Abstract
Paraneoplastic pemphigus was suspected in a 14-year-old Labrador retriever because of mucocutaneous erosions, microscopic suprabasal acantholysis, and keratinocyte apoptosis. In this patient, circulating IgG autoantibodies recognized plakin (envoplakin, periplakin) and desmoglein (desmoglein-1 and −3) antigens. Necropsy, however, failed to confirm the concurrent existence of hematopoietic or solid neoplasia. The diagnosis of pemphigus vulgaris therefore was proposed. This study illustrates that such a combination of clinicopathologic lesions and plakin/desmoglein-specific autoantibodies is not restricted to canine paraneoplastic pemphigus but can also be detected in another form of suprabasal pemphigus.
Reported in humans and one dog, 2 9 paraneoplastic pemphigus (PNP) is an autoimmune blistering disease associated with hematopoietic or solid neoplasia. It is characterized clinically by severe blistering and erosions located predominantly on mucosae or at mucocutaneous junctions. 2 9 Microscopically, human and canine PNP skin lesions exhibit a combination of suprabasal acantholysis, as seen in patients with pemphigus vulgaris (PV), and interface dermatitis or apoptotic keratinocytes indicative of erythema multiforme. 6 9 In humans and the dog with PNP, circulating autoantibodies are present and recognize a variable set of intracellular proteins from the plakin family (e.g., plectin, desmoplakin I and II, BPAG1, envoplakin, and periplakin). 2 4 Recent reports suggest that, in humans and the dog with PNP, there are also circulating autoantibodies that recognize desmoglein-3 (dsg-3), the main PV autoantigen. 1 4 These dsg-3-specific PNP autoantibodies are pathogenic; they can cause blistering in a murine passive transfer model. 1 Therefore, PNP is very similar to PV at clinical, histologic, and immunologic levels.
Here, we report the clinical and immunopathologic findings in a dog affected with PV. In this dog, the discovery of circulating autoantibodies directed against plakins and desmogleins led to the presumed diagnosis of PNP. After euthanasia, however, the search for neoplasia was unsuccessful. Our observations suggest that discovery of suprabasal acantholysis and apoptosis and of autoantibodies against plakins and desmogleins, albeit suggestive of PNP, are not pathognomonic for this rare canine autoimmune blistering dermatosis.
A 14-year-old male Labrador Retriever was presented with a complaint of hypersalivation of 4 days' duration. Physical examination revealed a severe erosive stomatitis affecting mainly the tongue and palate. Oral lesions responded to a regimen of spiramycin and anti-inflammatory doses of prednisone for 1 week. One month later, erosions and ulcers were observed around the claws, on interdigital spaces, at pressure points, and at mucocutaneous junctions. Histopathologic examination of lesional mucosal and haired skin biopsy specimens, performed on 2 separate occasions, revealed similar findings. Suprabasal acantholysis was associated with intraepidermal clefting (Figs. 1, 2). There was prominent keratinocyte apoptosis in the spinous epithelium, often observed with lymphocytic satellitosis (Fig. 2). Intraepidermal separation was associated with necrosis and suppuration of the superficial epithelium. In samples of the oral cavity, mild superficial submucosal lymphocytic and plasmacytic inflammation also was seen. Because of mucocutaneous erosive lesions and histologic demonstration of suprabasal acantholysis and keratinocyte apoptosis, canine PNP was suspected. 9 An attempt was made to discover an occult neoplasia. Hematology and a serum chemistry panel results were within normal limits. Cytologic examination of lymph node aspirates was not suggestive of hematopoietic neoplasia. Chest radiographs were unremarkable. Abdominal ultrasonography revealed a 3- × 3-cm hepatic nodule. Because of the animal's age, the owners declined laparotomy or liver biopsy, and the dog was prescribed prednisone (1 mg/kg/day). Skin lesions remained in partial remission with prednisone during the following 8 months. During this time, multiple complete blood counts and serum chemistry panels were evaluated, but no abnormalities were detected, other than those expected following high-dose corticosteroid therapy. Repeated liver ultrasound examinations confirmed that the nodule did not enlarge beyond its original size. Because the dog presented a relapse of oral lesions and severe symptoms of lethargy, he was euthanatized. Necropsy was performed and revealed diffuse enlargement of the liver. Serial sections of the liver did not demonstrate parenchymal nodules. Therefore, the identity of the nodule observed during ultrasound examination remained unknown. Multiple small nodules were found in the pancreas and the lungs. Gross examination of lymph nodes, spleen, digestive tract, urogenital tract, endocrine organs, prostate, and genitalia was unremarkable. Examination of the brain and spinal cord was declined by the owner for cosmetic reasons prior to burial. Specimens were collected from grossly abnormal organs and fixed in neutral buffered formalin. Histologic examination of liver, pancreas, and lung specimens revealed steroid hepatopathy, nodular hyperplasia, and focal mineralization, respectively.
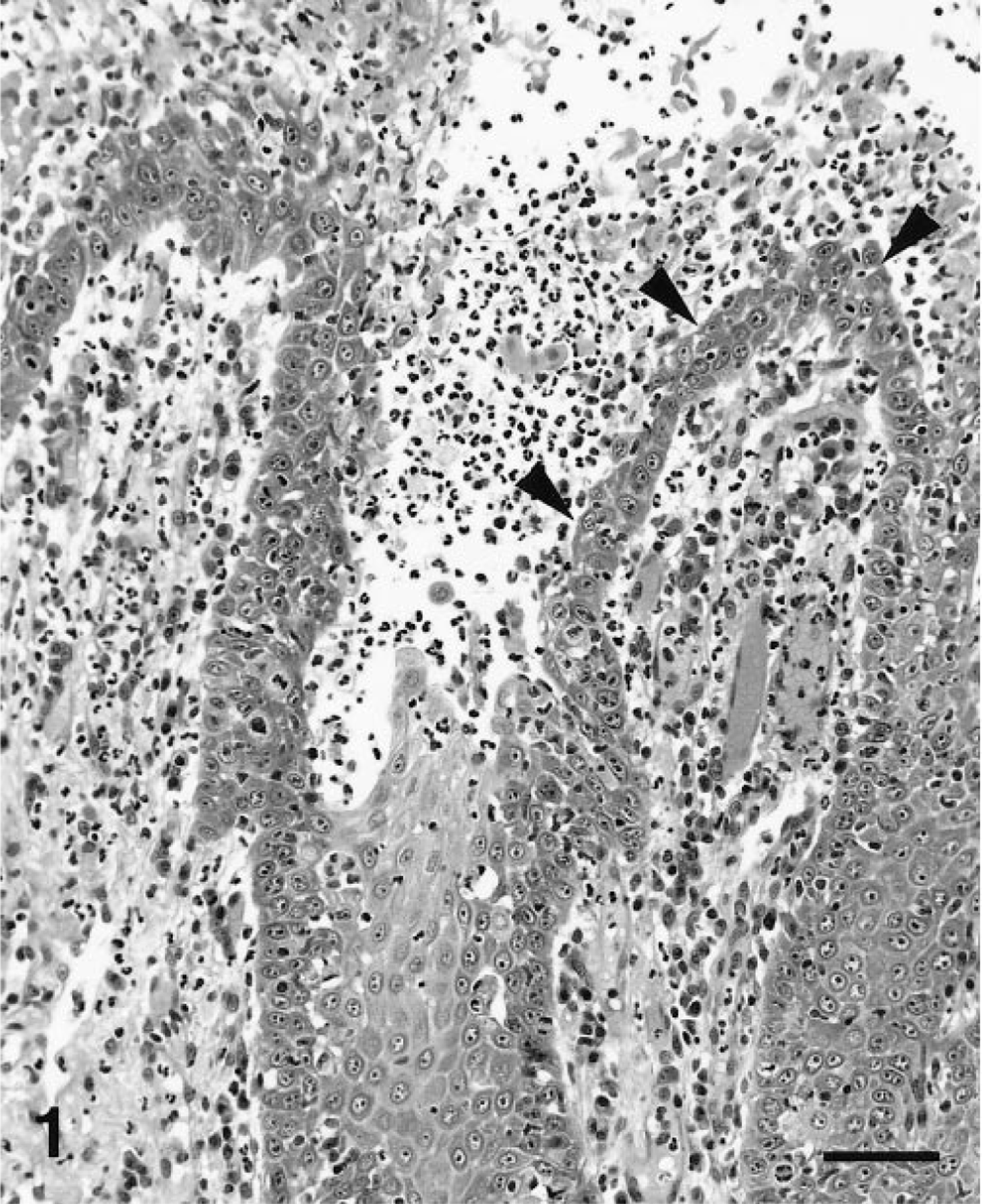
Oral mucosa; dog. Suprabasal acantholysis (black arrowheads) results in mucosal erosion and suppuration. HE. Bar = 80 μm.
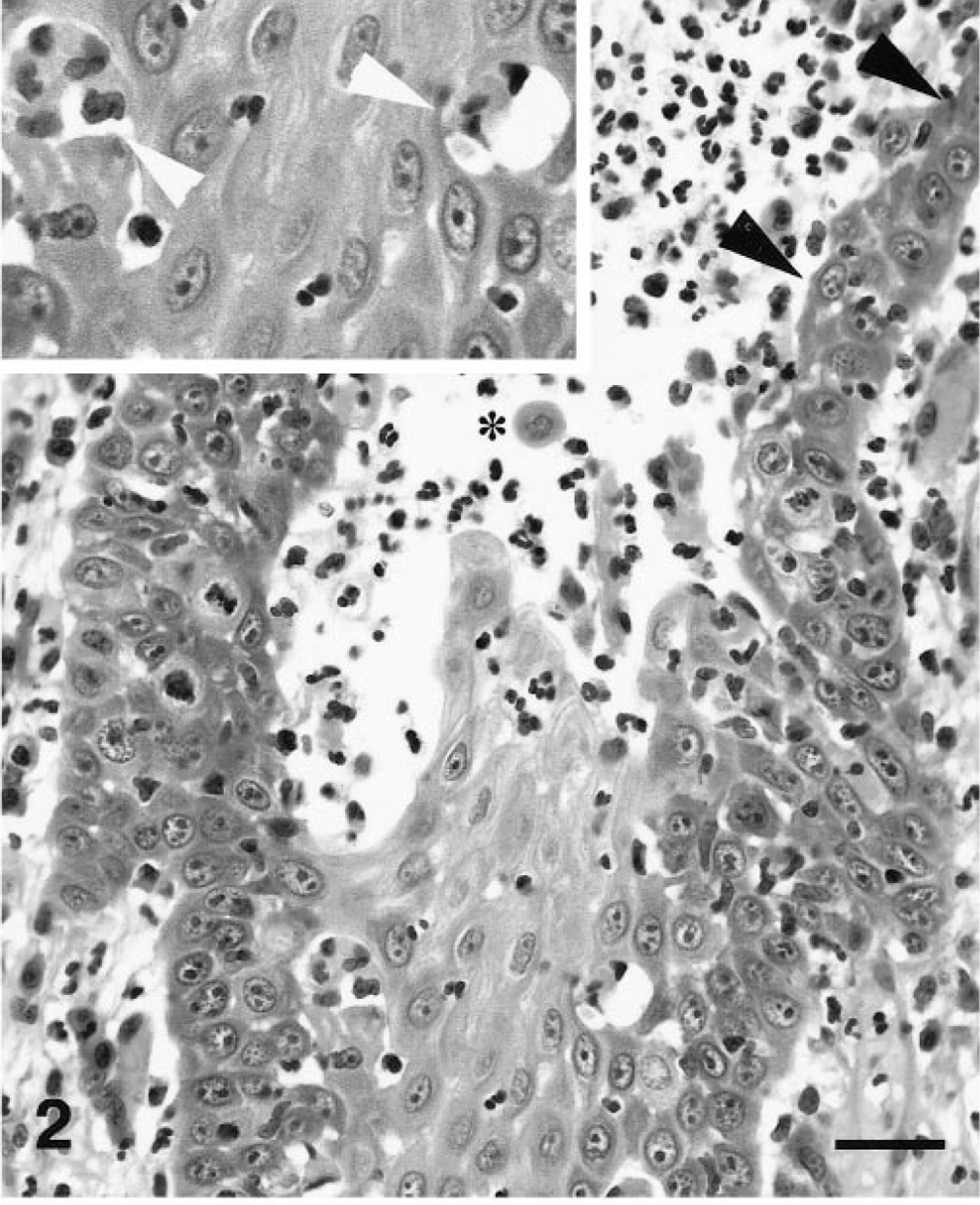
Oral mucosa; dog. Higher magnification of Fig. 1. Suprabasal acantholysis (black arrowheads) and mucosal erosion with suppuration can be seen. Occasional free-floating epithelial cells are present (asterisk). HE. Bar = 40 μm. Inset: Keratinocyte apoptosis with satellitosis can be observed at multiple levels of the epithelium (white arrowheads). Bar = 20 μm.
Detection of skin-fixed and circulating autoantibodies was carried out as previously described. 10 Direct immunofluorescence (IF) testing, performed on frozen specimens, revealed a strong intercellular deposition of IgG at all levels of the mucosal epithelium. Keratinocyte-bound IgA, IgM, and C3 were not seen. Indirect IF was performed twice, at the time of original diagnosis and after euthanasia, 8 months later. At both times, this method revealed a very high titer (> 1:1,000) of circulating anti-keratinocyte IgG autoantibodies using normal canine lip, tongue, and esophageal substrates. The pattern of intercellular fluorescence was strongest in the stratum spinosum and fainter around basal keratinocytes (Fig. 3). The serum titer of anti-keratinocyte autoantibodies was lower when a normal canine haired skin substrate was used. Indirect IF testing was negative with normal canine bladder (Fig. 4), a finding in contrast with those of other studies of human and canine PNP sera in which circulating autoantibodies bound to transitional epithelia. 2 9
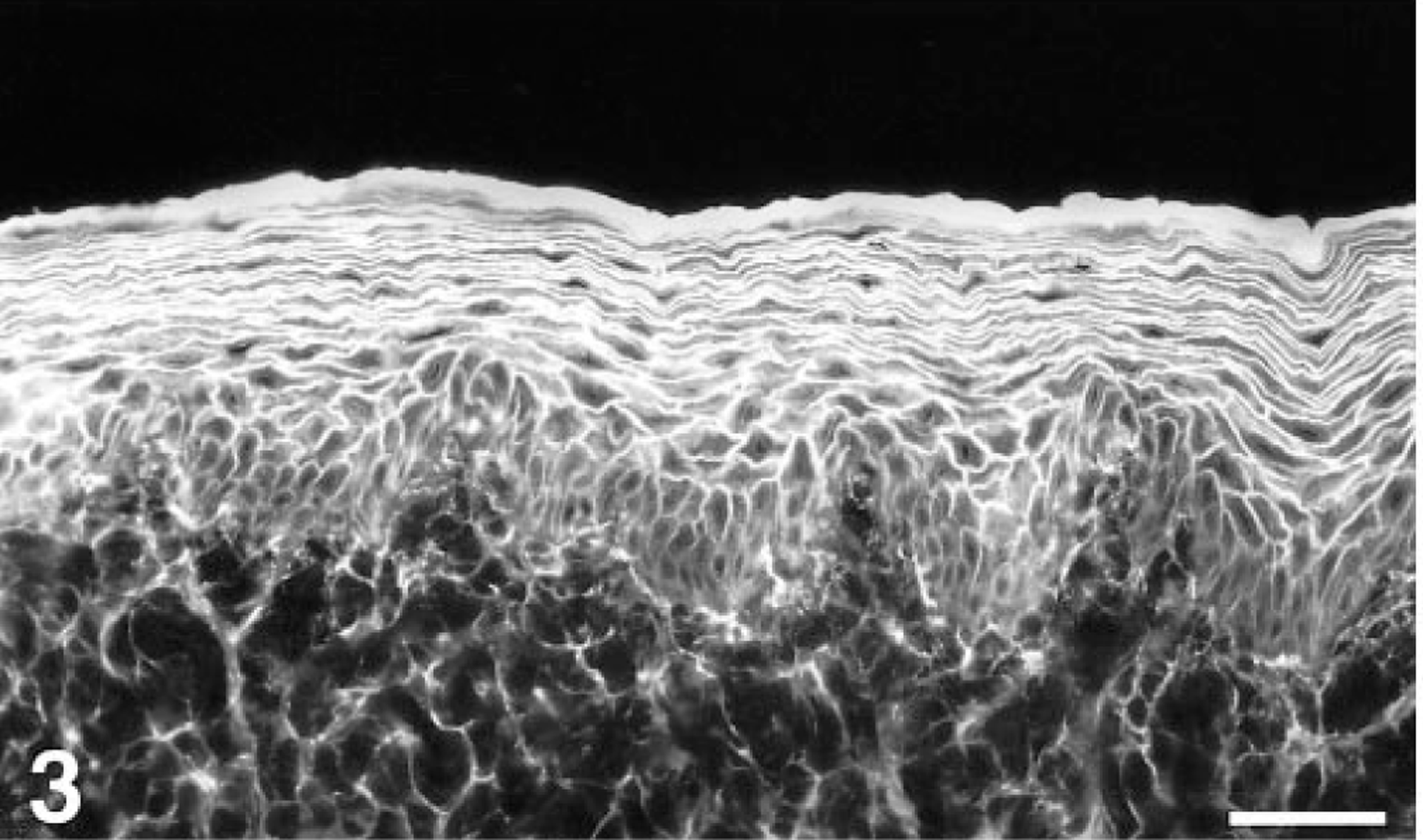
Esophagus; normal dog. Intercellular epidermal fluorescence characteristic of pemphigus. The staining is weakest around stratum basale keratinocytes. Indirect IF with normal canine esophagus substrate, serum from the affected dog, and anti-canine IgG-fluorescein. Bar = 60 μm.

Bladder; normal dog. There is no intercellular fluorescence in the bladder epithelium, in contrast to that seen with sera of dogs with PNP. Indirect IF with normal canine bladder substrate, serum from the affected dog, and anti-canine IgG-fluorescein. Bar = 60 μm.
Immunoblotting was done using cultured human keratinocyte extracts as described previously. 3 The dog's serum contained IgG autoantibodies that identified antigens of 210, 190, and 130 kd suspected to represent envoplakin, periplakin, and dsg-3, respectively (Fig. 5). An additional, an unidentified 105 kd antigen was also recognized by the patient's autoantibodies. To confirm the identity of targeted autoantigens, an enzyme-linked immunosorbent assay (ELISA) using the recombinant extracellular segments of human dsg-3 and dsg-1 7 was adapted for use in dogs by using anti-canine IgG secondary antibodies. This method established that this dog's serum, at a 1:1,000 dilution, contained autoantibodies directed against both dsg-1 and dsg-3 in amounts that exceeded that of a human PV serum sample. ELISA testing with normal canine serum yielded unremarkable results.
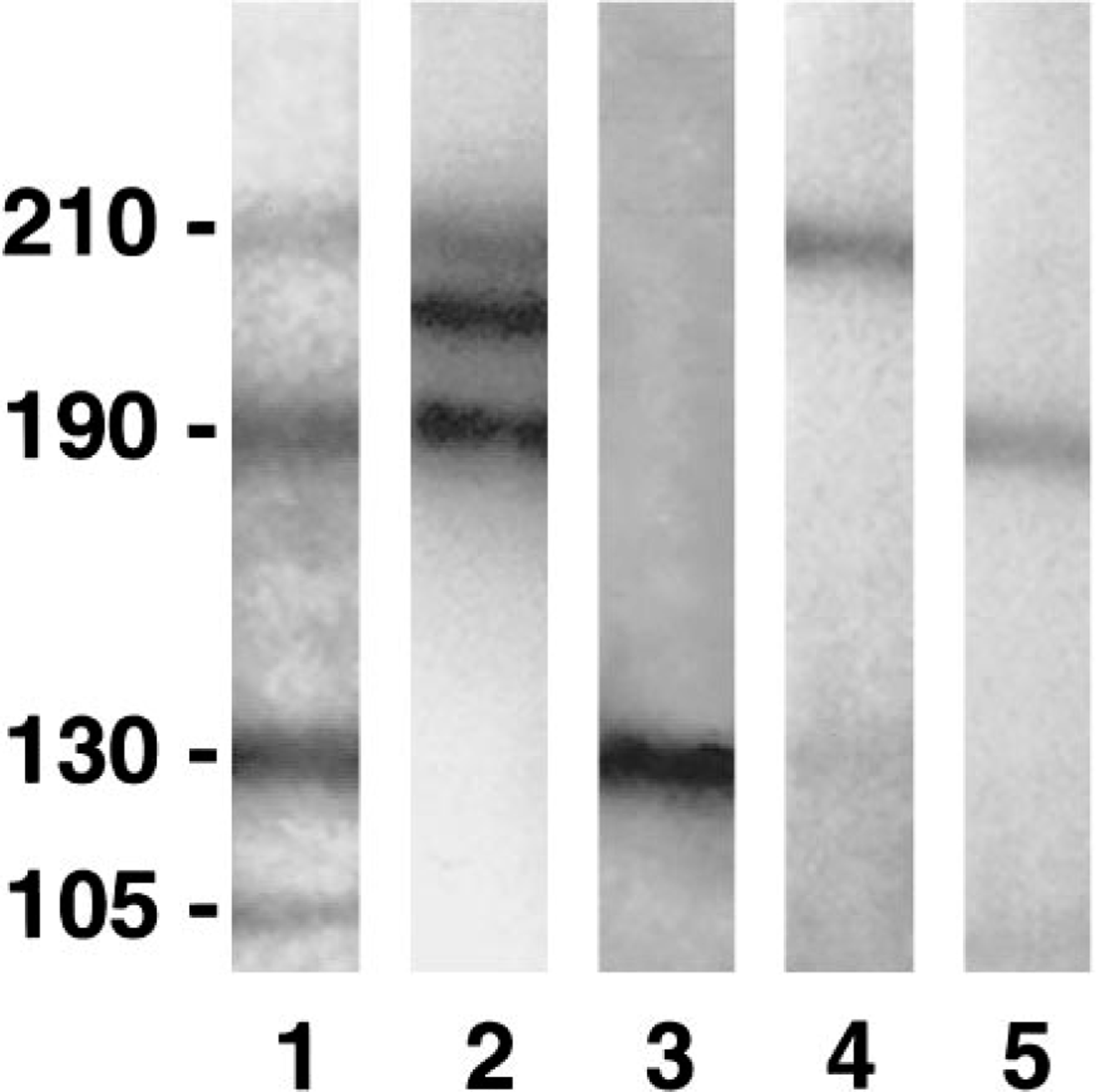
The affected dog's IgG autoantibodies (lane 1) recognize 210-, 190-, 130-, and 105-kd antigens. The 210-kd antigen comigrates with that recognized by a human PNP serum sample (lane 2) and a monoclonal antibody specific for human envoplakin (lane 4). The 190-kd antigen is suspected to represent periplakin, as recognized by human PNP IgG (lane 2) and monoclonal antibodies against this protein (lane 5). The 130-kd antigen probably is desmoglein-3, as targeted by IgG from a human with PV (lane 3). The identity of the 105-kd antigen remains unknown. Immunoblotting using an extract obtained from a human keratinocyte culture. Lane 1 was incubated with an anti-canine IgG-peroxidase, lanes 2 and 3 with an anti-human IgG-peroxidase, and lanes 4 and 5 with an anti-mouse IgG-peroxidase.
In conclusion, this dog's macroscopic lesions, histopathologic findings, and targeted autoantigens led to the tentative diagnosis of PNP. 9 However, repeated complete blood counts and serum chemistry panels, microscopic examination of lymph nodes aspirates, macroscopic examination of most organs at necropsy, and microscopic analysis of observed lesions did not reveal solid or hematopoietic tumors. Bone marrow, brain, and spinal cord were not examined during necropsy because of cosmetic concerns, but a neoplastic process of these tissues was not deemed likely because of lack of clinical or biologic abnormalities suggesting organic dysfunction. Because the search for an hematopoietic or a solid tumor was unsuccessful, the diagnosis of a variant of PV therefore was proposed. Even though the presence of an occult tumor in this dog was a possibility, we do not favor this hypothesis. First, the evolution of PNP in humans is most commonly fulminant, in spite of immune suppressive therapy, when malignant or benign neoplasms are not resected surgically. 2 In contrast, the lesions in this dog remained in partial remission for 8 months during prednisone therapy at low immunosuppressive doses. Second, this dog's circulating autoantibodies did not recognize bladder epithelial cells, a finding in contrast with IF results obtained for typical human and canine PNP sera. 2 9 Third, a diagnosis of PNP based on the sole observation of anti-plakin autoantibodies is not valid because such antibodies have been discovered in human patients with nonparaneoplastic forms of pemphigus. 5 8
Footnotes
Acknowledgements
We thank Dr. G. Haroutunian for the repeated ultrasonograms and Dr. J. Cullen for his help in the histopathologic review of necropsy specimens. We are grateful to W. Savage (Biomedical Communication, North Carolina State University College of Veterinary Medicine) for her help with the figures.
