Abstract
Objectives
Feline nasopharyngeal stenosis (NPS) is an uncommon disease in cats. The aim of this study was to compare the outcome with endoscopic and surgical treatments.
Methods
In this retrospective study, medical records were searched for cases of NPS and cases were divided into three groups: endoscopic treatment (balloon dilation); surgical treatment; or no treatment. Data are presented as median and range. Success rates and length of post-procedure corticosteroid treatment were compared between the two treated groups.
Results
A total of 21 cases were included: 10 males and 11 females (median age 6.7 years, range 0.5–14.4). Clinical signs at the time of initial presentation included permanent stertor (19/21), increased respiratory efforts (9/21), nasal discharge (9/21) and cough/reverse sneezing (6/21) with a duration in the range of 3–60 months. Diagnosis was based on endoscopy (21 cases) and CT (19 cases). In total, 12 cases were treated with endoscopic balloon dilation; 11 cases demonstrated a complete resolution of clinical signs (two cats had two dilations) and one case showed incomplete improvement after one balloon dilation but was successfully treated surgically. Eight cats were treated surgically (including one cat that was originally treated with balloon dilation), including seven cases with complete resolution of clinical signs and one case with a rostral NPS that failed to respond. Two cats were not treated and are still alive more than 3 years after diagnosis. The success rate was not different between the endoscopic (11/12) and surgically treated cases (7/8) (P = 0.85). The outcome was considered excellent in most of the cases as 18/23 procedures were successful, at least until the medium term, and 15/18 treated cats had no clinical signs of minimal stertor at follow-up.
Conclusions and relevance
This preliminary study suggests that minimally invasive endoscopic treatment of NPS has a high success rate and should be attempted in all cases with perforate stenotic membranes. When not achievable, extended palatoplasty can be recommended as a good alternative.
Introduction
Feline nasopharyngeal stenosis (NPS) is an uncommon disease.1–5 In most cases, it is secondary to chronic nasopharyngeal inflammatory disease; however, traumatic or congenital forms are reported. Underlying chronic inflammatory diseases include infection by feline herpesvirus or feline calicivirus and gastroesophageal reflux. 6
Common clinical signs include nasopharyngeal stertor, dyspnoea, sneezing and nasal discharge. 6 Diagnosis is mostly made using endoscopy of the nasopharynx. CT also allows visualisation of the stenosis, but if the CT slices are too thick or if mucopurulent material is present, the stenosis may be missed. Skull radiographs might also suggest NPS.7,8 Several treatment techniques have been described with various success and complication rates: Kelly forceps dilation or endoscopy-guided balloon dilation (BD) with or without temporary or permanent stent placement, and surgical excision of the stenotic membrane through longitudinal palatal incision with or without mucosal flap or surgical extended palatoplasty.6,7,9–11 Mitomycin or triamcinolone injections are sometimes added to these procedures to prevent reformation of the fibrous tissue. 10 In the available literature, the BD technique is often associated with a high rate of stenosis relapse; 6 however, Glaus et al 12 have reported a high success rate on a limited number of cases. The risk of relapse and the possible need of multiple procedures after BD or surgical treatment sometimes lead clinicians to rapidly choose permanent stenting. However, permanent stenting is technically demanding and associated with a relatively high rate of complications, including poor tolerance, tissue in-growth, chronic infections, stent fracture and oronasal fistula. 10 It was our impression that BD was associated with a lower complication rate than commonly reported, and that this less invasive technique should be attempted whenever possible. However, to the best of our knowledge, a comparison between endoscopic and surgical treatment has not yet been reported; therefore, the aim of this retrospective study was to compare endoscopic and surgical treatment success rates in feline NPS.
Materials and methods
Medical records were searched for cases of NPS that were divided into three groups: endoscopy-guided BD (B-group); surgical treatment (S-group); or untreated (U-group). Consent from every owner was obtained before inclusion of their cats in this retrospective study. The severity of clinical signs was retrospectively estimated using a semi-quantitative scale: no clinical sign associated with NPS (grade 0); intermittent stertor (grade 1); permanent stertor (grade 2); permanent stertor with sneezing and/or nasal discharge (grade 3); and dyspnoea (grade 4). Endoscopic or surgical treatment was performed based on the individual clinician’s decision, essentially depending on personal preference; however, all the imperforated stenoses were treated surgically. A similar BD procedure was used in all cases. Briefly, a 10–12 mm diameter and 55 mm length standard dilation balloon (VYTIL) was inserted under general anaesthesia through the ventral aspect of the nostril, pushed in the oesophagus, pulled back across the NPS and progressively filled with water from 3 to 4.5 to 6 atm, each pressure value being maintained for 5 mins. All surgeries were performed by the same diplomate of the European College of Veterinary Surgeons. The extended palatoplasty procedure has been described elsewhere. 11 Briefly, NPS and associated caudal soft palate are resected en bloc through a transoral approach with monopolar electrocautery. Post-procedure analgesia consisted of mostly methadone immediately postoperatively and then buprenorphine as needed; however, this was not reported in all files and therefore cannot be detailed in the paper. Success was defined as an improvement in clinical signs leading to either an absence of clinical signs or only mild stertor (grade 1). Immediate, medium-term and long-term success were assessed a few days, a few weeks to a few months, and 6 months or more after the procedure, respectively. Data are presented as median and range. Comparison between epidemiological data, clinical data and length of medical treatment between the B- and S-groups was performed using a Mann–Whitney U-test. A comparison of the success rate between the B- and S-groups was assessed using a χ² test. A P value <0.05 was considered statistically significant.
Results
In total, 21 cases were included (10 male cats, 11 female cats; median age 6.7 years, range 0.5–14.4; median weight 3.9 kg, range 2.6–7.4). Clinical signs included permanent stertor (19/21), increased respiratory efforts (9/21), nasal discharge (9/21), cough or reverse sneezing (6/21) and sneezing (5/21) with a duration in the range of 3–60 months. Gastrointestinal clinical signs were reported in some cases (dysphagia in 4/21 cats, vomiting or regurgitation in 2/21 cats). Two cats were presented with ear scratching, suggesting concurrent otitis. A total of 11 cats experienced their first clinical signs as adults, seven as kittens. Three cats were adopted as adults; therefore, the age of disease onset was unknown. Most of the cats were outdoor cats.
Diagnosis was based on endoscopy (21 cases) and CT (19 cases). Among the 19 cats that underwent CT examination, the NPS was located in the nasopharyngeal rostral third in one cat, in the nasopharyngeal middle third in two cats, in the nasopharyngeal caudal third in 14 cats and at the junction between the nasopharyngeal middle and caudal thirds in two cats. CT allowed a correct to clear visualisation of the NPS in 17/19 cases. Three cats had CT images compatible with concurrent chronic rhinitis and seven with otitis media. At initial presentation, most of the cats had previously received antibiotics or anti-inflammatory drugs, with variable and temporary improvement but none of them had any specific treatment of the NPS.
Of the cats, 12 were treated with BD (B-group), while eight were treated surgically (S-group), including one cat from B-group that was also surgically treated after a single unsuccessful BD. Age, weight and duration of clinical signs were not statistically different between the two treated groups (see the table in the supplementary material); the median preintervention clinical grade was 3 in both the B- and S-groups.
A total of 12 cases were treated with BD, with 11 successes and one failure that was treated surgically because of a perceived insufficient improvement (grade 4 to grade 2) a few days after the first BD procedure. Myringotomy was also planned for this cat owing to concurrent otitis media, and it might have hastened the decision to perform a palatoplasty under the same anaesthesia. This latter case was included in the two treated groups. Two cats had two BD procedures because of NPS relapse associated with a recurrence of clinical signs 5 and 10 months after the initial procedure, respectively. One of these cats had a caudal NPS and the other cat had an NPS located in the caudal part of the middle third of nasopharynx.
Eight cats were treated surgically, including one case with rostral NPS that was treated by transpalatal excision followed by temporary silicone stenting, and seven cases treated with extended palatoplasty. Among the cases treated with extended palatoplasty, all NPS were located in the caudal nasopharynx and three were imperforated. Treatment of the cat with the rostral NPS failed despite one additional surgery a few months later.
Most of the cats received a short course of antibiotics (mainly amoxicillin/clavulanic acid) and all received decreasing doses of corticosteroids after BD or surgery. The duration of corticosteroid treatment was not statistically different between the B-group and the S-group (median treatment length 16.5 days and 10 days, respectively; P = 0.23). Two cats received long-acting dexamethasone phenylproprionate and disodium phosphate injections (Dexafort; MSD) 0.15 mg/kg SC because of an expected poor treatment observance at home. When prescribed, median initial prednisolone dosage was not statistically different between the B-group and the S-group (median initial dosage 1 mg/kg/day and 0.75 mg/kg/day, respectively; P = 0.83).
Two cats were included in the U-group. Despite clinical grades of 3 and 4 at their initial presentation, they are still alive more than 3 years after diagnosis with stable disease. In one case, BD was attempted but failed as a result of the impossibility to push the balloon through the NPS and surgery was declined. In the other case, surgical treatment was advised after investigation of the concurrent chronic rhinitis, but owners refused because of financial constraints.
No complications directly related to the procedures were reported. The duration of hospitalisation was difficult to compare between the two groups because of two outliers in the S-group (one cat presented at the emergency department for life-threatening dyspnoea and regurgitations associated with concurrent megaoesophagus, and another cat with a rostral NPS), with each requiring more than 48 h of hospitalisation. Data were also missing for one case in the B-group. However, after excluding these three cases, the procedure was more frequently performed on an outpatient basis in the B-group (9/11) than in the S-group (1/5). Time to improvement after the procedure could not be compared between the two groups because of missing data; however, an improvement in clinical signs was seen in nearly all cats immediately after the procedure.
The immediate success rate was not different between the B-group (11/12) and the S-group (7/8) (P = 0.85). Complete improvement was reported in 7/12 cats in the B-group and 3/8 cats in the S-group; partial improvement was reported in 3/12 cats in the B-group and 5/8 cats in the S-group. The median post-intervention clinical grade was 0 in the B-group and 1 in the S-group.
Follow-up was available for 20/21 cases (11/12 cases from the B-group, 8/8 cases from the S-group and 2/2 cases from the U-group). Follow-up data were retrieved through a follow-up telephone call performed at various times after diagnosis. The median time from initial diagnosis to follow-up telephone call was 347 days (range 10–10,339). The long-term follow-up for BD cases was considered excellent, with 8/9 cats that had no clinical signs to minimal stertor. For the S-group cases, the long-term follow-up was considered excellent for 3/5 cats, while one cat was a grade 3 with a concurrent rhinitis and the last cat was the one with the rostral stenosis that died from an anaesthetic accident during a third surgical attempt at another institution. Data about follow-up and clinical grade evolution at different time points in treated cats are presented in Figures 1 and 2. For the two cats with untreated NPS, follow-up data were obtained 624 days and 1346 days after the initial diagnosis, respectively. Their owners reported courses of inhalations of essential oils and occasional antibiotic treatments; quality of life was considered acceptable by the owners despite persistent clinical signs.
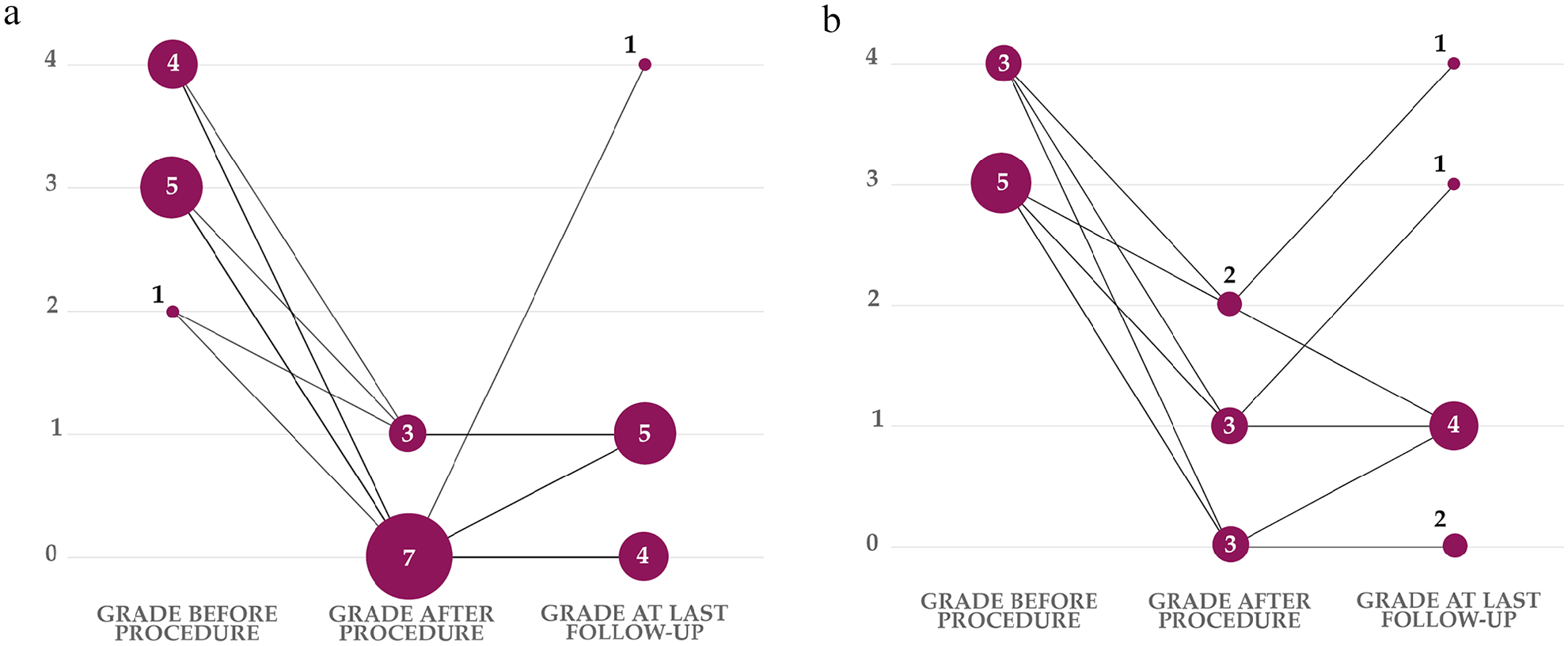
Clinical grade evolution before and after the initial procedure and at the last follow-up for cases with available follow-up data. The vertical axis indicates the clinical grade, while the numbers associated with the dots indicate the number of animals for each grade at each time point. (a) B-group; the cat that underwent surgical treatment a few days after the initial balloon dilation attempt appears only in panel b; the cat with a clinical grade of 4 at the last follow-up died at that time from suspected nasopharyngeal lymphoproliferative disease. (b) S-group: the cat with a clinical grade of 3 at last follow-up had concurrent chronic rhinitis
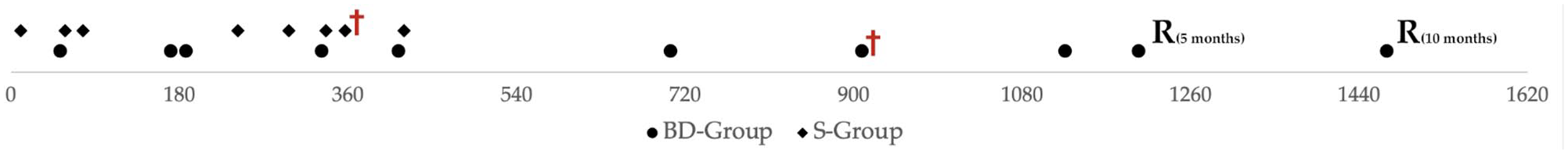
Time from initial diagnosis to last follow-up and follow-up details for treated patients with available follow-up data. The horizontal axis represents time in days. Each symbol on the timeline represents an individual patient at its last follow-up after initial treatment of nasopharyngeal stenosis; cats with relapse after balloon dilation (BD) are shown by the letter R with time to relapse indicated in brackets and time from initial BD to recurrence is indicated; for cats identified with the cross (
Only two cases had a follow-up endoscopic evaluation performed. The first cat was presented for investigation of a suspected chronic bronchopneumopathy. This cat had previously undergone two BDs. At that time, more than 2 years after the last BD, the owner reported slight intermittent stertor. An endoscopic evaluation showed a partial recurrence of NPS (Figure 3), but it did not seem to impact the cat’s quality of life; therefore, BD was not recommended. In another cat that was initially suspected of NPS recurrence, endoscopic evaluation led to a strong suspicion of nasopharyngeal lymphoma; however, it was not confirmed with a biopsy. During the follow-up period, two cats died from diseases unrelated to NPS, and the cat with rostral NPS died during a third surgical treatment attempt.

Endoscopic evaluation follow-up 2 years after last treatment showed partial recurrence of nasopharyngeal stenosis in 1/2 cats that already underwent two balloon dilation (BD) procedures. A new BD was not performed, because of the absence of significant clinical signs
Discussion
Several techniques have been described for NPS management, with various success and complication rates.6,7,9–11,13 Unfortunately, owing to a paucity of cases, a direct comparison of different techniques has not been previously reported.
Contrary to what has been reported in other studies,10,14 the NPS recurrence rate with BD was low in our study, despite a prolonged follow-up, and could be correctly managed by a new endoscopic BD. Our results are similar to those obtained by Glaus et al. 12 A certain amount of restenosis is likely to be frequent with BD even in the absence of clinical signs, as it was observed during the reported follow-up endoscopy.12,15 In our study, partial relapse (restenosis) has been suspected in a few cases because of the recurrence of a minimal stertor a few months after initial BD (Figure 1). Partial restenosis has only been endoscopically confirmed in one case in the B-group undergoing respiratory endoscopy for other reasons. Arguably, we believe that success should be defined as relief of clinical signs and long-term absence of clinically significant recurrence. Therefore, possible partial restenosis might have been missed because it might not significantly affect the clinical outcome.
It is difficult to compare the success rates reported in this study with other studies. In the largest study reported to date, 22 cats underwent BD, with a success rate of only 50%. 10 However, most of them were referred after one or several NPS dilation attempts, and three cats had imperforate NPS, which seems to be associated with a lower success rate for BD. In this study, none of the cats with imperforate NPS were successfully treated with BD. In our study, surgery was advised for all cats with imperforate stenosis, and no imperforate stenoses were included in the B-group, which also could have contributed to our success rate and may represent a selection bias.
Moreover, it is likely that concurrent diseases, such as chronic rhinitis, might also play a role in the risk for restenosis. In the case series by Pollack et al, 14 of the four cats that underwent CT evaluation, three had concurrent chronic rhinitis, which might have lowered the success rate. In the 19 cats that underwent CT scan evaluations in our study, only four had concurrent evidence of chronic rhinitis: three were in the S-group and one was in the U-group. Biopsies were not performed, however, except for the cat in the U-group. Of the S-group cats with concurrent rhinitis, one was the cat with the rostral stenosis that failed to respond to treatment, and another one had still marked nasal discharge and stertor despite an initial improvement after surgery (Figure 1b), but no additional examinations were performed.
Concurrent otitis media was often identified as previously described; 13 however, only 2/7 cats with fluid accumulation in the tympanic bullae showed associated clinical signs, questioning the clinical significance of this finding. In NPS, the abnormal membrane may induce Eustachian tube occlusion and mucous accumulation in the bullae and potential secondary otitis media. As for chronic rhinitis, it can be hypothesised that untreated otitis media might be a risk factor for recurrence because of possible sustained local inflammation. In our study, only one symptomatic case was treated with myringotomy. In the other symptomatic case, myringotomy was recommended after initial BD but was not performed because of resolution of middle ear disease-related clinical signs; this cat was diagnosed with recurrence of NPS 10 months later.
Localisation of NPS might also have influenced the success rate of BD. In our study, the cat that changed groups because of unsuccessful initial BD had a caudal NPS, and the two cats with relapse after BD had a caudal NPS. BD was successful in 8/9 cats with a caudal NPS and in the two cats with middle third nasopharynx NPS. In the study by Burdick et al, 10 71% of NPS cases were reported to be in the caudal third of the nasopharynx, 16% in the rostral third and 13% in the middle third. In that study, BD was not successful in any of the patients with a lesion in the rostral or middle third of the nasopharynx. The impact of NPS localisation on BD treatment success remains uncertain at that stage. Perhaps the etiopathogenesis of the disease, which is probably associated with the NPS distribution, is a better predictor of procedure success. Unfortunately, it cannot be determined in most cases, and the paucity of the cases limits the statistical analysis.
Because it was not clearly mentioned in all medical files, it is difficult to compare the BD technique used for each case. However, the same balloon size was used for all the cats treated with BD. In the first report of successful BD in a cat with NPS, a 15 mm valvuloplasty dedicated balloon was used. 15 In the study by Burdick et al, 10 the median balloon size was different in successful and non-successful BDs (10 mm vs 8.5 mm, respectively), but this difference was not statistically significant. Further prospective studies are needed to determine the ideal balloon size for BD of NPS. Concurrent fluoroscopy has been recommended during the endoscopy-guided BD procedure to evaluate correct fitting of the balloon in the nasopharyngeal cavity. 10 Fluoroscopy was not used in our study.
Another factor that may have impacted the success rate is post-procedure treatment. In our study, medical treatment after the procedure was not standardised. There is no clear recommendation concerning anti-inflammatory drugs after NPS treatment. Most authors5,6,11–14 used prednisolone 0.5–2 mg/kg PO q24h with decreasing doses; the benefit of higher dosages or prolonged therapy in preventing NPS recurrence is unknown, but this may exacerbate any viral infection. Triamcinolone or mitomycin injections have also been reported; however, there is no clear evidence supporting their systematic use. 10 These injections were not performed in our study.
The main procedure-related complication for BD was failure due to the impossibility of correctly placing the balloon across the NPS. In one case, because of the inability to push the balloon across the nostril, a surgical suture was placed across the NPS through the nostril in a retrograde fashion with a bronchoscope forceps and was used to pull the balloon across the nostril, to allow proper placement across the NPS. The initial use of a conventional dedicated guidewire might have helped prevent this difficulty, but it was not available. Surgical treatment was finally advised for two cases in which the BD procedure could not be completed: in one case because of non-visualisation of an extremely caudal NPS and in the other case because of the inability to place the balloon across the stenosis.
In our study, extended palatoplasty was especially recommended in cases where NPS was imperforated or very caudal, or in cases of financial constraints precluding several BDs. Food-material nasal foreign bodies, chronic rhinitis or frequent reverse sneezing might be anticipated with a large extended palatoplasty, although these have never been reported in either our study or that by Sériot et al. 11
Two cats left untreated were included in this study. Interestingly, the owners reported stable disease years after the initial diagnosis, with an acceptable quality of life. From a medical point of view, such assessment is questionable because NPS seems to cause major discomfort. In fact, many owners in this study did not realise the impact NPS had on their cat until it was treated. Of the treated cases, except for respiratory relief, two cats were reported to meow more, one cat purred for the first time since adoption and two owners reported their cat was more active after intervention. Food intake dramatically increased in other cats. In one cat that also had frequent regurgitations at initial presentation, radiographic examination identified a megaoesophagus. Although persistent, the frequency of regurgitations was reduced after intervention. Megaoesophagus resolution after correction of a marked obstructive upper respiratory disease has been previously reported in cats, but was not assessed in this case.16,17
The main limitations of our study are the limited size of our population of cats, its retrospective nature and the associated missing data or lack of standardisation. Some data are also missing for the long-term follow-up because some cats were lost to follow-up. Another limitation is the lack of randomisation. The allocation of the cases between the B-group and S-group is biased because all imperforated stenoses underwent surgery; however, as detailed in the table in the supplementary material, the distribution of the location of the stenosis is balanced between the two groups. The absence of a PCR test to assess for feline upper respiratory viruses is also a limitation because it could be possibly linked to a relapse; however, the sensitivity of such testing has not been assessed in an NPS setting and false-negative testing may occur as NPS is not an active disease but rather a post-inflammatory sequela.
Conclusions
This preliminary study suggests that minimally invasive endoscopic treatment of NPS has a high success rate and should be attempted in all cases considering its very low complication rate. However, surgical palatoplasty offers a safe and successful alternative for caudal or imperforate NPS; therefore, stents should probably be reserved for salvage procedures in the face of unsuccessful previous treatment attempts.
Supplemental Material
sj-docx-1-jfm-10.1177_1098612X241281902 – Supplemental material for Nasopharyngeal stenosis in cats: a retrospective study of 21 cases comparing endoscopic and surgical treatment (2018–2022)
Table 1. Epidemiological data for the two groups.
Footnotes
Supplementary material
The following file is available as supplementary material:
Table 1. Epidemiological data for the two groups.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Author note
These results were presented in part as an oral research abstract at the 2022 ECVIM-CA congress.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognized high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers, tissues and samples) for all procedure(s) undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
