Abstract
Objectives
CT is frequently utilised in the assessment of upper respiratory tract disorders. The aim of the present study was to further describe the CT features in cats with confirmed nasopharyngeal stenosis (NPS).
Methods
CT images of the heads of cats with NPS confirmed via retroflex nasopharyngoscopy between 2011 and 2023 were retrospectively reviewed to describe the imaging features of NPS and other concurrent findings.
Results
In total, 12 cats were included. All but one cat had pre- and post-contrast images available. All CT studies demonstrated focal, abrupt narrowing of the nasopharyngeal lumen by a variably thickened, homogeneous, soft tissue attenuating band. One cat appeared to have two separate NPS, giving a total of 13 NPS. All NPS were located in the caudal third of the nasopharynx, which resulted in a marked reduction in the luminal cross-sectional area. The narrowing of the nasopharyngeal lumen was either concentric (11/13) or lateral-to-lateral (2/13). Mild homogeneous contrast enhancement of the soft tissue band was observed in 8/11 cats (73%). The soft palate was focally dorsally deviated at the NPS site in 8/12 cats (67%) and was best seen in the sagittal plane. Other concurrent findings included non-contrast-enhancing soft tissue attenuating material within the nasal cavity (7/12) and tympanic bullae (4/12), and mild medial retropharyngeal lymphadenomegaly (2/12).
Conclusions and relevance
The most frequently observed CT features in cats with NPS are marked, focal, abrupt narrowing of the nasopharyngeal lumen by a mildly contrast-enhancing soft tissue band in the caudal third of the nasopharynx and focal deviation of the soft palate dorsally at the NPS site. Identifying any of these CT features in the presence of compatible clinical signs should prompt further evaluation with retroflex nasopharyngoscopy to confirm the diagnosis of NPS and to direct appropriate treatment.
Keywords
Introduction
Nasopharyngeal stenosis (NPS) is the narrowing of the nasopharyngeal lumen by a soft tissue membrane, resulting in partial or complete obstruction of the nasopharynx. 1 It may be congenital or acquired, caused by scar tissue formation. The aetiology of acquired NPS in cats is not entirely understood, but chronic rhinitis, upper respiratory tract infections and aspiration rhinitis from gastric reflux during anaesthesia have been implicated.1 –4 NPS is an uncommon condition in cats, representing approximately 4% or 6% of cats presenting with clinical signs of nasopharyngeal disease 5 or chronic nasal disease, 6 respectively. Similar to other upper respiratory tract disorders, NPS leads to signs of upper airway obstruction with stertorous breathing as a main presenting clinical sign. Other reported clinical signs include open-mouth breathing, dysphagia, gagging, sneezing and chronic nasal discharge.1 –3,6 –10
While retroflex nasopharyngoscopy allows visual confirmation and diagnosis of NPS,2,6,9 CT provides detailed information on the location and the extent of the stenosis and assessment for other concurrent abnormalities such as chronic rhinitis, otitis media or choanal atresia.3 –5,11 Additionally, the measurements obtained with CT can assist with treatment planning in selecting the appropriate balloon size and length for endoscopic-guided balloon dilation or the appropriate stent size.3,11 However, more importantly, CT allows assessment of the nasal cavity for other potential causes of upper respiratory tract signs. Therefore, the combination of retroflex nasopharyngoscopy and CT is useful for evaluating NPS.3,4,11,12
NPS presents similarly to other nasopharyngeal disorders, and CT is used with increasing frequency in the investigation of upper respiratory tract disorders in cats. A single recent case series describes the clinical and CT features of acquired and congenital NPS in 23 cats. 13 The objective of the current study was to further describe the CT features of confirmed NPS in cats.
Materials and methods
Study design and case selection
This was a retrospective, single-centre, case series, descriptive study approved by the RCVS Ethics Review Panel. The medical records were searched for cats with a final diagnosis of NPS between January 2011 and October 2023. Cases were included if a CT examination of the head was performed and NPS was confirmed with retroflex nasopharyngoscopy. The investigations were performed by a board-certified internal medicine specialist or an internal medicine resident under their supervision.
Age at presentation, sex, breed, body weight, presenting clinical signs, duration of the clinical signs, history of upper respiratory tract disease (if available), retroflex nasopharyngoscopy findings, endoscopic-guided balloon dilation procedure notes (if performed) and outcome (if available) were recorded. The duration of the clinical signs was considered chronic if the clinical signs were present for ⩾3 weeks. 6
Image evaluation
The CT studies were reviewed by a board-certified veterinary radiologist and a second-year veterinary radiology resident using DICOM-viewing software (OsiriX, version 13.0.1; Pixmeo). Both reviewers were aware that all cases had confirmed NPS. The images of all CT studies were reviewed using a soft tissue window for the medium-frequency spatial reconstruction algorithm, and bone and lung windows for the high-frequency spatial reconstruction algorithm. Multiplanar reconstructions in the transverse, sagittal and dorsal planes were used to evaluate all studies. Standardised locations for specific measurements of dimensions were established prior to image evaluation. Measurements of the dimensions of the nasopharyngeal lumen and evaluation of the shape of the NPS were recorded from images reconstructed in the high-frequency algorithm and displayed in a lung window for better contrast at the soft tissue–gas interface and better discrimination of the edges of the NPS. 14 The mouth position of the cats on the CT studies was recorded as open if the mouth was propped open with portions of a plastic syringe case or by the endotracheal (ET) tube connector, or closed if the mouth was in a neutral position with the ET tube in place. 15
The CT studies were evaluated for the presence of nasopharyngeal luminal narrowing with an associated soft tissue attenuating band. The location of the NPS was recorded as rostral (rostral third of the nasopharynx), middle (middle third of the nasopharynx) or caudal (caudal third of the nasopharynx), as previously described (Figure 1). 4 If post-contrast images were available, contrast enhancement of the band was subjectively evaluated as present or not. The presence and shape of a visible lumen within the stenosis were evaluated, and its height and width were measured using digital callipers at its narrowest point on transverse images, perpendicular to the longitudinal axis of the NPS. Measurements were also taken immediately rostral and caudal to the stenosis. Regions of interest were drawn around the lumen at the same locations described above to measure luminal cross-sectional areas (Figure 2). The length of the NPS was measured on sagittal images, as previously described. 3 The degree of NPS was determined by comparing the cross-sectional area at the narrowest point of the stenosis with the cross-sectional area of the nasopharyngeal lumen rostral to the stenosis, and recorded as mild (<25% reduction), moderate (25–50% reduction) or marked (>50% reduction).

Sagittal plane reconstruction non-contrast CT image of the head of a cat with a normal nasopharynx, displayed in a bone window (window level = 500 HU, window width = 2500 HU). This image shows the three different locations within the nasopharynx. Rostral = rostral third of the nasopharynx; middle = middle third of the nasopharynx; caudal = caudal third of the nasopharynx

Sagittal plane reconstruction (a), dorsal plane reconstruction (b) and transverse plane reconstruction (c–e) non-contrast CT images of the head of a cat (case 2) with a nasopharyngeal stenosis (NPS) in the caudal nasopharynx. The images are displayed in a lung window (window level = −500 HU, window width = 1400 HU). For the transverse images, the cat’s right is on the left of the images. The corresponding levels of the transverse slices are marked by the dashed lines on the sagittal plane and dorsal plane reconstructions, indicating the level of the nasopharynx rostral to the NPS (c), the level of the narrowest point of the nasopharynx (d) and the level of the nasopharynx caudal to the NPS (e). A region of interest (ROI) is drawn around the outline of the nasopharyngeal lumen (pink outline) immediately rostral to the NPS (c) and immediately caudal to the NPS (e) to measure cross-sectional areas. The narrowest point of the NPS (d) in this cat is too small for accurate placement of a ROI and the degree of reduction in the nasopharyngeal luminal cross-sectional area in this cat is marked
The nasopharynx was evaluated for the presence of luminal material, which was graded as a mild, moderate or marked amount. The position of the soft palate was evaluated for deviation, and the direction of any deviation was recorded.
The following subjective CT features of the nasal cavity were recorded as described in previous studies:16,17 the presence and laterality of abnormal soft tissue or fluid attenuating material, the degree of material, the location within the nasal cavity (rostral, middle or caudal), associated contrast enhancement, and the presence, laterality and severity of any nasal turbinate lysis.
The following subjective CT features of the tympanic bullae were recorded as described in a previous study: 18 the presence and laterality of soft tissue or fluid attenuating material within the tympanic bullae, contrast enhancement of the bulla contents, bulla thickness, and lysis or proliferation of the tympanic bullae.
The medial retropharyngeal lymph nodes were evaluated for size, attenuation and contrast enhancement pattern as described in previous studies.19,20 The maximal height and width of the lymph nodes were measured on the transverse images. The medial retropharyngeal lymph nodes were considered enlarged if the maximal width was greater than 6.5 mm based on previously reported normal values (mean of 4.7 mm plus 2 SDs of 0.9 mm). 19
Statistical analysis
The clinical data, CT imaging qualitative features and CT imaging quantitative measurements were entered into commercial spreadsheet software (Excel; Microsoft) by one of the authors (KK). The frequency of categorical data was reported as proportions and percentages, and continuous variables were expressed as medians and ranges (minimum to maximum).
Results
Study population and clinical findings
In total, 12 cats met the inclusion criteria. The median age at presentation was 9 years, ranging from 1.3 to 13 years. The median body weight was 3.5 kg, ranging from 2.9 to 6.1 kg. There were nine female cats (five entire and four spayed) and three male neutered cats. The breeds represented included domestic longhair (n = 2), domestic shorthair (n = 2), Birman (n = 2), and one each of Arabian Mau, Balinese, Burmese, European Shorthair, Maine Coon and Tonkinese. Clinical data of all 12 cats are summarised in supplementary material 1.
Presenting complaints included upper respiratory tract noise (11/12; 92%), intermittent open-mouth breathing (8/12; 67%), excessive swallowing or gulping (3/12; 25%), sneezing (2/12; 17%), gagging/retching (2/12; 17%), coughing (1/12; 8%) and weight loss (1/12; 8%). Clinical signs had been present since a kitten in two cats (18%) and progressive or worsening in nine cats (82%). The duration of clinical signs prior to presentation ranged from 2 weeks to 13 years, with a median of 2 years.
On clinical examination, a variable degree of stertorous breathing was observed in 11/12 cats (92%). Other clinical examination findings included mild expiratory wheeze (2/12; 17%) and stridor (1/12; 8%). One cat presented with severe dyspnoea.
An underlying cause of the NPS was not determined in all cats. However, the suspected cause of the NPS was chronic upper respiratory tract infection in one cat (case 7) and aspiration of gastric contents into the nasopharynx secondary to chronic vomiting in two cats (cases 9 and 12).
CT image acquisition parameters
All cats were imaged in sternal recumbency under general anaesthesia and all cats had an ET tube placed. The mouth of 2/12 cats was in a neutral position with the ET tube in place and the mouth of 10/12 cats was propped open with portions of a plastic syringe case (4/10) or by the ET tube connector (6/10). Six cats had CT examinations performed using a dual-slice CT unit (HiSpeed CT/e Dual; GE Hangwei Medical Systems) and six cats were evaluated with a 64-slice multidetector row CT unit (SOMATOM go.All; Siemens Healthcare). The images were reconstructed in the transverse plane using medium and high spatial frequency algorithms. Technical parameters of the individual CT examinations are provided in supplementary material 2.
Eleven of the 12 cats had pre- and post-contrast images acquired, and the remaining cat had only non-contrast images acquired. Iodinated contrast agent (Iohexol, Omnipaque 300 mg I/ml; GE Healthcare) was injected intravenously at 2 ml/kg using either manual injection or a power injector (CT motion; Ulrich Medical). The scan delay was 45–60 s following the start of contrast agent injection, independent of the delivery method.
CT features of nasopharyngeal stenosis
An abrupt, focal narrowing of the nasopharyngeal lumen caused by a variably thickened, homogeneous, soft tissue attenuating band, compatible with NPS, was present in all 12 cats (Figure 2 and Table 1). One cat (case 6) appeared to have two separate focal stenotic areas on CT, giving a total of 13 NPS. All NPS (13/13) were located in the caudal nasopharynx, of which three were located at the caudal margin of the soft palate. Eleven of 13 NPS resulted in concentric narrowing of the nasopharyngeal lumen (Figure 3a), while the remaining two resulted in lateral-to-lateral narrowing and an ‘hourglass-shaped’ lumen (Figure 3b) on transverse images. The lumen of the narrowest point of 10 NPS was too small or not visible for accurate placement of callipers and ROI for measurements of width, height and cross-sectional area (Figure 2). All NPS resulted in a marked reduction in the nasopharyngeal luminal cross-sectional area (Figure 2). Measurements of all NPS are summarised in Table 2. The stenosis showed mild homogeneous contrast enhancement in 8/11 cats.

Transverse non-contrast CT images of two different cats with nasopharyngeal stenosis (NPS) displayed in a lung window (window level = −500 HU, window width = 1400 HU). The cats’ right is to the left of the images. In (a), the NPS results in concentric narrowing of the nasopharyngeal lumen (white arrows) (case 2). In (b), the NPS results in lateral-to-lateral narrowing of the nasopharyngeal lumen (white arrows) (case 11)
CT features of the nasopharyngeal stenosis (NPS) and nasopharynx rostral to the NPS of 12 cats with confirmed NPS; one cat had two NPS, giving a total of 13 NPS

One cat had only non-contrast CT images acquired and therefore one NPS was not included in the total number
Distribution of the length of nasopharyngeal stenosis (NPS), dimensions of NPS at the narrowest point, dimensions of the nasopharynx rostral to NPS and dimensions of the nasopharynx caudal to NPS in 12 cats, measured on CT images
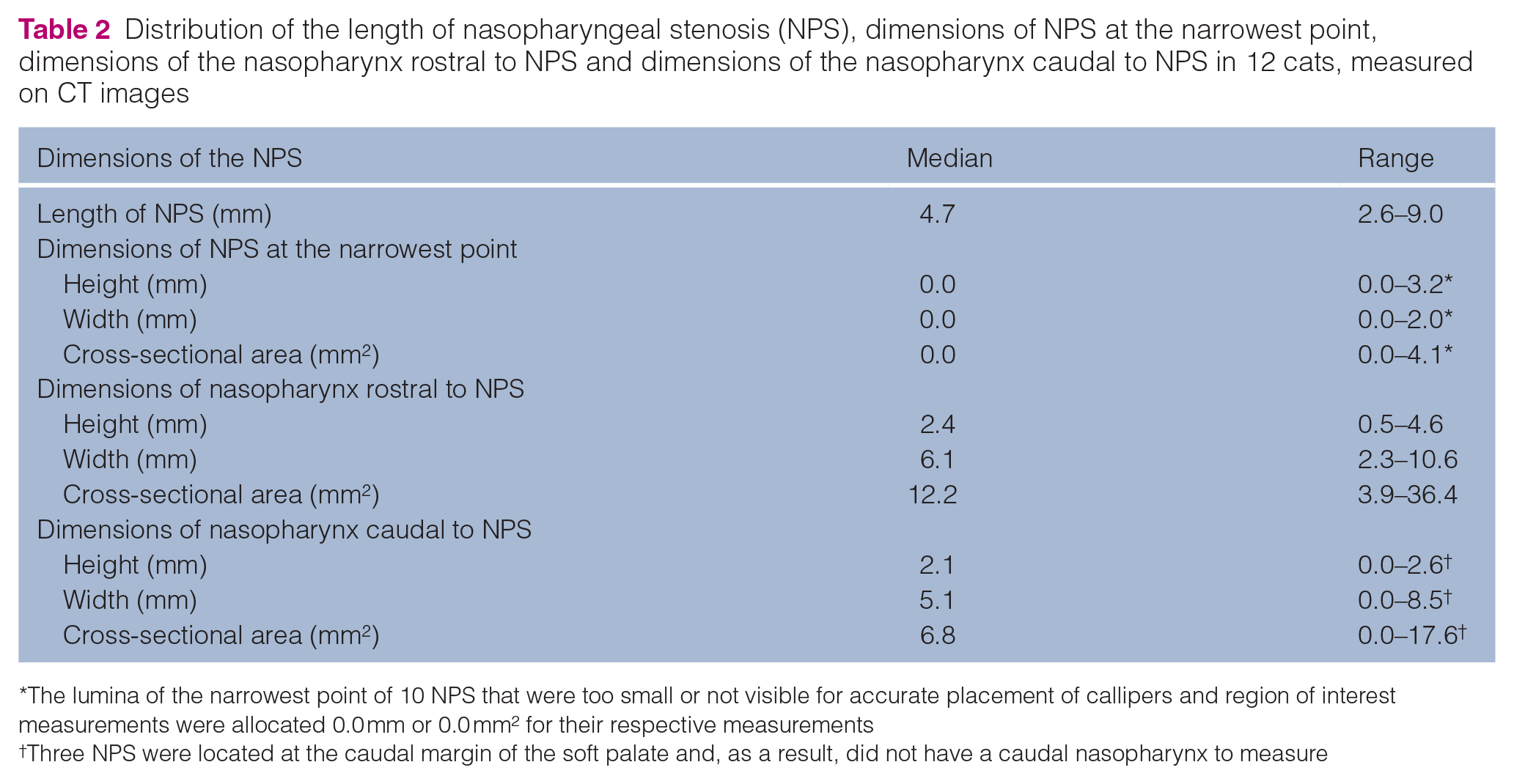
The lumina of the narrowest point of 10 NPS that were too small or not visible for accurate placement of callipers and region of interest measurements were allocated 0.0 mm or 0.0 mm2 for their respective measurements
Three NPS were located at the caudal margin of the soft palate and, as a result, did not have a caudal nasopharynx to measure
Focal dorsal deviation of the soft palate at the site of NPS was observed in 8/12 cats (67%) and was best seen in the sagittal plane (Figure 4a). The ET tube in the remaining four cats was in close contact with the soft palate, which hindered evaluation for focal dorsal deviation of the soft palate at the site of NPS (Figure 4b). Accumulation of non-contrast-enhancing fluid or soft tissue attenuating material was present rostral to only 4/13 NPS (31%) and was classified as either mild (n = 3) or moderate (n = 1). The CT features of NPS are summarised in Table 1, and the CT findings of the individual cases are summarised in supplementary material 3.

Sagittal plane reconstruction non-contrast CT image of the head of two different cats with a nasopharyngeal stenosis (NPS) (*) in the caudal nasopharynx displayed in a lung window (window level = −500 HU, window width = 1400 HU). In (a), there is focal dorsal deviation of the soft palate at the site of the NPS (white broad arrow) (case 2). The soft palate at the site of dorsal deviation is not in close contact with the endotracheal tube (#). In (b), the soft palate is in close contact with the endotracheal tube (#), which hinders evaluation for focal dorsal deviation of the soft palate at the site of the NPS (case 1)
Additional CT findings accompanying nasopharyngeal stenosis
The additional CT findings observed in the nasal cavity, tympanic bullae and medial retropharyngeal lymph nodes of all 12 cats are summarised in Table 3. The CT findings of the individual cases are summarised in supplementary material 4. Accumulation of non-contrast-enhancing fluid or soft tissue attenuating material was present within the nasal cavity of 7/12 cats (58%). This material was predominantly bilateral in distribution (6/7; 86%) and mild in severity (6/7; 86%). The distribution of the material was caudal (4/7), generalised (2/7) or rostral (1/7). All 11 cats that received intravenous contrast medium showed mucosal enhancement. Nasal turbinate destruction was not observed in any of the 12 cats.
Additional CT findings of the nasal cavity, tympanic bullae and medial retropharyngeal lymph nodes of 12 cats with confirmed nasopharyngeal stenosis
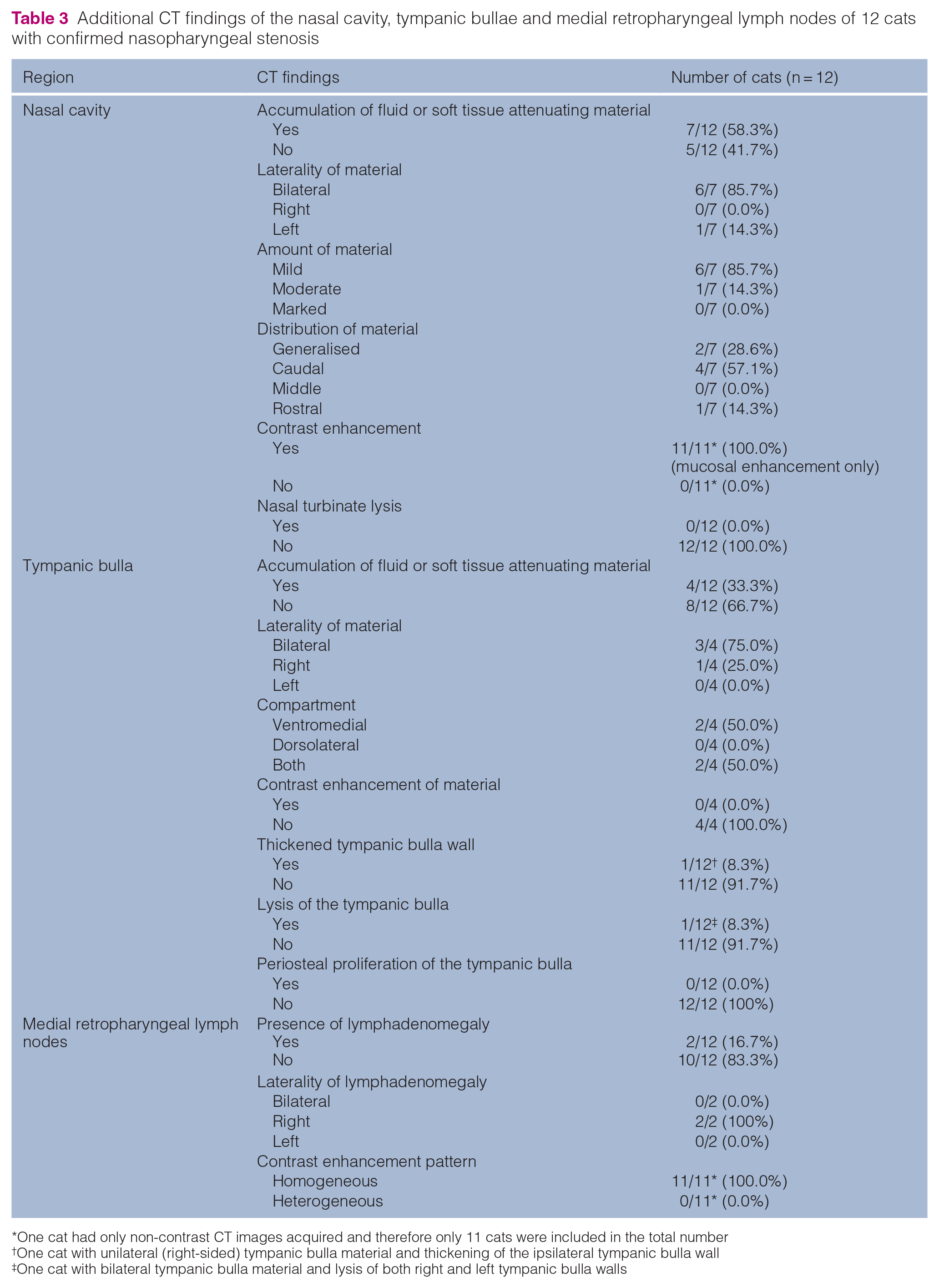
One cat had only non-contrast CT images acquired and therefore only 11 cats were included in the total number
One cat with unilateral (right-sided) tympanic bulla material and thickening of the ipsilateral tympanic bulla wall
One cat with bilateral tympanic bulla material and lysis of both right and left tympanic bulla walls
There was an accumulation of non-contrast-enhancing fluid or soft tissue attenuating material in the tympanic bullae of four cats (4/12; 33%), which was predominantly bilateral (3/4) in distribution; measurements are presented in supplementary material 5. The cat with unilateral tympanic bulla content had smoothly marginated thickening of the ipsilateral ventromedial bulla wall. One cat with bilateral tympanic bulla content had mild lysis of the ventral wall of both tympanic bullae.
Mild lymphadenomegaly was observed in two cats (2/12; 17%) and was unilateral in both cats; measurements for all 12 cats are presented in supplementary material 6. The medial retropharyngeal lymph nodes in all 11 cats that received intravenous contrast agent demonstrated homogeneous contrast enhancement.
Retroflex nasopharyngoscopy findings
The retroflex nasopharyngoscopic findings were available in the medical records of all cats. A narrowed nasopharyngeal lumen was described in 11/12 cats; in case 8, visibility of the nasopharynx was possible only following the passage of a balloon dilation catheter and was considered consistent with NPS. The degree of nasopharyngeal luminal narrowing was reported for only four cats and was described as marked in all cases. Similarly, the location of the NPS was reported for only five cats: in the caudal nasopharynx in four cats and the mid-nasopharynx in one cat. The cat (case 6) with two sites of NPS observed on CT had only one area of stenosis visible endoscopically in the caudal nasopharynx; in this cat, antegrade rigid rhinoscopy was not possible due to the narrow nasal passages.
Treatment and outcome
Eleven of the 12 cats had endoscopic-guided balloon dilation of the NPS performed. In the remaining cat (case 6), a narrow-gauge Foley catheter was used to dilate the stenosis with the Foley balloon because the nasal passages were too small for passage of a balloon dilation catheter. All 12 cats demonstrated improvement of clinical signs following dilation of the NPS and were reported as showing either significant improvement (4/12) or complete resolution (8/12).
Seven of the 12 cats re-presented with upper respiratory tract signs; the timeframe of recurrence of clinical signs ranged from 2 months to 12 months following initial dilation of the NPS (median 7 months). Retroflex nasopharyngoscopy confirmed recurrence of NPS in 5/7 cats and endoscopic-guided balloon dilation of the NPS was performed in all five cats, which resulted in improvement of the clinical signs. One of the cats (case 6) redeveloped upper respiratory tract signs a third time (31 months following the initial NPS dilation), and retroflex nasopharyngoscopy confirmed recurrence of NPS; dilation of the NPS was able to be performed with a balloon dilation catheter on both occasions of recurrence.
Of the 2/7 cats that did not have evidence of recurrence of NPS on retroflex nasopharyngoscopy, one cat (case 1) had a cluster of polypoid mucosal lesions causing occlusion of the nasopharyngeal lumen but the endoscope could pass beyond this region. A CT of the head was subsequently performed, which identified marked caudal nasopharyngeal luminal narrowing at approximately the same location as the previous study without evidence of a mass lesion. Surgical debridement via avulsion under direct visualisation was performed, and repeat retroflex nasopharyngoscopy confirmed marked improvement in nasopharyngeal luminal patency and no residual polypoid tissue. Histopathology of the polypoid tissue identified marked primarily neutrophilic and plasmacytic ulcerative nasopharyngitis with granulation tissue that was suspicious for a response to an infectious process or physiomechanical injury.
In the other cat (case 2) that did not have evidence of recurrence of NPS on retroflex nasopharyngoscopy, CT of the head was subsequently performed, which identified a retropharyngeal mass resulting in compression of the pharynx and larynx. Fine-needle aspirates of the retropharyngeal mass were obtained and cytology was consistent with carcinoma. The cat deteriorated clinically the following day post-procedures and was euthanased.
Discussion
All NPS in the present study were located in the caudal third of the nasopharynx on CT, consistent with previous studies that identified NPS most frequently in the caudal third (22/31 cats; 71%) 4 to caudal half (19/23 cats; 83%) 13 of the nasopharynx in cats. The location of NPS has been shown to have clinical implications in dogs and cats because balloon dilation was more likely to be successful for stenosis located in the caudal third of the nasopharynx. 4 In the present study, the location of the NPS on retroflex nasopharyngoscopy was reported for only five cats; in 4/5 cats, the NPS was described to be in the caudal nasopharynx, consistent with the CT finding. However, in one cat, the NPS was described as located in the mid-nasopharynx on retroflex nasopharyngoscopy. Compared with the tomographic overview that CT provides, it could be challenging to determine the exact location of the NPS endoscopically, especially if the lesion is not located at the caudal margin of the soft palate.
An associated focal dorsal deviation of the soft palate at the site of NPS, best seen in the sagittal plane, was observed in 8/12 cats (67%) and represents a potentially useful feature to help distinguish a NPS from nasopharyngeal luminal fluid or soft tissue. This finding is consistent with reported radiographic findings of cats with NPS, where dorsal deviation or deformation of the soft palate was observed on lateral projections accompanying a thin soft tissue opacity across the nasopharynx.8,21,22 In a recent CT study, partial or complete adhesion between the nasopharynx and soft palate was described in cats with NPS; however, neither the frequency of this observation nor dorsal deviation of the soft palate at the site of stenosis was specified. 13 In another study, dorsal deviation of the soft palate at the attachment site was present radiographically in 10/15 cats with NPS and was considered to be suggestive of NPS. 8 Therefore, the finding of dorsal deviation of the soft palate at the site of nasopharyngeal luminal narrowing on CT should raise suspicion for NPS. However, the position of the endotracheal tube should be considered in the interpretation of this feature as it may displace the soft palate dorsally. 15
Intravenous administration of contrast medium could be useful for the assessment of NPS as the contrast-enhancing membrane could be distinguished from nasopharyngeal luminal fluid or mucus that is accumulated within or rostral to the stenotic region; this could assist with recognition of NPS and assessment of the total length of the NPS.3,14 In the present study, there was no appreciable contrast enhancement of the NPS in 3/11 cats (36%) that received intravenous contrast medium. Although the lack of apparent contrast enhancement of the NPS in the three cats did not hinder their identification, the possibility of mucus accumulation interfering with the assessment of the appearance of the NPS or resulting in an artefactually thicker appearance in these cases cannot be excluded. As the reviewers were aware of the presence of NPS in all cases, this bias may have implications in detecting the stenosis, which could be missed under normal evaluation.
Reviewing the images using a high-frequency spatial reconstruction algorithm in a lung window helped detect and evaluate the NPS due to better discrimination of the edges of the NPS 14 and better contrast at the soft tissue–gas interface, and using this approach is therefore advised when reviewing CT images for NPS. Additionally, the images should be interpreted in combination with images reviewed using a medium-frequency spatial reconstruction algorithm in a soft tissue window, as well as on pre- and post-contrast images, if available, to help distinguish NPS from other soft tissue attenuating luminal material such as mucus or fluid.
One cat in the present study appeared to have two separate areas of NPS in the caudal nasopharynx on CT; however, only one area of stenosis in the caudal nasopharynx was visible on retroflex nasopharyngoscopy and antegrade rigid rhinoscopy was not possible. As a result, the CT finding of two separate areas of NPS could not be endoscopically confirmed. The possibility of the ET tube compressing the nasopharynx in this case 15 and creating the impression of a second area of stenosis is possible.
An underlying cause of the NPS was not determined in any of the cats in the present study. However, this is not uncommon, as demonstrated by a previous study where the underlying cause was not determined in 21/46 cases (46%) with NPS, of which 15 were dogs and 31 were cats. 4 In that study, the underlying cause was not determined in a higher proportion of cats (17/31; 55%) compared with dogs (4/15; 27%). 4 Acquired NPS is considered to occur more commonly than congenital NPS.4,7 In a previous study, 12/31 cats with NPS were suspected to have developed scar tissue secondary to either chronic rhinitis (7/12), post-anaesthetic aspiration rhinitis (1/12) or upper respiratory tract infection (4/12). 4 In our study, the suspected cause of the NPS was chronic upper respiratory tract infection in one cat and aspiration of gastric contents into the nasopharynx secondary to chronic vomiting in two cats. NPS can occur as a congenital anomaly, and choanal atresia, a congenital malformation of the caudal nasal cavity, has been reported to cause stenosis in the rostral nasopharynx. 23 Choanal atresia results in complete or incomplete obstruction of the choanae by a bony, membranous or mixed tissue, and has been reported in the dog and the cat.23–25 Although two cats in our study had displayed clinical signs since kittenhood, an acquired cause was presumed to be more likely for all 12 cats considering the caudal location of the NPS and the CT findings not being supportive for choanal atresia. Caudally located NPS have been reported with palatal dysgenesis in two cats;26,27 however, abnormalities of the soft palate were not identified on retroflex nasopharyngoscopy for all 12 cats in our study. Nevertheless, it can be challenging to distinguish congenital NPS from acquired NPS, as suggested by a recent study. 13
There is a clinical implication in distinguishing between a patent soft tissue membrane and an imperforate membrane because the outcome for patients with an imperforate membrane is less likely to be successful with balloon dilation. 4 A previous study found that imperforate membrane was infrequently identified in cats with NPS (3/31 cats) compared with dogs (8/15 dogs). 4 Interestingly, a more recent study identified a higher prevalence of imperforate membrane in its population of cats with NPS (11/23 cats; 48%), which was significantly associated with nasal obstruction and fluid in the tympanic bullae on CT. 13 The higher prevalence of imperforate membrane in that recent study may reflect the referral population and potentially a region-specific underlying aetiology (eg, infectious agent, congenital or developmental anomalies). In the present study, the nasopharynx in one cat was visible on reflex nasopharyngoscopy only following the passage of a balloon catheter, possibly representing an imperforate membrane. In this case, the lumen of the NPS was not visible on the CT images. Similarly, the majority of the lumen of the NPS was not visible on CT despite being visible on retroflex nasopharyngoscopy, suggesting that CT may not distinguish between NPS due to a patent soft tissue membrane or an imperforate membrane. The small number of cases in our study limits meaningful interpretation of this finding; however, we suggest that CT should not be considered the sole diagnostic modality for investigation of NPS, and the type of NPS should be characterised further endoscopically due to its clinical significance.
In the present study, findings compatible with non-destructive rhinitis were observed in 58% of cats and were considered mild in severity for the majority of cases. Changes compatible with rhinitis of varying degrees of severity have been reported in cats with NPS on CT,3,9,10,13 and this is suggested to be due to a decrease in nasopharyngeal mucus clearance from the nasal cavity. 13
In the present study, an accumulation of non-contrast-enhancing fluid or soft tissue attenuating material in the tympanic bullae was observed in only a small number of cats. None of the cats in the present study had clinical signs of ear disease. A previous study observed bilateral tympanic bulla effusion in 34% of cats with evidence of nasopharyngeal disease on CT. 28 The opening of the auditory tube is located near the region of the caudal nasopharynx; thus, pathology of the caudal nasopharynx could contribute to physical or functional alteration of the auditory tube and result in reduced clearance of material from the tympanic bulla or an ascending infection from the upper respiratory tract. 28
The present study has some limitations inherent to its retrospective nature. First, the CT acquisition and reconstruction parameters were not standardised, with two different CT scanners and differing intravenous contrast administration techniques used. The acquisition of images with thin slices (1 mm or less) is recommended for the investigation of NPS as the lesion could be missed or underestimated with thicker slices due to partial volume averaging.7,14 Additionally, the varied rate and duration of intravenous contrast injection, and timing between the injection of contrast medium and post-contrast imaging, could have altered the magnitude of contrast enhancement and the contrast enhancement pattern.20,29 However, the non-standardised acquisition and reconstruction parameters and intravenous contrast administration techniques likely reflect the variety of CT protocols encountered in clinical practice. Moreover, the goal of the present study was not to evaluate optimal scanning protocols for NPS.
A second limitation of the study was that the positioning of the head and neck was not standardised; this could have resulted in variations in the appearance of the NPS, the position of the ET tube, the position of the soft palate and the measurements made of the NPS. Future studies could standardise the position of the head and neck by positioning the head and neck of the animal as parallel as possible to the CT scanner table, and measuring the angle between the head and neck and table. Third, it was not standardised to have the mouth propped open by a plastic syringe case for assessment of the pharyngeal structures, and the positioning of the ET tube connector was not standardised. These factors resulted in variability in the mouth position of the cats (open vs closed) for the CT examination and the degree that the mouth was open. In a previous study, the visibility and assessment of pharyngeal structures including the nasopharynx was significantly improved with the mouth in an open position compared with closed. 15 As such, it was suggested that CT with the mouth open could be useful in the assessment of NPS. 15 However, as discussed above, the presence of the ET tube could displace the soft palate dorsally and compress the nasopharynx. 15 Thus, the presence of the ET tube could also potentially affect the appearance and measured dimensions of the NPS, and evaluation for the presence of dorsal deviation of the soft palate at the site of NPS. Future studies could evaluate the ideal degree to prop the mouth open, and the benefit of CT with or without an ET tube for the assessment of NPS. Finally, the number of cases included in the present study is small due to the uncommon nature of this condition.
Conclusions
This study highlights the CT features of NPS in cats. The most frequently observed features are marked, focal, abrupt, concentric narrowing of the nasopharyngeal lumen by a mildly contrast-enhancing soft tissue attenuating band in the caudal third of the nasopharynx and focal dorsal deviation of the soft palate at the site of stenosis. Identifying these CT features in the presence of compatible clinical signs should prompt further evaluation of the nasopharynx with retroflex nasopharyngoscopy to confirm the diagnosis and direct appropriate treatment.
Supplemental Material
Supplementary material 1
Clinical data of all 12 cats with confirmed nasopharyngeal stenosis.
Supplemental Material
Supplementary material 2
CT technical information and parameters of all 12 cats with confirmed nasopharyngeal stenosis.
Supplemental Material
Supplementary material 3
CT features of the nasopharyngeal stenosis of the 12 cats.
Supplemental Material
Supplementary material 4
Additional CT findings of the nasal cavity, tympanic bullae and medial retropharyngeal lymph nodes of the 12 cats.
Supplemental Material
Supplementary material 5
CT measurements of the right and left tympanic bulla wall thickness of the 12 cats.
Supplemental Material
Supplementary material 6
CT measurements of the right and left medial retropharyngeal lymph nodes of the 12 cats.
Footnotes
Author note
This paper was presented, in part, as an abstract at the 2024 European Association of Veterinary Diagnostic Imaging British and Irish Division (EAVDI-BID) Spring Meeting.
Supplementary material
The following files are available as supplementary material:
Supplementary material 1. Clinical data of all 12 cats with confirmed nasopharyngeal stenosis.
Supplementary material 2. CT technical information and parameters of all 12 cats with confirmed nasopharyngeal stenosis.
Supplementary material 3. CT features of the nasopharyngeal stenosis of the 12 cats.
Supplementary material 4. Additional CT findings of the nasal cavity, tympanic bullae and medial retropharyngeal lymph nodes of the 12 cats.
Supplementary material 5. CT measurements of the right and left tympanic bulla wall thickness of the 12 cats.
Supplementary material 6. CT measurements of the right and left medial retropharyngeal lymph nodes of the 12 cats.
Conflict of interest
Linnaeus Veterinary Limited supported the costs of the open access publication charges. The funder was not involved in the study design, collection, analysis, interpretation of data, the writing of this article or the decision to submit it for publication. The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as potential conflict of interest.
Funding
Linnaeus Veterinary Limited supported the costs of the open access publication charges.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognised high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers, tissues and samples) for all procedure(s) undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
