Abstract
Practical relevance
Cutaneous immune-mediated diseases in cats can present with clinical signs of erythema, hair loss, scaling/crusting, and erosions with or without pruritus, mimicking many differential diagnoses such as hypersensitivity dermatitis and infections (eg, parasitic and fungal dermatitis). Veterinarians are encouraged to become familiar with the characteristic clinical features of various immune-driven skin disorders to permit early diagnosis and appropriate treatment.
Clinical challenges
Diagnosis and therapeutic management of feline immune-mediated skin diseases can be challenging. Although immune-mediated skin diseases in cats are rare, these can be associated with severe systemic clinical signs, which can lead to poor quality of life and owners electing euthanasia.
Aims
In the second of a two-part article series, the clinical presentations of the feline immune-mediated skin diseases erythema multiforme, Stevens-Johnson syndrome/toxic epidermal necrolysis spectrum, plasma cell pododermatitis, proliferative and necrotizing otitis externa, pseudopelade, alopecia areata and auricular chondritis are reviewed and differential diagnoses, diagnosis and therapeutic approach are discussed. In Part 1, the equivalent is discussed for feline immune-mediated skin diseases related to the pemphigus complex, cutaneous lupus erythematosus, and thymoma- and non-thyoma-associated exfoliative dermatitis.
Evidence base
The information in this article is based on a review of the existing veterinary literature.

Introduction
In Part 1 of this two-part article series, we discussed feline immune-mediated cutaneous diseases of pemphigus complex, cutaneous lupus erythematosus (CLE), and thymoma-associated and non-thymoma-associated exfoliative dermatitis. Here, in Part 2, we review the pathogenesis, signalment, clinical signs, laboratory and histopathology findings, treatment and prognosis of further immune-mediated cutaneous diseases - erythema multiforme (EM), Stevens-Johnson syndrome (SJS)/toxic epidermal necrolysis (TEN) spectrum, pseudo-pelade/alopecia areata, plasma cell pododermatitis, proliferative necrotizing otitis externa (PNOE) and auricular chondritis.
Erythema multiforme, Stevens-Johnson syndrome and toxic epidermal necrolysis
EM had long been considered part of a spectrum of diseases that included SJS and TEN. 2 However, the current human and veterinary clinical classification defines SJS and TEN as variants of the same disease spectrum that differ from EM subsets in clinical appearance and causality.3,4 Drugs are thought to induce most cases of SJS/TEN,3-5 whereas EM is commonly associated with infectious triggers or has an unknown cause.3,4
Erythema multiforme
EM is an acute immune-mediated disorder that affects the skin and/or mucous membranes (oral cavity, genital, perianal and periocular). EM is divided into minor (EMm) and major (EMM) forms based on mucosal involvement and systemic signs of illness present in the latter.3,6,7 Only a few feline EM cases can be found in the veterinary literature.8-15
Pathogenesis
The pathogenesis of EM is still not completely understood. Still, it is likely immunologically mediated and involves autoreactive T-cell generation and activation by antigen (infectious, drug)-loaded epithelial cells resulting in epidermal damage from lysis of surrounding keratinocytes. 16 A drug causality was implicated in seven feline EM patients; however, there was preceding laryngotracheitis of unknown origin or vaccination with feline rhinotracheitis-calicivirus-panleukopenia virus vaccine reported in three of the cases.8-15 Similarly to human and canine EM patients, there are doubts regarding the accuracy of feline EM diagnosis in several reports of drug-induced EM, 12 as some of these cases may have exhibited SJS or SJS/TEN overlap and were inaccurately classified.


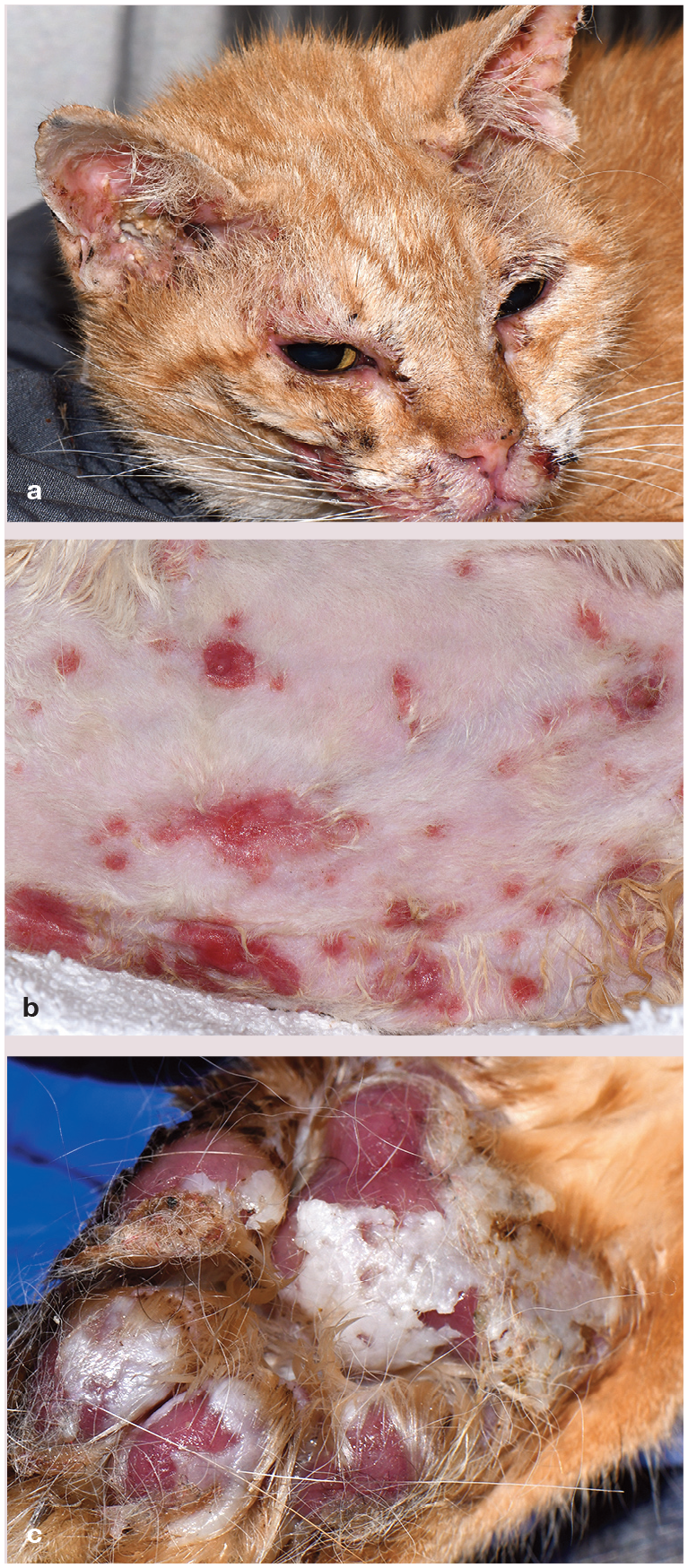
Presumptive feline herpes-associated erythema multiforme. (a) Widespread scaling, alopecia and multifocal erosions affecting the head and cranial trunk in a 2-year-old female domestic shorthair cat. (b) Erosions to ulcerations with alopecia affecting ventral periocular and perinasal areas in a 10-year-old male castrated Persian-mixed cat. Image (a) courtesy of Christine Prost and image (b) courtesy of Michele De Lucia
Interestingly, a putative feline herpes-associated EM was proposed in two cases where a feline herpes virus 1 DNA was isolated from skin biopsies of cats presenting with widespread exfoliative dermatitis, erosions, scaling and a history of recurrent upper respiratory tract infections.14,15 The lesions in human herpes simplex virus (HSV)-induced EM are virus-free (ie, viral cytopathic changes are not present) but contain HSV DNA fragments, most often comprising sequences that encode polymerase gene Pol. 16 Viral protein expression in the skin (notably Pol, rarely thymidine kinase) initiates lesion development through recruitment of a V beta-restricted population of virus-specific CD4 helper T cells, type 1, that produce interferon-gamma. 16 This early virus-specific response is followed by an amplified inflammatory cascade characterized by enhanced cytokine production and the accumulation of T cells that respond to auto-antigens, which are likely released by lysed or apoptotic virus-infected cells. 16 Although proposed as a counterpart of the human herpes-associated EM, there is a need for proposed cases of feline presumptive herpes-associated EM14,15 to be better characterized and described for viral replication and the presence of viral fragments and inclusion bodies that would differentiate between infection and ‘true’ EM-like pattern.
Diagnostic approach
The diagnosis of feline EM is based on a combination of the following criteria: (i) history and characteristic type and distribution of skin lesions based on the clinical classification; and (ii) supportive histopathology: cytotoxic interface dermatitis identifying transepidermal ker-atinocyte apoptosis with hydropic changes and dyskeratosis of basal keratinocytes (Figure 2).8-16 Importantly, histopathologic features are not pathognomonic and the diagnosis of EM across humans, dogs and cats is clinicopathologic.8-16 Depending on the biopsy site and the stage of the clinical disease, full-thickness necrosis may be the dominant lesion in a biopsy from the center of a target lesion, whereas interface dermatitis with vacuolar change of basal keratinocytes may be seen at the margin (‘zonal changes’). 16 EM may be challenging to differentiate histologically from other cytotoxic interface dermatoses in cats, particularly SJS/TEN, some cases of thymoma-associated/non-thymoma-associated exfoliative dermatosis and potentially some variants of CLE.
Histopathology of presumptive feline herpes-associated erythema multiforme in a cat. There is moderate to severe epidermal hyperplasia with transepidermal apoptotic keratinocytes (arrow), extravasation of lymphocytes into epidermis and lymphocytic satellitosis of apoptotic keratinocytes (arrowhead). Hematoxylin and eosin, χ 20
Clinical management
The treatment of EM varies according to disease severity and causality. Any drug suspected to have precipitated EM should be promptly discontinued. The clinical course of proposed feline herpes-associated EM appears self-limiting based on limited cases reported; antiviral therapy (eg, famciclovir at 90 mg/kg q12h) with or without systemic antimicrobials for secondary bacterial infection is recom-mended.14,15 In the severe form of EM, systemic glucocorticoids in conjunction with antiviral therapy may be advised, depending on the etiology.8-13 Some cats with the persistent form of EM may respond to oral immuno-suppressants such as ciclosporin (5-7 mg/kg q24h) or mycophenolate mofetil (10-12 mg/kg q12h) for long-term management.
Stevens-Johnson syndrome/toxic epidermal necrolysis spectrum
SJS and TEN are rare, predominantly drug-induced, severe cutaneous T-cell-mediated immune reactions characterized by widespread epidermis and mucosal epithelium sloughing.3,4 The two terms describe variants of the same disease spectrum; SJS is the less extensive (with less than 10% of the body surface affected), SJS/TEN overlap affects between 10 and 30% of the body surface, and TEN represents the more widespread form where more than 30% of the body surface is involved.3,4
Pathogenesis
Drugs are reported as the leading cause of SJS/TEN, with the risk of a hypersensitivity reaction developing in the first few weeks after drug administration. 17 A new disease-specific algorithm, the algorithm of drug causality for epidermal necrolysis (ALDEN), has been recently validated for human patients with SJS/TEN, and it shows superiority to previous algorithms.17,18 A recent feline SJS/TEN case report also utilized ALDEN for the drug causality evaluation. 5 Strong associations between SJS/TEN and several drugs exist in dogs; beta-lactam and trimethoprim-potentiated sulfonamide antibiotics, phenobarbital and carprofen have been implicated.4,17 In cats, a strong causal association has been reported for beta-lactam antibiotics, organophosphate insecticide and d-limonene.5,19-21
At this time, the precise molecular and pathogenic cellular mechanisms leading to the development of SJS/TEN are partially understood, and no focused pathogenesis studies have been conducted on feline SJS/TEN patients. The lesions of SJS/TEN are histopathologically characterized by widespread pan-epithelial keratinocyte apoptosis and necrosis, a process initiated by drug- or drug/peptide-specific cytotoxic T-lymphocytes and/or natural killer-cells. 22 Drugs can stimulate the immune system by directly binding to the class I major histocompatibility complex, resulting in the clonal expansion of a specific population of cytotoxic T-lymphocytes, which infiltrate the skin and secrete soluble proapoptotic factors like granulysin, Fas ligand, perforin and granzymes. 22
Diagnostic approach
Although the history and clinical signs may suggest a diagnosis of SJS/TEN, skin biopsy (see the ‘Tip for biopsy collection’ box in Part 1 1 ) is necessary to support the clinical assessment and exclude other erosive to ulcerative dermatoses. Histopathologic examination reveals lymphocytic interface dermatitis with apoptosis at multiple epidermal levels, progression to epidermal coagulation necrosis and epidermal detachment with ulcerations (Figure 4). 5 Similarly to human and canine SJS/TEN, there is a potential histologic overlap between feline EM and SJS/TEN. 5 Therefore, a pathologist’s microscopic interpretation should be restricted to an umbrella diagnosis of an EM-TEN epidermal necrotiz-ing disease, and the further subclassification of the different entities should depend upon patient history, clinical signs and skin lesion extent. 18

Feline Stevens-Johnson syndrome and toxic epidermal necrolysis spectrum. Multifocal to coalescing erythematous macules with erosions to ulcerations and sloughing affecting (a) periocular, perioral and oral areas as well as the (b) trunk and (c) footpads in a 14-year-old male castrated domestic shorthair cat. Courtesy of Dr Michaela Austel

Histopathology of feline Stevens—Johnson syndrome (SJS) and toxic epidermal necrolysis (TEN) spectrum in a 14-year-old male castrated domestic shorthair cat. Lymphocytic interface dermatitis with extravasation of lymphocytes into the epidermis, lymphocytic satellitosis of apoptotic keratinocytes and superficial epidermal hydropic degeneration can be seen and is the predominant inflammatory pattern in SJS-TEN spectrum. Within the epidermis, progression is observed to full-thickness epidermal necrosis (asterisk), which contains a few entrapped lymphocytes and keratinocytes apoptotic bodies. Hematoxylin and eosin, χ 20. Courtesy of Michaela Austel
In suspected feline cases of SJS/TEN, clinicians should be encouraged to take multiple biopsies since some skin biopsies in these patients may lack epithelium and may not be used for diagnosis. Clinicians are encouraged to work with an experienced der-matopathologist and sample lesions from a range of disease stages, with an emphasis on early to active lesions since ulcerated/ late-stage lesions have a lower diagnostic yield.
Clinical management and prognosis
Although rare, SJS/TEN is a devastating disease; the mortality for SJS is <10%, with the figure rising to 40% for TEN in humans. 17 Significant mortality is associated with TEN in cats, confirming TEN as one of the few dermatologic diseases that constitute a medical emergency.5,19-21 Early recognition, as well as prompt and appropriate management, are essential and can be life-saving. Immediate withdrawal of suspected medication (most commonly beta-lactams, sulfa drugs or non-steroidal anti-inflammatory drugs) and referral to an emergency center are crucial requirements for improving SJS/TEN prognosis. An extensive epidermal loss results in massive fluid, electrolyte and plasma protein losses; therefore, supportive care is required for patients (aggressive fluid replacement, anti-microbial therapy, wound care, analgesia and nutritional support). Immuno-suppressive agents (eg, glucocorticoids, ciclosporin, mycophenolate) have been controversial, but recent evidence shows a possible beneficial role for ciclosporin in humans during early disease development. 23 Considering the rare reports of feline SJS/TEN, there is a lack of information to make recommendations on immunosuppressive protocols for cats.

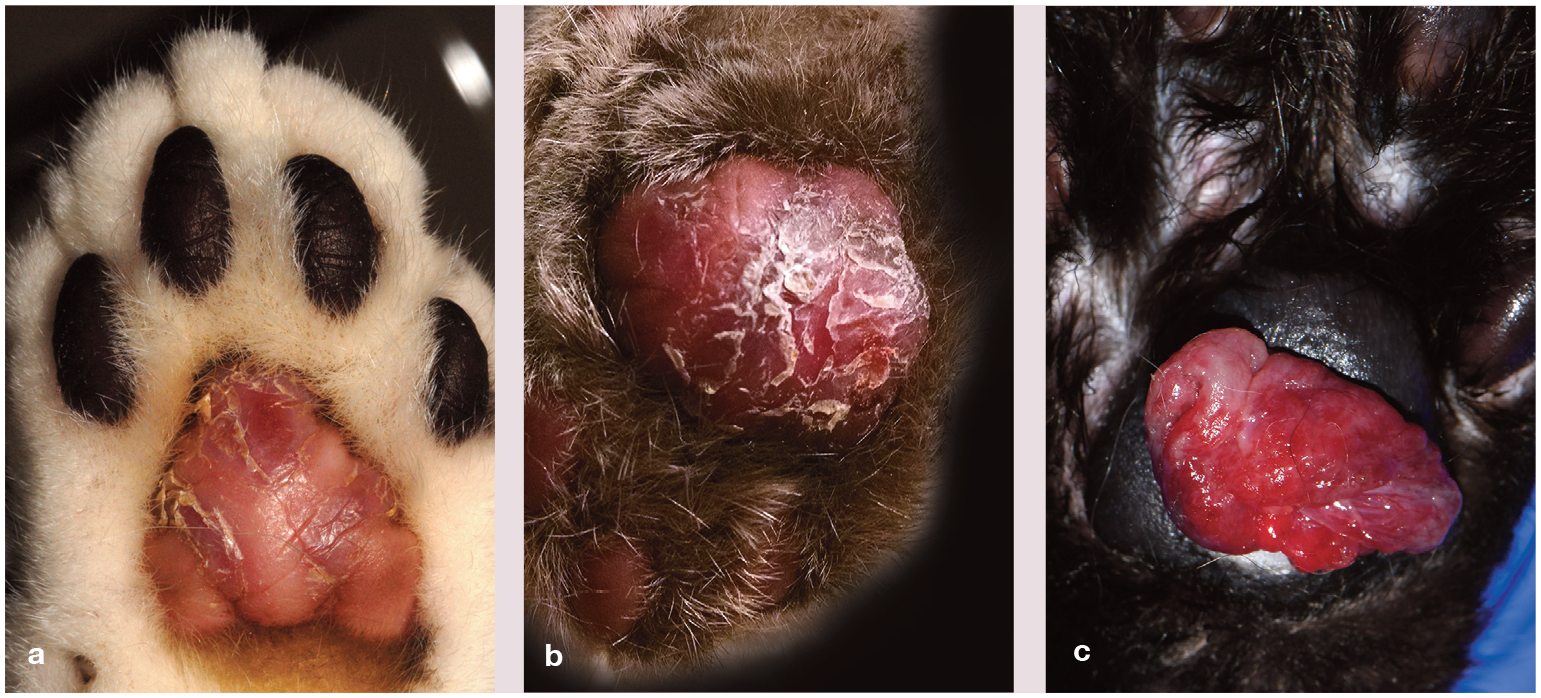
Feline plasma cell pododermatitis. (a,b) Swollen and flaccid metacarpal pads with depigmentation (a), erosions and scaling. (c) Swollen metacarpal pad with ulceration and proliferative granulation tissue
Plasma cell pododermatitis
Plasma cell pododermatitis is a condition that almost exclusively affects the footpads of cats.24-26 The first description and case series emerged in 1979 and 1980, respectively, with five feline patients exhibiting swelling and ulceration of the foot pads. 24 This uncommon condition is also called ‘pillow foot’ due to its highly characteristic clinical features.27,28
Pathogenesis and signalment
The cause and pathogenesis of plasma cell pododermatitis have not been completely elucidated; however, biopsy and laboratory findings suggest an immune-mediated process as the most likely culprit. 24 Consistent hypergammaglobulinemia, marked tissue plasma-cytosis and response to immunomodulatory therapy are hallmark features and support an immune system dysfunction.24,27,33
Plasma cells are mature B-lymphocytes that become activated and then secrete antibodies, usually in response to an antigenic stimulus such as an infection or an inflammatory process. 28 It has been hypothesized that feline plasma cell pododermatitis can be considered a cutaneous reaction pattern with numerous causes, including infections; however, this hypothesis is controversial.33-35 Multiple authors have reported a high incidence of feline immunodeficiency virus (FIV) and feline leukemia virus (FeLV) in cats with this condition.30,32,35 A single study showed a positive FeLV immunohistochemistry (IHC) of paw pad biopsies in a cat with plasma cell pododermatitis that was seropositive for both FeLV and FIV infectious diseases; ICH for FIV was negative in the skin samples, however. According to the authors, despite the FIV-negative results on IHC, a potential FIV involvement was still possible, considering that retroviruses can stimulate polyclonal B cells and secondary hypergammaglobuline-mia, altering the immune function of B-lymphocytes and plasma cells, thus triggering plasma cell pododermatitis. 30 A possible allergic etiology was also proposed as seasonal recurrence has been reported in some cases with active lesions during warm seasons and spontaneous regression in winter.24,25,36
The age of onset is variable and can range from 6 months to 12 years of age, with some authors reporting that male castrated domestic shorthair cats may be over-represented.25,28,32,35
Diagnostic approach
Although the clinical presentation for feline plasma cell pododermatitis is very characteristic, the differential diagnoses include eosinophilic, bacterial or fungal granulomas, foreign body reactions and nocardiosis affecting the footpads. 28 A tentative clinical diagnosis is based on compatible history, skin lesions and the demonstration of large numbers of plasma cells via aspirate, cytologic examination or skin biopsy. In addition, serum protein electrophoresis may reveal polyclonal gammopathy. 33 Histopathologic findings feature perivascular to diffuse infiltration of plasma cells affecting the dermis and the hypodermal adipose tissue (Figure 6).25,27,36 Mott cells -plasma cells containing spherical cytoplasmic inclusion bodies (Russell bodies) - are typically observed in the lesions (Figure 6b).27,33,37 There also may be epidermal acanthosis with erosion, ulceration and variable exudation. Low numbers of other inflammatory cells may be present, such as lymphocytes, macrophages, eosinophils and variable numbers of neutrophils. Chronic lesions may be fibrotic.27,34,36 Hematologic findings include mature neutrophilia, leukocytosis with mild left shift, thrombocytopenia, monocytosis and hypergammaglobulinemia. Anemia and lym-phopenia have also been reported.24,28,31,35,38 Cats with plasma cell pododermatitis may or may not be FIV and/or FeLV positive, and testing for these two viral infections is encouraged. 29

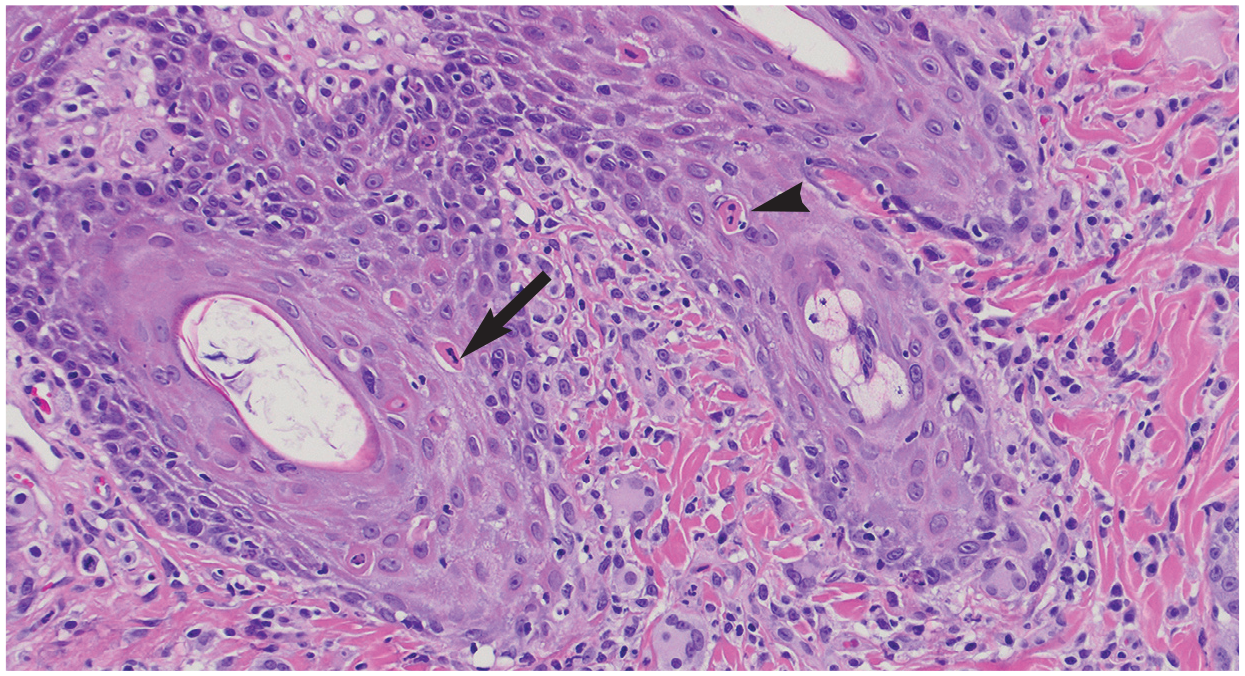
Histopathology of feline plasma cell pododermatitis. The dermis and often the underlying adipose tissue of the paw pad are diffusely infiltated with predominantly plasma cells, and some neutrophils and lymphocytes, obscuring normal architecture. (a) Hematoxylin and eosin, χ 4. (b) Russell body-containing plasma cells (Mott cells [arrows]) are observed as well. Hematoxylin and eosin, χ 40
Clinical management and prognosis
Immunomodulatory treatments and/or surgical intervention have been recommended to manage feline plasma cell pododermatitis. 30
Initial therapy with doxycycline at 10 mg/kg PO q24h can be effective due to its immunomodulatory effects.28,30,34,39 In a single study, 35% of patients (6/17 cats) treated with doxycycline monohydrate went into complete remission within 3 to 8 weeks, whereas 53% of cats (9/17 cats) had partial improvement and 12% (2/17 cats) did not respond. 39 Response to doxycycline may take up to 10 weeks, and treatment should be continued until clinical signs are resolved (Figure 7). Moreover, liquid doxycycline has been recommended to minimize doxycycline-associated tablet/capsule side effects such as esophagitis. 29

Plasma cell pododermatitis in a cat treated with a tapering dosage of oral prednisolone (0.5 mg/kg q24h for 2 weeks) and long-term oral doxycycline (5 mg/kg q24h). Within 5 weeks of treatment, the (a) initial swelling and depigmentation of metacarpal pad had (b) almost completely resolved
Given the increased concerns of antimicrobial resistance and following antibiotic stewardship guidelines, some authors may reason that antibiotics should only be used for confirmed bacterial diseases and not for immune-mediated diseases.38,40 Oral ciclosporin, a calcineurin inhibitor at a dosage of 7 mg/kg q24h until remission, is another immunomodulatory option for controlling the disease. 29 Aurothioglucose was reported to be effective in four cats. 31 In some patients, depending on the severity of the disease, some authors may additionally start systemic oral glucocorti-coids alone or in conjunction with other immunomodulatory treatments. 28 Oral daily prednisolone (2.2-4.4 mg/kg), triamcinolone acetonide (0.4-0.6 mg/kg) or dexamethasone (0.2 mg/kg) can be effective in inducing remission of skin lesions in feline plasma cell pododermatitis; the daily dose should be tapered following a favorable response. 29
Surgical excision of affected footpads is usually curative and reserved for cases that do not respond to medical therapy.25,28,30,32
The prognosis of feline plasma cell pododermatitis varies as some patients may have resolution of clinical signs spontaneously, whereas others may require immunomodulating agents and lifelong therapy.
Proliferative and necrotizing otitis externa
PNOE is an uncommon to rare disorder affecting the ear canals and concave pinnae of cats and kittens. 41 Extra-auricular lesions have also been reported to affect the face, eyelids and other areas of the skin.42,43
Pathogenesis and signalment
The pathogenesis involves an immune-mediated process associated with CD3+ T cells infiltrating the epidermis and inducing apoptosis in caspase 3-positive keratinocytes. However, the reason for generating these T cells remains unknown. 44 Infectious agents, including FIV and FeLV, have not been shown to induce this condition.45-48 PCR for feline herpesvirus 1 and IHC for papillomavirus and calicivirus have yielded negative results.47,49
Cats of any age can be affected, with reports ranging from 2 months to 14 years of age.50,51 Although no specific breed appears to be predisposed, most cases were described in domestic shorthair cats; 49 males may be overrepresented. 46


Proliferative and necrotizing otitis externa. (a,b) Well-demarcated erythematous plaques with adherent, thick dark brown to black keratinous debris are present in the medial aspect of the pinna and at the entrance to the auditory canal. (c) Extra-auricular lesions in the periocular areas on the face of a cat with proliferative and necrotizing otitis externa. Image (a) courtesy of Jenise Daigle
Diagnostic approach
Diagnosis is based on clinical features and histopathology. Erythematous plaques should be biopsied, taking care to preserve the adherent keratinous crusts. 49 The primary histologic feature is severe acanthosis of the outer root sheath of hair follicles with scattered single-cell necrosis of keratinocytes at different epithelial levels (Figure 9). The epidermis and follicular infundibulum present with ortho-keratotic to parakeratotic hyperkeratosis. Multifocal hydropic degeneration causes epidermal and follicular infundibular pallor. The dermis shows a mixed inflammatory infiltrate composed predominantly of lymphocytes, plasma cells and neutrophils, or eosinophils and mast cells.45,49,50

Histopathology of proliferative and necrotizing otitis externa. (a) Severe epidermal hyperplasia (asterisk) and intense superficial dermatitis with striking layered parakeratotic hyperkeratosis intermingled with lymphocytes and neutrophils can be seen. Hematoxylin and eosin, χ 10. (b) Scattered apoptotic-appearing keratinocytes (arrows) surrounded in some areas by lymphocytes are present within severely hyperplastic epidermis and superficial follicular epithelium. Hematoxylin and eosin. Bar = 20 µm
Clinical management and prognosis
Treatment with topical tacrolimus 0.1% twice daily is the initial treatment for most cases (Figure 10). Lesions may resolve after 3 to 12 weeks.41,44,50 A single case report noted no recurrence in a 2-year follow-up after discontinuing the treatment. 44 Tacrolimus is a topical macrolide lactone with good penetration within the stratum corneum. 52 As a calcineurin inhibitor, tacrolimus inhibits interleukin (IL)-2 and IL-4 gene transcription, downregulates the expression of other T-cell cytokines and prevents T-cell response to antigens. 50 Sole topical glucocorticoid therapy with fluocinolone acetonide, dexamethasone, prednisolone, betamethasone or mometasone furoate has demonstrated partial to no improvement.41,44,50 However, in one case report, a combination therapy of topical steroid lotion (mometasone furoate 0.1%) and oral glucocorticoids (initial triamcinolone ace-tonide with a switch to prednisolone later) was successful. 51 Oral prednisolone alone has shown partial improvement. 50

Feline proliferative necrotizing otitis externa (a) before and (b) after treatment with topical tacrolimus ointment 0.1% twice daily for 30 days
In one case, ciclosporin was used at 7 mg/kg to 10 mg/kg PO q24h, along with topical tacrolimus 0.1% for PNOE extra-auricular lesions. All PNOE-associated lesions resolved in 6 weeks; however, the PNOE lesions in the ear relapsed 2 weeks later. At that point, daily topical tacrolimus 0.1% was resumed, along with prednisolone 1 mg/kg PO q24h. Ear canal PNOE lesions healed once again and remained in remission under treatment. 43
A recent report described two cases successfully treated with off-label use of oclacitinib. Cat 1 was treated with oclacitinib 1.5 mg/kg PO q12h. Complete remission was obtained in 12 weeks. Cat 2, which also had extra-auricular lesions, was treated with oclacitinib 0.5 mg/kg PO q12h. Complete remission was achieved after 7 weeks of treatment. Upon follow-up, both cases remained in complete remission after the oclacitinib had been discontinued for 1 year and 1.5 years; no adverse effects due to the treatment were noted. Moreover, regular complete blood cell counts and serum chemistry profiles were unremarkable for both patients. 48 However, it is essential to note that a safe therapeutic index for oclacitinib has not been established in cats. 53 The elimination half-life is 2.5 h, which is shorter than the half-life in dogs, and therefore, a twice daily dosing of oclacitinib is necessary for immunomodulatory efficacy. 53 In addition, transcript quantification and protein levels of Janus kinase 2 (JAK2) are much lower in feline cells compared with canine cells, with significant implications for the safety profile in cats. Note that JAK2-dependent cytokines include erythropoietin and granulocyte-macrophage colony-stimulating factors. Therefore, over-inhibition of JAK2 cytokines may result in substantial adverse hematologic effects. 53
Spontaneous remission may occur in some cases, although it may take over 12 months. 50 The prognosis for PNOE is generally good, including for the extra-auricular lesions. 43
Pseudopelade and alopecia areata
Pseudopelade and alopecia areata are two rare immune-mediated diseases of cats characterized by non-scarring, non-inflammatory and non-pruritic alopecia.
Pseudopelade
The autoimmune process for pseudopelade has been described by the tropism of CD3+ CD8+ cytotoxic lymphocytes toward the hair follicle isthmus. In addition, high titers of IgG autoantibodies specific to structures of the lower hair follicle, such as trichohyalin and hair keratin, have been detected. 54 In essence, the pathogenesis involves a cytotoxic T-cell lymphocytic inflammatory infiltrate targeting the isthmus of the hair follicle, where the bulge - an area rich in follicular stem cells - is located. 55 Destruction of the stem cells in the bulge results in permanent alopecia. 56


Feline alopecia areata. Generalized, non-pruritic, non-inflammatory alopecia affecting the (a,b) head and (c) abdomen and legs. Courtesy of Morgan Matthai and Lori Thompson
Diagnostic approach
Clinical differential diagnoses may include alopecia areata, follicular dysplasia, traction alopecia, psychogenic alopecia, endocrinopathies, demodicosis and dermatophytosis. Punch biopsies should contain only abnormal tissue of maximal alopecia; 57 the margin of alopecic to healthy-haired areas and healthy-haired skin can also be submitted so that the dermatopathologist can observe changes in disease progression without the impact of disease chronicity.
Histologic examination demonstrates mild to severe, predominantly lymphocytic and histiocytic follicular inflammation in and around the isthmus. Hair bulb and adnexae are not affected. Apoptotic keratinocytes and follicular degeneration may be present with chronicity. Micro foreign body granulomas are seen surrounding fragments of free hair shafts. Late stage of chronic lesions are characterized by follicular atrophy and fibrotic tissue (angio -fibrotic stellae). 54
Clinical management
In cats, treatment for pseudopelade is limited due to the small number of patients reported. This condition usually does not respond to glucocorticoids or chlorambucil. 55 However, modified ciclosporin with or without gluco-corticoids may result in transient hair growth. The alopecia will return if treatment is discontinued.54,55
Alopecia areata
Alopecia areata-like dermatosis has been described in a 10-year-old domestic short-haired cat with non-inflammatory alopecia on the ventrum and legs. 58 The cat also developed onychomadesis. Histologically and immunohistochemically, the affected skin presented with moderate to severe mural folli-culitis and perifolliculitis at the level of the follicular isthmus, composed of cytotoxic T-lymphocytes. 58 It is important to note that alopecia areata is defined as immune-mediated destruction of the follicular bulb (ie, bulbitis), not the isthmus (Figure 12). However, in some cases, the inflammation can progress superficially and affect the isthmus to a lesser extent. 57 This is in contrast to the histologic findings of the one cat reported with aopecia areata-like dermatosis. For this cat, clinical pathology tests were unremarkable. Likewise, antinuclear antibody, FIV/FeLV, dermatophyte culture and skin scrapings were all neg-ative. 58 In cats, no treatment has been reported for this condition. Nevertheless, oral immuno-suppressive doses of glucocorticoids and/or modified ciclosporin (5-7 mg/kg) can be used for treatment.

Histopathology of feline alopecia areata reveals peri- and intrabulbar lymphocytic infiltation of anagen hair follicle with effacement of the bulb (arrow). (a) Hematoxylin and eosin, χ 4. (b) Hematoxylin and eosin, χ 20. Courtesy of Morgan Matthai and Lori Thompson
Auricular chondritis (‘relapsing polychondritis’)
Auricular chondritis is a rare disease of cats characterized by inflammation and destruction of the auricular cartilage and occasionally non-articular cartilaginous structures.55,59 The term ‘relapsing polychondritis’ (RPC), which is adapted from a similar condition in humans, seems controversial in cats, given that in most feline patients only the auricular cartilage is affected. Moreover, even when additional chondral tissues are involved, they do not appear to be relapsing in nature.55,59,60


Feline auricular chondritis. Severe erythema, thickening, crusting and swelling of the pinnae with progression to tissue fibrosis and deformation. Image (a) courtesy of Dr Niksa Lemo and images (b) and (c) courtesy of David Senter
Pathogenesis is considered to be the result of an immune-mediated process that primarily targets type II collagen, which is restricted to car-tilage. 61 It has also been speculated that chronic otitis, trauma or damage to the cartilage may play a role in the development of the disease. 55 Age of onset varies, ranging from 1.5 to 14.5 years. However, most affected cats are young to middle-aged with a median of 3 years. 61 No sex or breed predilection has been reported.
Diagnostic approach
The clinical differential diagnoses for auricular chondritis include aural hematoma with fibrosis, otoacariasis with resulting scarring of the pinnae, actinic keratosis with deformity and scarring of the pinnae, frostbite, immune-mediated vasculitis and hypercortisolism with curled pinnae.55,60 The diagnosis of auricular chondritis is based on clinical signs. If necessary, a full-thickness biopsy of the pinna, including the cartilage, should be submitted for histopathology. The histologic lesions are characterized by inflammation, degeneration, necrosis, loss of basophilic staining of the cartilage matrix, perichondrial edema, and fibrocyte and capillary endothelial cell proliferation (Figure 14). The inflammatory infiltrate in the cartilage is predominantly lymphocytic with few multinucleated giant cells. Neutrophilic infiltration in and around the auricular cartilage can be observed in different stages of the disease. Focal areas of der in humans, there are two sets of diagnosticmal mixed lymphocyte, plasma cell and mast cell aggregates have also been reported.55,59-61

Histopathology of feline auricular chondritis shows degenerated cartilage with perichondrial mixed inflammation consisting of lymphocytes, macrophages and neutrophils; moderate fibrosis surrounds the cartilages. (a) Hematoxylin and eosin, χ 4. (b) Hematoxylin and eosin, χ 20
In humans, there are two sets of diagnostic criteria for RPC. The first requires proven inflammation of at least two out of three cartilages, including the auricular, nasal or laryn-gotracheal cartilages. The second requires proven inflammation in one cartilage plus two additional clinical signs, including ocular inflammation, vestibular dysfunction, seronegative inflammatory arthritis or hearing loss. 62 In cats, a diagnosis of feline RPC may be more appropriate when at least two other organs are proven to be affected as listed for humans and/or at least two different anatomic sites having histologically proven chondritis. 55 In one case report, a 3-year-old Japanese domestic cat with auricular chondri-tis was also diagnosed with chondral changes in the costae, larynx, trachea and limb joint. Even though the polyarthritis in this patient was erosive and more consistent with chronic progressive polyarthritis of cats, this case met the criteria for RPC. 59 In another case report, a 3-year-old Siamese cat with auricular chondri-tis was also diagnosed with bilateral corneal opacification and suspected valvular disease. While humans with RPC can also present with ocular inflammation and cardiac abnormalities, it is unclear if this was a genuine case of feline RPC, considering that the ocular histo-logic results were inconclusive and other possible causes of ocular and cardiac diseases were not completely ruled out.61,63
Clinical management and prognosis
Response to treatment is variable. One case went into remission within 3 weeks when prednisolone was administered at a dosage of 3 mg/kg q24h. At that point, the medication was gradually tapered and then discontinued. No relapse was reported during the 14-month follow-up period. 60 Other cases failed to respond to prednisolone monotherapy at doses ranging from anti-inflammatory to immuno-suppressive. 61 Dapsone at a dosage of 1 mg/kg PO q24h with or without glucocorticoids induced remission in four cats with no relapse after the therapy was discontinued.55,61,63 Pinnectomy was curative in one case. 61 One case failed to respond to modified ciclosporin at a maximum dosage of 7.5 mg/kg PO q24h and dapsone at 1 mg/kg PO q24h. 59 For the patients that respond to treatment, relapse may occur if therapy is discontinued. 61 Improvement without treatment and spontaneous remission have also be reported.55,61 Permanent cartilage deformity is expected with or without treatment. 55
Key Points
✜ The history and physical examination findings of any cat presenting with clinical signs of erythema, hair loss, scaling/crusting and erosions with or without pruritus should be screened for signs that are less consistent with common skin-restricted dermatoses (eg, hypersensitivity dermatitis, parasitic and fungal dermatitis) and that may raise suspicion for cutaneous immune-mediated diseases. In this scenario, a broad knowledge of cutaneous immune-mediated dermatoses will help with the recognition of less consistent historical or lesional clues to raise the profile of specific immune-mediated diseases, and accurate diagnostic skills and tools will help exclude common differential diagnoses.
✜ Cytology and skin biopsies are often most valuable for diagnosing cutaneous immune-mediated diseases and distinguishing between infectious, hypersensitivity and neoplastic causes.
✜ EM, SJS and TEN had long been considered part of an identical spectrum of immune-mediated diseases; however, the current human and veterinary clinical classification defines SJS and TEN as variants of the same disease spectrum that differ from EM subsets in clinical appearance and causality. Drugs are thought to induce most cases of SJS/TEN, whereas EM is commonly associated with infectious triggers or has an unknown cause.
✜ Similarly to human and canine SJS/TEN, there is a potential histologic overlap between feline EM and SJS/TEN. Therefore, a pathologist’s microscopic interpretation should be restricted to an umbrella diagnosis of an EM-TEN epidermal necrotizing disease, and the further subclassification of the different entities should depend upon patient history, clinical signs and skin lesion extent.
✜ Plasma cell pododermatitis is a condition that almost exclusively affects the foot pads of cats, which are often swollen, soft and exhibit a white, scaly, silvery, crosshatched striated appearance.
Clinically distinctive lesions of PNOE are well-demarcated proliferative dark brown to black necrotic plaques covering the concave pinnae and opening of the vertical canal. Extra-auricular lesions affecting the face, with severe crusting and ulcerative dermatitis, tissue edema and alopecia, have been described.
✜ Pseudopelade and alopecia areata are two rare immune-mediated diseases of cats characterized by non-scarring, non-inflammatory and non-pruritic alopecia.
✜ Cats with auricular chondritis exhibit characteristic pinnal lesions encompassing swollen, thickening, deformation, pain and intense erythema. If necessary, a full-thickness biopsy of the pinna, including the cartilage, should be submitted for histopathologic examination.
Footnotes
Correction (May 2025):
This article has been updated to include the current address of Paulo Gomes.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and / or publication of this article.
Funding
The authors of this commissioned Clinical Spotlight review received an honorarium; as for all ]FMS articles, this Clinical Spotlight article went through peer review.
Ethical approval
This work did not involve the use of animals and therefore ethical approval was not specifically required for publication in JFMS.
Informed consent
This work did not involve the use of animals (including cadavers) and therefore informed consent was not required. For any animals or people identifiable within this publication, additional informed consent for publication was obtained.
