Abstract
Objectives
Cyclooxygenase-2 (COX-2), a pivotal enzyme in the cyclooxygenase family, plays a critical role in carcinogenesis. While its expression is well documented in various neoplasms in humans and dogs, data on COX-2 expression in feline neoplasms, particularly nasal malignant epithelial tumours, is limited. This study aimed to evaluate COX-2 expression in feline nasal malignant epithelial tumours through immunohistochemistry. We hypothesised that these tumours would exhibit COX-2 expression, consistent with findings in humans and dogs.
Methods
Formalin-fixed, paraffin-embedded biopsy samples from feline nasal malignant epithelial tumours were retrospectively analysed for COX-2 expression by immunohistochemistry. Biopsies from cats previously treated with non-steroidal anti-inflammatory drugs were excluded. Immunohistochemistry was performed with a monoclonal rabbit antibody, with feline renal macula densa cells serving as the positive control. The immunoreactive score (IRS) combined a semiquantitative estimation of immunolabelled neoplastic cells with labelling intensity. Scores in the range of 0–1 were classified as negative, 2–3 as low, 4–8 as intermediate and greater than 8 as high COX-2 expression levels.
Results
A total of 18 feline nasal biopsies (nine adenocarcinomas, seven carcinomas, one squamous cell carcinoma and one mucinous carcinoma) were included. Clinical signs included nasal discharge, sneezing, epistaxis and inspiratory dyspnoea. COX-2 expression was not detected in any case (IRS = 0). Follow-up data were available for 7/18 cats. The overall median survival time after diagnosis in our cohort was 667 days (range 0–1642).
Conclusions and relevance
In contrast to canine nasal malignant epithelial tumours, COX-2 expression was not observed in feline nasal malignant epithelial tumours. These results suggest species-specific differences in COX-2 expression in nasal malignant epithelial tumours. Further studies evaluating other carcinogenesis pathways, such as vascular endothelial growth factor or platelet-derived growth factor, seem crucial to better understand feline nasal malignant epithelial tumours and to improve their therapeutic management.
Introduction
Cyclooxygenase-2 (COX-2), a pivotal enzyme within the cyclooxygenase family, plays a crucial role in the synthesis of prostanoids, which serve as key mediators of inflammation and pain.1,2 COX-2 expression, akin to cyclooxygenase-1, is induced in response to specific stimuli, including hormonal triggers, inflammatory cytokines, growth factors and oncogenes. 1
In carcinogenesis, COX-2 primarily acts on the tumoral microenvironment.3 –7 It exhibits antiapoptotic properties by inhibiting B-cell lymphoma 2, enhances angiogenesis through stimulation of vascular endothelial growth factor (VEGF) production, promotes vascular sprouting, migration and tube formation, activates epidermal growth factor (EGF) and induces matrix metalloproteinases.3,4 In addition, it plays a role in modulating the tumour microenvironment immunologically, allowing tumour cells to evade host immune defences by modulating cytokine production, impairing dendritic cell function and suppressing lymphocyte proliferation.5 –7
In both humans and dogs, COX-2 expression has been observed in a wide range of malignant neoplasms, including pulmonary adenocarcinoma, squamous cell carcinoma, colonic adenocarcinoma, prostatic carcinoma and urothelial carcinoma.8 –11 However, research on COX-2 expression in feline neoplasms remains limited, with only mild expression reported in urothelial carcinoma, oral squamous cell carcinoma, mammary carcinoma, olfactory neuroblastoma and low-grade alimentary lymphoma.12 –15 Moreover, expression of COX-2 has been identified as a negative prognostic factor, associated with malignancy criteria and shortened survival time in various canine neoplasms (mammary carcinoma, mast cell tumour, melanoma, osteosarcoma and renal carcinoma) and feline mammary tumours. 13
In both humans and dogs, COX-2 overexpression has been documented in nasal malignant epithelial tumours.9,16 –19 Consequently, non-steroidal anti-inflammatory drugs (NSAIDs) have become a component of therapeutic management, along with radiation therapy, chemotherapy, tyrosine kinase inhibitors (toceranib) and surgery.20 –23
In cats, sinonasal neoplasms account for 1–8.4% of all tumours, with 90% being malignant.24 –26 Among these, lymphoma comprises the majority, accounting for 50% of cases, while fewer than half originate from epithelial tissues.24 –28 For nasal lymphoma, one study including only six cases did not find COX-2 expression. 12 However, no study to date has investigated COX-2 expression in feline nasal malignant epithelial tumours.
The aim of this study was to evaluate COX-2 expression in feline nasal malignant epithelial tumours using immunohistochemistry. We hypothesised that feline nasal malignant epithelial tumours would express COX-2, as reported in humans and dogs. A secondary aim of this study was to describe the outcomes of cats diagnosed with nasal malignant epithelial tumours.
Materials and methods
Study population
A retrospective search was conducted on the electronic veterinary medical records database of the veterinary teaching hospital of Alfort, École nationale vétérinaire d’Alfort (France), focusing on cats diagnosed with nasal malignant epithelial tumours between 1 January 2007 and 1 January 2020. The inclusion criteria comprised a histopathological diagnosis of nasal malignant epithelial tumour retrieved from our biobank. Cats treated with NSAIDs, steroids, radiation therapy, chemotherapy, surgery or tyrosine kinase inhibitors before biopsy were excluded. Informed consent was obtained from the owner or legal custodian of all animals described in this work.
The following data were recorded for each cat at diagnosis: signalment; clinical signs; duration of clinical signs; prior treatments; body weight and body condition score; complete blood count (CBC); blood smear; biochemistry results; and histological analysis. In addition, results from diagnostic imaging examinations and rhinoscopy were recorded. If a CT scan was performed, the images were reviewed by a diplomate of the European College of Veterinary Diagnostic Imaging. Reports of rhinoscopy performed at the time of diagnosis by a diplomate of the European College of Veterinary Internal Medicine (ECVIM) or an internal medicine resident under the supervision of an ECVIM diplomate were reviewed. Retrograde endoscopy was performed using a flexible bronchoscope (BFQ190, 4.9 mm, 60 cm; Olympus), while antegrade endoscopy was performed using a rigid rhinoscope (HOPKINS 0° 2.7 mm 18 cm; Storz). Staging was determined with a clinical stage assigned using the modified Adams clinical staging method for nasosinal tumours whenever possible. 29 For each cat, treatment following diagnosis was recorded.
To investigate COX-2 expression in the nasal mucosa of healthy cats, a retrospective search was performed using the electronic veterinary medical records database of the veterinary teaching hospital of Alfort, École nationale vétérinaire d’Alfort (France), to select cases of nasal biopsies with no pathological findings and no history of previous NSAID treatment investigated between 1 January 2007 and 1 January 2020.
Histopathological analysis and immunohistochemistry
All samples were fixed in 10% formalin and embedded in a paraffin block. The final diagnosis of nasal malignant epithelial tumour was established by a diplomate of the European College of Veterinary Pathologists (ECVP) on histological sections stained with haematoxylin-eosin-saffron.
Sections were prepared from paraffin-embedded tissues for immunohistochemical examinations. Expression of COX-2 was assessed immunohistochemically. Sections with a thickness of 5 μm were placed on Superfrost Plus microscopy slides (Thermo Fisher Scientific). Automated immunohistochemistry was performed on the Discovery ULTRA system (Roche; Ventana Medical Systems). Slides were deparaffinised in aqueous-based detergent solution (Discovery Wash; Ventana). Heat-induced antigen retrieval was performed using CC1 solution (Discovery Cell Conditioning Solution 1, pH 7.8) at 95°C for 8 mins then at 100°C for 12 mins. Primary antibody COX-2 (D5H5 XP Rabbit monoclonal antibody #12282, 1:500; Cell Signaling Technology) was incubated at 37°C for 32 mins. Secondary antibody (OmniMap anti-Rabbit HRP; Roche) was incubated at 37°C for 16 mins. The immunoreaction was revealed with a ChromoMap DAB kit (Roche) and counterstained with haematoxylin. A section from a feline kidney, known to express COX-2 in the macula densa cells, was used as a positive control, as previously described. 30 Negative control was assessed using phosphate-buffered saline instead of the primary antibody.
Evaluation of immunoreactivity
A semiquantitative estimation of immunolabelled neoplastic cells was conducted by an ECVP diplomate according to a previously published technique, 18 based on the intensity of the labelling and the immunoreactivity. The immunoreactive score (IRS) was determined from the percentage of positively labelled tumour cells and their average staining intensity (Table 1). This assessment was performed at a magnification of × 200, with scores averaged across five fields. The IRS was then calculated by multiplying percentage and intensity scores, resulting in a range of 0–12. Scores in the range of 0–1 were classified as negative, 2–3 as low, 4–8 as intermediate and greater than 8 as high COX-2 expression levels. Tumour samples with an IRS of 2 or higher were defined as having positive expression. 18
Immunoreactive score (IRS) used to characterise COX-2 expression
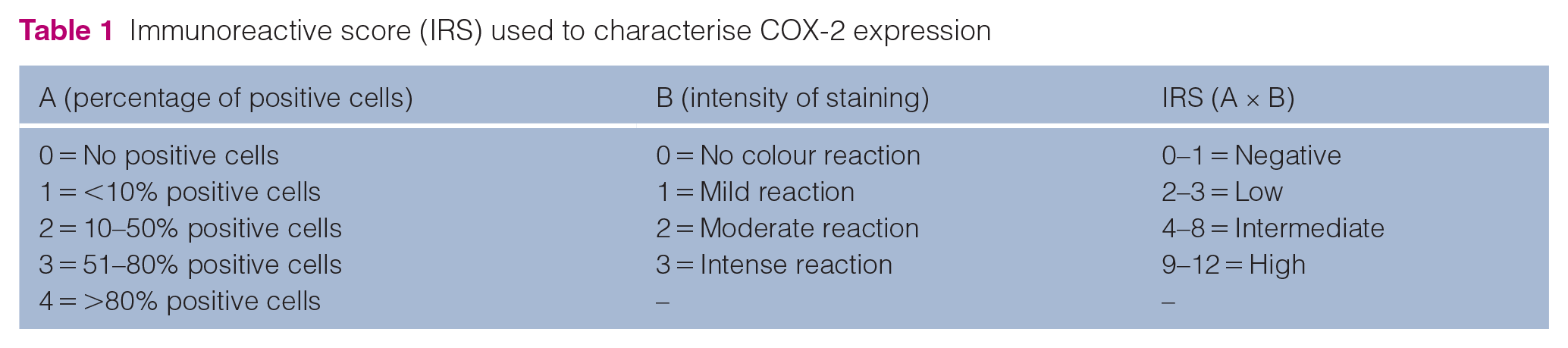
Survival time
Survival time was calculated from the date of diagnosis until death. Cats were censored from the survival analysis if they were lost to follow-up, if they died of another disease, or if they were alive at the time of data analysis.
Results
Case selection
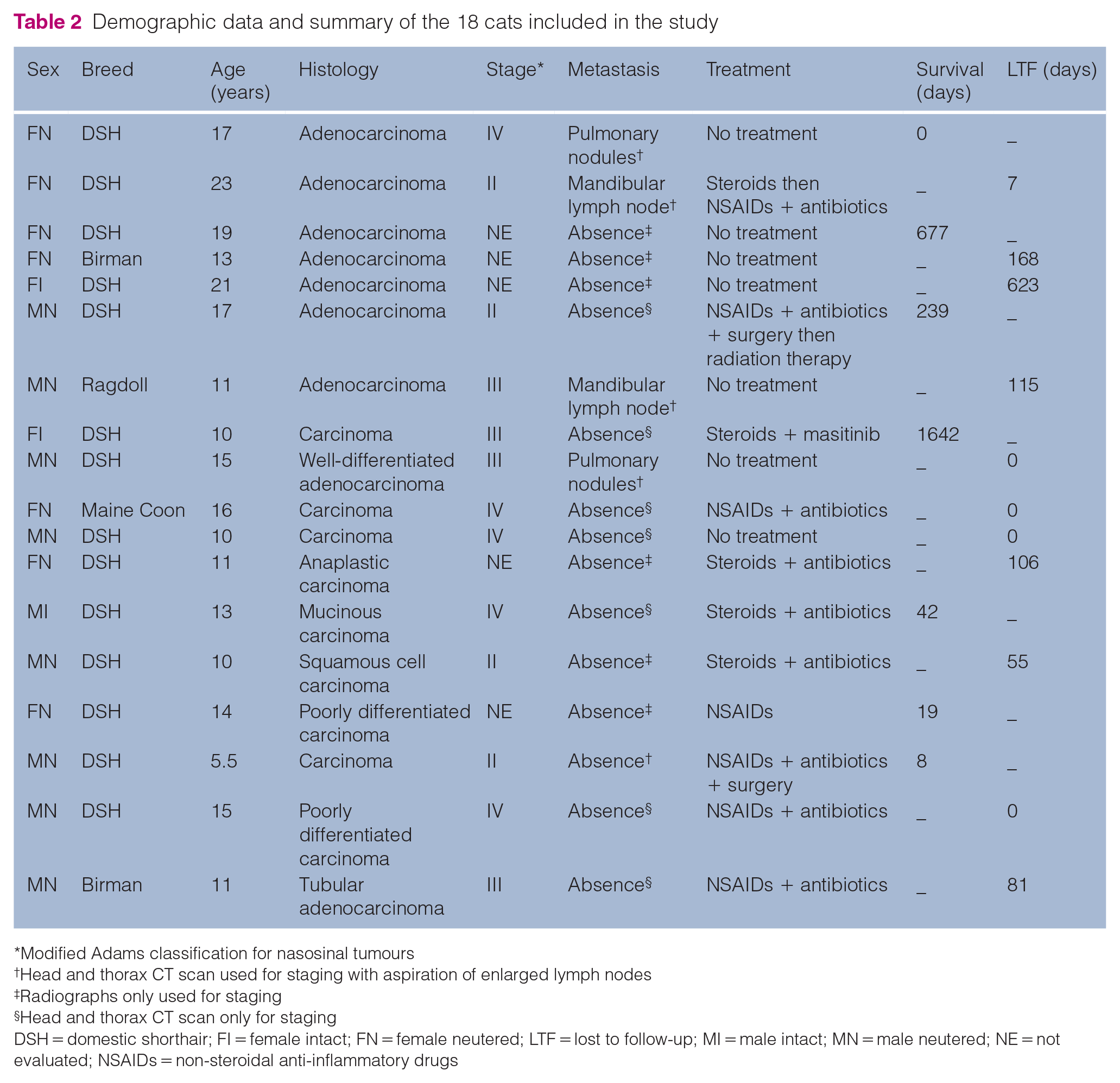
A total of 18 cats diagnosed with a nasal malignant epithelial tumour met the inclusion criteria (Table 2).
Demographic data and summary of the 18 cats included in the study
Modified Adams classification for nasosinal tumours
Head and thorax CT scan used for staging with aspiration of enlarged lymph nodes
Radiographs only used for staging
Head and thorax CT scan only for staging
DSH = domestic shorthair; FI = female intact; FN = female neutered; LTF = lost to follow-up; MI = male intact; MN = male neutered; NE = not evaluated; NSAIDs = non-steroidal anti-inflammatory drugs
Breeds included 14 (77.8%) domestic shorthair cats, two (11.1%) Birman cats and one (5.6%) each of Maine Coon and Ragdoll. The median age at diagnosis was 13.5 years (range 5.5–23). Nine cats were male, including one intact male, and nine were female, including two intact females.
The most common clinical signs observed at presentation included unilateral or bilateral serous (8/18, 44%) or mucopurulent (4/18, 22%) nasal discharge, sneezing (11/18, 61%), epistaxis (9/18, 50%) and inspiratory dyspnoea (9/18, 50%). The mean duration of clinical signs before diagnosis was 4.3 months (range 0.5–24).
Comorbidities included stage III chronic kidney disease (International Renal Interest Society classification) (n = 1) and hyperthyroidism treated with methimazole (n = 3).
Clinicopathological findings
Staging
A CBC was performed in 12/18 cats. Abnormalities included mildly regenerative anaemia (4/12, 33%), leukocytosis (4/12, 33%), leukopenia (3/12, 12%), thrombocytopenia (1/12, 9%) and thrombocytosis (1/12, 9%). A serum biochemistry analysis was available for 12/18 cats. Abnormalities included an increase in serum total protein concentration (6/12, 50%) and an increase in serum creatinine concentration (5/12, 42%). Electrolyte levels were evaluated in 5/18 cats, with one cat showing mild hypernatraemia. Ionised calcium levels were assessed in 4/18 cats, and all measurements fell within the reference intervals.
A full-body CT scan was performed in 13/18 cats. Tumour lesions were unilateral in 7/13 (54%) cases and bilateral in the remaining six cases. Other abnormalities detected included lytic bone lesions of ethmoturbinates, the cribriform plate and the hard palate (9/13, 69%; 5/13, 38%; 2/13, 15%, respectively). In total, 13 cats were staged using the modified Adams clinical staging method: five cats were stage IV, four were stage III and four were stage II.
Locoregional lymphadenopathy was observed on CT scans in 6/13 (46%) cases. Among those cases, mandibular lymph nodes were bilaterally enlarged in all cases and medial retropharyngeal lymph nodes were enlarged in two cases. All lymph nodes were sampled for cytological examination. Only two were diagnosed with metastasis from a nasal adenocarcinoma.
Pulmonary nodules were noted on CT scans in 2/13 (15%) cats, both diagnosed with nasal adenocarcinoma. For one cat, numerous nodules with a diameter of 2 mm were observed throughout the lung parenchyma, while a single nodule of 3.5 mm (right caudal lung lobe) was detected for the other one. None of the nodules were sampled for further analysis. For these two cats, metastasis to locoregional lymph nodes was not observed. For five cats, thoracic CT was not carried out and staging was performed with three-view thoracic radiographs. No thoracic metastasis was identified in those cases.
Rhinoscopy was performed in 8/18 (44%) cats, showing a bilateral nasal mass in 4/8 (50%) cases and choanal obstruction in 5/8 (63%) cats.
Histopathological analysis
Histopathological samples were obtained by CT-guided biopsy in 10/18 cases and endoscopy-guided biopsy in 8/18 cases. Histopathological subtypes included adenocarcinoma (n = 9/18), carcinoma (n = 7/18), squamous cell carcinoma (n = 1/18) and mucinous carcinoma (n = 1/18).
Immunoreactivity and COX-2 expression
A homogeneous cytoplasmic expression of COX-2 was noted for the positive control (feline macula densa cells) (Figure 1).

Immunohistochemistry using an anti-COX-2 rabbit monoclonal antibody demonstrates intense positive cytoplasmic staining of the feline macula densa (arrow), used as a positive control
All feline nasal malignant epithelial tumours were negative for COX-2 expression (IRS = 0) (Figure 2).

Immunohistochemistry using an anti-COX-2 rabbit monoclonal antibody on a feline nasal carcinoma. Lack of staining indicates an absence of COX-2 expression
Nasal tissue samples from 10 healthy cats tested negative for COX-2 expression (IRS = 0).
Treatment
Of the 18 cats, 11 received treatment after diagnosis. Six cats were treated with NSAIDs (meloxicam 0.05 mg/kg PO q24h or piroxicam 0.3 mg/kg PO q24h) and four cats received oral prednisolone (0.5–1 mg/kg PO q24h), and one cat was switched to meloxicam because of a lack of response to prednisolone. Two cats underwent surgical debulking (ventral rhinotomy and dorsal sinusotomy). One of these cats received external beam radiation therapy (six fractions of 5 Gy over 6 weeks) owing to tumour recurrence 2 months after surgical excision.
One cat received masitinib (9 mg/kg PO q48h for 1 month then q72h for 50 months). Antibiotics were prescribed for 9/18 cats (amoxicillin clavulanate n = 6, doxycycline n = 2, metronidazole/spiramycin n = 1).
Outcomes
The overall median survival time after diagnosis was 667 days (range 0–1642) (Figure 3; Table 2). Eleven cats were lost to follow-up at the time of study completion. One cat was euthanased at the time of diagnosis. Euthanasia was performed in four cases due to tumour progression at 19, 42, 677 and 1642 days following diagnosis. For the two remaining cats, euthanasia was elected due to urethral obstruction 8 days after diagnosis and multiple traumas 239 days after diagnosis.

Kaplan–Meier survival curve for the 18 cats in the study. The median survival time in the population was 667 days. Tick marks represent patients lost to follow-up
Discussion
To the best of the authors’ knowledge, this study is the first to establish the absence of COX-2 expression using immunohistochemistry in feline nasal malignant epithelial tumours.
COX-2 expression was not observed in any of the neoplastic tissues. These results show a difference between feline and canine nasal malignant epithelial tumours regarding COX-2 expression. In dogs, expression of COX-2 was found in 71–81% of nasal malignant epithelial tumours, with a mean IRS of 7.5 (range 2–12), using the same method as described in our study.9,31 Another study investigated the impact of COX-2 expression on outcomes for dogs diagnosed with nasal carcinoma treated with a hypofractionated radiation protocol. Among samples from 42 dogs, expression of COX-2 was shown in 91% of cases, with a mean IRS of 6.6 (range 0–12). 18 However, expression of COX-2 was not found to be associated with outcome. No study has specifically investigated the influence of histological subtypes on COX-2 expression in canine nasal malignant epithelial tumours, but the limited available data suggest that there is no difference in expression profiles.9,18,20,32
In humans, expression of COX-2 was found in 45–60% of nasal malignant epithelial tumours.16,17 Differences in COX-2 expression were assessed depending on histological subtype, with a marked expression found in tobacco-associated nasal adenocarcinoma (92%) and weak expression in nasal squamous cell carcinoma (8%). 16
Studies on COX-2 expression on feline neoplasia are limited to a small number of cases. COX-2 expression was reported for urothelial carcinoma, head or neck cutaneous squamous cell carcinoma and low-grade alimentary lymphoma.12,14,33 Conversely, the absence of COX-2 expression was reported in feline pulmonary adenocarcinoma, intestinal adenocarcinoma, lymphoma and vaccine-associated sarcoma. 10
Possible explanations for the lack of COX-2 expression in feline nasal malignant epithelial tumours observed in this study include the actual absence of COX-2 expression, unrepresentative tissue sampling and issues with the fixation method. Since biopsies represent only a small portion of the tumours compared with excisional biopsies, our findings may not fully capture the overall expression if COX-2 expression is heterogeneous within the tumours. However, multiple biopsies were taken at the time of diagnosis and several slides from these biopsies were analysed. In addition, formalin fixation and/or tissue processing may damage or denature proteins, potentially impairing antigen recognition by immunohistochemistry.34,35 False-negative results due to overfixation are unlikely, as all samples were fixed for less than 5 days. Although the monoclonal antibody used in our study had not previously been tested in cats, it has been widely employed in human research with similar methodologies.36 –38 Given the strong and specific labelling of the positive control (feline macula densa cells) in our study, a lack of reactivity on feline tissues is considered unlikely. However, additional studies using other commercially available anti-COX-2 antibodies are needed to confirm our preliminary findings.10,12,39
Various techniques and methodologies, such as western blot or immunohistochemistry, have been described to assess COX-2 expression in cancer, each with varying degrees of accuracy. 10 This may explain the significant variability in the results of studies investigating COX-2 expression in the same type of cancer and within the same species. Although western blot could have been considered for identifying COX-2 expression in our study, this technique has limitations. 40 Specifically, western blot does not allow for the distinction between COX-2 expression in neoplastic cells and inflammatory cells. This limitation can complicate interpretation, particularly given that nasal tumours can be associated with peritumoral inflammation.
COX-2 expression can be induced by tumour development or inflammatory processes, although some organs constitutively express COX-2. Its expression in healthy nasal epithelium has been demonstrated in humans and, occasionally, in dogs.41 –43 To the best of the authors’ knowledge, no studies have investigated the constitutive COX-2 expression in nasal epithelium of healthy cats. In this study, none of the nasal biopsies from healthy cats showed COX-2 expression; however, further studies are needed to confirm this initial observation.
In our study, most of the cats were treated with NSAIDs only. Radiation therapy remains the standard treatment for the management of feline nasal malignant epithelial tumors.44,45 A recent study using definitive intent external beam radiation therapy (10 daily fractions of 4.2 Gy) reported a median time to progression of 269 days and a median overall survival time of 452 days. 44 Definitive radiation protocol was associated with improved progression-free survival (504 days) and overall survival (721 days) when compared with palliative protocol (198 days and 284 days, respectively). 45 Cyclical hypofractionated protocols (QUAD Shot) have also shown similar survival time (460 days). 46 The longest survival in our population was observed in a cat treated with masitinib. In one study, 4/9 (44%) dogs with nasal carcinoma treated with toceranib showed clinical benefit, though none achieved a complete response and only two had a partial response. 22 The median survival time was 298 days. 22 To the authors’ knowledge, no studies have investigated the potential of tyrosine kinase inhibitors in the treatment of feline nasal malignant epithelial tumours.
While COX-2 expression was not observed in our study population, cats with nasal malignant epithelial tumours may still benefit from anti-COX-2 therapy for its anti-inflammatory and analgesic properties.
This study has several limitations because of its retrospective nature and the small number of cases. In particular, given that the calculated median survival time is based on a small sample size, caution should be exercised before drawing any conclusions. The sampling method may not always capture a representative portion of tumour tissue, potentially leading to false negatives. Moreover, marker expression within a tumour sample may be heterogeneously distributed.
Further investigation into additional biomarkers is necessary to determine optimised therapeutic management for cats with nasal malignant epithelial tumours. A subset of canine nasal carcinoma expresses platelet-derived growth factor receptors, EGF receptors or VEGF receptors, offering potential therapeutic options with tyrosine kinase inhibitors.20,22,47,48 Recent studies have shown prognostic significance of EGF receptor expression in canine nasal carcinoma, underlining the importance of conducting further studies in a feline population. 20
Conclusions
Our study found an absence of COX-2 expression in feline nasal malignant epithelial tumours, contrasting with findings in dogs. Despite this, anti-COX-2 treatment may still help alleviate clinical signs and improve the comfort of cats with these tumours. Further investigation through larger prospective studies is needed to deepen our understanding of the role of COX-2 in the development of feline nasal malignant epithelial tumours and its potential impact on prognosis.
Footnotes
Acknowledgements
We would like to extend our sincere appreciation to Dr Laura Barrett DVM, DACVIM-Oncology, for her English language editing.
Author note
This paper was presented as a poster at the 2022 European Society of Veterinary Oncology congress (Siracusa).
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approvement
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognised high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers, tissues and samples) for all procedure(s) undertaken (prospective or retrospective studies). For any animals or people individually identifiable within this publication, informed consent (verbal or written) for their use in the publication was obtained from the people involved.
