Abstract
Objectives
The objective of this study was to compare the use of the feline Glasgow Composite Measured Pain Scale (CMPS-f) at home and in a veterinary hospital. The hypothesis was that pain-free cats would score higher in the CMPS-f when in a stressful situation than when calm and relaxed; that is, healthy but stressed cats could appear to be in discomfort or pain.
Methods
Healthy, non-painful adult cats owned by clinical staff were included in a prospective clinical trial with two observers (caregiver [CG] and researcher). Cats were scored by their CG at home (H), after arrival at the clinic (C1) and after a routine health check (C2). A researcher pain-scored the cats at C1 and C2 concurrently with the CG. Friedmann’s test with Dunn’s multiple comparison test was used. The level of significance was set to an alpha of 5%.
Results
Data from 17 cats were included in the statistical analysis. Scores by the CG and researcher at C2 were higher compared with H (P <0.01 and P <0.01, respectively) and C1 (P = 0.02 and P <0.01, respectively). The mean increase in CMPS-f scores from H to C2 and from C1 to C2 was 5.8 and 4.1, respectively. At C2, the CMPS-f intervention level of 5/20 and above, indicating pain, was reached in 11/17 cats. There was no significant difference in the scores assigned by the CG and researcher within each time point.
Conclusions and relevance
Pain scores recorded after examinations in the clinic were significantly higher than those recorded at home. This suggests that stress may lead to a misinterpretation of the CMPS-f, potentially affecting the recognition of pain in cats during clinical assessments.
Introduction
Pain is a subjective multidimensional experience affecting a patient’s individual health and welfare. 1 It includes complex sensory, affective-emotional as well as cognitive domains. 2 This makes pain recognition and rating challenging, especially in non-verbal individuals like animals. In feline patients, the difficulty in clearly recognising pain has been described as one of the main reasons for inadequate pain therapy. 3 In addition, the possible influence of an animal’s personality and demeanour on pain evaluation is discussed.4 –7 This is important because, to date, pain rating in cats widely relies on behavioural observation eventually combined with evaluation of response to interaction.7 –10 On these grounds, different scales have been developed and validated to detect pain in cats.7,8,11 –13 One of these, the feline Glasgow Composite Measured Pain Scale (CMPS-f), 13 has been validated in several languages14,15 and is therefore a widely used tool for the assessment of acute pain in veterinary practice. 14 It enables veterinary professionals to evaluate the level of pain a cat might be experiencing by rating categories including behaviour, vocalisation, mobility, response to interaction, posture and state of arousal.
However, it was shown that when evaluating hospitalised cats, including those not expected to have a painful disease, some animals exhibit pain scores above the intervention level. This might falsely indicate the need for analgesia, despite the absence of a painful condition. 14 Difficulties in accurately discerning and interpreting behaviour and expressions traditionally associated with pain might contribute to this, because the appearance of pain might overlap with that caused by other (negative) emotional states. Changes in physiological parameters, restlessness, sleep disturbance, vocalisation, lethargy and aggression are all described as indicators for pain as well as for (dis)stress.10,16 –18 Positioning of the ears is one important point evaluated in the CMPS-f 13 and the Feline Grimace Scale. 12 However, the position indicative of pain (flattened ears, rotated outwards) might also be present during anxiety or fear. 17 Interestingly, in human medicine, discomfort scores rather than pure pain scores are sometimes utilised for non-verbal patients, acknowledging the difficulty in clearly separating between pain and other negative states.19,20
At present it is not clear if and how negative emotional states other than pain can alter the CMPS-f. Following the CMPS-f guidelines, the scores obtained in healthy non-painful cats should be less than 5/20. 13 Hence, the aim of this study was to identify if the behaviour in non-painful animals could affect the CMPS-f measurement and lead to their misclassification. The hypothesis is that pain-free cats score higher in the CMPS-f in a stressful than in a calm situation.
Materials and methods
The study design was critically reviewed by the ethical review group of the Association of Veterinary Anaesthetists (number 2022-005).
The study was designed as a prospective clinical trial. Each cat was examined by two individuals concurrently: the caregiver (CG) of the individual cat and 1/2 researchers.
Using the freely available software G*power 3.1.9.7 (University of Duesseldorf, Germany), the sample size was calculated for a difference of 1 between pain scores between measurement points, setting beta to 0.8 and alpha to 0.05. A minimum sample size of 12 cats was defined.
Cats privately owned by veterinary staff of the small animal hospital, University of Veterinary Medicine Hannover, Foundation, scheduled for their routine annual health check were recruited. The hospital is a referral centre with an included first opinion service, which is regularly used by the clinical staff for their own animals. Exclusion criteria were age younger than 6 months, medication needed to facilitate transport, highly anxious animals and cats unable to be examined without sedation.
The purpose and sequence of the study were verbally explained to volunteer participating owners before enrolment. Furthermore, owners received a written copy with information about the research and a guide on performing the cat’s examination using a prototype online mobile application for pain scores. This included a summary of the steps of pain scoring (observe from distance, approach the cat, palpation, take photos). As all CGs were veterinary professionals, no further formal training in pain scoring was conducted. CGs were encouraged to read the instructions and ask open questions before pain scoring their cat at home. Owners could withdraw their consent at any point during the study without providing a reason. Cat friendly techniques and handling were followed during the whole trial, including limited access to room, maximum number of individuals in the room restricted to three, towel wrap techniques, use of pheromones and highly palatable treats.
Pain score evaluation was divided into two parts: (1) scoring at home performed by the CG, and (2) scoring at the hospital performed by the CG and the researcher concurrently (Figure 1).

Study timeline. The light person represents the caregiver and the bold person represents the researcher. Clocks on top of the arrows indicate acclimatisation times allowed before starting pain scoring (15–60 mins on arrival at the clinic; 5–15 mins before the clinical examination). The car indicates the cat’s transport to the clinic, and the stethoscope, the clinical examination and the syringe represent the blood sampling performed
Scoring at home
Scoring at home was performed on the day of the hospital visit or the day before. This long timeframe was chosen to enable the CG to score their cat when it appeared most relaxed. This was defined as time point home (H). Manipulation and interaction with the cat were limited to the steps necessary for pain scoring according to the CMPS-f.
Scoring at the hospital
The second part of the study was conducted in a separate cat-dedicated ward at the veterinary hospital, which was reserved for only the study participants on that day. A pheromone plug-in was placed in the room at least 24 h before the cat’s arrival. Only the individuals directly involved (CG and researcher) had access to the room throughout the study.
After arrival at the hospital, the CG placed the cat in a kennel. The kennel was equipped with water, food, pheromone-sprayed blankets and a fresh litter tray. No den was provided as visualisation of the animal was needed while the CMPS-f was performed. An acclimatisation period of 15–60 mins was allowed until the owner judged the cat to be calm and relaxed. During acclimatisation, the cat was left undisturbed but with the CG and researcher in the room. Once the animal had settled, both observers performed the first pain score in the clinic (C1). When manipulation was requested on the CMPS-f, it was carried out by the CG, while the researcher judged according to their visual impression. A pause of 5–15 mins was allowed before the health check started. The health check included assessment of mucous membranes and capillary refill time, mouth inspection, chest auscultation, pulse palpation, abdominal palpation, evaluation of lymph nodes and measurement of rectal temperature. A blood sample for haematology and biochemistry analysis was aseptically collected via venepuncture. Once the physical examination and blood sampling were completed, the CG placed the cat back in the kennel for immediate observation and pain scoring by both observers (C2).
Statistical analysis
Software Graph Pad Prism 10 (GraphPad Software) was used for the statistical analysis. Demographic data were analysed using descriptive statistical methods.
Pain scores at different time points and between observers were compared with Friedmann’s test and Dunn’s multiple comparison test. Spearman correlation was calculated for time points C1 and C2 between the observers. Interrater reliability was calculated via calculation of linear weighted Cohen’s kappa coefficient. The level of significance was set to P <0.05.
Results
A total of 22 cats were recruited. All cats had visited the first opinion or the referral service of the hospital before. As a result of technical difficulties with the prototype online application, the data sets of five cats were incomplete and had to be excluded from the analysis. The study was therefore completed with the traditional paper form of the CMPS-f. Data from 17 cats were analysed. Four cats were measured with the online format and 13 with the printed version of the CMPS-f. Demographic data of all cats are shown in Table 1.
Demographic data of 17 cats included in the study

BSH = British Shorthair; DSH = domestic shorthair; FS = female spayed; IRIS = International Renal Interest Society; M = male; MC = male castrated
Mean acclimatisation time from arrival at the clinic until C1 was 18.4 ± 2.7 mins and mean acclimatisation time after C1 until the clinical examination was 6.4 ± 1.2 mins.
Pain scores given by both the CG and researcher for the individual cats at H, C1 and C2 are shown in Table 2. The mean scores increased from H to the subsequent measurements (C1 and C2), with the highest increases observed at C2 (researcher) (Table 3). Pain scores given by both the CG and researcher were different between time points H and C2 (P <0.01 and P <0.01, respectively) and between C1 and C2 (P = 0.02 and P <0.01, respectively). Within time points C1 and C2, no significant differences between the scores given by the CG and researcher could be detected (P >0.99) (Figure 2).
Feline Glasgow Composite Measured Pain Scale scores for 17 cats, evaluated by the caregiver (CG) and researcher (R) at time points home (H), arrival at the clinic (C1) and after the health check (C2)

Scores above the interventional threshold (⩾5/20) are indicated in bold
Mean increase of pain scores between measurement points home (H), in clinic 1 (C1) and in clinic 2 (C2) for the caregiver (CG) and researcher (R)
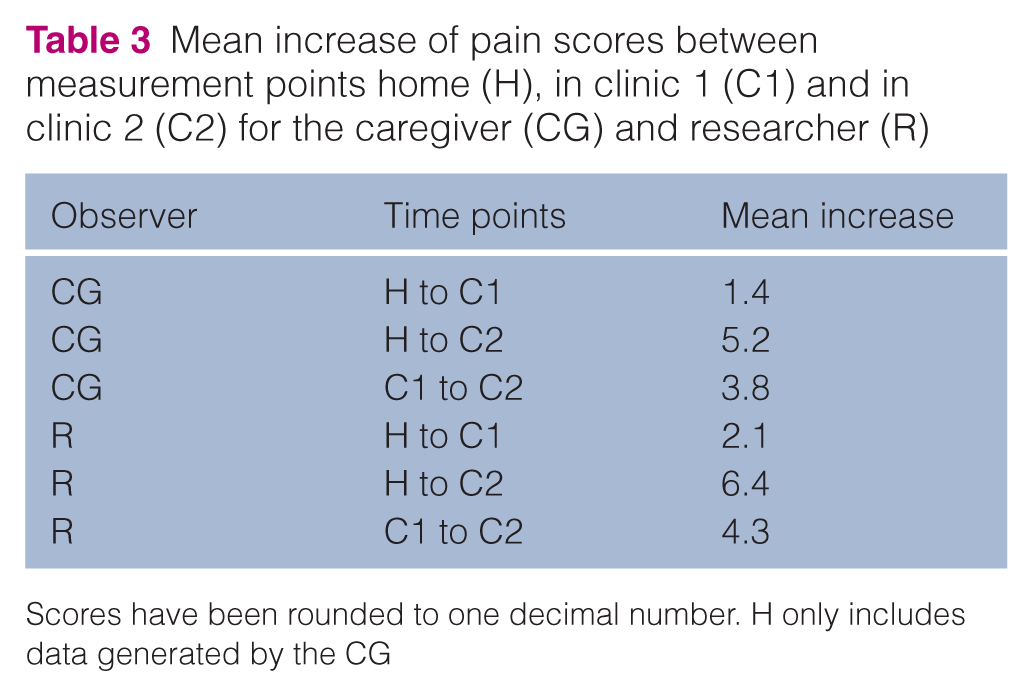
Scores have been rounded to one decimal number. H only includes data generated by the CG

Scatter plot of feline Glasgow Composite Measured Pain Scale scores for 17 cats, evaluated by the caregiver (CG) and researcher (R) at time points home (H), arrival at the clinic (C1) and after the health check (C2). Different colours represent individual cats. The horizontal line indicates the median of all scores. Significant differences towards H are marked with an asterisk (*)
Correlation of ratings between the CG and researcher at C1 was moderate (r = 0.6946; P <0.01) and at C2 was strong (r = 0.7271; P <0.01). Interrater reliability between the CG and researcher was moderate (Κ = 0.561; P <0.01).
The performed clinical examination did not indicate any unknown disease or necessity for treatment.
Discussion
During this trial, healthy cats received significantly higher pain scores after a possibly stressful experience (C2) than before (H and C1). Therefore, the hypothesis that pain-free cats score higher in the CMPS-f in stressful situations than in a calm environment was supported.
Discerning painful animals from stressed ones is of clinical importance as treatments will be altered based on pain scoring.21 –23 At C2, at least 1/2 observers gave 16/17 healthy cats a pain score of 5 or higher. In a clinical situation, this would have warranted unnecessary analgesic treatment, including the possible risk of adverse drug effects.
Venepuncture using a hypodermic needle is a clinical routine in veterinary practice and is usually expected to be non- to only mildly painful during the actual event of puncturing, but not afterwards or long-lasting. Furthermore, as none of the included cats suffered from known chronic pain states, possibly leading to hyperalgesia or allodynia, venepuncture as the possible reason for raised scores at C2 seems to be unlikely but cannot be excluded with certainty. In addition, none of the cats was known to have osteoarthritis, but no attempts were made during this study to definitely rule out osteoarthritis in the participating cats. Hence, possibly undiagnosed osteoarthritis, causing pain after manipulation during the clinical examination, might also be an explanation for raised scores at C2 in individual cats. Therefore, the distinct reason for the increase in scores cannot be firmly established. However, as study participants were healthy and in all probability pain-free, (dis)stress seems to be likely. A paucity of data exists about whether and to what degree changes in behaviour and facial expression due to other negative emotions (stress, nausea, dysphoria) can influence pain score results.4,5,22,24 Reviewing the items that are evaluated in the CMPS-f from all parts of the score (observation from distance, facial expression and interaction with the cat) answer options (eg, vocalisation, flattened ears, unresponsiveness, dull, grumpy) can also be found in texts describing other negative emotional states, such as as anxiety, fear or stress.16 –18
In human medicine, the overlap between stress and pain is well-documented. The same common neurological pathways and brain regions, particularly within the limbic system, which are involved in emotional responses and memory, including previous experiences of pain, modulate both stress and pain.25 –27 For example, stress activates the hypothalamic-pituitary-adrenal (HPA) axis, triggering the release of adrenal glucocorticoids, which affect emotional and cognitive functions, including the perception of pain. 28 This activation also stimulates the autonomic nervous system, leading to physiological changes such as increased heart rate and blood pressure, 28 which are often used as a clinical indicator of pain or stress. Furthermore, the limbic system and the HPA axis form an interconnected loop that regulates the stress response and glucocorticoid release. This loop influences how the body and brain respond to stress, while also modulating pain perception. 29
In the present study, the intention was to assess the immediate behavioural response of the cats to an acutely stressful situation. Previous veterinary experiences were not considered, although these could also influence the animal’s behaviour. In rats, it was shown that repeated stress could alter pain perception over a prolonged time even after the termination of the stimulus.30,31 Therefore, it cannot be excluded that some learnt behaviour or excessive sensitivity might have altered the scores measured.
Furthermore, the personality of the cats was not formally assessed by a validated scale. This can be seen as a limitation as data are available showing a higher increase in postoperative pain scores in cats with high demeanour scores than in animals with low demeanour scores. 5 However, in both groups of that study demeanour scores changed as well as pain scores, although in different magnitude. 5 Reassessing the items used in the pain and demeanour scores, it is evident that in some categories rather similar things are rated, possibly leading to overlap and chances of misinterpretation. Interestingly, a canine study could not find any relationship between preoperative anxiety levels and pain scores. 6 At present, available demeanour scores are only studied in a clinical, often perioperative, environment.4 –6 No data about possible changes in demeanour scores of individual cats due to different environments are available. Therefore, it remains unclear whether, or to what extent, transport from home to the clinical environment may have influenced the cats’ demeanour. The focus of the present study was to determine whether, in stable cats without painful conditions, other negative emotions could lead to falsely elevated CMPS-f scores. As all but one cat exceeded interventional thresholds at C2, this was confirmed. Exploring whether the magnitude of such emotional influences on CMPS-f scores varies among cats with different personality types could be an interesting topic for future research.
The CMPS-f is a widely recognised pain assessment tool for cats.7,32 Its structured design incorporates assessment of both behavioural and physiological indicators.13,24 However, it was designed to examine possible painful cats in a veterinary environment,13,24 and was validated and often used for the rating of cats postoperatively when these animals likely could have been in pain.13,24 In the context of a veterinary environment and after surgery, behaviour such as vocalisation, immobility, altered posture or facial expression likely will indicate pain and will correctly be interpreted as such. However, as mentioned earlier, these behavioural alterations could also indicate stress, fear or anxiety,16 –18 which might be present concurrently with pain or in other situations exclusively without pain. Therefore, to minimise the number of wrongly interpreted cats, on one hand, the sensitivity of the CMPS-f to (dis)stress-induced behaviours demonstrates the need for interpretation of the scores in the context of the cat’s individual situation and disease. On the other hand, it highlights the importance of complementary assessment of stress and the use of stress-reduction strategies, such as low-stress handling, acclimatisation period and appropriate housing, including hiding options, to mitigate the effects of (dis)stress on pain scores. 33 Despite using stress-reducing measures in the present study, a change in CMPS-f scores was detected. Whether cats would have scored even higher without these strategies cannot be foreseen.
The current study incorporated an acclimatisation time after arrival at the clinic; still, the variable length of it could be seen as a study limitation. In contrast, other studies describe standardised acclimatisation periods of 5 or 10 mins.16,34 In those studies, differences in heart rate, respiratory rate, blood pressure or blood cortisol between assessment at home and in clinic could be documented, possibly indicating that acclimatisation periods were not long enough for some cats. Therefore, the acclimatisation time in the present study was subjectively adjusted to the CG’s perception of the cat. If none or a standardised acclimatisation period had been used, some of the pain scores at C1 might have been different. Furthermore, in the present study, approach, contact and handling of the cat at time points H and C1 were performed solely by the cats’ familiar CG, and handling was limited to actions essential for pain scoring. In contrast, in other studies, a veterinarian performed a full clinical examination and blood collection.16,34 This more extensive and potentially unpleasant interaction with an unfamiliar person may have already elevated stress levels in the cats, even at home, and could have diminished the differences observed between study locations. However, in the present study, no significant differences were found between scores at H and C1, suggesting that the time given by the CGs was likely sufficient for the cats to settle after transport. Furthermore, the presence of a familiar CG may have helped the cats to relax in the clinical environment. Using video-based observations, it has been shown in horses that discomfort behaviour decreases in the presence of a caretaker. 35
Similar to other studies,7,13,16 agreement between the measurements of the two observers before and after stress (C1 to C2) was good. This might be partly due to the structured format of the CMPS-f, which minimises subjective variability. In addition, all observers were veterinary professionals; this might also have led to more homogeneity, as trained observers tend to demonstrate higher consistency, especially in clinical settings where untrained individuals might overlook subtle behaviours. 33
During the first days after ovariohysterectomy in a home environment, it has been demonstrated that laypersons are able to recognise changes in their cat’s behaviour that might indicate pain. 36 Supported by this finding and to get a real baseline of the individual cat while avoiding stress due to an unfamiliar person at home, the study was designed with only CGs’ evaluation of the cat at H. As pain scores of CGs and researchers did not differ significantly at C1 and C2, it was decided that it was reasonable to also compare the measurements of the researchers to H. The concordance between CGs and researchers further demonstrated the robustness of the scale for use across different evaluators as researchers achieved scores comparable to CGs without prior familiarity with the animal.
As an additional indication of the robustness of the scale, not only were the final scores assigned to each cat by both observers similar, but the increases across all CMPS-f items at each time point also followed a concurrent pattern.
The design of the current trial as a clinical study has some limitations. Cats were scored only once at each time point by two observers. To minimise stress for the animals and comply with cat friendly practice standards, the presence of additional investigators in the examination room was not permitted. However, video recording during pain scoring followed by offline analysis could have allowed for the inclusion of more observers with varying levels of experience. In addition, blinding of investigators to time points C1 and C2 would have been feasible. Because of the study design, cats had spent the longest time in the clinic and underwent repeated handling at C2. To assess the effects of time and repeated handling on scores, additional scoring points at different times after arrival would have been valuable, but this was not possible in the clinical setting. Nevertheless, the main objective of this study was achieved under the current conditions. Similar to other studies,36,37 the present trial demonstrates that stress can influence behavioural parameters. Consequently, the clinical recognition of pain in feline patients using the CMPS-f is likely to be affected by this emotional state.
Merola and Mills2,38 caution about the challenge of identifying shared indicators of pain and other emotions in cats and conclude that no specific signs that can reliably identify pain have been reported. In human medicine, the challenges of accurately recognising pain in non-verbal individuals have led to the development of discomfort scales rather than pure pain scales.17,39,40 These scales acknowledge that distinguishing between negative emotions as the cause of a particular behaviour or physiological change may be impossible. Nevertheless, higher scores on such discomfort scales – whether due to pain or other factors – prompt CGs to take action to support the wellbeing of the affected individual.19,39,40
Considering these points, the question arises whether it might also be sensible in veterinary medicine to shift from using pure pain scales towards discomfort/distress scales. This approach could be particularly important outside the postoperative period, when pain is not presumed and when no specific pain assessment tool exists for patients in a given situation. Until this issue is resolved, it remains essential to interpret pain scores within the context of the individual cat’s circumstances to avoid false-positive or false-negative results.
Conclusions
This study suggests that (dis)stress can affect pain score measurements obtained using the CMPS-f. To minimise false-positive ratings and potentially unnecessary analgesic treatment, pain scores in hospitalised patients should always be interpreted in the context of the underlying disease and any analgesia already administered. A concurrent evaluation of factors that may lead to (dis)stress – particularly in cases where conventional pain assessment is inconclusive – seems to be a sensible approach.
Footnotes
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors were employees of the University of Veterinary Medicine Hanover, Foundation and acknowledge financial support by the Open Access Publication Fund of the University of Veterinary Medicine Hannover, Foundation.
Author note
Preliminary results were presented as a poster at Kölner Katzentage 2023 (16–17 September) in Köln (Germany) and the Association of Veterinary Anaesthetists autumn meeting 2024 in London (UK).
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognised high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers, tissues and samples) for all procedure(s) undertaken (prospective or retrospective studies). For any animals or people individually identifiable within this publication, informed consent (verbal or written) for their use in the publication was obtained from the people involved.
