Abstract
Objectives
The aim of the present study was to establish a reference interval (RI) for urine kidney injury molecule-1 (KIM-1) in healthy cats.
Methods
History, physical examination, blood pressure, and feline immunodeficiency virus and feline leukemia virus serology status were determined. A complete blood cell count, serum biochemical profile, urinalysis and kidney ultrasound were performed, and N-terminal pro-brain natriuretic peptide, total thyroxine (TT4) and urine KIM-1 were measured. An RI was calculated and the effect of age, sex, body condition score (BCS), blood pressure, symmetric dimethylarginine (SDMA), serum creatinine concentration (SCr), phosphorus, TT4, urine specific gravity (USG) and mid-sagittal kidney length on urine KIM-1 was evaluated using a general linear model.
Results
Of 69 recruited cats, 50 met the inclusion criteria. There were 35 male cats and 15 female cats, with a median age of 4.3 years (range 1.0–12.3), median weight of 5.11 kg (range 2.52–8.45) and median BCS of 6/9 (range 3–8). The median serum concentrations were SDMA 11.0 µg/dl (range 2–14), SCr 88.5 µmol/l (range 47–136), phosphorus 1.41 mmol/l (range 0.8–2.2) and TT4 32.0 nmol/l (range 17–51). Median USG was 1.057 (range 1.035–1.076), mid-sagittal left kidney length was 3.50 cm (range 2.94–4.45) and mid-sagittal right kidney length was 3.70 cm (range 3.06–4.55). The derived RI for urine KIM-1 was 0.02–0.68. USG was a significant (P <0.001) predictor of urine KIM-1. Individually, age, sex, blood pressure, BCS, SDMA, SCr, phosphorus, TT4 and mid-sagittal kidney length were not significant predictors of urine KIM-1. In a multivariate model, if combined with USG, SDMA concentration was predictive (P = 0.030) of urine KIM-1.
Conclusions and relevance
Urine concentration was significantly correlated with urine KIM-1, which will be an important consideration when interpreting findings in cats with potential kidney injury.
Introduction
Chronic kidney disease (CKD) is one of the most common diseases in cats, affecting ⩾30–40% aged over 10 years.1–3 In both humans and cats, CKD is postulated to result from the accumulation of multiple, undetected acute kidney injuries (AKIs) throughout life.4–12 Current assays for assessing kidney disease in cats, including increased serum creatinine concentration (SCr) and decreased urine specific gravity (USG), are reflective of reduced kidney function; however, they are insensitive for the detection of kidney injury. Increases in SCr occur only days to weeks after kidney injury or unilateral nephrectomy in healthy cats, and may not exceed the upper limit of the reference interval (RI) until approximately 50% of kidney function is lost. 13 In addition, AKI in cats is not always associated with a significant decrease in USG. 12 Furthermore, SCr and USG may be influenced by factors independent of renal function or injury, including muscle mass and hydration status, respectively.14,15 Symmetric dimethylarginine (SDMA), while potentially more sensitive than SCr in the detection of AKI, may be influenced by non-renal diseases, including neoplasia, nephrolithiasis and diabetes mellitus.16 –18 More sensitive and specific indicators of kidney injury have been identified in humans, rodents and cats.19 –23 Among these, urine kidney injury molecule-1 (KIM-1) is a type I transmembrane glycoprotein that facilitates the removal of apoptotic debris from the tubular lumen and promotes cell-to-cell and cell-to-matrix adhesion.24–29 KIM-1 is rapidly and markedly upregulated in regenerating epithelial cells in the S3 segment of the proximal tubules after ischemic, toxic, septic, hypovolemic, hypotensive or transplant-related injury.12,30,31 Conversely, KIM-1 is detected at low levels in healthy feline kidney tissue, likely reflecting normal turnover of proximal tubular cells.12,32 An extracellular portion of KIM-1 is released into urine, allowing for non-invasive measurement.32,33 Studies in multiple species, including cats, have shown that injured renal tubules upregulate and shed KIM-1 into urine before SCr increases above the RI.26,31,32,34 A point-of-care lateral flow assay (LFA) that detects urine KIM-1 in cats with monoclonal antibodies was developed specifically for feline KIM-1. 35 This LFA can be interpreted visually or with a reader that provides a numerical density reading in relation to an internal control. The assay is robust and suitable for research and clinical use. 36 Healthy cats had low but consistently detectable urine KIM-1, which is similar to findings in humans and rodents.30,31,37 However, urine from only 13 cats was previously evaluated, which was an insufficient sample size to generate an RI. An RI for urine KIM-1 in healthy cats would be useful for the interpretation of test results in disease states.
Materials and methods
Sample population
Healthy cats belonging to staff and students at the Ontario Veterinary College, University of Guelph and the Mississauga Oakville Veterinary Emergency and Specialty Hospital were recruited between June 2021 and March 2022 with informed consent. The health status of each cat was determined based on a review of clinical signs of hyporexia, polyphagia, polyuria, polydipsia, vomiting, diarrhea, lethargy, weight loss, coughing or sneezing in the 12 months preceding enrollment. Each cat was physically examined, blood pressure was measured, and serum N-terminal pro-brain natriuretic peptide (NT pro-BNP), SDMA and total thyroxine (TT4) were determined. A complete blood cell count (CBC), serum biochemical profile, urinalysis, kidney ultrasound, and feline immunodeficiency virus (FIV) and feline leukemia virus (FeLV) serology testing were performed. Animal use and procedures were approved by the University of Guelph Animal Care Committee (protocol number 4513) in accordance with the standards set by the Canadian Council on Animal Care and the Ontario Animals for Research Act.
Data collection
Owners completed a form that asked for the cat’s signalment and temperament as well as current and prior medication, adverse drug reaction, vaccination, anesthetic, environment (indoor vs outdoor) and diet history. Owners also indicated if their cat had experienced hyporexia, polyphagia, polydipsia, polyuria, diarrhea, weight loss, vomiting, lethargy, coughing or sneezing in the past 12 months. Cats received oral gabapentin (median dose 30.36 mg/kg, range 0–79.4) alone or in combination with trazodone (median dose 0 mg/kg, range 0–10.5) at least 1–2 h before examination and were allowed to acclimate to a quiet examination room for at least 10 mins. The cats were then physically examined by a veterinarian. This included a fundic examination with a penlight (Professional PenLite; Welch Allyn) and lens (Pans Retinal 2.2; Volk). Body condition score (BCS) and muscle condition score were determined according to the descriptions and illustrations provided by the World Small Animal Veterinary Association. 38 Hydration was determined by assessment of mucous membrane moisture and skin turgor. Blood pressure was measured from the forelimb or tail with the cat in lateral recumbency. Eight blood pressure measurements were obtained using a blood pressure cuff (Medical Neonatal Soft Disposable Blood Pressure Cuff; SunTech Medical or Disposable Cuffs; SoftCheck), sphygmomanometer (DS65 Durashock; Welch Allyn) and Doppler flow detector (Ultrasonic Doppler Flow Detector 811-B; Parks Medical Electronics). The first measurement was discarded and the remaining seven measurements were averaged. 39 A total of 4–5 ml of blood was then collected from either the jugular or saphenous vein, depending on the technician’s preference. Two milliliters of EDTA blood and 1 ml of serum were collected for CBC, biochemical profile, TT4 (AHL; Animal Health Laboratory, University of Guelph), and FeLV and FIV serology and NT pro-BNP assay (SNAP FIV/FeLV Combo and SNAP Feline proBNP tests; IDEXX; performed in house). Another 1 ml of serum was submitted to IDEXX for measurement of the SDMA concentration. Images of the kidneys (both left and right in the mid-sagittal and transverse planes) were then obtained via ultrasound using a linear (Lumify 12-4 MHz; Philips) or curvilinear (iU22 C8-5 MHz; Philips) ultrasound probe. A minimum of 2 ml of urine was collected via antepubic cystocentesis and/or from litterboxes containing non-absorbent litter (NOSORB; Catco Vet Products). The ultrasound images were reviewed by a board-certified radiologist using a standardized form to assess kidney length in the mid-sagittal plane and for loss of corticomedullary definition, presence of hyperechoic cortices, mineralization of the calices, nephrolithiasis, renal infarction, pelvic dilation, perirenal effusion and perirenal hyperechoic fat.40 –45
KIM-1 measurement
An aliquot of urine was submitted for urinalysis at the AHL and 20 µl was used to perform the feline KIM-1 LFA (Bioassay Works) as detailed previously. 35 The KIM-1 value was determined by dividing the test result by the control result. The use of a ratio with a standardized positive control accounts for slight variations in environmental humidity and ambient temperature, which may affect immunoassays.
Exclusion criteria
Cats were excluded if they were not amenable to gentle restraint despite gabapentin and trazodone administration or if they had a history of hyporexia, polyphagia, polyuria, polydipsia, vomiting, diarrhea, lethargy, weight loss, coughing or sneezing in the 12 months preceding enrollment. A history of anesthesia in the 2 months preceding enrollment, detection of a heart murmur or serum NT pro-BNP concentration >100 pmol/l,46–50 a previous diagnosis of hypertrophic cardiomyopathy or systolic blood pressure >160 mmHg 39 also disqualified cats from inclusion. Other exclusion criteria were evidence of blindness, retinal detachment, retinal hemorrhage or retinal edema on ophthalmologic examination, 51 a TT4 outside the RI, 52 evidence of pyuria (⩾5 white blood cells/high-power field), 53 casts in urine sediment, alanine transaminase (ALT) or alkaline phosphatase (ALP) above the RI, USG <1.035, 54 thrombocytopenia in the absence of platelet clumping, 55 blood urea nitrogen (BUN), SCr or SDMA above the RI, positive FeLV and/or FIV serology test results, or ultrasonographic abnormalities of the kidneys.
Statistical analysis
To investigate potential predictors of urine KIM-1, a general linear model was constructed, which included the continuous explanatory variables of age, SDMA, SCr, USG and kidney length, and the dichotomized variables of sex (male, female), blood pressure (<140 mmHg, ⩾140 mmHg), phosphorus (<1.5 mmol/l, ⩾1.5 mmol/l), TT4 ⩽30 nmol/l, >30 nmol/l) and BCS (<7/9, ⩾7/9). Normality was tested using a Shapiro–Wilk test and examination of the residuals.
Tolerance limit formulae from Krishnamoorthy and Mathew 56 using SAS software (SAS/STAT 9.4; SAS Institute) were used for the KIM-1 RI calculation.
Results
Sample population
A total of 69 cats were recruited, of which 50 met the inclusion criteria. Eight cats were excluded because SDMA was >14 µg/dl as the only abnormality, three were excluded because USG was <1.035 as the only abnormality and an additional two cats were excluded due to renal infarcts identified on ultrasound as the only abnormality. One cat each was excluded due to the following: (1) positive FeLV and FIV status; (2) SDMA >14 µg/dl, SCr >190 µmol/l and USG <1.035; (3) SDMA >14 µg/dl and TT4 <13 nmol/l; (4) TT4 >55 nmol/l; (5) ALT >105 U/l; and (6) ALP >60 U/l and reduced corticomedullary definition on ultrasound. In total, 49 cats had trace to moderate proteinuria but were retained in the study. Of the 50 cats that met the inclusion criteria, 35 were male (34 castrated, one intact) and 15 were female (all spayed). Domestic shorthairs were most common (n = 35); additional breeds were Sphynx (n = 1), Manx mix (n = 1), domestic mediumhair (n = 4), domestic longhair (n = 4), Himalayan (n = 1), Ragdoll/Siamese mix (n = 1), Munchkin (n = 1), Persian mix (n = 1) and Ragamuffin (n = 1). Three cats (6%) were aged >10 years and 32 (64%) were aged 1–6 years. None of the cats received regular prescription medications. All cats had normal hydration and muscle mass. Additional data, including further physical examination findings, oral sedation doses, laboratory results and ultrasound measurements, are summarized in Table 1.
Characteristics of study cats

IDEXX Laboratories
Animal Health Laboratory, University of Guelph
SDMA = symmetric dimethylarginine
Urine KIM-1
Urine KIM-1 results were normally distributed (Shapiro–Wilk, P >0.3 for all models) (Tables 2 and 3, Figure 1). The USG (slope = 6.8) was a significant (P <0.001) predictor of urine KIM-1 (Figure 2). When analyzed in conjunction with USG, SDMA (slope = 0.012; P = 0.03) was a significant but weak predictor of urine KIM-1. Individually, age (P = 0.51), sex (P = 0.85), BCS (P = 0.52), blood pressure (P = 0.71), phosphorus (P = 0.83), SDMA (P = 0.32), SCr (P = 0.48), TT4 (P = 0.88) and mid-sagittal kidney length (left, P = 0.31; right, P = 0.42) were not significant predictors of urine KIM-1. A urine KIM-1 RI of 0.02–0.68 was calculated to include 99% of values from the population of cats with 95% confidence.
Descriptive statistics of urine KIM-1 results

KIM-1 = kidney injury molecule-1
Tests for normality

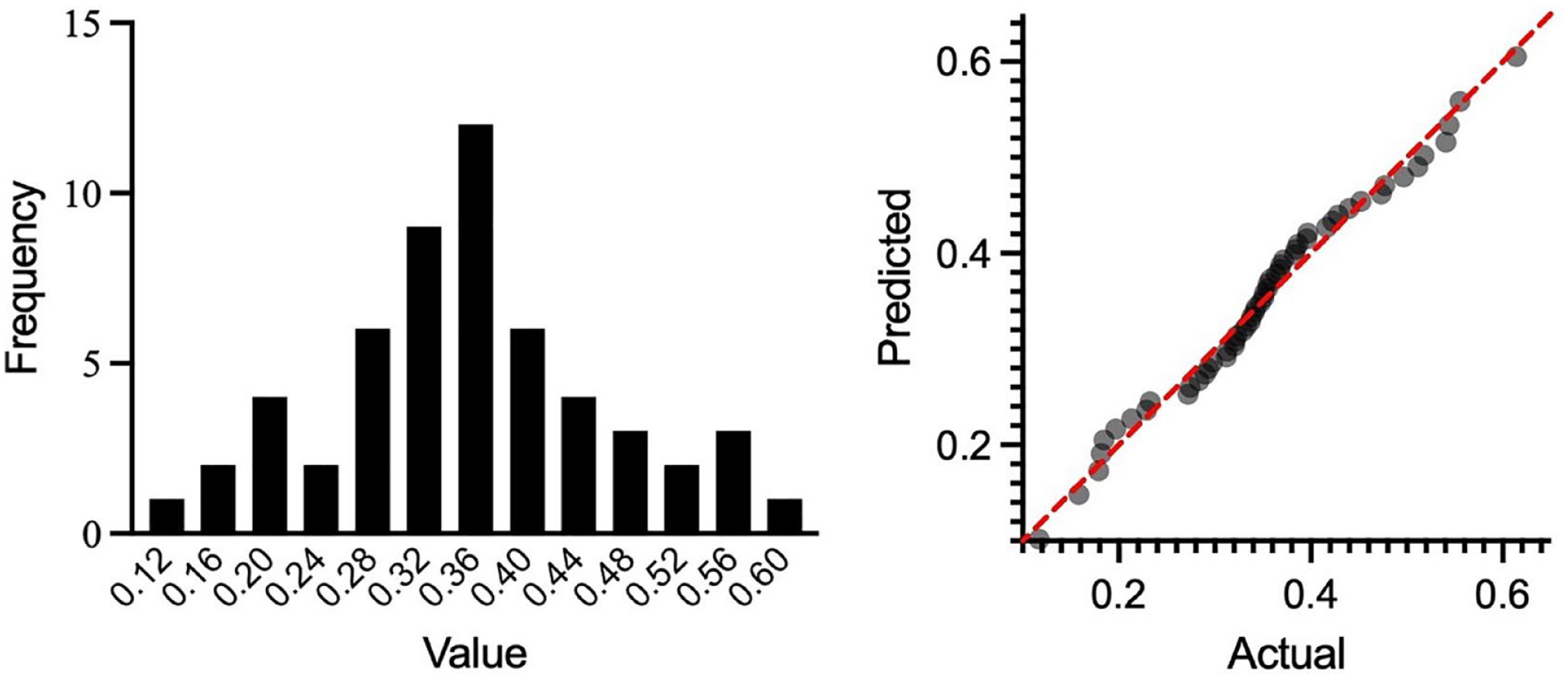
Left: Relative frequency distribution of urine KIM-1 values in healthy cats. Right: Quantile–quantile plot to illustrate the normal distribution of residual urine KIM-1 values. KIM-1 = kidney injury molecule-1

Urine specific gravity (USG) as a significant predictor of urine KIM-1 in 50 healthy cats. The formula for predicted data (predicted urine KIM-1 = 6.86 [USG] + 0.012 [SDMA] − 7.03) incorporates both USG and SDMA in the determination of urine KIM-1. KIM-1 = kidney injury molecule-1; SDMA = symmetric dimethylarginine
Discussion
This study generated an RI for urine KIM-1 in healthy cats. To ensure that participants represented a healthy population with normal renal function, specific inclusion and exclusion criteria were applied. This resulted in the exclusion of eight cats with increased SDMA as the only abnormality. It was unexpected to exclude 12% of otherwise healthy cats based on a high SDMA concentration. Pre-renal subclinical dehydration, early kidney disease undetectable with other tests or a slightly low RI may have yielded these results, since neither body condition nor dietary protein is thought to affect SDMA concentration. 57 Of these eight cats, six had USG >1.050, including one with USG of 1.072, which could suggest dehydration; however, 40/50 study cats had USG >1.050 and SDMA within the RI. According to IDEXX guidelines, cats with an SDMA concentration in the range of 15–17 µg/dl as the only abnormality and without polyuria, polydipsia, an-orexia, weight loss, hypertension, increased BUN, SCr or phosphorus, inappropriate USG, active urine sediment, proteinuria, weight loss or abnormal renal palpation and imaging should have a re-evaluation of SDMA, BUN, SCr, phosphorus and urinalysis after 2–4 weeks. 58 This was not within the scope of this study; therefore, the development of subsequent overt kidney disease could not be entirely ruled out.
Three additional cats were excluded due to USG <1.035 as the sole abnormality, which may have been a normal finding in relation to water intake, insensible water losses or renal water excretion, or due to loss of concentrating ability associated with early renal disease. Two cats were excluded due to the detection of renal infarcts on ultrasound. These cats had SDMA, SCr and USG within the RIs; therefore, the infarcts may have been incidental without a detectable impact on kidney function. A single cat was excluded due to positive FeLV and FIV test results without concurrent hematologic abnormalities or clinical signs. Another cat was excluded because its SDMA and SCr values were above the RI and USG was <1.035, supportive of renal dysfunction. SDMA was >14 µg/dl and TT4 was below the RI in another cat, resulting in exclusion, even though the TT4 result was suspected to be spurious considering the lack of clinical signs and rarity of hypothyroidism in cats. Other excluded individuals had TT4 >55 nmol/l, consistent with hyperthyroidism, and either slightly increased ALT or ALP, both considered to be likely clinically inconsequential variations. 52
Urine KIM-1 was detectable in the urine of all healthy cats, consistent with results in humans and likely due to normal turnover of proximal tubular cells.12,32,35 The results of the present study are not directly comparable to prior results due to optimization of the LFA from the initial prototype. 32 USG was found to be a significant predictor of urine KIM-1, which is expected since high urine solute concentrations also increase KIM-1. In a previous study, a marked decrease in USG was associated with a slight decrease in KIM-1, which may reflect a dilutional effect or altered shedding of KIM-1 from proximal tubular cells. 35 In humans, urinary biomarkers are often indexed to urinary creatinine concentration (uCr) or urine osmolality (uOsm) to account for the variation in urine concentration since oliguria can cause an increase, and polyuria a decrease, in the absolute concentration of a biomarker.59–61 This practice is controversial since uCr may vary between individuals and may be affected by non-renal factors, such as muscle mass, sex and diet, 62 and uCr fluctuates since the glomerular filtration rate changes throughout the course of AKI. 59 Indexing to uOsm has been proposed as an alternative in humans in the light of these limitations; however, uOsm also changes throughout the course of AKI, reflecting variable urine electrolyte, glucose and other solute concentrations. 63 In a study of dogs with and without leptospirosis, dogs without kidney disease (n = 5) had lower urine KIM-1 than dogs with leptospirosis (n = 25), but a relationship between urine KIM-1 and USG in the healthy dogs was not noted. 64 The assay used had not been validated for canine samples. It is possible that the positive association identified in this study reflects the use of an assay validated for cats, assessment of a greater number of samples and the wider range of USG in healthy cats; however, more specific investigations of a range of USG values in relation to urine KIM-1 and uCr may be insightful.
SCr concentration was not a significant predictor of urine KIM-1. In humans with active tubular injury, the amount of KIM-1 detected by immunohistochemistry (IHC) in renal biopsies correlated with SCr. 65 While similar studies in naturally occurring AKI have not been performed in cats, experimentally induced AKI increased renal tubular KIM-1 detected by IHC as well as SCr. 12 Likely for similar reasons, SDMA by itself was not predictive of urine KIM-1, but in conjunction with USG there was weak prediction of urine KIM-1. The clinical utility of the latter association remains to be determined.
Neither age, sex, BCS, blood pressure, phosphorus, TT4 nor mid-sagittal kidney length was a significant predictor of urine KIM-1. Similar to the findings in this study, Bland et al 35 also reported low amounts of urine KIM-1 in samples from healthy young and older cats. In contrast, in humans, a linear relationship between urine KIM-1 and age, and a quadratic relationship if urine KIM-1 was normalized for uCr, was reported.66,67 The lack of correlation between urine KIM-1 and cat age in the present study may be due to the inclusion of relatively few older cats, limited sample size or different physiological properties of KIM-1 in cats relative to humans. 35
A significant relationship between sex and KIM-1 has not been reported in humans and was also not identified in this study.66,68,69 Most cats were neutered, hence the potential influence of sex hormones was largely ablated. A positive correlation between KIM-1 and systolic blood pressure has been reported in humans but was not noted in this study, which may be because only normotensive cats were included. 70
The absence of a significant correlation of urine KIM-1 with TT4, kidney length, phosphorus or BCS was expected due to the strict inclusion criteria. However, the effect of chronic hyperthyroidism on renal tubular health is of great interest, and assessing urine KIM-1 in relation to the severity and duration of hyperthyroidism should be a focus of future research.
The present study has some limitations. First, according to the American Society for Veterinary Clinical Pathology guidelines, a minimum sample size of 120 is recommended to determine RIs using non-parametric methods with 90% confidence. 71 Hence, a sample size of 50 cats may not reflect the full range of potential values observed in healthy cats. However, the recruitment of additional cats was not feasible due to the specific inclusion criteria, time and funding constraints. A gold standard to assess adequate glomerular filtration, such as inulin clearance, was not included. Inulin clearance is not readily performed and was considered impractical to include in this baseline study. While cats aged 1–15 years were eligible for the study, 32/50 (64%) cats were aged <6 years. This may impact the generalizability of the RI to geriatric individuals and tempers the conclusion that urine KIM-1 does not vary with age in cats. Urine protein:creatinine (UPC) ratios were not determined in this study, which is a limitation since proteinuria may be associated with AKI. If proteinuria had been used as an exclusion criterion, several cats would have been excluded. In addition, since UPC ratios were not determined, the effect of proteinuria on KIM-1 could not be evaluated. However, proteinuria has been identified in 25–50% of cats otherwise deemed healthy and thus, if used as an exclusion criterion, may have resulted in the elimination of healthy cats. 72 Furthermore, given the high variability in UPC ratios, it is recommended to assess three or more urine samples, obtained at least 2 weeks apart, to confirm renal proteinuria, which was not within the scope of this study. 73 There was a lack of standardized fasting for participants, which might have affected USG. 15 Urine KIM-1 was not normalized for USG or urine creatinine concentration (uCC), which is of debated value in humans and undetermined for cats. However, in healthy, normally hydrated individuals, urine creatinine excretion is stable, and such normalization may therefore not improve the interpretation of a urine biomarker.66,68 Lastly, urine was collected at differing times throughout the day. In humans, urine KIM-1 may vary diurnally, and standardized timing of urine collection should be considered in future studies. 68
Conclusions
Findings from this study suggest an RI for urine KIM-1 in healthy cats. Further investigation is warranted to define more closely the effect of age, acute and chronic kidney disease, and comorbidities, such as hyperthyroidism, on urine KIM-1 in cats.
Footnotes
Acknowledgements
The authors thank the owners of the cats for participating in this study.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Funding for this study was provided by the Ontario Veterinary College Pet Trust Foundation.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognised high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animals described in this work (experimental or non-experimental animals, including cadavers) for the procedure(s) undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
