Abstract
Intestinal mucositis is a frequent side-effect of chemotherapy treatment. Many oncological research programs aim to identify novel treatments for this distressing condition, and these programs frequently use rat models. Little is known about the presence and progression of pain in these models and how this can best be treated by analgesic therapy. We used a number of behaviour-based methods of pain assessment to determine which tools were best suited for pain identification. Baseline measures for behavioural assessment, rat grimace score and sociability were determined through analysis of continuously recorded video data and an applied social interaction test (n = 16). Mucositis was then induced by intraperitoneal injection of 5-fluorouracil (150 mg/kg) and further behavioural analyses undertaken. An assessment of enrichment interaction was also made by determining the mass of a plastic chew toy gnawed both pre- and post-chemotherapy injection. Behavioural scoring was performed 1, 6, 12, 24 and 48 h after injection, with facial expression being scored at the 12, 24 and 48 h time-points. Sociability testing was performed once during the post-injection period. No significant differences were found in grimace scores between baseline and later daily measures. Behaviours similar to those previously reported post-laparotomy were observed. Writhing, twitching and back-arching behaviours were most evident in rats affected by mucositis and were increased in frequency (respective P values: 0.002, 0.004 and 0.008) 48 h after chemotherapy injection compared with baseline, implying that pain onset occurred around this time-point. Social investigatory behaviour was also increased (P = 0.002) following disease onset. Each day, rats post-5FU injection gnawed a greater percentage of their nylabone enrichment by weight than the saline-injected control rats (P = 0.046). These data suggest that, of the tools tested, behavioural assessment scoring may find greatest utility in rodent models of intestinal mucositis and should be investigated further.
Mucositis is an ulcerative disorder that affects the digestive system, manifesting as a frequent consequence of chemotherapy treatment; significantly impacting on quality of life in cancer patients treated with chemotherapeutic agents.1,2 The pathogenesis of mucositis involves the direct inhibition of DNA replication and mucosal cell proliferation by the chemotherapy agent, leading to gastrointestinal tract damage. Such damage includes apoptosis of cells, crypt cell hypoproliferation, villous atrophy, collagen breakdown, ulcerations and increased intestinal permeability. 3 As a result, serious clinical symptoms occur which include abdominal bloating, pain, nausea, diarrhoea and weight loss. 4 Pain largely arises from inflammation and consequent ulceration of the gastrointestinal tract, with the mouth (oral mucositis) and small intestine (intestinal mucositis) most frequently affected. 5 The distressing nature of these symptoms leads to the mucositic condition being a major dose-limiting factor in the treatment of many cancers. 1 Consequently, a number of research programs are aimed at investigation of mucositis disease mechanisms, and identification of novel therapeutic targets. Animal models are widely employed in these research investigations. 6
Rats are commonly used as animal models for mucositis disease investigations. Typically, mucositis is induced through an administration of a chemotherapeutic agent. Chemotherapy administration leads to the rapid-onset of mucositis as shown by body weight loss, clinical scoring, and retrospective histological analysis.7,8 Clinical scoring typically records the presence or absence of clinical signs including piloerection, diarrhoea and body condition score in order to assign a value for each, which is summed to arrive at a total clinical score. However, despite these measures being objective in nature, they are typically retrospective, and therefore do not aid in humane endpoint determination or analgesic therapy modification. This inability to alleviate pain or distress poses both an animal welfare and ethical issue, and could potentially impact on the research data obtained.
It is assumed that rats subjected to mucositis experience similar symptoms to those reported by human patients. Abdominal pain is one such symptom that may be amenable to amelioration through analgesic administration. As such, determining a method of evaluating both presence of, and level of pain would be of value for animal welfare reasons as well as improving the validity of therapeutic testing. Pain tends to evoke changes in behaviour which could potentially be measured quantitatively; 9 however pain assessment in rodents remains challenging, since as prey species they need to mask signs of pain to avoid predator attention. 10 Nevertheless, there has been significant progress in determining visceral signs of pain in rats, 11 although much of this previous work has been carried out in a laparotomy model.2,12,13
More recently, changes in facial expression have been characterized as a potential tool for rodent pain assessment, the so-called ‘grimace scales’.14,15 This method may offer considerable advantages over other methods of behavioural assessment scoring since human observers tend to focus on the facial area, 16 and scoring is likely to be less time-consuming than traditional behaviour-based techniques. This technique has largely been tested in the evaluation of short-lived acute pain,17,18 as opposed to longer-term pain states. The social interaction test is generally regarded as a measure of anxiogenic effect, 19 rather than pain. However, pain has been shown to impact upon social interaction and vice versa. 20 Hence, the social interaction test may be a valuable adjunctive measure under controlled experimental conditions.
The aim of this exploratory study was to investigate behavioural assessment scoring, facial grimace scoring, and the social interaction test, to determine which tool (if any) might be most appropriate for the assessment of pain in rats subjected to mucositis, given the unique nature of this condition and the accompanying environmental conditions (metabolism-cage housed). We also sought to identify observation time-points that allowed effective pain assessment. An assessment of enrichment interaction was also made to provide a non-specific indicator of welfare state. Specifically we were interested in methods that could show the presence of pain and chart its progression.
Materials and methods
Animals
This study used 16 male Hsd:Sprague Dawley (SD) rats (University of Adelaide, Laboratory Animal Services, Adelaide, Australia). Rats were housed individually in metabolism cages (Tecniplast, Exton, PA, USA) throughout the study. A standard casein-based diet 21 and potable RO water were provided ad libitum. All animals were provided with a half plastic bone toy (Nylabone Products, Neptune, NJ, USA) as enrichment. A five-day period of acclimatization was provided prior to commencement of behavioural observations. The animal room was maintained at 21–23℃ with a 12 h reversed light/dark cycle. Red light (approximately 650 nm wavelength) was provided during darkness to facilitate video recording.
This study was conducted as part of a larger program of work investigating novel therapeutic agents for the treatment of chemotherapy-induced mucositis; the animals forming the disease control group of this larger study. The research was approved by the Animal Ethics Committee of the University of Adelaide and conducted in accordance with the provisions of the Australian code for the care and use of animals for scientific purposes. 22
Experimental design
Animals acted as their own controls to minimize the effects of inter-individual variability in behavioural data interpretation.
Rats were orally gavaged with water once daily for nine days (between days 0 and 8). On day 6, all animals were injected with 5-fluorouracil (5-FU) (Mayne Pharma Pty Ltd, Mulgrave, Victoria, Australia; 150 mg/kg) via the intraperitoneal route. Rats were humanely killed 72 h following chemotherapy injection by carbon dioxide asphyxiation.
Behavioural measures
The rats were video-recorded continuously in their home cages by closed-circuit television (CCTV) cameras (OzSpy CLOC 600IR, Brisbane, Australia). Cameras were mounted on a rack directly behind the metabolic cages (one camera per cage) and at approximately 20 cm distance from the cage. These data were used for later rat behavioural analysis and facial image collection. A social interaction test (modified from Sams-Dodd 23 ) was performed on one occasion pre- and post-mucositis induction (days 2 and 7). This test used a circular homogeneously illuminated arena with the base and sides made of plastic sheeting and hessian (50 cm radius). The floor was covered with a grey bedding material (Animal Bedding; FibreCycle Pty Ltd, Yatala, Queensland, Australia) which had been exposed to other rats’ odour prior to testing. Behaviour during the test was recorded using a video camera (Aiptek ADD T2 Pro; Aiptek International Inc, Hsinchu, Taiwan) from above the arena. The tests were performed in a separate room free from personnel entry.
To perform the test, the experimental rat was placed into the arena simultaneously, and approximately 40 cm apart, from an unfamiliar SD male rat which was not part of this study. The experimental rat was marked with a non-toxic black marker pen to enable recognition from video footage. Behaviour was recorded for 10 min.
In order to assess enrichment use, the method described by Turner et al. was used. 24 The Nylabone™ toy was weighed at the start of the study, just prior to the 5-FU injection and at the completion of the study.
Behavioural data analysis
Home-cage observations
Ethogram for in-cage behavioural analysis (adapted from Roughan and Flecknell 2 ).

Facial image analysis
At each of the time-points used for behavioural scoring during the light phase of the cycle, 10 still images of the face of the rats were retrieved from the video footage. These images were taken when the rats directly faced the camera or, on occasion, when a clear side-on image could be obtained. Images were cropped to show the face alone. For scoring, three images at each time-point were selected using a random number generator. The images were scored by two treatment-blinded coders using the method described by Sotocinal et al. 15 This method scores each image based on four action units: orbital tightening, nose/cheek flattening, ear change and whisker change. Each facial unit was scored from 0–2 (0 = not present, 1 = moderate and 2 = severe). However, whisker position proved difficult to visualize from the photographs and hence this was excluded from the analysis. Therefore, the maximum score obtainable was 6. The four action unit scores were summed to produce the total score. The mean of the total scores obtained from the three images for each rat was used in the analysis. The mean of both coders’ scores was used in the analysis.
Social interaction test
Video recordings of the social interaction test were analysed by one trained observer using behavioural event recorder software (Sit & Wait, Faculty of Veterinary Sciences of Szent Istvan University, Budapest, Hungary). The ethogram used was as described by Sams-Dodd. 23 The frequency and total duration of each behaviour over the 10 min experimental period was recorded for each rat.
Statistical analysis
Statistical analyses were conducted using SPSS (Version 21; SPSS Inc, Chicago, IL, USA). Numerical data were expressed as mean ± standard error of the mean. Statistical analysis of the percentage weight loss of the toy and the sociability test data were analysed using a paired student’s t-test. In-cage behavioural data and grimace scores could not be assumed to be normally distributed and hence were analysed non-parametrically using the Friedman test. Post-hoc analyses were performed using the Wilcoxon matched-pairs test. Results were deemed significant when P was less than 0.05, but where appropriate, a Bonferroni correction was performed to account for multiple comparisons.
Results
Behavioural assessment scoring
Mean ± SEM of the frequency of selected behaviours before and during mucositis disease progression scored during the dark phase of the circadian cycle.
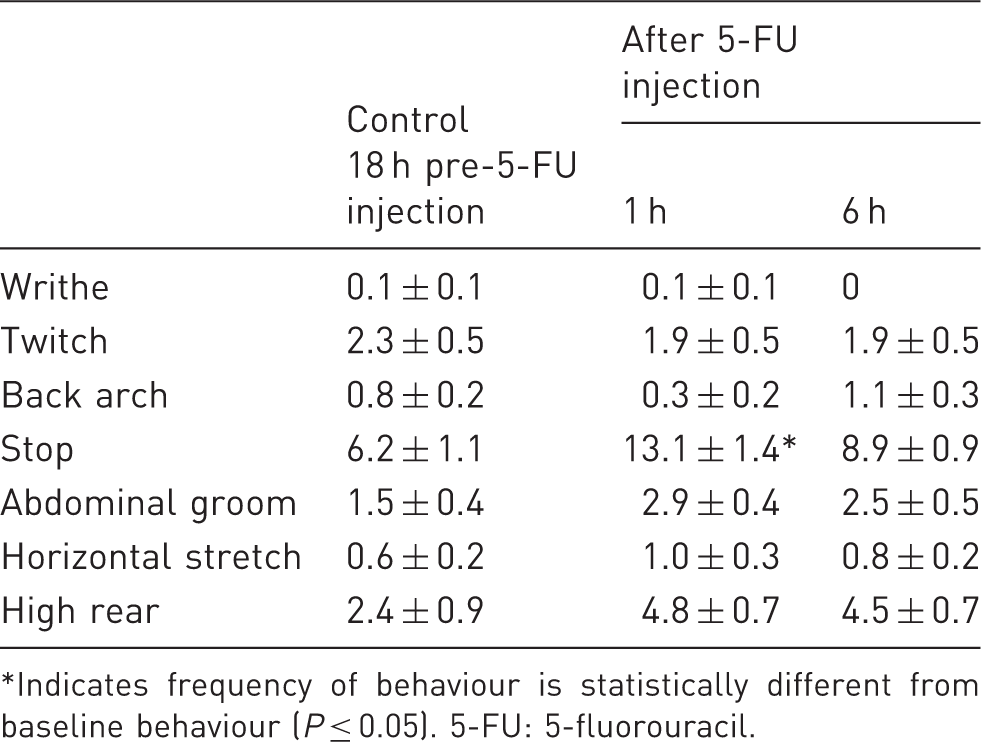
Indicates frequency of behaviour is statistically different from baseline behaviour (P ≤ 0.05). 5-FU: 5-fluorouracil.
In the days following mucositis induction, changes in frequency or duration of a selected number of behaviours were detected. Other behaviours in the ethogram were scored but showed no statistically significant changes and hence these data are not presented. Behaviours which were subject to changes included sleep and back-arch duration, and frequency of transient cessation of activity (stop), writhing, twitching and back arching (Figures 1 and 2). The general trend was that all behaviours increased in frequency or duration over time, with the exception of duration of sleep which decreased. There was also a decrease in the duration of sleep bout over time (i.e. duration of all occurrences/frequency). This showed statistical significance from the baseline value by 48 h post-chemotherapy injection (209 ± 12 s and 133 ± 15 s respectively; P = 0.002). All of these behaviours except for stop frequency demonstrated a statistically significant change in the value of the behaviour by 48 h after mucositis induction (Figures 1 and 2). Significant changes in values from baseline were often not exhibited until 24 h after chemotherapy injection and values were then deviated further from baseline at 48 h post-injection.
Mean ± SEM of the duration of (A) back arches; (B) sleep before and during mucositis disease progression scored during the light phase of the circadian cycle. Bar data not sharing the same letter are significantly different (P ≤ 0.05; Bonferroni corrected). Mean ± SEM of the frequency of (A) writhing; (B) occurrences of cessation of activity (stop); (C) twitching; and (D) back arch before and during mucositis progression scored during the light phase of the circadian cycle. Bar data not sharing the same letter are significantly different (P ≤ 0.05; Bonferroni corrected).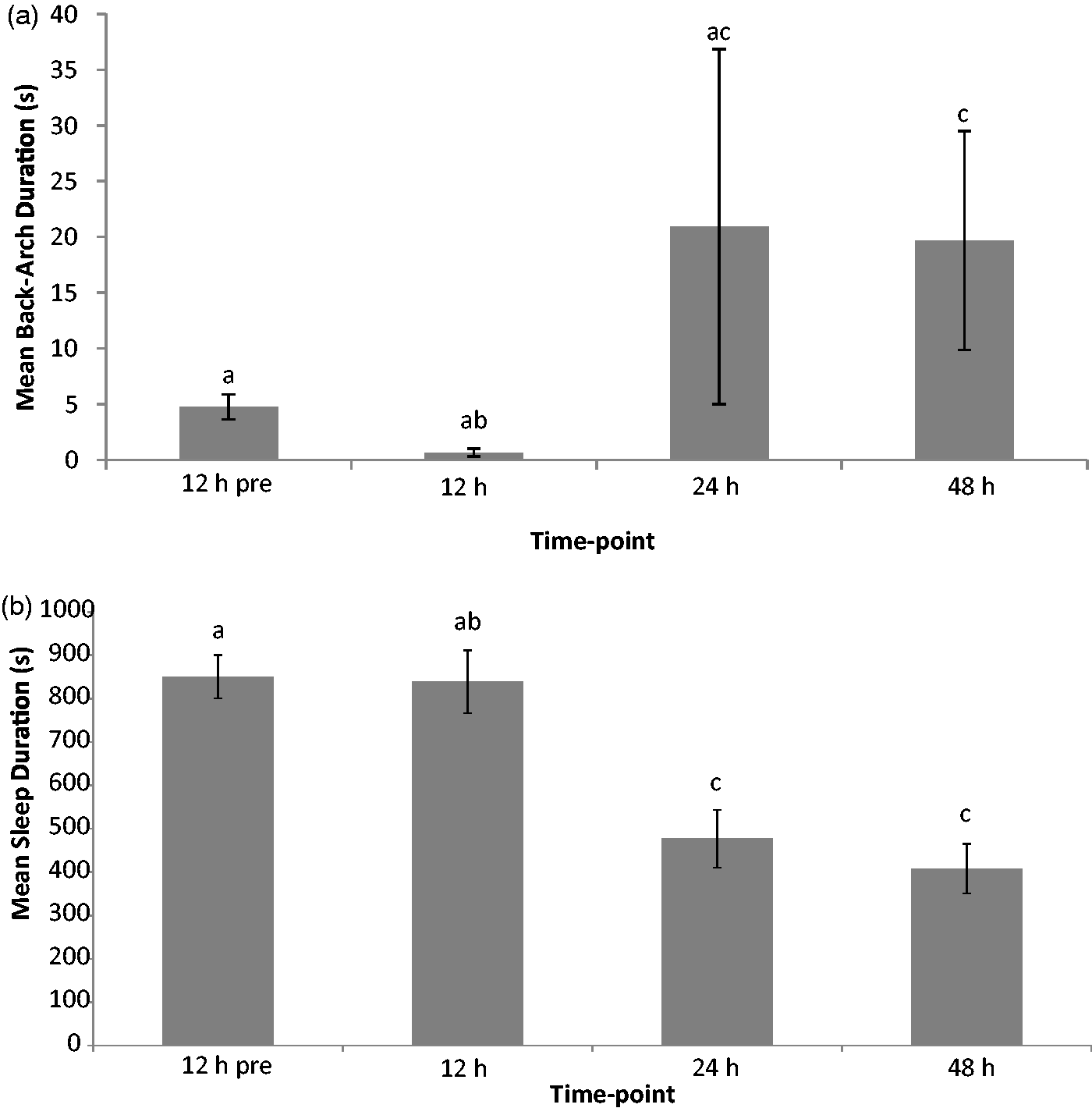
Enrichment interaction
All animals made use of the chew toys provided (see Figure 3), although the percentage loss of bone weight over the study period was minimal.
Nylabone chew toy at the completion of the study.
Each day, rats (post-5FU injection) gnawed a greater percentage of their nylabone enrichment than saline-injected control rats (0.13 ± 0.16 and 0.83 ± 0.22 respectively; P = 0.046).
Rat grimace scores
Mean grimace score, averaged across all time-points, was 2.9 ± 0.2 (out of a maximum of 6). There were no significant differences between scores at any of the time-points (P = 0.19).
Social interaction test
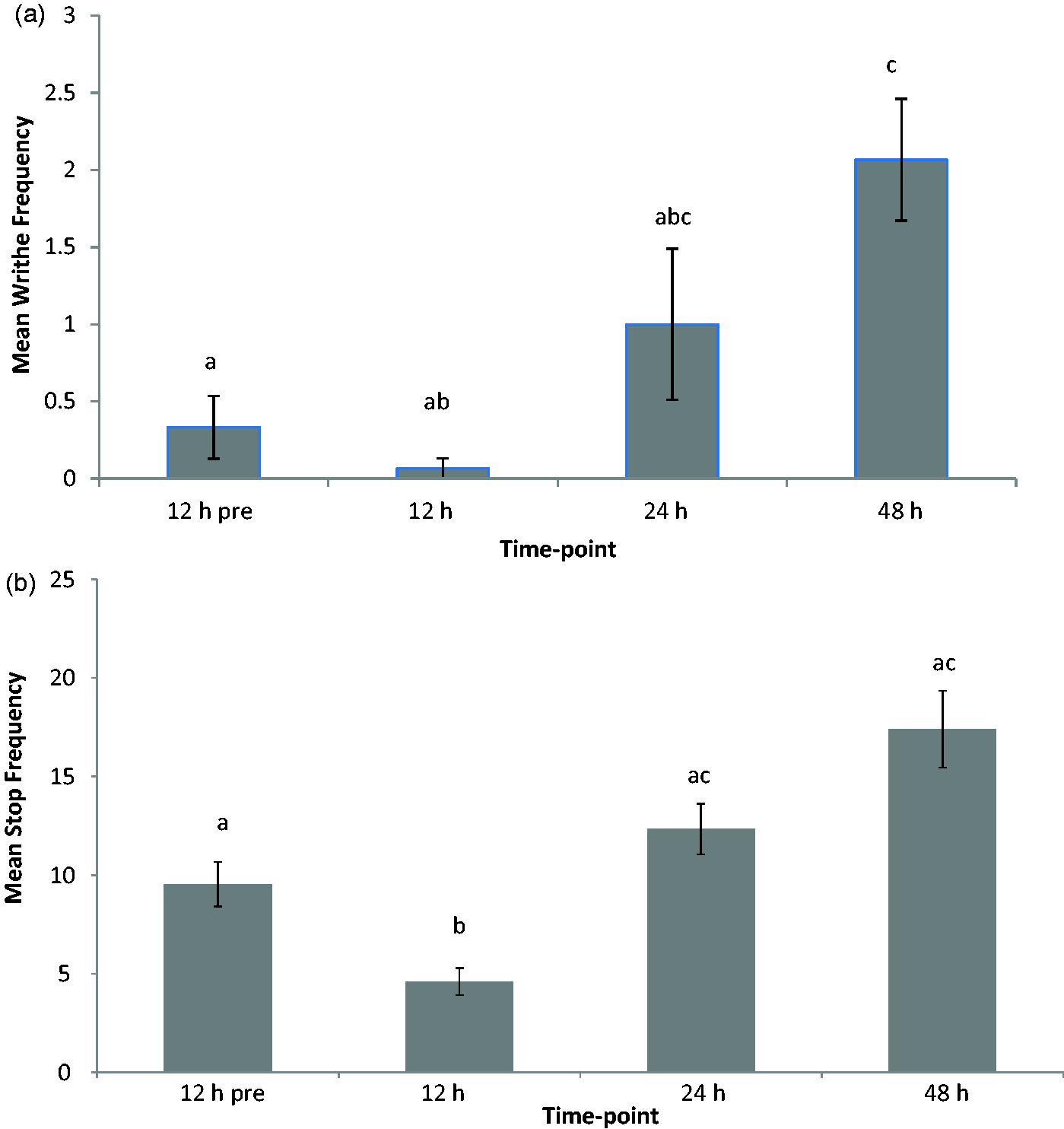
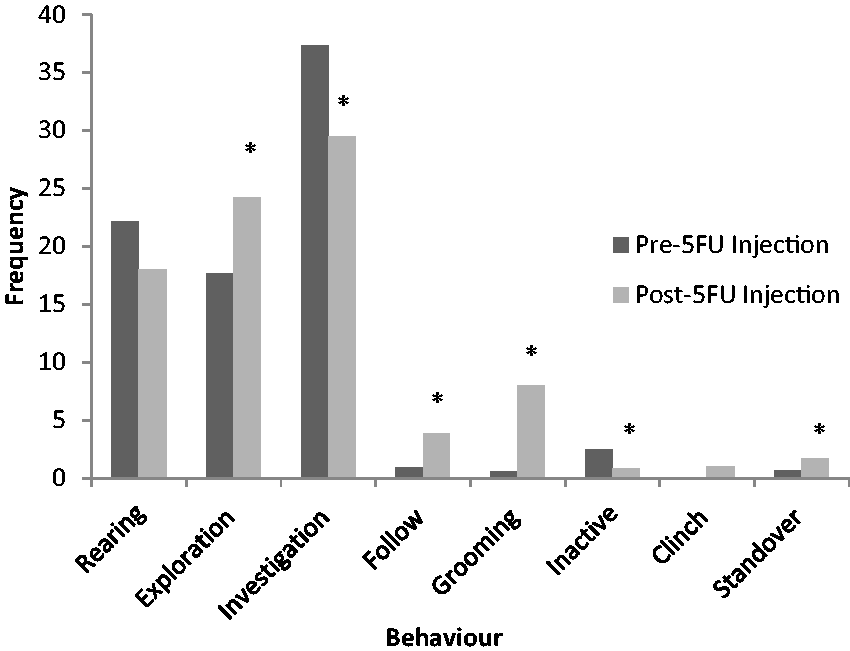
After 5-FU injection, rats spent a greater proportion of their time engaged in affiliative behaviours such as investigation, and following of the other rat (Figure 4). Time spent in grooming was also increased. Frequency data were shown separately due to the weak correlation demonstrated between investigation behaviour frequency, and the duration of the behaviour (r = 0.24, P = 0.37) (Figure 5). This negative correlation was manifested as a decreased frequency of investigation events in comparison to the healthy animal and a corresponding increase in exploration frequency. As illustrated from the video footage, this likely reflected the frenzied behaviour shown by the experimental rat to interact, and to remain with its novel conspecific, hence reducing the number of individual interaction events.
Graph of mean percent time spent engaging in categories of behaviour during 10 min sociability test (n = 16). *Denotes significant difference from the same baseline behaviour in non-mucositis affected animals (P ≤ 0.05). Graph of mean frequency of engagement in categories of behaviour during the 10 min sociability test (n = 16). *Denotes significant difference from the same baseline behaviour in non-mucositis affected animals (P ≤ 0.05).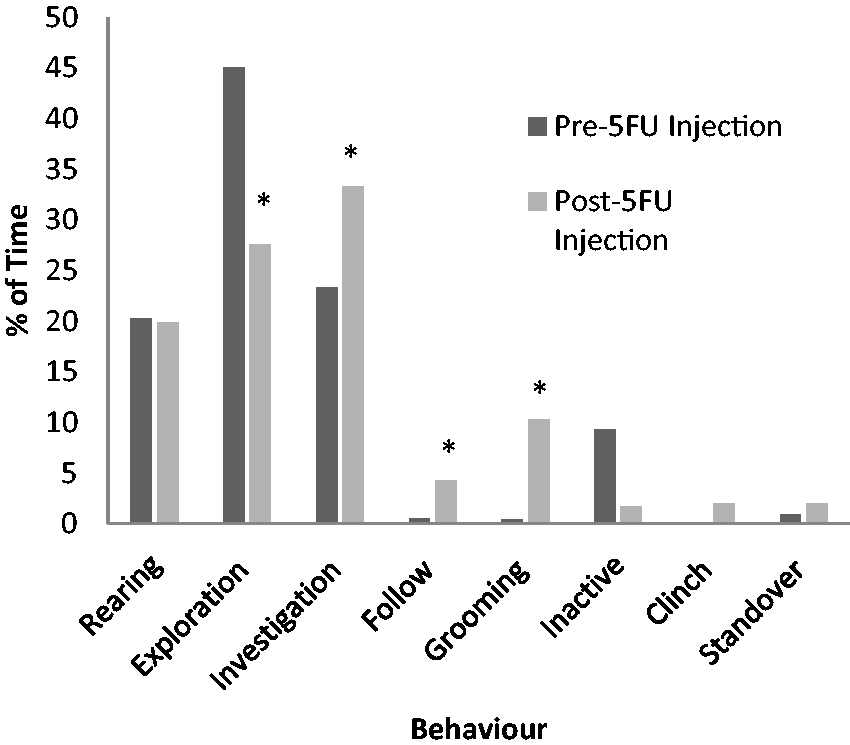
Discussion
This exploratory study was designed to establish whether a number of behavioural assessment tools, previously determined to represent abdominal pain, could be employed in rats believed to be suffering from abdominal pain caused by intestinal mucositis. Methods evaluated included behavioural assessment scoring, facial grimace scoring, and the social interaction test, in addition to enrichment interaction. In summary, writhing, twitching and back-arching behaviours were most evident in rats affected by mucositis and were increased in frequency by 48 h after chemotherapy injection compared with baseline, implying that pain onset occurred around this time-point. In the social interaction test, the social behaviour ‘investigation’ was also increased following disease onset. Rats with the mucositic condition were found to gnaw a greater percentage of their nylabone enrichment by weight than the saline-injected control rats.
Behaviours that had been previously reported post-laparotomy,2,13 were observed both before and after chemotherapy injection; including twitching, writhing, rearing and stretching, among others. We did not identify any novel behaviours that could have been specific to this type of pain. While comparing between studies is challenging, general activity levels are likely to be slightly reduced in comparison to previous studies in both the healthy and diseased animal. For example, in the current study, during the dark period of the circadian cycle, healthy animals performed high rearing and abdominal licking at a mean frequency of 2.4 and 1.5 (Table 2) in comparison to 8.1 and 4.1 respectively, in the study by Roughan and Flecknell, over a 30 min observation period. 2 Activity levels were not however reduced to the extent that behavioural changes could not be identified. A general lack of behavioural activity was similarly reported in a rat abdominal tumour growth model. 27 This inactivity may be associated with moderate or severe pain. 9 However, given that in the current study low activity levels were apparent in healthy animals, this is more likely to have been brought about through the nature of the metabolism-cage housing set-up. It has previously been reported that singly-housed animals in metabolism cages have reduced activity in comparison to animals housed alone in standard open-top cages. 28 Animals in this study were also orally gavaged and singly housed as part of the biomedical study of which they were also a part. This has the potential to be a confounding influence since both of these conditions may modify behavioural responses. However, Turner et al. performed a welfare investigation of oral gavage and demonstrated no significant changes in corticosterone levels, body weight, food consumption or adrenal gland:body weight ratios, implying that welfare was not negatively affected by gavage. 24 Furthermore, if as suggested in previous studies, the behaviours observed are truly pain-specific then oral gavage would not be expected to impact on them greatly since it can be assumed to be a distressing rather than a painful insult. In investigating the same animals, using a repeated measures experimental design, these factors have been controlled for since in both arms of the experiment (pre- and post-chemotherapy injection) animals were orally gavaged and singly housed. This design does however pose an issue when comparing these data with that obtained in future studies. Researcher evaluation of behavioural indices should consider the animals’ environment and associated procedures when undertaking any form of behavioural study. Furthermore, a pilot study should be performed to account for intricacies of experimental design unique to the model used.
Despite this reduced activity some behaviours were identified that showed a significant change from baseline values following chemotherapy injection. Interestingly, all behaviours except transient inactivity remained statistically unchanged in frequency during the period of dark cycle observations at 1 and 6 h after the 5-FU injection (Table 2). This could imply that animals were not experiencing any, or only minor, pain during these few hours after injection, including any pain or discomfort arising as a result of the injection itself. This is feasible since substance absorption via the intraperitoneal route of administration could be 50% slower than the intravenous route, 29 and may also be subject to hepatic metabolism before becoming available systemically. 30 Thus the commencement of intestinal damage could occur after 6 h. Alternatively, the absence of significant changes in behavioural frequency may have resulted from the reduced ability of the behavioural coder to identify these subtle behavioural changes from the video recordings filmed in dim light. Given this possibility, we feel that behavioural scoring conducted in the light part of the diurnal cycle is preferable.
Behavioural observations conducted in the light cycle and continuing until 48 h after chemotherapy injection identified a number of behaviours which showed statistically significant changes in their frequency and/or duration. These included sleep and back-arch duration and writhe, stop, twitch and back-arch frequency. The most convincing of these behaviours for future study, based on their ability to chart disease progression, are the duration of sleep bouts, and frequency of back arching, twitching and writhing. These data tended to show significant changes from baseline at 48 h post-chemotherapy injection, and thus this may represent the day after injection when pain is most likely to be experienced. This contradicts body weight loss data in the 5-FU mucositis models, since a loss is typically recorded from 24 h after chemotherapy injection, and the loss becomes progressively greater until day 8 of the study. 8 This finding implies that body weight measures, while quite possibly indicating sickness behaviour are not necessarily associated with the presence of pain. Duration of sleep decreased significantly, by almost half, from 24 h after chemotherapy injection. This implied reduced sleep efficiency since less total time was spent sleeping. We also demonstrated a decrease in the duration of sleep bouts, implying a greater level of restlessness. These changes to sleep patterns in the face of pain are corroborated by previous studies using brain-implanted electrode electroencephalogram (EEG) recordings.31,32 These findings render sleep valuable as a behavioural marker since it is relatively simple to observe and measure. However, it may not be specific to pain and is likely to be influenced by analgesic agent administration; thereby making its use limited in studies comparing analgesic agents. Frequency of the behaviours twitch, back arch and writhe are therefore most useful for further studies of pain in chemotherapy-induced mucositis, such as the 5-FU model presented here.
The finding that rats proportionately gnawed more of their chew toy post-5-FU injection was unexpected since it has been shown that rats experiencing stress interact less with their environments. 33 Possibly, an increase in gnawing may have resulted as a redirected behaviour in the face of a painful insult. The gnawing of the bone may act as a distraction from any pain the animal is experiencing. Redirected oral behaviours have been reported in rats as a result of the non-specific (but likely painful) stimuli of tail-pinch and tail electrical stimulation. 34 However, there do not appear to be any reports of such redirection as a specific response to pain; and this would be an interesting area for further investigation. Nevertheless, in view of the small mass of bone gnawed this test is not likely to show disease progression with any accuracy, and would require further evaluation to determine whether such a response is repeatable, and that it is indeed attributable to pain.
The rat grimace scale has been shown to be a reliable tool for evaluation of pain in rats.15,35 We failed to show any change in grimace score between any of the time-points investigated. This could imply that the rats were not experiencing any pain. However, it is more likely that the time-points chosen were too long after the onset of pain. There is a paucity of studies evaluating grimace scores in rats, however the murine literature, 14 and the study of Sotocinal et al. have suggested that the grimace scale is only effective at detecting pain for up to approximately 6 h after the initial insult. 15 This does not necessarily indicate that pain is no longer present, but reflects the evolutionary adaptation to inhibit a pain face to reduce the likelihood of predator interest. 15 In the alternative, Liao et al. have demonstrated changes in rat grimace score over a period of days in an orthodontic model of experimental tooth movement. 35 However, it is possible that the area of pain localization is important in grimace scoring, since it would be expected that oral pain is more likely to influence facial expression due to its proximity to the muscle effecting the facial change, than pain of a visceral origin.
We also encountered practical problems in assessing the grimace scores that may have impacted on the assessment scores we obtained. Previous studies have removed animals from their home-cages and placed them in clear cubes for filming.15,17 Since we used continuously recorded video footage of the rats in their home-cages our image resolution was likely to be poorer. Due to this, we were unable to score the action unit for whisker change and as a consequence scored out of a maximum of 6 which likely reduced the sensitivity of the test. Additionally, it was not possible to score the images during the dark period of the circadian cycle when animals may have shown facial expression changes due to the close proximity in time to chemotherapy drug administration.
Rats with mucositis demonstrated an increase in social interaction (investigation), following behaviour and grooming in comparison with the values in the healthy animals. To the video coder this increase in social interaction was brought about by frenzied actions to remain in close contact with the novel rat which has not been reported in previous studies. The extent of this activity was so great as to allow the observer to quickly identify group membership from the video footage despite blinding. Such an increase without a corresponding increase in motor activity has been deemed to indicate an anxiolytic effect. 36 However, this explanation appears counterintuitive in the face of mucositis.
Isolation has been shown to increase social interaction, 37 although these rats were housed alone for the pre-injection test and had been singly housed throughout the acclimatization period so the only changed factor was the disease presence. It has been shown in male mouse sibling pairs that physical affiliative contact correlates with an increase in nociceptive threshold since stimuli provided by the partner induces analgesia. 38 This response appears to be opioid-mediated since the pain reactivity change was blocked by naloxone. 39 Relatedness or previous contact does however seem to be an important factor in this response and there are no reports of this phenomenon occurring in animals that are completely unknown to each other, as in the current study. Nevertheless, the notion that physical proximity to another animal may mediate pain responses through a self-analgesic, opioid-mediated mechanism, remains an intriguing hypothesis for further investigation.
This study has evaluated a range of tools that could be employed to indicate pain or distress in a visceral, but non-surgical model; intestinal mucositis. We believe this to be the first rat study to assess behaviours in an abdominal pain model not involving laparotomy. The parameters that show most promise for future investigation are behavioural assessment scoring and specifically the frequency of twitching, writhing and back-arching behaviours. Future studies should confirm that these behavioural changes are pain-related by applying an analgesic test which would be hypothesized to attenuate the behaviour. While we failed to detect any changes in the rat grimace scores over the duration of the study it would be valuable to repeat these assessments, making more frequent observations over a shorter time period during early disease onset to avoid data confounding by the possible inhibition of the pain face over time.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
