Abstract
Objectives
Serum galectin-3 (sGal-3) is a protein present in renal tubules and increases in experimental rodent models of acute kidney injury. The aim of this study was to compare sGal-3 concentrations in healthy cats and cats with ureteral obstruction (UO).
Methods
This was a retrospective study. Banked serum was used for sGal-3 evaluations in 15 healthy control cats and 22 cats with UO. For the control cats, creatinine and symmetric dimethylarginine were within reference intervals and ultrasound showed minimal to no kidney changes. A feline-specific sGal-3 ELISA was used to determine sGal-3 concentrations. Samples were analyzed in duplicate, and results were included if the coefficient of variation between samples was <20%. Shapiro–Wilk testing was used to evaluate for normality and parametric statistics were performed. P <0.05 was considered significant.
Results
Mean (±SD) sGal-3 was lower in healthy cats (274.3 ± 146.5 pg/ml) than in cats with UO (707.7 ± 223.3 pg/ml; P <0.0001). There was no difference in sGal-3 concentrations between cats with unilateral or bilateral UO (P = 0.24) and no correlation between sGal-3 and creatinine, body weight or age. With a cutoff of 500 pg/ml, sGal-3 had a sensitivity of 86% (95% confidence interval [CI] 0.67–0.95) and specificity of 100% (95% CI 0.79–1.00) for differentiating cats with UO from healthy cats.
Conclusions and relevance
An early diagnosis of UO is critical, given its associated morbidity and mortality. Given the differences in sGal-3 concentrations in healthy cats and those with UO in this study, sGal-3 shows potential for an early UO diagnosis; however, further research is needed.
Introduction
The diagnosis of acute kidney injury (AKI) using traditional biomarkers, such as creatinine, has several limitations. The reliance on serum creatinine alone can overestimate kidney function in a variety of physiological states 1 and significant kidney injury can occur while creatinine is still within the reference interval (RI). 2 Novel biomarkers of kidney injury may provide earlier AKI diagnosis, thereby allowing for earlier intervention. This may be especially beneficial in cats with ureteral obstruction (UO) where more rapid renal decompression may lead to better preservation of kidney function and thereby improved outcomes. Galectin-3 (Gal-3) is a lectin protein expressed by macrophages, epithelial cells and endothelial cells in a wide range of mammalian tissues. 3 In adult mice, Gal-3 expression has been detected in the proximal tubule, the thick ascending limb of the loop of Henle, as well as in principal and intercalated cells. 4 Galectin proteins have been shown in humans and non-feline species to play a role in both intracellular and extracellular regulation of cell growth, 5 inflammation, 5 apoptosis 5 and immune responses to infectious agents. 6 Gal-3 expression within the kidneys is upregulated in both ischemic and toxic AKI; 7 in humans, Gal-3 levels upon admission to the intensive care unit were higher in patients with AKI 8 and in those who developed a major adverse kidney event. 8 Gal-3 inhibition attenuated hypertensive nephropathy in a rat study, 9 leading to a concurrent decrease in renal damage and proteinuria.
It is well documented that significant kidney injury develops within 24 h after UO, and that prolonged UO leads to progressive and irreversible fibrosis and chronic kidney disease (CKD).10,11 UO is a common cause of AKI in cats,12,13 which causes significant morbidity and mortality. In addition, it is common for non-azotemic subclinical AKI to occur in feline patients when the contralateral kidney is healthy. In addition, in early or partial UO, misdiagnosis can occur when significant dilation of the ureter or renal pelvis is not present on ultrasonographic examination. As such, novel biomarkers that allow for early UO diagnosis are critical. Gal-3 has shown promise in its correlation with UO-induced AKI, where increased expression was found in mice with iatrogenic unilateral UO. 14 We therefore aimed to measure and compare serum galectin-3 (sGal-3) concentrations in healthy control cats and in cats with UO. We hypothesized that sGal-3 concentrations would be higher in cats with UO compared with healthy control cats.
Materials and methods
Animals
Cats with UO
A retrospective study that used stored banked serum from cats previously diagnosed with UO that presented to University of California, Davis William R Pritchard Veterinary Medical Teaching Hospital between March 2014 and May 2019 was analyzed. Cats included in the study underwent abdominal ultrasonography and were diagnosed with UO by a board-certified radiologist or radiologist in training under the guidance of a board-certified radiologist. Cats were further categorized as having unilateral or bilateral UO. The presence of concurrent CKD was also determined for each cat and was defined as a previous diagnosis of CKD based on known historical elevations of creatinine above the reference baseline more than 3 months before presentation to the hospital or ultrasonographic evidence of CKD at the time of presentation. Urine output was estimated by the attending clinician at the time of sGal-3 sampling, and oliguria was defined as <1 ml/kg/h over a 12–24 h period. Serum samples were collected at the time of the initial presentation to the hospital and were stored at −80°C until analysis.
Healthy control cats
Banked serum from healthy control cats stored at −80°C was used. Clinically healthy cats were recruited from the University of California, Davis William R Pritchard Veterinary Medical Teaching Hospital. Cats were included as healthy controls if patient history, physical examination by a board-certified internist, clinicopathologic parameters and ultrasonographic evaluation were consistent with a healthy cat as outlined below. To be included, cats had to have serum concentrations of creatinine, blood urea nitrogen (BUN), symmetric dimethylarginine (SDMA), phosphorus, total calcium, ionized calcium and total T4 within established RIs. Normal urine dipstick analysis and urine specific gravity >1.030 were also required.
Ultrasonographic evaluation of the kidneys was performed in each control cat by a board-eligible veterinary radiologist, with images reviewed by a board-certified radiologist before inclusion. Kidneys were assessed for the following abnormal characteristics: renal length <2.9 cm; reduced corticomedullary distinction; heterogeneous renal parenchyma; diffuse or focal cortical or medullary hyperechogenicity; medullary rim sign; renal parenchymal mineralization; or evidence of >1 renal infarct. Control cats were excluded from the study if more than one of these abnormalities were detected on ultrasonography.
Measurement of sGal-3 concentration
sGal-3 concentrations were measured using a feline-specific sGal-3 ELISA. The sGal-3 ELISA assay was performed and interpreted according to the manufacturer’s instructions. 15 Briefly, all reagents and samples were brought to room temperature (18–25°C). Serum was diluted 100-fold and 100 μl standard of the diluted cat serum specimen was added to each well and incubated for 2.5 h at room temperature. Subsequently, 100 μl of prepared biotinylated anti-feline sGal-3 antibody was added to each well and incubated for 1 h at room temperature. Then, 100 μl of prepared streptavidin solution was added to each well and incubated for 45 mins. Between each step, the ELISA plates were washed four times with the provided wash solution. Consequently, 100 μl TMB One-Step Substrate Reagent was added to each well and incubated for 45 mins at room temperature. After the final incubation, 50 µl of stop solution was added to each well and the plate was read at 450 nm immediately (Multiskan FC Microplate Photometer; Thermo Scientific). The assays were performed in duplicates. Mean sGal-3 concentrations were recorded. If the coefficient of variation (COV) between the duplicate samples (intra-assay COV) was above 20%, the results were discarded. For values below the minimum detectable level of feline sGal-3 of 4.5 pg/ml, 15 a numerical value of 0 was given. For values above the maximal detectable level of feline sGal-3 of 1000 pg/ml, a numerical value of 1001 pg/ml was given.
Statistical analysis
Analyses and statistics were performed using commercially available software (Prism version 10.0; GraphPad). Shapiro–Wilk testing was used to evaluate for normality and parametric statistics were performed. Normally distributed data were expressed as mean ± SD and non-normally distributed data as median (interquartile range [IQR]). Demographic data were summarized with descriptive statistics. Two-sample unpaired t-tests were performed to assess the differences in sGal-3 concentrations between UO cats and healthy cats. A receiver operator characteristic (ROC) curve was created and the area under the ROC curve (AUC) calculated for sGal-3 prediction of UO status. An optimal cutoff value was determined using Youden’s J statistic. The corresponding sensitivity, specificity and likelihood ratios with 95% confidence intervals (CIs) are reported. Spearman’s rank correlation coefficient was used to assess for correlations between sGal-3 and creatinine, age, BUN and body weight. Spearman (r) values of 0–0.4 were considered to have a weak association, values of 0.5–0.7 a moderate positive association and values >0.7 were considered to have a strong correlation. 16 Among the UO group, sGal-3 concentrations between cats with unilateral or bilateral UO, cats with and without CKD, and cats with and without oliguria were assessed using two-sample unpaired t-tests after evaluation of normality using Shapiro–Wilk testing. P <0.05 was considered significant.
Results
A total of 24 adult cats with UO were included (13 castrated males, 11 spayed females). Breeds consisted of domestic shorthair (n = 14), domestic mediumhair (n = 3), domestic longhair (n = 2), Siamese (n = 2), Ragdoll (n = 1), Savannah (n = 1) and Burmese (n = 1). The mean age was 7.6 ± 2.7 years, with a mean body weight of 4.6 ± 1.1 kg. Median serum creatinine was 6.9 mg/dl (IQR 3.1–15.85) and mean serum BUN was 129 ± 83.5 mg/dl. In total, 15 cats had unilateral UOs and nine cats had bilateral UOs. One cat with a unilateral UO had concurrent renal agenesis of the contralateral kidney.
In total, 15 healthy control adult cats were included in the study (seven castrated males, eight spayed females). Breeds consisted of domestic shorthair (n = 7), domestic mediumhair (n = 2), domestic longhair (n = 4), Siamese (n = 1) and Bengal (n = 1). The mean age was 5.6 ± 3 years, with a mean body weight of 4.8 ± 1.4 kg. Mean serum creatinine was 1.3 ± 0.2 mg/dl and mean serum BUN was 26 ± 4.2 mg/dl.
Of the initial 24 cats with UO, 2/9 cats with bilateral UO had duplicate sGal-3 concentrations that had a COV >20% and were excluded from further analysis. Mean sGal-3 concentrations were therefore determined for 22 cats with UO (15 unilateral, seven bilateral) and 15 healthy control cats.
The average intra-assay COV was 4.1%. The mean sGal-3 concentrations were lower in healthy cats (274.3 ± 146.5 pg/ml) compared with cats with UO (707.7 ± 223.3 pg/ml; P <0.0001) (Figure 1). The optimal cut-point to discriminate between UO and healthy cats was 500 pg/ml, yielding a sensitivity of 86% (95% CI 0.67–0.95) and specificity of 100% (95% CI 0.79–1.0) for the detection of UO (Figure 2). The AUC was 0.95 (95% CI 0.88–1.0).

Serum galectin-3 concentrations from cats with ureteral obstruction (UO) and healthy cats. Each solid circle represents a single cat and the horizontal red line represents the mean for each group

Receiver operator characteristic curve for the detection of ureteral obstruction using serum galectin-3 compared with ultrasonographic detection of UO. Area under the curve = 0.95 (95% confidence interval 0.88–1.0)
Age was higher within the UO group (P = 0.02). There was no difference in body weight between cats with UO and healthy control cats (P = 0.65). Among cats with UO and healthy control cats, there was no strong correlation between sGal-3 and age (r = 0.3, P = 0.05) or body weight (r = 0.07, P = 0.65). Among the cats with UO, there was no strong correlation between sGal-3 and creatinine (r = 0.43, P = 0.05) or between sGal-3 and serum BUN (r = 0.37, P = 0.09). There was no linear correlation between increasing serum creatinine and sGal-3 concentrations (Figure 3).
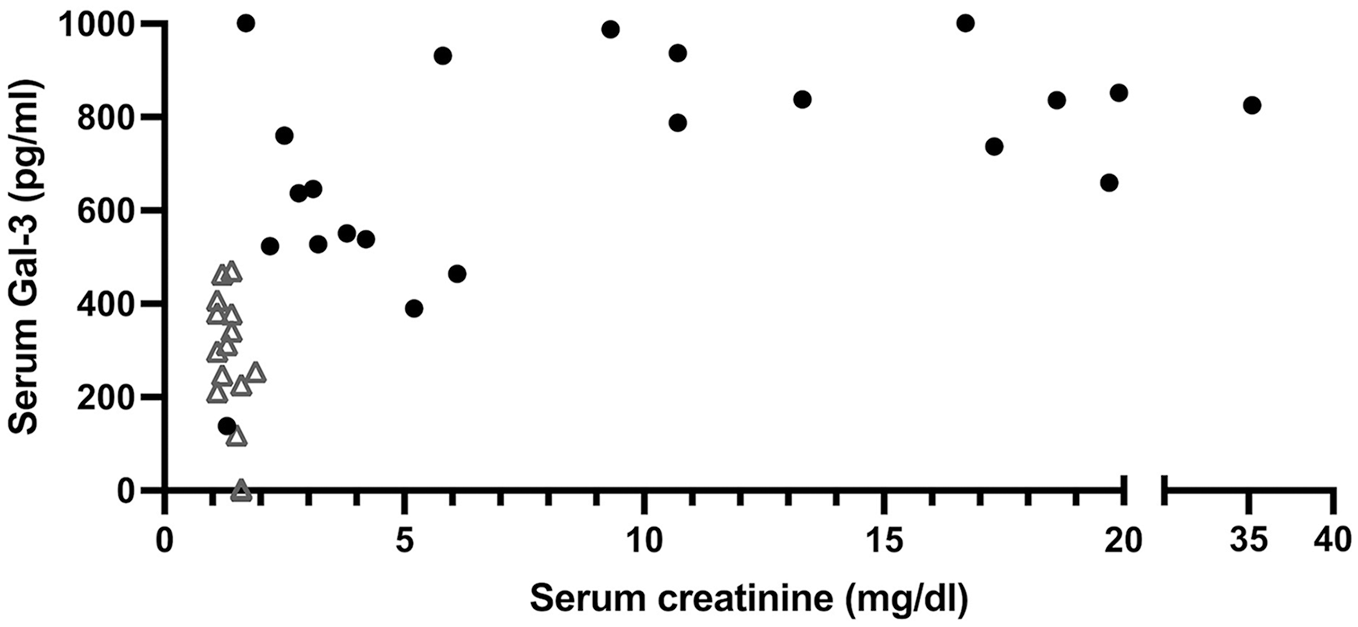
Plot demonstrating serum creatinine concentration vs serum galectin-3 concentration for cats with ureteral obstruction (black solid circles) and healthy cats (open triangles)
Among the cats with UO, there was no difference in sGal-3 concentrations between unilateral (n = 7) and bilateral (n = 15) UO (P = 0.24), cats with (n = 11) and without (n = 8) pre-existing CKD (P = 0.28), or cats with (n = 6) and without (n = 12) documented oliguria (P = 0.39) (Figure 4).

Serum galectin-3 concentrations in cats with ureteral obstruction subcategorized by (a) presence or absence of chronic kidney disease (CKD), (b) presence or absence of oliguria and (c) unilateral vs bilateral UO. Each solid circle represents a single cat and the horizontal red line represents the mean for each group
Discussion
This preliminary pilot study demonstrated that sGal-3 concentrations were higher in cats with UO than in healthy cats. In this cohort of cats, using a cutoff value of 500 pg/ml, sGal-3 had a sensitivity of 86% and specificity of 100% in differentiating between cats with UO and healthy cats. As opposed to humans, where elevations in sGal-3 concentrations have been shown to correlate inversely with estimated glomerular filtration rate (GFR),8,17,18 there was no linear association between sGal-3 and serum creatinine in this study, indicating that sGal-3 is unlikely to be a filtration marker or marker of GFR in this cohort of cats. This study found no difference in sGal-3 concentrations between subcategories of cats with UO, including unilateral vs bilateral disease, presence or absence of CKD, or urine output status. However, sGal-3 concentrations were higher in cats with UO than in healthy cats, suggesting that this marker may potentially be a good candidate as a marker for cats with AKI secondary to UO.
UO has been shown to initiate marked inflammation and fibrosis, 19 and the inflammatory response to UO may itself cause substantial kidney parenchymal injury. 12 In a murine model, renal Gal-3 expression correlated with renal inflammation and fibrosis. 20 Increased glomerular expression of Gal-3 was found in individuals with lupus who also had concurrent kidney injury and inflammation. 21 Further studies have demonstrated Gal-3 to be involved in not only the production of pro-inflammatory cytokines, but also the induction of reactive oxygen species. 22 As above, unilateral UO is often used as a model for renal fibrosis induction, 23 and elevated renal Gal-3 correlates with the development of renal fibrosis in mice.24,25 Gal-3 knockout mice showed less renal fibrosis after kidney transplantation compared with controls, 26 elucidating Gal-3 as having a role in the development of renal fibrosis. Urinary Gal-3 levels have also been examined in humans who underwent kidney biopsies, with higher urinary Gal-3 levels associated with more severe renal fibrosis. 27 The elevated sGal-3 in cats with UO in the present study may represent renal inflammation secondary to tissue and cellular damage that occurs with UO, although currently no studies have assessed this link in feline patients.
No differences in sGal-3 concentrations were noted between several subcategories of cats with UO. While sGal-3 was performed on stored serum specimens initially obtained at presentation to our institution, the duration of clinical signs and any administration of treatment before presentation was not consistently known. In the study by Okamura et al 14 using mice with unilateral UO, peak levels of Gal-3 expression within the kidney occurred 7 days after obstruction. Another experimental study in rats showed that Gal-3 mRNA expression was elevated above baseline for 28 days after kidney injury. 7 Further studies evaluating changes in sGal-3 after successful renal decompression may give insights into the processes of kidney repair and recovery, or to persistent ongoing renal inflammation.
The study had some limitations. This was a retrospective study using previously banked serum and therefore the sample size was small for both the healthy cats and those with UO. As banked serum samples were used, the effects of storage at −80°C and handling on sGal-3 levels are not known. It is possible that degradation of sGal-3 may have occurred, although a prior study in humans demonstrated the stability of SGal-3 in stored samples for 12 months. 28 Samples from both cats with UO and healthy controls were handled and stored in a similar way and stored for a similar period. Study participants were not age matched between groups, with a median higher age within the UO group (P = 0.02). The effects of age on sGal-3 levels in cats are unknown; however, no relationship between sGal-3 and age was identified in one human study. 29 Although the ELISA assay used in this study was preliminarily assessed by the company, 15 external independent validation of the assay was not performed as part of this study. A further limitation of this study is the potential for overestimation of diagnostic accuracy of the ELISA in differentiating the groups, given the inherent bias of a two-gate accuracy test. 30 Elevations of Gal-3 have been documented in cats with hypertrophic cardiomyopathy 31 and hence may not be specific to renal injury in cats with UO. Potential cardiac changes secondary to anemia, uremia or fluid overload could occur in cats with UO. 32 Given the retrospective nature of enrolment in this study, echocardiography was not performed in all cats with UO in this study. It is therefore important to note that some of the Gal-3 elevations in these cats could have been secondary to a cardiac-related etiology.
Conclusions
Serum Gal-3 may be a useful biomarker for the diagnosis of UO in cats. Further studies are needed to better elucidate the role of Gal-3 in renal injury and recovery, and to understand the utility and specificity of sGal-3 as an early marker of non-azotemic AKI secondary to UO and other causes of AKI in cats.
Footnotes
Author note
This paper was presented in part at the 2023 ACVIM Forum, Philadelphia, Pennsylvania, USA.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Funding for this study was provided by a private donor in memory of our cherished patient, Star Hat.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognized high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers, tissues and samples) for all procedure(s) undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
