Abstract
Practical relevance:
Cutaneous immune-mediated diseases in cats can present with clinical signs of erythema, hair loss, scaling/crusting and erosions with or without pruritis, mimicking many differential diagnoses such as hypersensitivity dermatitis and infections (eg, parasitic and fungal dermatitis). Veterinarians are encouraged to become familiar with the characteristic clinical features of various immune-driven skin disorders to permit early diagnosis and appropriate treatment.
Clinical challenges:
Diagnosis and therapeutic management of feline immune-mediated skin diseases can be challenging. Although immune-mediated skin diseases in cats are rare, these can be associated with severe systemic clinical signs, which can lead to poor quality of life and owners electing euthanasia.
Aims:
In the first of a two-part article series, the clinical presentations of feline immune-mediated skin diseases related to the pemphigus complex, cutaneous lupus erythematosus, and thymoma- and non-thymoma-associated exfoliative dermatitis are reviewed and differential diagnoses, diagnosis and therapeutic approach are discussed. In Part 2, the equivalent is discussed for erythema multiforme, f Stevens-Johnson syndrome/toxic epidermal necrolysis spectrum, plasma cell pododermatitis, proliferative and necrotizing otitis externa, pseudopelade, alopecia areata and auricular chondritis.
Evidence base:
The information in this article is based on a review of the existing veterinary literature.

Introduction
The terminology of immune-mediated and autoimmune diseases has been used interchangeably as closely related terms throughout the past three decades. Immune-mediated skin diseases are characterized by a common theme of dysregulation of the normal immune response, and some of these diseases are categorized as autoimmune diseases, implying a direct attack of the immune system on normal tissues. Such autoimmune attacks can be caused by autoantibodies (eg, pemphigus complex) or by autoreactive T lymphocytes (eg, cutaneous lupus erythematosus [CLE]). Immune-mediated and auto immune skin diseases in cats are rare. 1 Although feline immune-mediated diseases share similarities to humans and dogs in clinical manifestations, pathogenesis and treatment approaches, less is generally investigated and known in cats than in other species. As the spectrum of feline immune-mediated cutaneous diseases has expanded markedly over the years, veterinarians are encouraged to become familiar with the characteristic clinical features of various immune-driven skin disorders to permit early and accurate diagnosis and appropriate treatment.
Part 1 of this two-part article series discusses feline immune-mediated cutaneous diseases of pemphigus complex, lupus erythematosus, and thymoma- and non-thymoma-associated exfoliative dermatitis. The article will review the pathogenesis, signalment, clinical signs, laboratory and histopathology findings, treatment and prognosis in these feline immune-mediated skin diseases.
Feline pemphigus foliaceus
Pemphigus foliaceus (PF), which is considered the most common autoimmune skin disease in cats, is characterized by erythematous acan-tholytic pustules, erosions and crusts that predominantly affect the face, nipples and feet.
Pathogenesis and signalment
PF in humans and animals is characterized by the binding of pathogenic autoantibodies to desmosomal epidermal adhesion proteins, resulting in acantholysis and recruitment of inflammatory cells, with subsequent blistering of the superficial epidermis. 3 The auto-antibody desmosomal target in PF is well characterized in humans and dogs.3,4 In contrast, the investigations in cats are limited to the detection of antikeratinocyte IgG in cats with PF using direct and indirect immuno-fluorescence. The desmosomal target is currently unknown but likely different than in dogs due to the detection of positive immuno-staining on buccal mucosal tissue.5,6
PF has been reported in different breeds of cats (ie, domestic short- and mediumhair, Siamese, Persian and Persian-crossbred, Burmese and others) without a specific breed or sex predisposition.7–10 Based on the published extensive case series findings,7–10 PF usually affects adult cats with a median age of 5-6 years, though the range varies between studies from 5 months to 17 years.
Similar to canine PF, most cats are not found to have underlying triggers associated with PF development.7–10 Historically, there have been rare reports of proposed drug-induced PF11–13 and a single cat has been reported with both thymoma-induced exfoliative dermatitis and PF as a part of paraneoplastic syndrome (PNS). 14
Diagnostic approach
Currently, the diagnosis of feline PF is based on a combination of the following criteria: (i) history and characteristic type and distribution of skin lesions; (ii) exclusion of other acan-tholytic neutrophilic pustular dermatoses (eg, staphylococcal superficial pyoderma, pustular dermatophytosis); and (iii) supportive cytology and/or histopathology confirming acan-tholytic pustular dermatitis with acantholytic keratinocytes (Figure 2a).7–10,15 Immunologic tests (ie, direct and indirect immuno-fluorescense, serologic detection of autoanti-bodies against epithelial cell surface antigens) are part of the diagnostic criteria for human pemphigus. 3 In contrast, the sensitivity/specificity of immunologic pemphigus tests in dogs and cats is unknown and these tests are not commercially available to practitioners.5,6
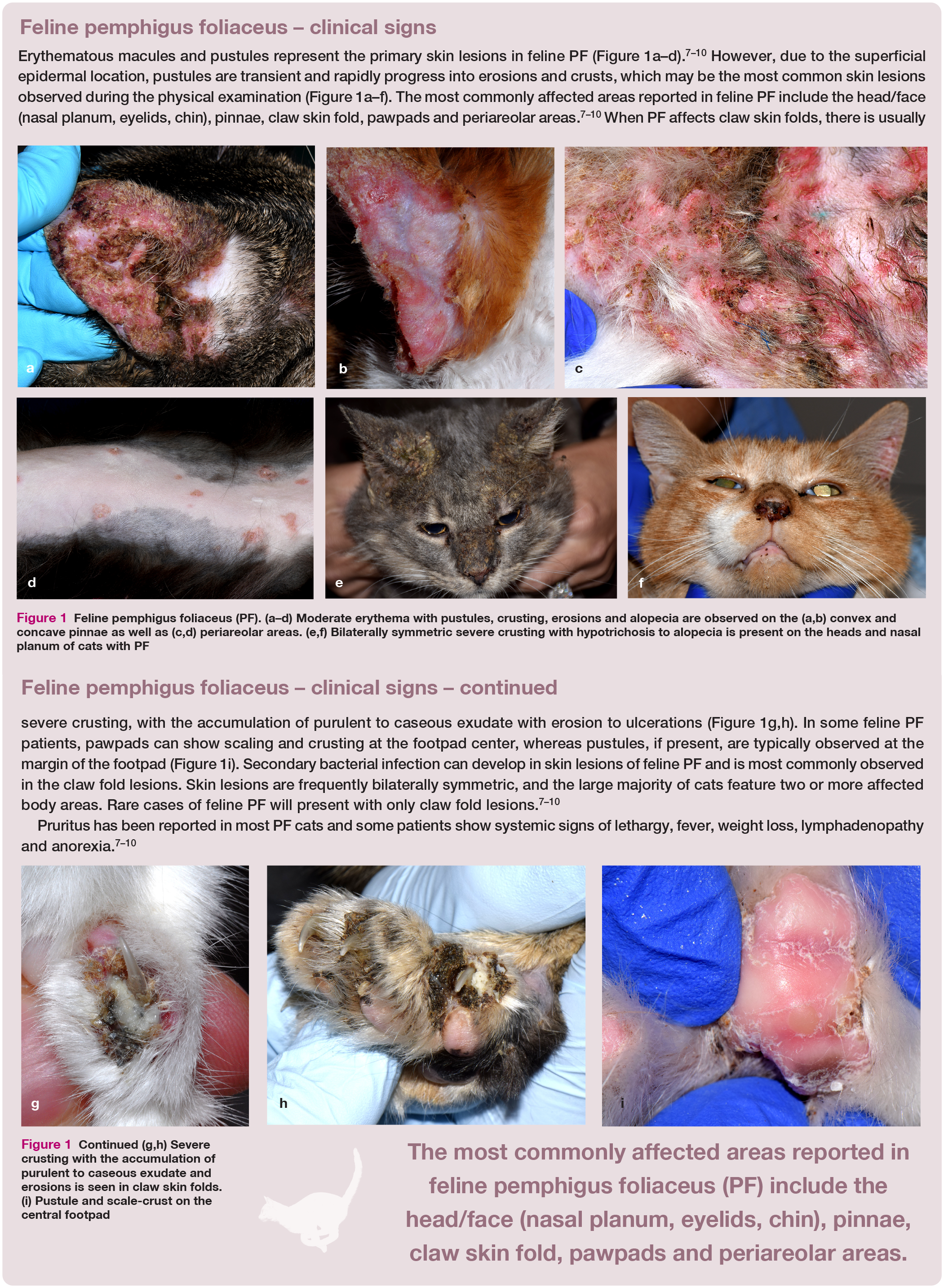

Feline pemphigus foliaceus (PF). (a-d) Moderate erythema with pustules, crusting, erosions and alopecia are observed on the (a,b) convex and concave pinnae as well as (c,d) periareolar areas. (e,f) Bilaterally symmetric severe crusting with hypotrichosis to alopecia is present on the heads and nasal planum of cats with PF (g,h) Severe crusting with the accumulation of purulent to caseous exudate and erosions is seen in claw skin folds. (i) Pustule and scale-crust on the central footpad
As mentioned, feline PF is primarily characterized by subcorneal pustules with acantholy-sis (Figure 2b) that progress into erosions and crusts. Several erosive and crusting skin diseases in cats could represent differential diagnoses; however, only PF, pustular der-matophytosis (eg, superficial fungal dermatitis due to Trichophyton species) and bullous impetigo (eg, staphylococcal dermatitis with exfoliative toxins) can cause primary subcorneal pustules with acantholysis.16-18 Pustular dermatophytosis has been reported in dogs and the skin lesions usually lack symmetry and do not affect multiple body areas.16,18 Bullous impetigo has not been described in cats19,20 and, in general, superficial pyoderma does not typically feature bilateral and symmetric distribution and/or affect multiple claw folds and pawpads.19,20 In patients with infection, aerobic bacterial culture with susceptibility testing can be considered to make the decision on the best systemic antimicrobial therapy. For patients with suspected dermatophytosis, fungal culture should be obtained by collecting crusts and hair from the skin lesions. In addition, special stains to assess the presence of bacterial and fungal organisms should be routinely performed on histopathologic slides when PF is suspected.

(a) Cytology obtained from under the crusts of a cat with pemphigus foliaceus demonstrates round acantholytic keratinocytes (asterisk) surrounded by well-preserved neutrophils (arrowhead) in the absence of cocci or nuclear streaming. Standard Diff-Quik stain, χ 100. (b) An intact subcorneal pustule (asterisk) with abundant acantholytic keratinocytes (arrowhead), active acantholysis and well-preserved neutrophils within the epidermis. Hematoxylin and eosin, bar = 50 μm

The presence of acantholysis in suspected PF lesions can be evaluated using cytology (standard Diff- Quik stain) from intact pustules, underneath newer moist crusts or from the purulent exudate in claw folds and/or biopsy of these lesions (see the ‘Tips for biopsy collection’ box). The classical cytology of PF lesions reveals the presence of acantholytic keratinocytes with variable numbers of well-preserved neutrophils and/or eosinophils (Figure 2a). Histopathologic examination features subcorneal or intragranular pustules with acantholytic keratinocytes and a neutro-philic or mixed neutrophilic and eosinophilic perivascular to interstitial infiltrate.7–10 In PF patients where it is feasible, based on clinical signs, glucocorticoids should be discontinued for at least a week before biopsy collection (see the ‘Glucocorticoid withdrawal for biopsy of suspected PF lesions’ box).

Treatment and prognosis
Although the majority of PF cats have no underlying trigger (eg, drugs), eliminating any suspect causative factor should be implemented immediately. Ultraviolet (UV) light exposure has been associated with exacerbating PF skin lesions in humans and dogs.21,22 Although there have been no reports of UV-induced flares in feline PF patients, owners should be made aware of this possibly contributing disease factor in the long-term management.
Therapeutic management of feline PF remains challenging and commonly requires immunosuppressive medications to achieve clinical remission and long-term maintenance of the disease. Monotherapy with oral gluco-corticoids has been considered the mainstay of therapy for feline PF (eg, prednisolone 2-4 mg/kg/day; triamcinolone acetonide 0.2-2 mg/kg/day; dexamethasone 0.1-0.2 mg/kg/ day).7–10 Prednisone, a prodrug metabolized to active prednisolone, is not recommended in cats due to lower absorption and/or decreased prednisone conversion to prednisolone. 23 Subcutaneous injections of methylprednisolone acetate, commonly used to treat feline allergic dermatitis, 24 have not been evaluated in feline PF.7–10 Furthermore, conflicting data exist on the use of methylprednisolone acetate in cats regarding the dosages, frequency of administration and the risk of side effects (eg, steroid-induced diabetes mellitus and congestive heart failure).24,25
Although most cats with PF achieve complete remission (absence of new lesions with healing of previous original lesions) within a few weeks of recieving glucocorticoid monotherapy, only a minority of cats (4-15%) maintain disease remission if glucocorticoid administration is discontinued.7–10 Therefore, steroid-sparing adjuvants, such as ciclosporin (5-10 mg/kg/day) and chlorambucil (0.1-0.3 mg/kg/day), have been suggested to induce earlier clinical remission and provide long-term PF disease control.7–10 In addition to the treatments mentioned above, oclacitinib (1 mg/kg q12h PO) 26 and pentoxifylline (26.5 mg/kg q12h PO) in combination with topical hydrocortisone aceponate have been shown to be beneficial for the management of feline PF in two case reports. 27
The early usage of steroid-sparing agents would potentially lower the risk of developing glucocorticoid-associated side effects, including iatrogenic hyperadrenocorticism. However, selecting the optimal steroid-sparing adjuvant in feline PF is challenging due to the lack of prospective comparative clinical trials. Clinicians need to consider that every steroid-sparing adjuvant has specific advantages and disadvantages. Most reports regarding feline PF treatment utilized oral ciclosporin or chlorambucil as steroid-sparing agents; however, both drugs can cause adverse gastrointestinal side effects (eg, vomiting, hypersalivation, anorexia, diarrhea).7–10 When treating with chlorambucil, regular bloodwork monitoring for possible bone marrow suppression and idiosyncratic liver enzyme elevation is also required.28,29
The safe therapeutic index for oclacitinib, a Janus kinase inhibitor (JAK) registered for the treatment of canine allergic dermatitis, has not been established in cats and the manufacturer has shown that over-inhibition of JAK2 cytokines may result in substantial adverse hematologic effects. 30
Historically, azathioprine (1.1 mg/kg q48h) and aurothioglucose (0.5 mg/kg/week) have been also utilized for the control of feline PF.7–10 ,31,32 However, clinicians should be familiar with azathioprine-related side effects (eg, myelosuppression at 1.1 mg/kg q48h) in cats, which may be dose-dependent, presumably due to low thiopurine methyltransferase activity.31,32 Although initial reports on azathioprine usage for managing inflammatory bowel disease revealed that lower dosages (eg, 0.3 mg/kg q48h) can be successful,30,31 starting at a lower dosage and routine blood-work monitoring are mandatory to avoid severe and fatal adverse effects.
In general, feline PF has a good prognosis, as most cats achieve complete remission with medical management (eg, glucocorticoid monotherapy) within a median time of 22-36 days.7-10 However, disease flare-ups on the maintenance therapy are frequent, especially with treatment reduction or discon-tinuation.7–10 Unlike dogs, 33 cats with PF are rarely euthanized due to progressive skin lesions despite treatment, treatment-associated adverse effects or poor quality of life.7-10
Pemphigus vulgaris
In contrast to PF, little information is available for feline pemphigus vulgaris (PV). 34 The clinical and histopathologic features of feline PV resemble canine and human PV and, therefore, a similar pathomechanism of desmosomal antibody targeting is proposed for feline PV. 34 Human and canine PV investigations have shown desmoglein-3 as the major autoantigen.35,36 Currently, the autoantibody targets in feline PV skin lesions remain unknown.
Diagnostic approach
Definitive diagnosis is based on history, clinical signs (eg, skin lesion type, mucosal involvement) and skin biopsy, which shows suprabasalar acantholysis, cleft formation and tombstoning of the basal cell layer. Multiple biopsies are needed to capture diagnostic PV areas for histopathologic examination, with intact vesicles and/or margin of erosions to ulcers with adjacent ‘normal’ skin being commonly collected. Ideally, clinical photographs should also be submitted to the pathologist to aid differentiation between deep PF and PV. Depending on the lesion location, skin cytology can also be utilized to show the presence of acantholytic keratinocytes. A positive direct Nikolskiy sign (ie, an epidermal detachment caused by mechanical pressure on the normal skin far from the lesions) or marginal Nikolskiy sign (ie, an epidermal detachment caused by mechanical pressure of normal skin at the edge of PV vesicles/erosions), observed in human 37 and canine PV, 36 should be evaluated in feline PV patients.
The possible differential diagnoses include erosive to ulcerative diseases of the oral cavity and mucocutaneous junctions such as viral diseases (eg, herpesvirus, calicivirus), chronic ulcerative stomatitis, drug reactions such as Stevens-Johnson syndrome/toxic epidermal necrolysis group and rare subepidermal blistering skin diseases.
Treatment and prognosis
Treatment of reported feline PV patients with oral glucocorticoids (4-6 mg/kg/day of pred-nisolone) resembles the approach used in humans and dogs and shows some success in controlling the disease. 34 However, in refractory cases of feline PV, steroid-sparing immunomodulators (eg, chlorambucil, ciclo-sporin) should be considered, similarly to the treatment of feline PF.

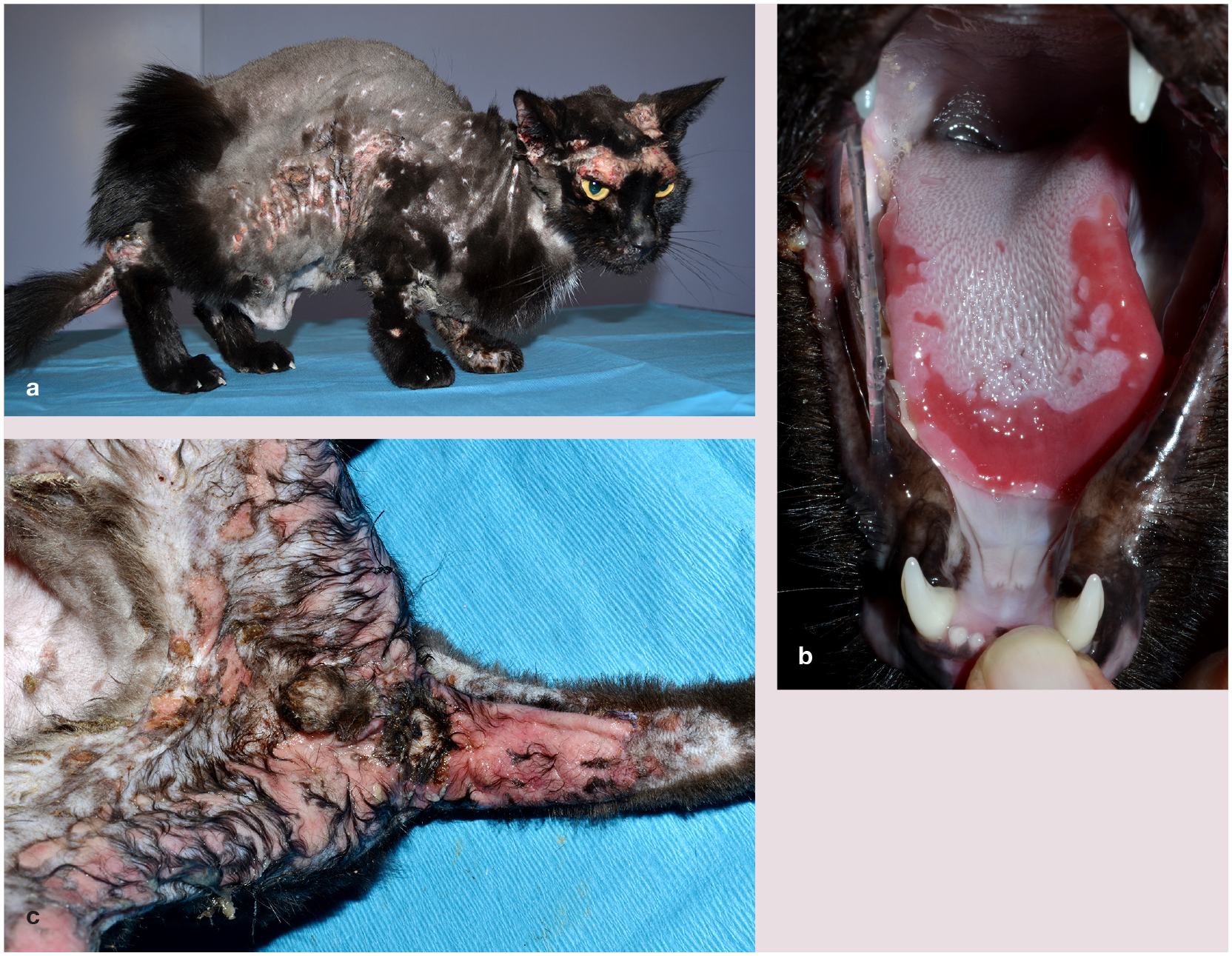
Feline pemphigus vulgaris (PV). Deep erosions to ulcerations with crusting are present in a cat with PV in the (a) periocular region, thorax and feet, (b) lips, oral cavity and tongue and (c) ventral tail, perianal and genital area. Courtesy of Luc Beco
Lupus erythematosus
CLE can affect only the skin or be present as part of a diverse range of potentially life-threatening and debilitating clinical signs in patients with systemic lupus erythematosus (SLE). 38 In contrast to humans and dogs,38,39 SLE and CLE variants such as discoid lupus erythematosus (DLE) have rarely been published in cats.40,41
Diagnostic approach
In the SLE and CLE/ DLE feline cases described in the box, histologic examination from skin biopsies revealed CLE-specific lymphocyte-rich interface dermatitis with basal keratinocyte vacuolar (hydropic) degeneration as well as lymphocytic mural interface folliculitis. The possible clinical differential diagnoses include dermatophytosis, thymoma-associated and non-thymoma-associated exfoliative dermatitis, cutaneous epitheliotropic lymphoma, erythema multiforme and PF. Definitive diagnosis is based on history, clinical signs, skin biopsy and, in cases with exfoliative dermatitis, ruling out the presence of thymoma (eg, thoracic radiographs, CT).
Treatment
Treatment of reported SLE/CLE feline patients has resembled the approach in humans and dogs,38,39 which involves sun avoidance, utilization of topical glucocorticoids/tacrolimus for localized lesions (eg, nasal lesions) and systemic immunomodulating drugs (eg, prednisolone 1-2 mg/kg q24h with taper, ciclosporin 5 mg/kg q24h, then taper) for generalized lesions.40-43


Feline cutaneous lupus erythematosus (CLE). (a) Depigmentation, deep erosions to ulceration on the nasal planum with bilaterally symmetric periocular alopecia and mild scaling/crusting. (b) Generalized head, neck and thorax alopecia with scaling and multifocal round ‘disc-like’ lesions. Courtesy of Claude Favrot
Thymoma- and non-thymoma-associated exfoliative dermatitis
An exfoliative dermatitis syndrome associated with thymoma, as well as one of a similar nature but without association with thymoma, has been reported in cats -’thymoma-associated exfoliative dermatitis’ and ‘non-thymoma-associated exfoliative dermatitis’, respectively.

Thymoma-associated exfoliative dermatitis
Thymoma-associated exfoliative dermatitis is a rare PNS where the cutaneous signs are frequently noted first, despite the likely initial presence of the neoplastic process. 46 Non-cancerous neoplasm-related skin lesions occur at a site distinct from the primary tumor or its metastases. 47 In cats, thymoma is the most common thymic neoplasm that arises from thymic epithelial cells in the cranial mediastinum and the second most frequent tumor in this anatomic location after lymphoma.48,49 While the term ‘thymoma’ has been used to denominate thymic epithelial tumors (TETs), some authors debate that this terminology can be deceptive as it often implies a benign process despite occasional malignant behavior and metastasis. 48 In general, feline thymomas are benign and remain confined to the cranial mediastinum; however, metastases to the pericardium, trachea, lungs, thoracic wall, diaphragm, lymph nodes, liver, spleen and kidney have been reported.50,51 In addition, thymomas have been associated with exfoliative dermatitis, PF 14 and other PNSs such as myasthenia gravis, myocarditis, autoimmune granulocytopenia and polymyositis.52,53
Pathogenesis and signalment
The pathogenesis of feline thymoma-associated exfoliative dermatitis has not been elucidated, but it is suspected to be an immune-mediated process similar to graft-vs-host disease.58,59 It has been proposed that thymoma-associated exfoliative dermatitis in cats is the result of a CD3+ T-cell-mediated process caused by abnormal antigen presentation of neoplastic thymic epithelial cells that cross-react with epidermal keratinocytes.55,59 In humans with thymoma-associated paraneoplastic autoimmune multi-organ syndrome with simultaneous occurrence of myasthenia gravis and PF, it appears that loss of tolerance by autoreactive Τ cells in the neoplastic thymus leads to the production of autoantibodies that cross-react with homologous epidermal antigens. 60 Moreover, the number of regulatory Τ cells in thymomas are inversely correlated with the occurrence of associated autoimmune disorders. 60


(left column) Thymoma-associated and (right column) non-thymoma-associated exfoliative dermatitis. Dry large adherent scales with alopecia and mild hyperpigmentation on the thorax of cats with (a) thymoma-associated and (b) non-thymoma-associated exfoliative dermatitis. Multifocal alopecia and hypotrichosis with thick adherent scales on the head, face and preauricular areas in cats with (c,d) thymoma-associated and (e) non-thymoma-associated exfoliative dermatitis. (f) Footpad and interdigital skin involvement with scaling in a cat with thymoma
This disease usually affects middle-aged to older cats; however, it has been reported in cats as young as 4 years of age. There are no reported sex or breed predispositions.46,49,56
Diagnostic approach
The diagnosis of thymoma-associated exfoliative dermatitis can be considered based on history, clinical and histopathologic findings and the presence of a cranial mediastinal mass on imaging examination. Cutaneous cytology assists in the diagnosis of secondary infections, and in the absence of acantholytic cells rules out PF. For diagnostic purposes, multiple areas of the skin should be biopsied, making sure to include scales, crusts and exfoliative lesions. 45 It is important to note that, as mentioned earlier, the skin should not be prepped prior to biopsy in order to preserve the lesions intact for sampling. Histopathology demonstrates: marked orthokeratosis to focal parakeratosis; mild to moderate epidermal hyperplasia, with hydropic degeneration of basal cell keratinocytes and transepidermal apoptotic keratinocytes; cell-poor to cell-rich interface dermatitis composed mainly of CD 3 + lymphocytes with fewer plasma cells and low numbers of mast cells and neutrophils; occasionally, infundibular lymphocytic interface inflammation, or infiltrative mural folliculitis with scattered basal cell apoptosis; pigmentary incontinence; and absent or atrophic sebaceous glands, which may contribute to the hyper-keratosis (Figure 6).44,45,47,50,56

Histopathologic examination in a cat with thymoma-associated exfoliative dermatitis shows characteristic changes of severe, orthokeratotic hyperkeratosis with lymphocytic cytotoxic dermatitis (interface changes with individual keratinocyte apoptosis [arrowheads] occurring at all levels of the living epidermis [asterisks]) and loss of sebaceous glands. Hematoxylin and eosin, bar = 50 μm
A range of thoracic radiology techniques, including ultrasound, CT and/or MRI, have been reported to reveal cranial mediastinal masses, which are usually suggestive of thymoma or thymic lymphoma.49,56,61 In the authors’ experience, radiographs are also useful in diagnosing cranial mediastinal masses as the thymus is not visible on thoracic radiographs in normal cats. Subsequently, transthoracic ultrasound-guided fine needle aspiration and biopsy of the cranial mediastinal mass can be performed. 49 Cytology of the aspirate usually reveals predominantly thymic epithelial cells and small mature lymphocytes with varying amounts of mast cells.44,55,57 In humans, CT has been the modality of choice for imaging the cranial mediastinum. Most importantly, for surgical planning purposes, advanced imaging such as CT or MRI is recommended and helps to distinguish space occupying from locally invasive masses. 61
Treatment and prognosis
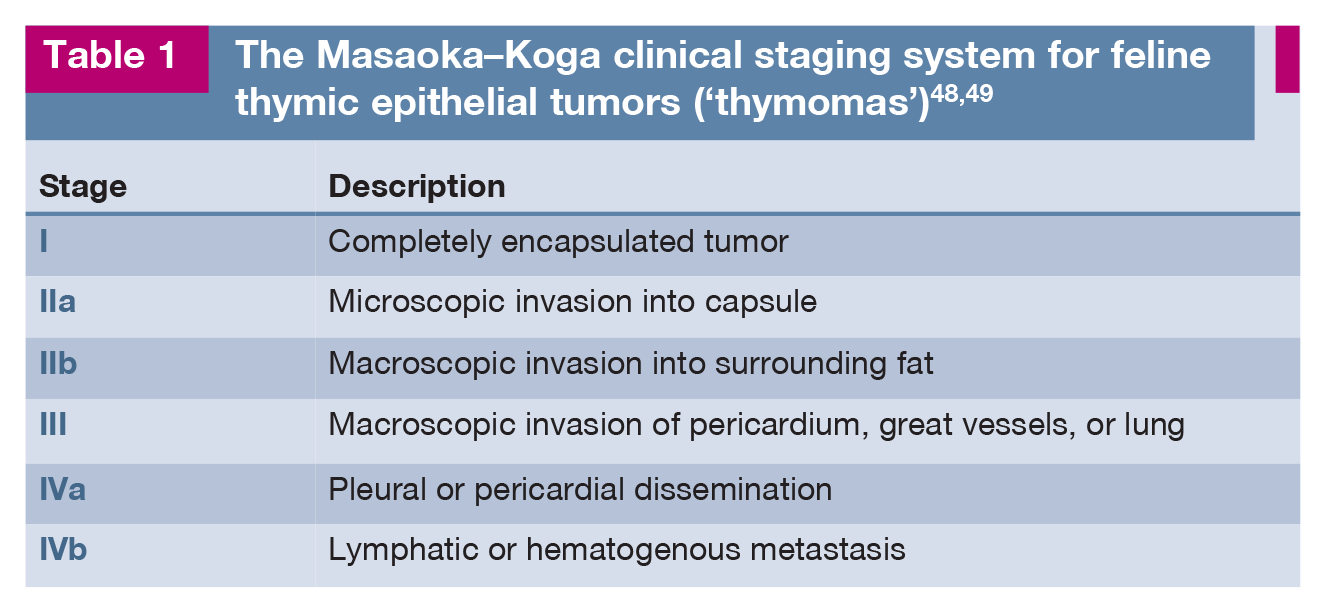
The diagnosis of paraneoplastic pathogenesis can only be proven by resolution of the skin lesions following surgical excision of the tumor. 58 In humans, a clinical staging system for TETs has been described based on the degree of tumor invasion into the capsule and surrounding tissues, and lymphatic and hematoge-nous metastasis.62,63 The Masaoka- Koga staging system is useful in determining treatment recommendations and prognosis in cats with thymoma (Table 1).48,49 In most cats with suspected thymoma-associated exfoliative dermatitis, tumor removal is the treatment of choice. 52
Adjuvant multimodal treatment is attempted when surgical margins are incomplete or local recurrence or metastatic disease is identified. Various chemotherapy protocols have been used; however, efficacy of this treatment modality is questionable.49,64 In contrast, radiation therapy alone or combined with surgery have shown promising results with rapid tumor reduction observed in cats submitted to adaptive radiotherapy.48,65
The prognosis for animals with non-invasive, resectable thymomas is good and skin lesions will gradually resolve after tumor excision.14,59 In cats treated by surgical excision alone, a 3-year overall survival rate of 74% has been observed. 61 In contrast, cats with invasive thy-momas have higher rates of recurrence and, postoperatively, mortality ranging from 11% to 22%.48,61,64 Many cases with large thoracic masses and severe debilitation are euthanized. 46
Non-thymoma-associated exfoliative dermatitis
A similar exfoliative dermatitis syndrome to thymoma-associated exfoliative dermatitis has been reported in cats but without a determined etiology.58,66,67 This condition is referred to as non-thymoma-associated exfoliative dermatitis because the skin lesions (Figure 5b,e) and histopathologic examination are indistinguishable from thymoma-associated exfoliative cases. In cats that had long-term follow-up, there was no development of thymoma. 58
Like for thymoma-associated exfoliative dermatitis, an immune-mediated process is suggested for non-thymoma exfoliative dermatitis, with CD3+ T-cell infiltration and epidermal cytotoxicity on histologic examination. 66 At the time of writing, a total of 20 cases have been reported in the literature.58,66,67 Taken together, most affected cats are middle-aged to older, with no apparent sex or breed predisposition.58,66,67 However, out of the initial case series, 3/18 cats were Somali, with two being littermates and living in the same household. 58 For the two isolated case reports,66,67 despite the presence of skin lesions, the cats did not display signs of general illness.
Diagnosis
In one of the studies described above, 58 12/18 cats presented with marked lethargy and depression. In addition, four cats presented with inconclusive abnormalities such as lymphoplasmacytic hepatomegaly, splenomegaly, lymphadenopathy or pancreatitis. Changes in the blood cell counts and serum biochemistry were also inconclusive and inconsistent. 58 All cats were tested, and were negative, for FIV and FeLV.58,66,67 PCR for feline herpesvirus DNA was negative in tested cats. 58
As already mentioned, for all cats with exfoliative dermatitis, imaging examination is crucial to confirm or refute the presence of a cranial mediastinal mass as the treatment protocol radically changes for non-thymoma-associated exfoliative dermatitis. 66
Management and prognosis
Cats with non-thymoma-associated exfoliative dermatitis respond to immunosuppressive therapy and most patients require long-term therapy to maintain remission.58,66,67
Most cases achieve remission with modified ciclosporin (6.75-7.5 mg/kg q24h) alone or combined with prednisolone (2-4 mg/kg q24h).58,66,67 Cats that occasionally do not respond to initial treatment with ciclosporin and/or prednisolone can still respond to dexamethasone (0.2-0.5 mg/kg q24h). 58 If glucocorticoids alone have controlled the disease, a gradual transition to ciclosporin alone, or combination therapy with low doses of glucocorticoids to maintain remission, is recommended by the authors. Topical therapy with anti-seborrheic shampoos and oral antibiotics have also been used with variable response.58,67 Relapse may occur, especially if the immunosuppressive therapy is discontinued.58,66,67 In contrast, one cat has been reported to go into spontaneous remission. 58
Key Ponits
✜ The history and physical examination findings of any cat presenting with clinical signs of erythema, hair loss, scaling/crusting and erosions with or without pruritus should be screened for signs that are less consistent with common skin-restricted dermatoses (eg, hypersensitivity dermatitis, parasitic and fungal dermatitis) and that may raise suspicion for cutaneous immune-mediated diseases. In this scenario, a broad knowledge of cutaneous immune-mediated dermatoses will help with recognition of less consistent historical or lesional clues to raise the profile of specific immune-mediated diseases, and accurate diagnostic skills and tools will help exclude common differential diagnoses.
✜ Cytology and skin biopsies are often the most important tools to diagnose cutaneous immune-mediated diseases and distinguish between infectious, hypersensitivity and neoplastic causes.
✜ PF is considered the most common immune-mediated, vesicobullous to pustular skin disease of cats.
✜ Therapeutic management of feline PF remains challenging and commonly requires a long-term combination of immunosuppressive medications (glucocorticoids and steroid-sparing agents); this can be frustrating due to cost, recurrence of the illness and possible adverse effects of the medications.
✜ In contrast to humans and dogs, studies of SLE and CLE, such as DLE, have rarely been published in cats.
✜ Exfoliative dermatitis, a clinical syndrome characterized by severe, generalized skin desquamation with occasional erythema, has been reported in cats with or without association with thymoma.
✜ The prognosis for cats with non-invasive, resectable thymomas is good and skin lesions will gradually resolve after tumor excision.
✜ Cats with non-thymoma-associated exfoliative dermatitis respond to immunosuppressive therapy but most patients require long-term therapy to maintain remission.
Footnotes
Correction (May 2025):
This article has been updated to include the current address of Paulo Gomes.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors of this commissioned Clinical Spotlight review received an honorarium; as for all JFMS articles, this Clinical Spotlight article went through peer review.
Ethical approval
This work did not involve the use of animals and therefore ethical approval was not specifically required for publication in JFMS.
Informed consent
This work did not involve the use of animals (including cadavers) and therefore informed consent was not required. For any animals or people identifiable within this publication, additional informed consent for publication was obtained.