood samples were drawn from vascular access ports (VAPs; CompanionPort CP-202K; Norfolk Vet Products) that were previously implanted for another study. The VAPs were surgically placed under general anesthesia into the jugular vein of each cat at least 3 months before beginning the experiment. VAP patency was maintained by weekly heparinized saline (10 U/ml) flushing followed by a 0.5 ml (100 U/ml) heparin lock injection, which was aspirated and discarded before sample collection.
BG concentrations were measured using a handheld glucometer validated for use in cats (AlphaTrak 2 Blood Glucose Monitoring System; Zoetis). Blood samples for glucagon measurement were collected in chilled EDTA tubes and placed on ice until the end of each experiment. The samples were then centrifuged, and the plasma was separated and frozen at −80°C until analysis. Plasma glucagon concentrations (pGlucagon) were measured with a glucagon ELISA (Glucagon ELISA; Mercodia AB) validated for use in cats 19 and plasma potassium concentration (K+) was measured with a point-of-care i-STAT Alinity v (Zoetis) with i-STAT CG8+ cartridges (Abbott).
Drug and placebo
Cats were passively restrained by one handler and Baqsimi (3 mg) was either administered into one nostril or rectally. For nasal and placebo application, the applicator tip was placed against the nostril so that the nostril was completely covered by the applicator. During rectal application, the tip of the applicator was inserted 5–10 mm rectally. The applicator was held between finger and thumb and the plunger was pressed as instructed by the manufacturer. After administration, empty applicators were cleaned and subsequently used for the mock application (placebo group). During placebo administration, the applicator was held against the nostril and then the back of the applicator was tapped to mimic the impact and sound created during the Baqsimi application. The Baqsimi applicator is not reusable, thus an inert powder could not be placed in the system to simulate a more accurate placebo experience.
Study design
This was a randomized, controlled, crossover study. Each cat was randomly assigned to receive one of three treatments: Baqsimi intranasally (nasal group); Baqsimi rectally (rectal group); and mock intranasal (placebo group). There was a 7-day washout period between treatments (see table in the supplementary material for the order in which each cat received the treatments). All cats were fasted overnight (>12 h) before data collection. For the treatment groups, BG concentrations were measured at −15 and −1 mins before administration and 5, 15, 25, 35, 45 and 60 mins after. pGlucagon was measured at −15, −1, 5 and 15 mins in 5/6 cats. pGlucagon was not measured in the sixth cat as it did not complete all arms of the study (see below). K+ was measured at −1, 5, 15, 25 and 60 mins. A spectrum of fear, anxiety and stress (FAS) score 20 was measured at all time points including at the time of administration (0 mins). An FAS spectrum score of 1 indicates mild/subtle signs of fear/anxiety/stress, scores of 2 and 3 indicate moderate signs, a score of 4 indicates severe signs, where the cat may actively try to escape or may freeze, and a score of 5 indicates severe signs, where a cat may exhibit confrontational or repelling behaviors. 14 Cats were observed for 1 h after administration of the glucagon for adverse effects (sneezing, nausea, vomiting). For the placebo group, BG concentrations were measured at −15, −1, 5, 15 and 25 mins. K+ was measured at −1, 5, 15 and 25 mins. pGlucagon was not measured in the placebo group.
Statistical analysis
All data were presented as median and ranges and analyzed with non-parametric tests using commercially available computer software (GraphPad Prism; GraphPad Software). Non-parametric repeated measure analyses were used for all comparisons. Results from time points T−15 and T−1 were averaged and presented as T−8. T−8, T15 and T25 were compared within each treatment group using Friedman tests with adjusted P values (Dunn’s multiple comparison tests) reported. Statistical significance was defined as P <0.05.
Results
Drug application and tolerability
A total of six cats were enrolled in this study. All cats completed the entire study except for one that did not receive the rectal administration after its first two procedures owing to reasons unrelated to the study protocol (see supplementary figures 1–3 showing individual cat data for each treatment).
Two of six cats (cats 5 and 6) in the nasal treatment group had unsuccessful medication applications, during which most of the powder aerosolized. In cat 5, the powder was pushed intranasally but was forcibly exhaled 1 s later. In this cat, there was only a marginal change in BG concentration. Cat 6 turned its head during intranasal deployment of the Baqsimi applicator and the authors were unsure if any medication successfully entered the cat’s nose. This failed application was associated with no change in BG concentrations.
There was a trend toward increased FAS scores from baseline (median 0, range 0–1) to time of application of nasal (median 4, range 2–5; P = 0.06) and placebo (median 3, range 2–4; P = 0.06), but not during rectal administration (median 0, range 0–2; P = 0.5). Five minutes after administration, all FAS scores were in the range of 0–2 for all groups. No additional restraint aids aside from the person passively restraining the cat were required in any treatment groups.
Adverse reactions are reported in Table 1. Sneezing was the most common reaction for cats receiving intranasal administration (4/6 cats), followed by hypersalivation (3/6 cats) and blepharospasm (2/6 cats). Sneezing occurred within seconds of nasal administration of the medication and resolved within 5 mins in all cats. One of the cats that sneezed was cat 6, which turned its head during drug administration. After rectal administration, 2/5 cats vomited; one cat vomited 11 mins after administration and the other vomited 60 mins after administration. There was one episode of vomiting in the placebo group 34 mins after administration.
Summary of adverse reactions by study group

Data are n
Drug efficacy
BG concentrations were available for cats at all time points. K+ was available for all scheduled time points, except for T−1 in two cats in the nasal treatment group, due to malfunction of the i-STAT.
Baseline BG, K+ and pGlucagon did not differ between treatment groups. Overall median baseline BG concentrations were 101 mg/dl (range 91–110). Overall median baseline K+ was 3.6 mmol/l (range 3.5–4.1). Overall median baseline pGlucagon concentration was 8.5 pmol/l (range 2.9–41.2).
pGlucagon was only tested in the treatment groups because of limited study funds. Cat 6 (the cat that did not complete the rectal portion of the study and turned its head during the nasal portion of the study; see supplementary figures 1 and 2) did not have glucagon measured after nasal administration. In the nasal group (n = 5), there was an increase in median pGlucagon from 12.2 pmol/l (range 3.5–44.1) at baseline (T−8) to 260.1 pmol/l (range 7.2–349.8) at T5 (P = 0.05) and to 218.5 pmol/l (range 7.9–349.8) at T15 (P = 0.02). In the rectal group (n = 5), there was an increase from 6.9 pmol/l (range 2.9–21.1) at baseline (T−8) to 349.8 pmol/l (range 30.5–349.8) at T5 (P = 0.08) and to 349.8 pmol/l (range 67.4–349.8) at T15 (P = 0.01) (Figure 1).

Median values and ranges for plasma glucagon concentrations in the nasal (N) and rectal (R) treatment groups at time points T−8 (average of T−15 and T−1), T5 and T15 mins
BG concentrations increased in both the nasal and rectal treatment groups as early as 5 mins after administration but did not change after placebo (Figure 2; supplementary figures 2 and 3). After nasal Baqsimi administration (n = 6), BG concentrations increased at 15 mins (P = 0.0064) to 137.5 mg/dl (range 104–251). After rectal Baqsimi administration (n = 5), BG concentrations increased at 15 mins (229 mg/dl, range 99–285; P = 0.002) and 25 mins (223 mg/dl, range 94–291; P = 0.002). There was no increase in BG in 3/6 cats after nasal administration (two of which were observed to not receive the full dose, as explained above) and in 1/5 cats after rectal administration (supplementary figure 3). In the cats that did respond to the nasal administration of Baqsimi, BG increased by ⩾20 mg/dl within 5 mins. In the four cats that responded to rectal administration, BG concentration increased by ⩾20 mg/dl within 15 mins; in 3/4 cats, this was observed in 5 mins.
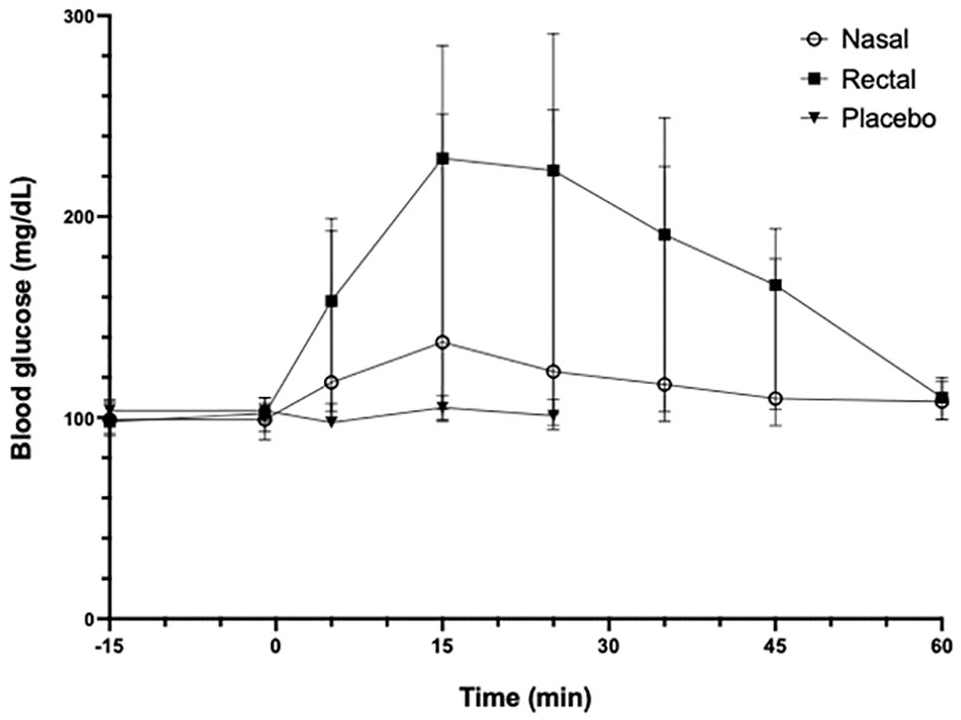
Median and ranges of blood glucose concentrations for nasal group (open circle), rectal group (black square) and placebo (black triangle)
In the placebo group, there was no change in K+ (Figure 3). In the nasal group (n = 4; cats 1 and 6 were excluded from the statistical analyses owing to an error in baseline K+ measurements), there was a mild decrease in K+ at T15 from 3.8 mmol/l (range 3.6–4.1) to 3.4 mmol/l (range 3.1–3.6; P = 0.02) (Figure 3). In the rectal group (n = 5), there was a mild decrease in K+ from 3.7 mmol/l (range 3.5–3.9) to 3.2 mmol/l (range 3.1–3.6) at T15 (P = 0.04) and to 3.1 mmol/l (range 2.9–3.4) at T25 (P = 0.01) (Figure 3).

Median and ranges of plasma potassium concentrations for nasal group (open circle), rectal group (black square) and placebo (black triangle)
Discussion
This study shows that Baqsimi, when administered intranasally and rectally, can increase BG concentrations within minutes and is tolerated in healthy cats with only mild and very transient adverse effects. FAS scores were similar between the placebo group and the nasal administration group, and lower in the rectal administration group, demonstrating greater tolerability of the rectally administered medication. The inclusion of an intranasal control group with demonstratable increases in stress without corresponding changes in BG was utilized to show that BG changes after drug administration (nasal and rectal) were the result of the medication itself rather than stress. Rectal administration of Baqsimi was associated with greater increases in BG and glucagon concentrations; however, it is unclear to what extent this was related to failed drug administration in the nasal group vs decreased absorption. In both groups, the rapid response to Baqsimi was similar to that previously reported for IV and intramuscular glucagon administration, with a peak in BG concentrations after 15 mins. 13
The frequency of vomiting seen with Baqsimi administration was lower than in studies describing IV glucagon administration in cats. 13 In human trials of Baqsimi, the most common adverse effects included nausea, headache, vomiting and upper respiratory irritation.13–15 Vomiting is an important side effect of glucagon administration given that hypoglycemic, obtunded patients may not be able to protect their airways. Larger studies, of hypoglycemic diabetic cats, are required to further investigate the frequency of vomiting in this population and to further evaluate the risk-benefit ratio in cats suffering from life-threatening hypoglycemia.
In people, hypokalemia requiring supplementation has been noted as a possible adverse event of glucagon overdoses. 14 Although clinically relevant decreases in K+ were not observed in our study, it is important to recognize that our study population was small and relatively homogenous; therefore, potential complications of this drug might be underestimated. Larger studies, of client-owned, ideally diabetic, cats are required to further investigate the risk of hypokalemia and the risk-benefit ratio in cats suffering from life-threatening hypoglycemia. Glucagon administration has also been reported to cause a transient hyperkalemia, which was not observed in this study but might become clinically significant in a larger, more heterogenous population. 21
Two of six cats in the nasal treatment group had unsuccessful medication applications, during which most of the powder aerosolized. During rectal administration, however, one of these cats showed a strong increase in BG and pGlucagon, while the other cat did not receive the rectal administration. In the authors’ opinion, the lack of compliance by the cat, rather than a failure of drug absorption, may have played a role in the ineffectiveness of nasal administration in these two cases. The lack of compliance may be a minor issue in the clinical setting, as cats experiencing severe, clinical hypoglycemic episodes might be too obtunded to resist a Baqsimi application.
While the cost of Baqsimi might deter some owners, it is important to emphasize that its purchase is likely a one-time expense. The shelf-life of Baqsimi is approximately 2 years from the manufacture date, and owners should be properly educated to use it only if their cat is suffering from a life-threatening insulin overdose with clinical signs such as severe obtundation, coma or seizures.
Sneezing might have decreased drug availability for absorption after nasal application. However, even if the drug was available, decreased absorption after successful administration might also contribute to lack of effect. In cat 3, in which BG and pGlucagon did not increase substantially after uneventful nasal administration, there was a substantial increase in both analytes after rectal administration. This was likely the result of a substantial difference in glucagon absorption through the nasal vs rectal mucosa. The difference in absorption could be related to differences in surface area or differences in the properties of the mucosa. Interestingly, in people, rectal administration of glucagon did not result in a significant change in BG concentrations, despite a significant increase in pGlucagon. 22 This might be explained by differences in absorption into systemic and portal circulations or by stimulation of gut and pancreatic hormones that might occur with one route of administration but not the other. This could potentially be further elucidated by measuring insulin concentrations at the time of administration. Because our study was attempting to answer a clinical question on the feasibility of the use of Baqsimi, and because of limited resources available for this study, insulin concentrations were not measured. In practice, the effect of glucagon, administered by any route, on insulin secretion, is irrelevant for the following reasons: (1) by definition, insulin concentrations are expected to be excessive in the target population; and (2) within the target population, the insulin concentration is not expected to change with glucagon administration as the source of excess insulin is exogenous.
As mentioned above, one limitation of this study is the small sample size, which inherently increases the chance of a type II error. In addition, we studied purpose-bred cats that are fairly homogenous in their genetic makeup and very homogenous in their environment (including diet). Sample size and heterogeneity are especially important in assessing the array of potential side effects to a medication. The small sample size also limits the study’s ability to determine whether Baqsimi treatment would routinely be useful for the treatment of hypoglycemia in cats because not all cats had a change in BG despite apparently successful medication administration. This also highlights that one dose may not be enough to restore euglycemia, if this drug restores it at all in hypoglycemic diabetic cats. The exact frequency and timing of vomiting, for example, might be an important question to investigate in future studies, as well as the exact benefit of a rapid at-home response to hypoglycemia. It is possible that the risk of glucagon-induced vomiting might outweigh the benefit of rapid resolution of hypoglycemia. Another limitation is the inability to account for how much medication was administered to each cat. This can lead each patient to receive different amounts of medication, which could lead to variation in study results.
Conclusions
This study showed that transmucosal glucagon powder administration was effective in raising BG concentrations rapidly in healthy cats with minimal side effects. Future larger studies are needed to quantify the efficacy and safety of transmucosal glucagon in diabetic cats, especially during hypoglycemic crises.
Supplemental Material
sj-docx-4-jfm-10.1177_1098612X241280516 – Supplemental material for Transmucosal glucagon rapidly increases blood glucose concentration in healthy cats
Table 1: Order of treatment administration by cat.
Supplemental Material
sj-pdf-1-jfm-10.1177_1098612X241280516 – Supplemental material for Transmucosal glucagon rapidly increases blood glucose concentration in healthy cats
Figure 1: Individual data for placebo administration.
Supplemental Material
sj-pdf-2-jfm-10.1177_1098612X241280516 – Supplemental material for Transmucosal glucagon rapidly increases blood glucose concentration in healthy cats
Figure 2: Individual data for intranasal administration.
Supplemental Material
sj-pdf-3-jfm-10.1177_1098612X241280516 – Supplemental material for Transmucosal glucagon rapidly increases blood glucose concentration in healthy cats
Figure 3: Individual data for rectal administration.
Footnotes
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors were partially supported by Merck Animal Health.
Supplementary material
The following files are available as supplementary material:
Table 1: Order of treatment administration by cat.
Figure 1: Individual data for placebo administration.
Figure 2: Individual data for intranasal administration.
Figure 3: Individual data for rectal administration.
Ethical approval
The work described in this manuscript involved the use of experimental animals and the study therefore had prior ethical approval from an established (or ad hoc) committee as stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers, tissues and samples) for all procedure(s) undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
