Abstract
Two outbreaks of cutaneous pythiosis caused by Pythium insidiosum were diagnosed in two herds of crossbred hair wool sheep of different ages in the semiarid region of Northeastern Brazil. In one herd of 120 sheep, 40 were affected. The other outbreak affected six sheep out of 80. Local swellings with ulcerative lesions were observed in the limbs and abdominal and prescapular regions. Three sheep were necropsied. Two of them had lung metastasis characterized by multifocal nodules measuring 0.5–2 cm. In one animal, the prescapular lymph node was also affected. In another, the cutaneous lesion extended to the sesamoid bone. Microscopically, there were multifocal granulomas with intralesional P. insidiosum hyphae and Splendore-Hoeppli material surrounding the hyphae. The diagnosis was based on the histologic lesions, immunohisto-chemical identification, and culture of the etiologic agent. One sheep treated with potassium iodide recovered. Standing in swampy water for long periods in a warm aquatic environment seems to be the reason for the high prevalence of the disease.
Pythium insidiosum is a pseudofungus that belongs to the kingdom Cromista, class Oomycete, family Pythiaceae. 7,12–14,16 Some species of Pythium are plant pathogens, and only P. insidiosum has been recognized as causing lesions in mammals. 13 Cutaneous pythiosis affects horses, causing ulcerative lesions mainly in the lower portion of the extremities or ventral portion of the abdomen. These anatomic regions are more often affected because they are more in contact with aquatic environments where P. insidiosum occurs. Pythiosis develops when open wounds are infected by the zoospores of Pythium, which are chemotactically attracted to decayed organic matter in the wounds. The disease is more prevalent in tropic and subtropic regions because P. insidiosum needs warm water to maintain its life cycle. 7,15 Cattle can also be affected by cutaneous pythiosis. 8,17,21 P. insidiosum also causes lesions in the skin and gastrointestinal tract in dogs, 16 dermatitis and keratitis in humans, 10,23 and nasal infection in cats. 3
Pythiosis in horses is characterized by large ulcerations composed of whitish yellow tissue with multiple draining tracts that discharge bloody exudates. Yellow gray to tan gritty concretions (kunkers), 2–10 mm in diameter, are observed within the draining tracts or may be extruded from the wounds. Histologically, pythiosis is characterized by eosinophilic granulomatous inflammation; kunkers are composed of sequestered vessels that have undergone coagulative necrosis, Pythium hyphae, and necrotic eosinophils. 7
In cattle the lesion is characterized by epidermal ulcerations associated with dermal thickening. Microscopically, there are multifocal granulomas with intralesional P. insidiosum hyphae, but kunkers are not observed. 17,21
In Brazil, pythiosis has been reported in horses from the states of Mato Grosso 5 and Rio Grande do Sul 11,19,20 and the semiarid region of the state of Paraíba 22 and in cattle in the state of Mato Grosso do Sul. 21 This report describes two outbreaks of pythiosis in sheep in the semiarid region of the state of Paraíba, northeastern Brazil.
One outbreak affected 40 sheep in a flock of 120 hair wool crossbred sheep in the municipality of Catingueira, state of Paraíba. The outbreak occurred when the flock stayed in a paddock surrounding a large pond, grazing for long periods at the border of the pond, where green grass was always present. The farmer reported that 36 sheep of different ages died in a period of 240 days. The clinical manifestation period was 60–90 days. In June 2001, two dead (sheep Nos. 1 and 2) and one live (sheep No. 3) adult sheep were sent to the Veterinary Hospital of the Federal University of Campina Grande.
A second outbreak was observed in the municipality of Coremas, state of Paraíba. Six of 80 crossbred hair wool sheep of different ages were affected during a 60-day period. One dead sheep (sheep No. 4) was sent in November 2002 to the Veterinary Hospital. The flock had a history of standing for long periods, during the dry season, in swampy water at the border of a large pond.
Clinical signs were characterized by ulcerative lesions of the limb (Fig. 1), ventral abdomen, and prescapular region (Fig. 2). Limb lesions measured 2.5–24 cm dorsoventrally and 2.7–5 cm craniocaudally. The ulcers were dry or wet and had a dark red or brown to black surface.
Right hind limb; sheep No. 4. An ulcerated lesion is observed in the region of the metatarsus.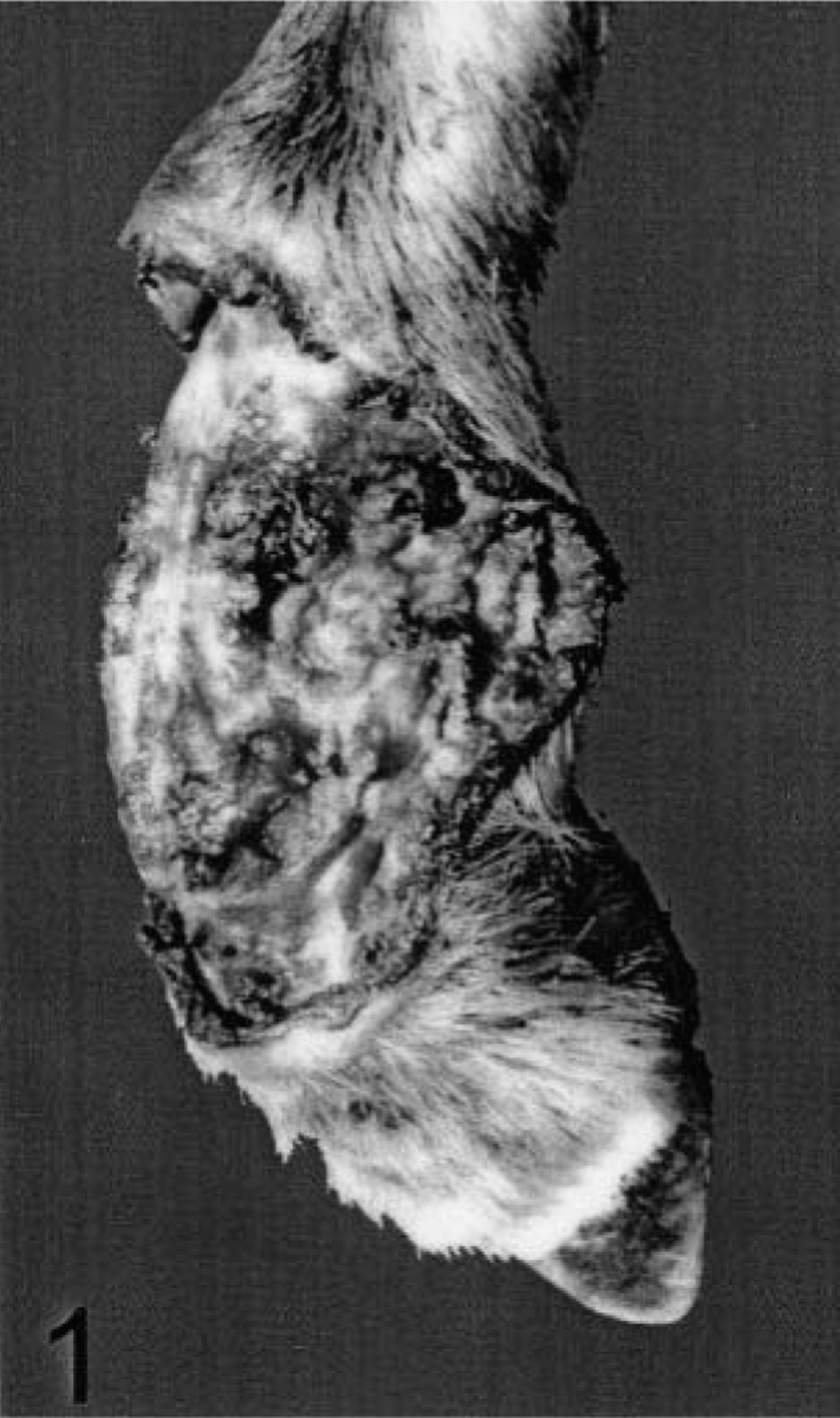
Sheep No. 4. A swelling measuring 6.8 cm craniocaudally and 11.5 cm dorsoventrally, with an ulcerated area is observed in the left prescapular region.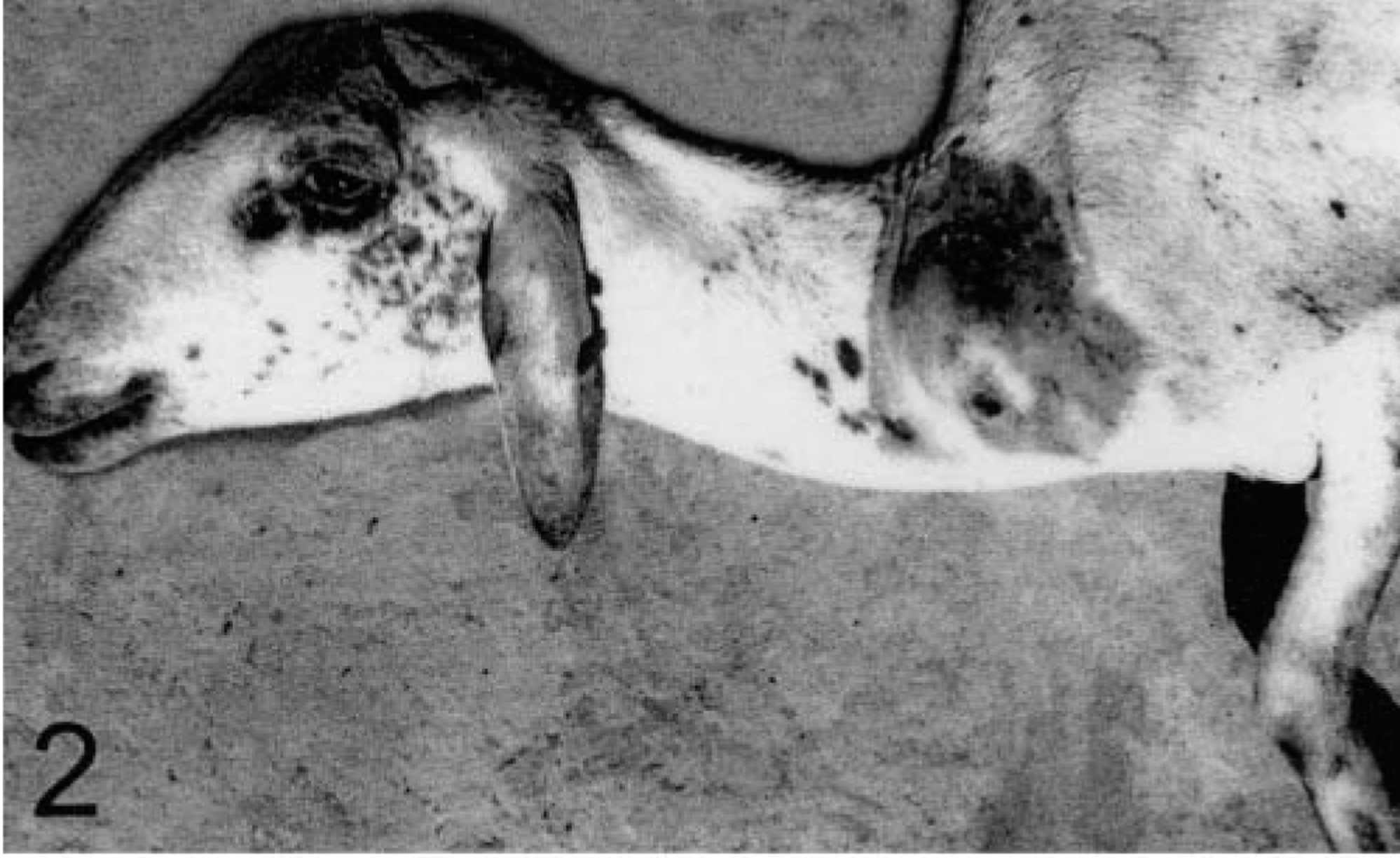
Sheep No. 1 had large lesions on the right metatarsal and left metacarpal regions and a smaller lesion on the right carpal region. At necropsy, this sheep was emaciated and had multifocal subpleural nodules in the lung, mainly in the caudal lobes. These lesions were characterized by several well-demarcated, slightly raised, nonulcerated nodules, 2 × 0.5 cm in diameter, with a gray to yellow center surrounded by a red halo at the periphery; the cut surface was whitish with multifocal yellow areas bordered in red.
Sheep No. 2 had an ulcerated lesion on the tibial region of the right hind limb and an ulcerated mass on the ventral abdominal skin. At necropsy, the sheep was emaciated. The mass in the abdomen measured 15 cm craniocaudally, 6 cm dorsoventrally, and 7 cm transversely. The cut surface was characterized by a yellowish soft tissue surrounded by a thin capsule of white tissue.
Sheep No. 3, which arrived alive, had a 4.2- × 3-cm ulcerated lesion on the left metacarpal region. A biopsy sample from this lesion including the skin and the sesamoid bone was obtained for histologic analysis. This sheep was treated orally with 7 mg/kg of body weight of potassium iodide, daily, for 7 days. Fifteen days after the start of the treatment the sheep recovered completely.
Sheep No. 4, which came from the second farm, had two large lesions: one affecting the skin of the left metatarsus and tarsus and another on the skin of the left metacarpus (Fig. 1). This sheep had also a swelling in the prescapular region (Fig. 2), affecting the prescapular lymph node. At necropsy the sheep was emaciated. The cut surface of the prescapular swelling was characterized by a 14-cm-deep soft yellow tissue surrounded, in the deep layers, by a white consistent connective tissue with some hemorrhagic areas. Pulmonary multifocal lesions, similar to those observed in sheep No. 1, were scattered in the caudal lobes.
In the dermis, the lesions were characterized by multifocal, sometimes coalescent, granulomas. In the center of these granulomas hyphaelike structures were surrounded by eosinophilic Splendore-Hoeppli material. Deposits of eosinophilic granules, presumably the remnants of eosinophils, were also observed. Necrotic eosinophils, neutrophils, and undifferentiated mononuclear cells were observed around the Splendore-Hoeppli material. These central areas were surrounded by epithelioid macrophages, giant cells, undifferentiated mononuclear cells, and less frequently eosinophils and plasma cells (Fig. 3). Well-vascularized loose connective tissue formed by interlacing fibers of immature collagen, sometimes infiltrated by undifferentiated mononuclear cells, eosinophils, and plasma cells, was observed between the granulomas. Occasionally, fibrinous thrombi were observed in the dermis below the ulcer. In the legs, the lesion extended frequently to the tendon sheaths. Occasionally, focal areas of necrosis were observed within the tendons. In the abdominal lesion of sheep No. 2, the lesion extended to the panniculus adiposus. In sheep No. 4, the lesion extended to the panniculus adiposus and the prescapular lymph node. The prescapular lymph node was diffusely necrotic with little identifiable lymphatic tissue. Numerous large foci with Splendore-Hoeppli material, eosinophilic granules, and necrotic eosinophils were observed within the remnants of lymphatic tissue. Loose connective tissue infiltrated by many eosinophils, neutrophils, and mononuclear cells was also present. Rarefaction of the lymphatic tissue and infiltration by eosinophils were observed in the remnants of medullary sinuses. There was marked proliferation of the collagenous capsule of the lymph node with multifocal accumulation of Splendore-Hoeppli material and infiltration by plasma cells, eosinophils, and undifferentiated mononuclear cells.
Sheep No. 1. Dermis below an ulcerated lesion. A granuloma containing hyphaelike structures (black arrow) surrounded by eosinophilic Splendore-Hoeppli material and numerous eosinophils was observed. This central area is surrounded by epithelioid macrophages. A giant cell is also observed (arrow head). HE. Bar = 100 µm.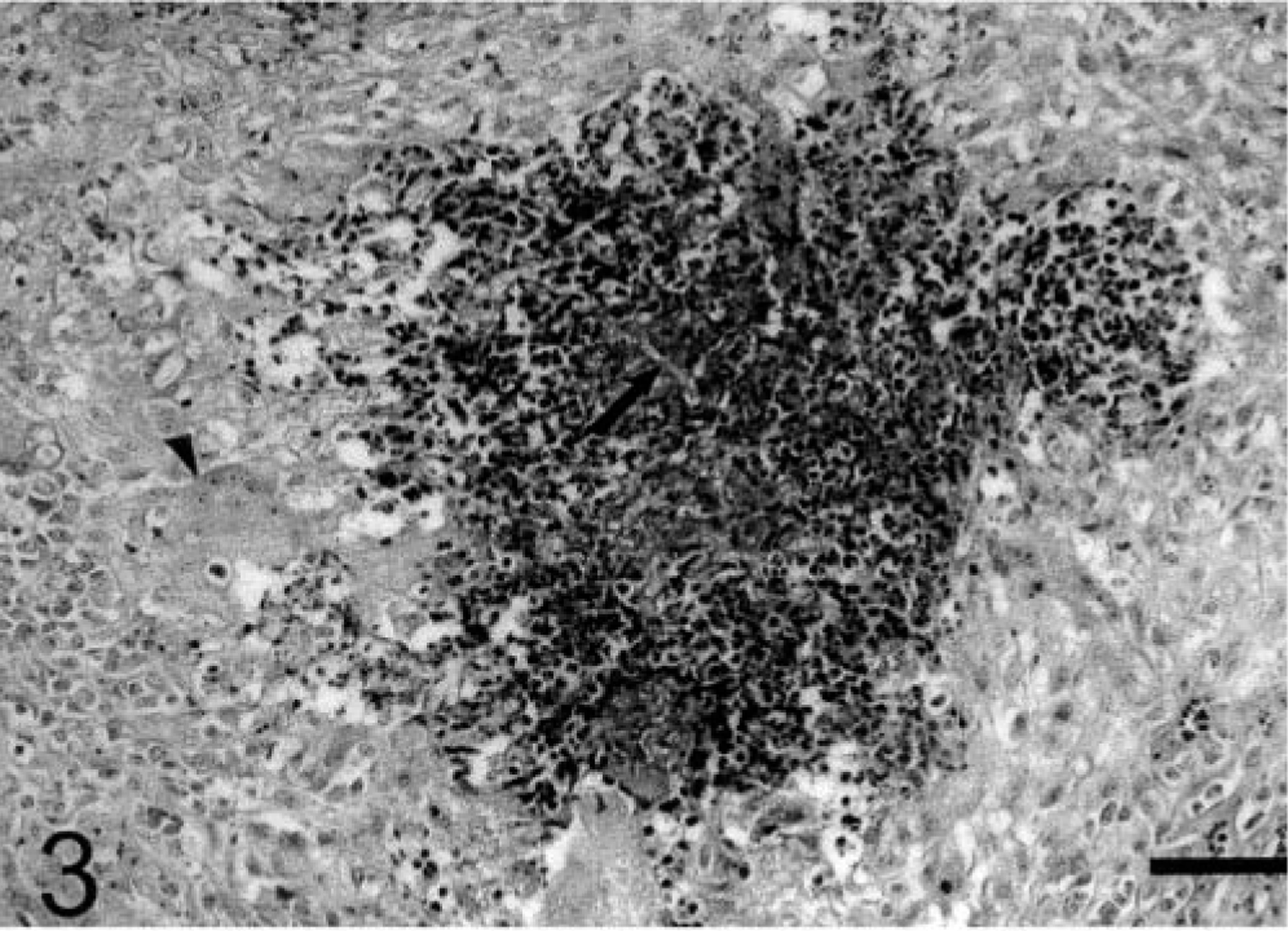
Lung lesions were characterized by large necrotic centers surrounded by plasma cells, undifferentiated mononuclear cells, and rarely giant cells and degenerated and necrotic eosinophils and neutrophils. Splendore-Hoeppli material was observed scattered within the necrotic centers. In the lung tissue around the lesion, the septa were thickened by edema, severe congestion, and infiltration by mononuclear cells. Edema and some macrophages were observed within the alveoli. The pleura was thickened by edema and proliferation of the connective tissue infiltrated by mononuclear cells, plasma cells, macrophages, and some eosinophils. In the sesamoid bone from sheep No. 3, the yellow bone marrow was partially substituted by necrotic unidentifiable tissue; eosinophilic granules were observed scattered within this necrotic tissue.
In the dermis, lung, and prescapular lymph node, in the centers of the granulomas, within the Splendori-Hoeppli material, Gomori methenamine silver stain revealed thick-walled, irregularly branched, rarely septate hyphae, 3–10 µm in diameter (Fig. 4). Numerous hyphae were observed also in the crust covering ulcers and in the necrotic marrow of the sesamoid bone. No hyphae were observed within the fibrinous thrombi in the dermis.
Sheep No. 1. Dermis below an ulcerated lesion. Thick-walled, irregularly branched, occasionally septate hyphae observed at the center of a granuloma. Gomori methenamine silver. Bar = 50 µm.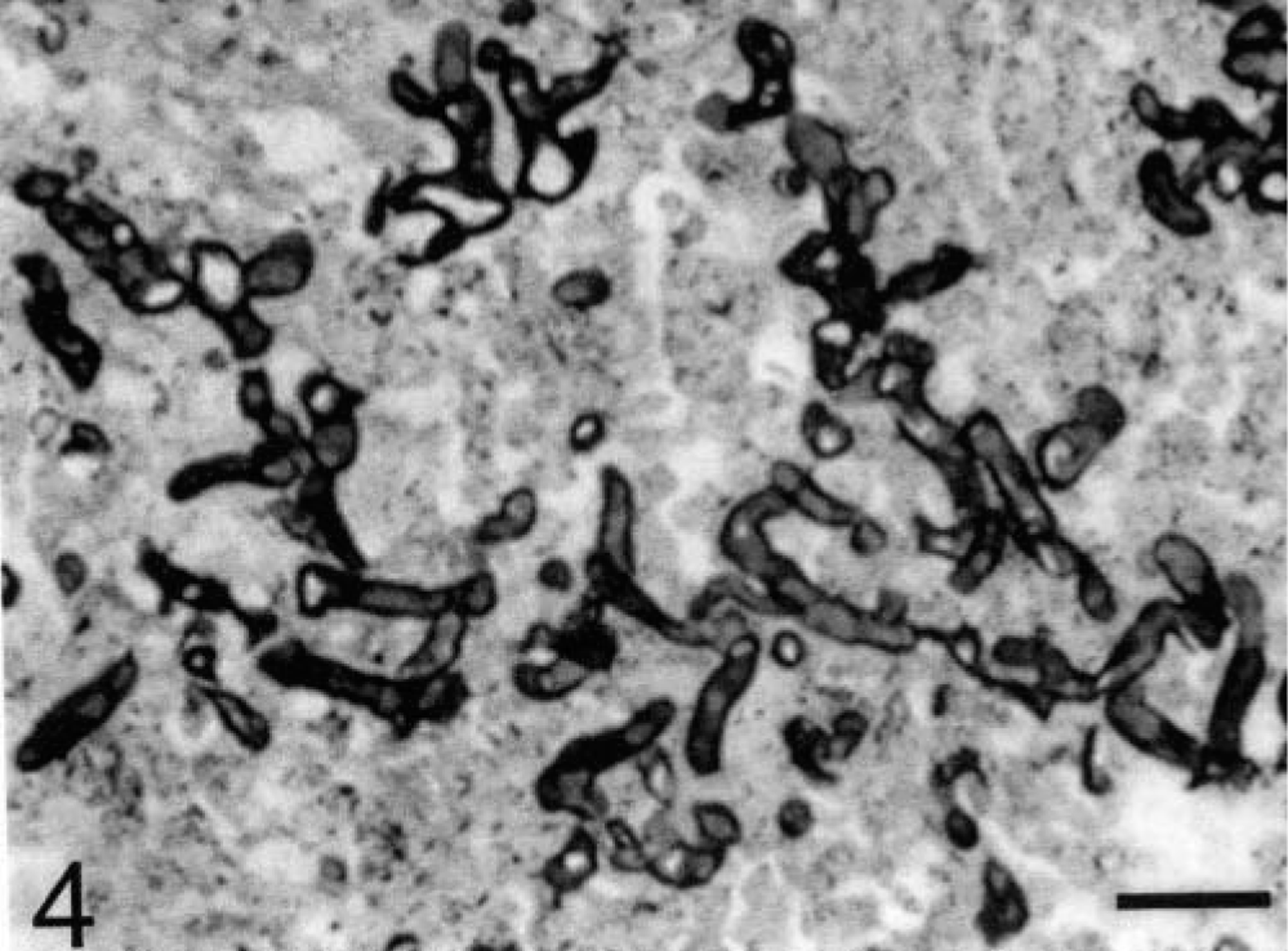
Immunohistochemical staining was performed on sections of lesions from two sheep (sheep Nos. 1 and 4) with specific antibodies (sent by Joe Newton, Ausburn, AL). 4 The slides were submitted for 20 minutes to a 3.5% H2O2 solution to inactivate endogenous peroxidase activity. Then, they were treated with 2% goat serum for 30 minutes. The sections were incubated with the primary antibody (anti-Pythium antibody produced in rabbits, diluted in phosphate-buffered saline at 10%, with 0.1% bovine serum albumin) for 45 minutes. A commercial streptavidin–biotin complex–peroxidase kit (Large volume Dako LSAB 2 kit Peroxidase Universal K675, Dako Corporation) was used according to the manufacturer's instructions. After addition of the substrate 3-amino-9-ethylcarbazol (DAB, Sigma), the sections were stained with hematoxylin, dehydrated through successive ethanol and xylene baths, and mounted for light microscopy. All samples were processed with Pythium-positive samples as control, and a case of cutaneous habronemiasis was used as the negative control. Lesions from both sheep were positive for P. insidiosum, Paraffin-embedded samples from the two sheep sent to Dr. Leonel Mendoza from the Medical Technology Program, Michigan State University, were negative by immunohistochemical staining with primary antibodies against Conidiobolus coronatus and Basidiobolus haptosporus and positive to P. insidiosum,
To cultivate P. insidiosum, specimens of cutaneous lesions were vigorously washed many times in sterile distilled water, minced into approximately 1-mm cubes, implanted into Sabouraud dextrose agar, and incubated at 37 C for 48 hours. 13 After a 48-hour incubation, cottonlike mycelial growth was observed. On microscopic examination, after clarification with 10% potassium hydroxide, the hyphae were branched at right angles and rarely septate. Although zoosporogenesis was not performed, the organisms were morphologically similar to P. insidiosum,
Despite the large number of sheep that died with similar clinical signs, 40 sheep in the flock of 120 and 8 in the flock of 80, only a few animals were subjected to pathologic examination. Because of the similar clinical signs and clinical manifestation period observed by the farmers in sheep affected previously, it was assumed that the nonexamined sheep also died of pythiosis. After the diagnosis of pythiosis the farmers removed the sheep from the paddocks, and no more cases were observed. A return visit was made to the farms after the withdrawal of the flocks from the paddocks, and no lesions were found in sheep examined.
This article reports the first few cases of pythiosis in sheep. A presumptive diagnosis of pythiosis was based on the histologic lesions with thick-walled, irregularly branched, occasionally septate hyphae 3–10 µm in diameter. A definitive diagnosis was made by immunohistochemical staining with specific antibodies and culture of the etiologic agent.
Clinical signs and pathology in sheep pythiosis have aspects similar to those observed in horses, but kunkers, which are characteristic in equine pythiosis, were not observed in sheep. Also, in cattle, kunkers are not observed. 17,21 In horses the disease is accompanied by intense pruritus, 7,24 which was not observed in the affected sheep and has not been reported in cattle. The clinical evolution of the disease seems to be different within species; in horses, the disease is refractory to treatment, and the prognosis is guarded in lesions of more than 4 weeks. Complete excision with 2-cm margins and regional perfusion of topical antifungal agents are recommended. 24 On the other hand, spontaneous healing has been recorded in cattle. 21 In our observations the only sheep that was treated with potassium iodide fully recovered in 15 days, suggesting that treatment of affected sheep may be effective. Spontaneous recovery, as it occurs in cattle, probably does not occur in sheep because all affected nontreated sheep in both farms in our study died.
As reported in horses and cattle, most lesions in sheep are localized in the extremities because the infection occurs when skin is in contact with contaminated water. After initial infection of the skin, metastasis to the lungs and regional lymph nodes and extension of the lesion to the underlying bone and tendon sheath occur in sheep. In horses, metastases are occasionally observed in the intestinal tract, 2,18 bone, 1,9,14 lung, 6 joints, and tendon sheath. 1,14 The high prevalence of disease in both sheep flocks, the occurrence of metastases in the regional lymph nodes and lung, and the spreading of the lesion to the underlying bone and tendon sheath suggest that sheep are highly susceptible to pythiosis and that the occurrence of the disease depends on the exposure and the ecologic conditions for multiplication and transmission of the agent.
In both outbreaks, the occurrence and high prevalence of pythiosis in sheep seem to have been strongly related to the ecology of the aquatic organism and the requirements of high environmental temperatures for rapid growth and asexual reproduction. In the semiarid region of Paraíba, temperatures are high throughout the year (mean of 28 C), and rains usually only occur from January or February to April or May. To overcome the long dry period, the construction of large ponds for water reservation is very common. During the dry season, green pastures are only found at the borders of these ponds, and animals frequently stand in these swampy waters for long periods. High prevalence of pythiosis is reported also in horses in this region, 22 and similar ecologic conditions are associated with the high prevalence of pythiosis in horses and cattle in other Brazilian regions 11,21 and tropic and subtropic regions in other countries. 1
Footnotes
Acknowledgements
We are grateful to Dr. Leonel Mendoza from the Medical Technology Program, Michigan State University, for assisting in the immunohistochemical staining with primary antibodies against Conidiobolus coronatus, Basidiobolus haptosporus, and Pythium insidiosum, This work was financially supported by the Programa de Apoio a Núcleos de Excelência (PRONEX), Grant N° 7697102600.
