Abstract
Objectives
The aim of this study was to prospectively evaluate in a randomized, triple-masked, placebo-controlled trial, outcomes for kittens with ocular manifestations of infectious upper respiratory disease (IURD) treated with an ophthalmic and oral antibiotic only vs those also treated with famciclovir.
Methods
Kittens were stratified into three age (1 to <4, 4 to <8 or 8–12 weeks) and two disease severity (‘mild’ [total disease score 1–11] or ‘severe’ [total disease score 12–23]) groups and randomized to receive approximately 5 mg/kg doxycycline either with placebo (group D) or with approximately 90 mg/kg famciclovir (group DF) PO q12h. Caregivers scored clinical signs once daily for 21 days. Ophthalmic examinations and scoring by veterinarians were completed on days 1 and 21. Ophthalmic and clinical resolution were defined as scores of zero for all ocular signs and all ocular and respiratory signs, respectively. Ophthalmic and clinical recovery were defined by absence of active inflammation.
Results
For kittens with mild disease, those in group D were slower than those in group DF to achieve clinical recovery (P = 0.049) and clinical resolution (P = 0.030), but not ophthalmic recovery (P = 0.064) or ophthalmic resolution (P = 0.089). Kittens with mild disease and receiving famciclovir achieved predicted 75% clinical resolution 4–5 days earlier than kittens with mild disease and receiving doxycycline alone, and kittens with severe disease (regardless of treatment group). Significantly fewer kittens in group DF developed corneal disease than in group D (P = 0.016). All five kittens whose clinical signs worsened sufficiently to be removed from the study were in group D.
Conclusions and relevance
The addition of famciclovir to standard antibiotic treatment may reduce corneal disease, length of stay and time to adoption for shelters and rescue groups, thereby increasing capacity for care. Early administration of famciclovir in kittens exhibiting mild ocular manifestations of IURD may be preferable to delaying this treatment until the disease progresses to a severe stage.
Keywords
Introduction
Infectious upper respiratory disease (IURD) is a common and major health problem for kittens worldwide, especially those housed in multi-cat environments, such as animal shelters, rescue groups and breeding catteries.1,2 Although many infectious agents may be involved, major pathogens include feline herpesvirus type 1 (FHV-1), feline calicivirus (FCV), Chlamydia felis, Bordetella bronchiseptica and Mycoplasma felis. 3 Of these, herpetic syndromes are considered most common, severe and persistent/recurrent, causing 100% morbidity and notable mortality in susceptible kittens. 4 Methods to control IURD in multi-cat settings include quarantine (which is consumptive of time, resources and space), 5 reduced crowding (which requires changes in shelter policies regarding euthanasia or admissions) and vaccination (which is not usually recommended in kittens aged <4 weeks).1,2,6 Therefore, IURD is a significant welfare and financial concern, and a major cause of increased length of stay (LOS), enucleation, euthanasia or difficulty rehousing cats and kittens from multi-cat settings.1,2
Treatment of IURD is typically symptomatic and heavily reliant on adequate supportive care; however, administration of antimicrobial agents with activity against known etiological agents is also indicated. Doxycycline is commonly administered for its reliable efficacy against C felis,7–9 M felis 10 and B bronchiseptica, 11 as well as other common respiratory bacterial pathogens,12–14 and is preferred over azithromycin 7 and topically applied agents 8 for C felis specifically. The tetracyclines may also exhibit some desirable anti-inflammatory and antiprotease effects distinct from their antimicrobial effects. 15 Meanwhile, for FHV-1, data regarding the safety and efficacy of famciclovir administered at 90 mg/kg are promising.16–21 However, there are only two studies reporting famciclovir treatment of neonatal and juvenile kittens with IURD.16,22 In one retrospective study, 16 neonatal/juvenile kittens showed marked improvement in clinical signs, and demonstrated fewer adverse events and underwent a shorter duration of treatment than older cats. 16 In another study, a significant difference in disease score was not detected between kittens receiving doxycycline and famciclovir vs those receiving doxycycline only; however, outcomes were reported for only 12 kittens/group, medication dosages were not adjusted as the kittens gained body weight, kittens were treated for variable periods and disease was scored with an emphasis on respiratory signs. Therefore, in the present study, we assessed the comparative efficacy of doxycycline only vs doxycycline-famciclovir in combination in a large population of neonatal and juvenile kittens treated for 21 days using doses adjusted weekly for change in body weight and using a scoring system that included respiratory and ophthalmic signs. We hypothesized that the addition of orally administered famciclovir would reduce disease duration and severity relative to that seen in kittens treated only with orally administered doxycycline.
Materials and methods
Data collection
This prospective, randomized, triple-masked, placebo-controlled trial enrolled kittens cared for by four tax-exempt animal rescue organizations and one animal shelter within Northern California between March 2019 and October 2020, and was approved by the University of California’s Animal Care and Use Committee. Informed consent was obtained for each kitten enrolled in the study. To assess eligibility for enrollment and to stratify kittens into treatment groups, all eligible kittens underwent a complete physical examination by a veterinarian at the William R Pritchard Veterinary Medical Teaching Hospital of the University of California Davis (UCD). At this time, kittens were also weighed and their age was estimated using physical characteristics, body weight and known eruption times for deciduous and permanent teeth. 23 In addition, clinical signs of IURD were graded according to a published scoring system, 24 modified only by removal of the single score for sneezing such that the total disease score was in the range of 0–23 (see Appendix A in the supplementary material, page 4). All kittens were also tested for the presence of serum feline leukemia virus (FeLV) antigen (SNAP test; IDEXX).
To be enrolled in this study, kittens were required to have clinical signs compatible with IURD (ie, total disease score ⩾1), weigh ⩽1.36 kg (3 lbs), have an estimated age of ⩽12 weeks, be free of circulating FeLV antigens and to have complete day 1 ophthalmic scores for both eyes. Eligible kittens were stratified at study entry into one of three age groups (1 to <4, 4 to <8 or 8–12 weeks) and one of two disease severity groups (‘mild’ [total disease score 1–11] or ‘severe’ [total disease score 12–23]). To ensure treatments were balanced across all age and disease severity strata, a block randomization was implemented with a block size of 4 and kittens randomized to receive one of two treatments. Kittens in group DF (comprising a total of six age and disease severity groups) received doxycycline monohydrate oral suspension ~5 mg/kg PO q12h (Lupin Pharmaceuticals) and famciclovir ~90 mg/kg PO q12h (Macleods Pharmaceuticals). For ethical reasons, an untreated group could not be included in this clinical trial. Kittens in group D (a further six age and disease severity groups) received doxycycline ~5 mg/kg PO q12h and, as a placebo, powdered lactose monohydrate PO q12h (Medisca). Kittens in both treatment groups also received one drop 0.3% ofloxacin ophthalmic solution (Akorn) in both eyes q12h. All kittens in both groups were treated for 21 days. Doses of doxycycline and famciclovir were considered approximate because kitten body weights changed rapidly throughout the study. For this reason, each kitten’s dose of famciclovir and doxycycline was adjusted and new prescriptions of medication were dispensed every 7 days according to a formula based on their body weight and designed to deliver at least 90 mg/kg famciclovir and 5 mg/kg doxycycline (see Tables 1 and 2 in the supplementary material). Doxycycline monohydrate suspension was reconstituted with water according to the manufacturer’s instructions. According to the package insert, this suspension was stable for 14 days at room temperature, at which time it was replaced with a newly reconstituted bottle of doxycycline. Famciclovir was prepared using commercially available tablets that were crushed, weighed and placed into gelatin capsules, as previously described.18,20 For the placebo, powdered lactose was placed into identical gelatin capsules using a similar volume to that used for the famciclovir capsules. Caregivers were given verbal and printed instructions to open the prescribed famciclovir- or lactose-containing gelatin capsules immediately before each dose, mix the contents with the doxycycline suspension and administer the mixture orally using a dosing syringe. All medications were dispensed by the UCD pharmacist who was the only study member unmasked to treatment. All caregivers, study veterinarians and staff, study authors and statisticians remained masked until after data analysis was complete.
Kittens were housed individually or in small groups in private foster homes. Shelter staff and foster coordinators as well as all caregivers completed a training program before caring for the kittens enrolled in this study. Kittens in foster groups were given colored collars if they could not be identified by study personnel using coat color and sex. Kittens in foster groups shared one bottle of doxycycline but had individualized prescriptions of placebo or famciclovir capsules based on their treatment randomization. Caregivers were given separate oral dosing syringes for each kitten and were instructed to clean the syringes with water after use and to replace them if they became difficult to use. Other than the administration of the three study-related medications, caregivers followed all shelter/rescue group protocols. Once daily for 21 days at the same time of day, trained caregivers recorded the body weight and completed a clinical disease scoring sheet (see Appendix A in the supplementary material) for each kitten in their care. Both eyes were scored using the same modified scoring system as that used by the admitting veterinarian 24 for severity of ‘conjunctivitis (conjunctival redness)’, ‘blepharospasm (squinting)’, ‘ocular discharge’ and ‘nasal discharge’ with a total possible disease score of 0–23. Caregivers were required to report any concerns or worsening of clinical signs to the study manager and shelter/rescue group when they occurred, and to submit their scoring sheets to the study manager every 7 days.
At study entry and study end (day 21), all kittens had a general physical examination performed by a study veterinarian and slit lamp biomicroscopy performed by a board-certified veterinary ophthalmologist or a veterinary ophthalmology resident. Both eyes were evaluated and scored according to a standardized scoring system more detailed than the one used by the admitting veterinarian and caregivers, which included ocular discharge, conjunctival hyperemia, chemosis, density and extent of corneal opacities, extent of corneal vascularization, extent of corneal fluorescein retention and the presence/absence of symblepharon, globe rupture and dendritic ulcers (see Appendix B in the supplementary material). This produced a total ocular disease score in the range of 0–52. On two occasions, the entire globe could not be visualized and some clinical features could not be scored. For data analysis, maximum possible scores were assigned to all ungraded categories in these instances.
Kittens could be removed throughout the study for any of the following reasons: caregivers’ or veterinarians’ failure to follow all protocols; incorrect study medication dispensed; non-fatal medical diseases other than IURD; death; or worsening of clinical signs of IURD (defined as those that failed to improve over any 5 consecutive days or worsened over 3 days). Regardless of the reason for censorship, all kittens were appropriately treated by the responsible veterinarian. Kittens removed from the study before 21 days or that did not experience the outcome event by day 21 were considered censored for analytical purposes.
Data analysis
Data generated through veterinary and caregiver examinations differed in several ways. Caregiver data consisted of limited clinical criteria, scored once daily for 21 days, by multiple trained lay observers, whereas veterinary data consisted of extensive clinical criteria, scored on days 1 and 21 only, by a board-certified veterinary ophthalmologist or a veterinary ophthalmology resident. For the caregiver data set, data from each kitten were included until the day of censorship; therefore, some caregiver data sets were incomplete. By comparison, kittens were required to contribute complete data from days 1 and 21 for inclusion in the veterinary data set. Therefore, kittens could be censored from one data set but contribute data to the other. For this reason, the two data sets were analyzed and reported separately and in different ways. However, to assess the alignment of caregiver and veterinary data, scores for ocular discharge assigned by caregivers and veterinarians on days 1 and 21 were compared using Stuart-Maxwell marginal homogeneity tests.
For caregiver data, the primary outcome measure was the rate of improvement in the clinical signs of kittens in the two treatment groups as evaluated once daily for 21 days by the caregivers. For analysis, four distinct definitions of improvement in clinical signs were used. The first two (termed ‘resolution’) assessed time to achieve a clinical score of zero. ‘Clinical resolution’ was defined as achievement by day 21 or earlier of a total clinical score of zero, based on the sum of all scores, including nasal discharge (ie, ocular discharge, blepharospasm, conjunctival hyperemia, nasal discharge). ‘Ophthalmic resolution (caregiver)’ was defined as achievement by day 21 or earlier of a total clinical score of zero for ophthalmic signs only (ocular discharge, blepharospasm and conjunctival hyperemia) without regard to nasal discharge. To account for the fact that serous nasal discharge or epiphora is a minor and, in many instances, tolerable sign of chronic prior IURD, a second definition (termed ‘recovery’) was also used. ‘Clinical recovery’ required scores of zero for conjunctival hyperemia and blepharospasm, and ⩽1 for nasal and ocular discharge. ‘Ophthalmic recovery (caregiver)’ required scores of zero for conjunctival hyperemia and blepharospasm, and ⩽1 for ocular discharge, with nasal discharge ignored. For all four definitions, time-to-resolution/recovery analyses were conducted using Kaplan–Meier plots and Cox proportional hazard regression models, with results presented as hazard ratios (HRs) and 95% confidence intervals (CIs). Interactions between variables were evaluated using likelihood ratio tests to assess the effect modification of the HR. The linearity assumption of the relationship between continuous covariates (severity, weight and age) and the response variable (log hazard rate of time to clinical sign resolution) was tested using link tests. Data from kittens with incomplete caregiver data sets were analyzed until the date of censorship. Time-to-event regression models were constructed to primarily evaluate the main effect of treatment for each of the four clinical outcomes, while evaluating age, weight and disease severity as confounders and effect modifiers. Because weight did not meaningfully (>10%) change the regression coefficients, it was omitted from further models. Although age was retained as a confounder in all models because of its wide variation in study kittens, there was no statistical interaction evidence supporting its inclusion as an effect modifier.
Veterinary data were handled similarly to those generated by caregivers except that data were recorded on days 1 and 21 only, ophthalmic data were more detailed and nasal discharge was not recorded. Similar to the handling of caregiver data, ‘ophthalmic resolution (veterinary)’ was defined as achievement of a total clinical score of zero for every clinical sign on day 21. However, to account for the fact that some clinical signs, such as symblepharon, corneal scarring, epiphora due to cicatrization of nasolacrimal drainage structures and so on, are likely to be permanent but not indicate active disease, a second endpoint of ‘ophthalmic recovery (veterinary)’ was defined as achievement of all of the following clinical scores by day 21: ocular discharge ⩽1; conjunctival hyperemia = 0; chemosis = 0; corneal opacity ⩽1; area of corneal opacity ⩽1; corneal vascularization = 0; fluorescein = 0; and dendritic ulcers = 0.
For caregiver and veterinary data sets, proportions of (1) kittens censored, (2) eyes in which various clinical signs persisted, developed or resolved, (3) kittens achieving clinical/ophthalmic resolution/recovery and (4) kittens changing from unilateral to bilateral disease or vice versa were compared between the two treatment groups and between the two disease severity groups using Fisher’s exact test and among the three age groups using the Cochrane–Armitage test.
All statistical analyses were performed using Stata/BE 17.0 statistical software (StataCorp). For all analyses, P values <0.05 were considered statistically significant.
Results
A total of 373 kittens entered the study and were stratified with respect to age and disease severity, and then randomized with respect to treatment (D or DF) such that they formed 12 groups (Figure 1).

A total of 373 kittens entered the study and were stratified into one of three age groups (1 to <4, 4 to <8 or 8–12 weeks) and one of two disease severity groups (‘mild’ [total disease score 1–11] or ‘severe’ [total disease score 12–23]) before being randomized into one of two treatment groups (doxycycline and famciclovir [group DF] or doxycycline and placebo [group D]). (a) For the caregiver data set, no data were available on day 1 for 23 kittens. (b) For the veterinary data set, 42 kittens were censored at various points throughout the study
Caregiver data
Of the 373 eligible kittens, no caregiver data were available on day 1 for 23 kittens. In addition, 21 kittens were censored at various points throughout the study (Table 1) and, therefore, contributed only partial caregiver data. Thus, caregiver data were available for analysis from 350 kittens (329 with complete data and 21 with partial data) (Figure 1a). No significant differences were detected in the proportion of kittens censored regardless of whether these data were assessed by treatment, age or disease severity groups assigned at study entry (Table 1).
Number of kittens removed from analyses of caregiver data and reasons for removal considered by treatment group, age groups and severity groups at study entry

No significant difference was detected for any comparison. Treatment and disease severity groups were compared using Fisher’s exact test, and age groups were compared using the Cochran–Armitage test
D = doxycycline; DF = doxycycline and famciclovir; IURD = infectious upper respiratory disease
Considering recovery and resolution data for all kittens for which caregiver data were available and without regard for treatment group or censorship, clinical resolution (ie, scores for ocular discharge, blepharospasm, hyperemia and nasal discharge all = 0) was achieved in 309/349 (89%) kittens, and clinical recovery (ie, scores of zero for conjunctival hyperemia and blepharospasm, and ⩽1 for nasal and ocular discharge) was achieved in 318/349 (91%) kittens. Ophthalmic resolution and recovery (ie, ignoring nasal discharge scores) were achieved in 314/350 (90%) and 320/350 (91%) kittens, respectively. The proportion of kittens that achieved clinical or ophthalmic resolution or recovery (as judged by caregivers) did not differ significantly by treatment group (P = 0.242–0.650) (Table 2).
Proportion of kittens achieving bilateral ophthalmic or clinical resolution or recovery as scored by veterinarians and caregivers and considered by treatment group

Caregiver-generated clinical resolution and recovery data were available for 349 kittens and ophthalmic resolution and recovery data for 350 (one caregiver failed to score nasal discharge). Veterinarian-generated ophthalmic resolution and recovery data were available for 331 kittens. No significant difference in the proportions of kittens was detected for any comparison (Fisher’s exact test). See ‘Materials and methods’ section for definitions of caregiver and veterinary ophthalmic recovery and resolution, and caregiver clinical recovery and resolution
D = doxycycline; DF = doxycycline and famciclovir; NA = not applicable
Disease severity was strongly associated with time to outcomes, with severely affected kittens taking longer than mildly affected kittens to achieve ophthalmic or clinical recovery or resolution (HR 0.66–0.67, P <0.001 for all four outcomes). When evaluating severity as an effect modifier, there was a consistent pattern of interactions between treatment and severity after adjustment for age, with those differences being significant for ophthalmic recovery (P = 0.048) and close to significant for clinical recovery (P = 0.055). Differences for clinical resolution (P = 0.102) and ophthalmic resolution (P = 0.104) were not significant. For kittens with mild disease, those in treatment group D were slower than those in group DF to achieve clinical recovery (HR 0.73, 95% CI 0.54–1.00, P = 0.049) and clinical resolution (HR 0.71, 95% CI 0.52–0.97, P = 0.030) but not ophthalmic recovery (HR 0.75, 95% CI 0.55–1.02, P = 0.064) or ophthalmic resolution (HR 0.76, 95% CI 0.56–1.04, P = 0.089). No meaningful difference between treatments was detected when disease was severe (P = 0.48–0.94). Using clinical resolution as an example, kittens with mild disease and receiving famciclovir achieved the predicted 75% resolution of clinical signs 4–5 days earlier than kittens in the other three groups (ie, kittens with mild disease and receiving doxycycline alone or kittens with severe disease in either treatment group). Kittens with mild disease receiving famciclovir achieved the predicted 75% resolution of clinical signs 5 days earlier than kittens receiving doxycycline alone, for all outcomes (clinical resolution, clinical recovery, ophthalmic resolution and ophthalmic recovery). These findings are visually demonstrated in Figure 2.

Kaplan–Meier survival curves showing the probability of achieving (a) ophthalmic recovery or (b) resolution or (c) clinical recovery or (d) resolution for kittens with infectious upper respiratory disease defined at study entry as mild (dashed lines) or severe (solid lines) and treated twice daily for 21 days with doxycycline alone (black lines) or with famciclovir (blue lines)
Veterinary data
Regarding the veterinary data set, 42 kittens were censored at various points throughout the study. Thus, complete veterinary data were available for analysis from 331 kittens (Figure 1b). Although no significant differences in the proportion of censored kittens were detected when assessed by treatment, age or disease severity groups assigned at study entry, no kittens in group DF required censoring due to worsening of IURD differences and the difference between this proportion and the proportion in group D (n = 5) neared significance (P = 0.061) (Table 3).
Number of kittens removed from analyses of veterinary data and reasons for removal considered by treatment group, age groups and severity groups at study entry

Data are n. Although some comparisons approached significance, no significant difference was detected for any comparison. Treatment and disease severity groups were compared using Fisher’s exact test, and age groups were compared using the Cochran–Armitage test
D = doxycycline; DF = doxycycline and famciclovir; IURD = infectious upper respiratory disease
According to the veterinary definitions, ophthalmic resolution (veterinary) (ie, total clinical score = 0) was achieved in both eyes of 206/331 (62%) kittens, and unilaterally in a further 55/331 (18%) kittens (Figures 3 and 4). The median disease score in the 195 eyes not achieving ophthalmic resolution was 1 (range 1–29), with 108 (55%) eyes achieving a score of 1 (Figures 5 and 6). Ophthalmic recovery (veterinary) (ie, minimal/inactive disease signs) was achieved in both eyes of 221 (67%) kittens, and unilaterally in a further 50 (15%) kittens (Figures 7 and 8). The proportion of kittens that achieved ophthalmic resolution or recovery (as judged by veterinarians) did not differ significantly by treatment group (P = 0.312) (Table 2).

External photographs of (a,d) both eyes, and the (b,e) right and (c,f) left eyes of a domestic shorthair kitten aged approximately 4 weeks (a sibling of the kitten shown in Figure 4) on (a–c) day 1 and (d–f) day 21 of treatment with famciclovir and doxycycline. The kitten’s total clinical score on day 1 was 16; by day 21, it had achieved clinical resolution with a score of zero

External photographs of (a,d) both eyes, and the (b,e) right and (c,f) left eyes of a domestic shorthair kitten aged approximately 4 weeks (a sibling of the kitten shown in Figure 3) on (a–c) day 1 and (d–f) day 21 of treatment with doxycycline only. The kitten’s total clinical score on day 1 was 15; by day 21, it had achieved clinical resolution with a score of zero

External photographs of (a,d) both eyes, and the (b,e) right and (c,f) left eyes of a domestic shorthair kitten aged approximately 4 weeks on (a–c) day 1 and (d–f) day 21 of treatment with famciclovir and doxycycline. The kitten’s total clinical score on day 1 was 44; by day 21, it had not achieved clinical resolution/recovery (total clinical score of 15) and remained ulcerated in the left eye

External photographs of (a,d) both eyes, and the (b,e) right and (c,f) left eyes of a domestic shorthair kitten aged approximately 4 weeks on (a–c) day 1 and (d–f) day 21 of treatment with doxycycline only. The kitten’s total clinical score on day 1 was 35; by day 21, it had not achieved clinical resolution/recovery (total clinical score of 11) but was non-ulcerated in both eyes
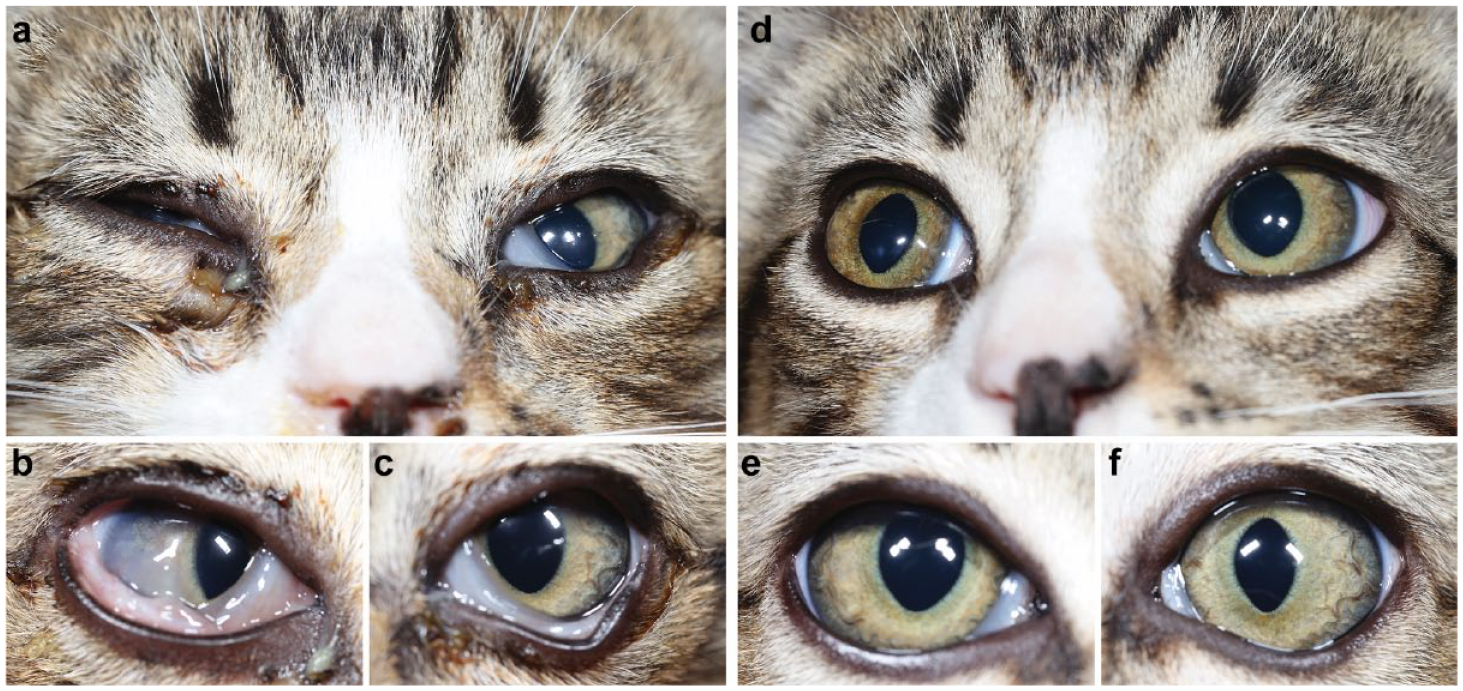
External photographs of (a,d) both eyes, and the (b,e) right and (c,f) left eyes of a domestic shorthair kitten aged approximately 4 weeks on (a–c) day 1 and (d–f) day 21 of treatment with famciclovir and doxycycline. The kitten’s total clinical score on day 1 was 23; by day 21, it had achieved clinical recovery with a score of 2 in the right eye (which was non-ulcerated) and zero in the left eye

External photographs of (a,d) both eyes, and the (b,e) right and (c,f) left eyes of a female domestic shorthair kitten aged approximately 8 weeks on (a–c) day 1 and (d–f) day 21 of treatment with doxycycline only. The kitten’s total clinical score on day 1 was 14; by day 21, it had achieved clinical recovery with a score of 2 in the right eye (which was non-ulcerated) and zero in the left eye
Because only two time points were assessed in the veterinary data sets, the rate of disease progression/regression could not be assessed; however, the proportions of kittens experiencing resolution, development or persistence of ophthalmic signs were assessed. Proportions of kittens in which corneal ulceration, corneal disease, conjunctivitis, symblepharon or globe rupture resolved, developed or persisted by day 21 are summarized in Table 4. Significantly fewer kittens receiving famciclovir developed corneal disease over the 21-day observation period (P = 0.016). Meanwhile, symblepharon persisted in more kittens receiving famciclovir and doxycycline than in those receiving doxycycline alone; however, this difference did not achieve significance (P = 0.077) and was not noted for the development or resolution of symblepharon. Looking collectively at unilateral vs bilateral outcomes, no significant difference was detected between treatment groups (P = 0.193–0.954) (Table 5).
Number of eyes in which specified clinical signs resolved, developed or persisted based on analysis of veterinary data and considered by treatment group

Data are n. Data were analyzed using Fisher’s exact test. The one significant difference is shown in bold. For the purpose of these analyses, ‘corneal ulceration’ was defined as any eye with a dendritic or geographic ulcer; ‘corneal disease’ was defined as any eye with corneal opacity, ulceration (dendritic or geographic) and/or corneal vascularization; ‘conjunctivitis’ was defined as any eye with hyperemia and/or chemosis
D = doxycycline; DF = doxycycline and famciclovir
Progression or resolution of ocular disease based on analysis of veterinary data and by treatment group

Data are n. No significant difference in the number of kittens was detected for any comparison (Fisher’s exact test)
D = doxycycline; DF = doxycycline and famciclovir
On day 1, there were occasional but notable differences in the scoring of ocular discharge between caregivers and veterinarians (P = 0.0036). Discordant scoring arose when caregivers assigned a score of 2, while veterinarians were more likely to assign scores in the range of 3–5 (ie, 1–3 points higher). On day 21, however, there was much greater concordance between scores assigned by veterinarians and caregivers (P = 0.851), particularly because 82% of both scorers assigned scores of zero and only 1% of scores were 3 or greater.
Discussion
This study reports clinical outcomes for more than 330 kittens aged <12 weeks, diagnosed with ophthalmic manifestations of IURD and managed by trained caregivers in foster homes under the close supervision of study veterinarians. All kittens received standard nursing care, topically applied ofloxacin (one drop in both eyes q12h) and doxycycline (5 mg/kg PO q12h). The principal goal of this study was to assess the effect of adding famciclovir (90 mg/kg PO q12h) to this 21-day treatment protocol. Although the majority of kittens in the study (regardless of treatment, age or disease severity at study entry) achieved the defined resolution or recovery endpoints, kittens receiving famciclovir along with antibiotics had a number of statistically significant or near-significant outcomes superior to those seen in kittens receiving antibiotics only. Most importantly, kittens with mild disease at study entry and receiving famciclovir in addition to doxycycline achieved clinical resolution (complete absence of clinical signs) and clinical recovery (absence of active disease) significantly faster than kittens receiving doxycycline only did. The rates at which they achieved ophthalmic resolution (P = 0.089) and recovery (P = 0.064) also approached significance. Moreover, compared with those receiving only doxycycline, significantly fewer kittens treated with famciclovir developed corneal disease. Finally, all kittens whose clinical signs worsened sufficiently to be removed from the study received doxycycline only, although this difference was not large enough to achieve significance (P = 0.108). The results of the present study are in alignment with those from one previous study 16 but differ from those of another study. 22 These disparate results may reflect the fact that the present study enrolled many-fold more kittens than in prior studies, restricted enrolment to kittens aged ⩽12 weeks and with a body weight ⩽1.36 kg, and adjusted medication dosages every 7 days to account for the rapid changes in a kitten’s body weight at this age.
Collectively, the results from the present study support the use of famciclovir along with doxycycline in kittens with IURD, especially when considered on a population basis, since reducing LOS for kittens at a shelter improves the shelter’s capacity for care. Assuming a shelter houses just 20 kittens, a reduction in time to 75% resolution of 4–5 days, as predicted in the present study, would amount to a reduction of 80–100 kitten days within that establishment – along with consequent savings in time, resources and associated personnel. This, in turn, allows more kittens to be cared for over time and lowers the daily shelter population, potentially reducing the risk of euthanasia due to lack of space. 25 Reducing LOS in foster homes would likely be associated with reduced workload and burnout in caregivers as well as shelter staff managing foster homes, potentially improving caregiver retention and increasing the capacity for care for rescue groups. Therefore, the addition of famciclovir may save the lives of kittens. For several reasons, reductions in LOS and improvement in capacity for care are especially important regarding kittens vs older cats or dogs and puppies. Kittens are routinely the smallest and sometimes the most vulnerable of patients cared for in multi-cat settings, and even numerically small improvements in the reliability or speed of recovery can produce clinically significant changes. Likewise, capacity for care issues are particularly pertinent in kittens vs puppies since cats are seasonal breeders, meaning shelters and rescue groups have times of the year when their resources are likely to be overwhelmed, making even a small decrease in LOS potentially meaningful, especially since some believe that younger kittens are easier to adopt than older kittens. Furthermore, kittens with IURD are typically quarantined or isolated within foster homes and/or shelters, further limiting space and resources. Shorter times in hospital or isolation wards allow more kittens or cats in need to be treated and permits kittens to be moved to group housing with more social interactions, thereby encouraging normal behavioral development and earlier adoption.
These findings are especially important given the ease of administration, low cost and high tolerability of famciclovir in the present study. Herein, 90 mg/kg famciclovir was administered by crushing a tablet and combining it with doxycycline suspension for oral administration. These two drugs were combined immediately before administration and were not pre-mixed. This is critical given that famciclovir compounded as a pre-mixed oral suspension showed poor accuracy, precision and consistency of formulation.26,27 Although this study did not specifically assess the safety of famciclovir administration in kittens, the overall excellent and similar rates of ophthalmic and clinical resolution and recovery in kittens receiving or not receiving famciclovir appear to confirm earlier work showing that cats in general16–21,28–30 and younger kittens in particular 16 tolerate famciclovir very well. Even at 90 mg/kg (the dose required to achieve plasma and tear drug concentrations likely to have an antiviral effect against FHV-1), 30 famciclovir is an inexpensive option in small kittens. Although the purchase price varies widely across global markets, at the time of performing this study, the client cost in our practice for a typical daily dose for a 10-week-old kitten weighing 1 kg cost just US$0.33. Considering multi-cat settings and given the reduced LOS and increased capacity of care afforded by famciclovir administration, this represents a relatively small and justifiable investment. Managers of other multi-cat environments would need to conduct similar analyses considering all important factors, especially pharmaceutical, housing and staffing costs.
Although this study demonstrated multiple outcomes that were significantly better for kittens receiving famciclovir, no significant difference was noted between treatment groups for the majority of outcomes. This was largely because of the very high proportion of kittens that improved in this study regardless of treatment. Overall, 87% of kittens achieved the defined ‘resolution’ of ophthalmic and overal clinical disease as assessed by their caregiver, and 63% and 69% achieved clinical resolution and clinical recovery, respectively, as assessed by a veterinarian. Strikingly, only two kittens in this study died or were euthanized (one each from treatment groups D and DF), and no eyes were enucleated. Taken together, these results speak to the importance of excellent supportive care (nutrition, hydration, temperature control, and parasite therapy and prophylaxis) and appropriate antimicrobial support as was provided by the caregivers caring for kittens in their homes in this study.
Conclusions
IURD is a significant welfare and financial concern for kitten owners, animal shelters and rescue organizations and their caregivers, and remains one of the major reasons for enucleation, euthanasia and difficulty in finding homes for kittens, thereby increasing LOS and reducing the capacity for care. In many animal shelters and rescue organizations, famciclovir is not prescribed unless kittens have recurrent or severe disease. As mildly affected kittens in the present study experienced significantly more rapid resolution of clinical signs when treated with famciclovir and doxycycline than did kittens treated with doxycycline only, this study supports the early treatment of kittens with famciclovir, rather than waiting until they develop severe or recurrent disease. Although the administration of famciclovir may increase pharmaceutical costs and require increased staff time for medication administration, the decrease in LOS and increase in positive outcomes will improve animal welfare and may economically justify the use of famciclovir in conjunction with doxycycline for the treatment of IURD in kittens within shelters and other multi-cat settings.
Supplemental Material
sj-docx-3-jfm-10.1177_1098612X241278413 – Supplemental material for Doxycycline with or without famciclovir for infectious ophthalmic and respiratory disease: a prospective, randomized, masked, placebo-controlled trial in 373 kittens
Table 1: Dose of famciclovir administered to kittens per body weight.
Supplemental Material
sj-docx-4-jfm-10.1177_1098612X241278413 – Supplemental material for Doxycycline with or without famciclovir for infectious ophthalmic and respiratory disease: a prospective, randomized, masked, placebo-controlled trial in 373 kittens
Table 2: Dose of doxycycline administered to kittens per bodyweight.
Supplemental Material
sj-pdf-1-jfm-10.1177_1098612X241278413 – Supplemental material for Doxycycline with or without famciclovir for infectious ophthalmic and respiratory disease: a prospective, randomized, masked, placebo-controlled trial in 373 kittens
Appendix A: Foster parent daily scoring chart.
Supplemental Material
sj-pdf-2-jfm-10.1177_1098612X241278413 – Supplemental material for Doxycycline with or without famciclovir for infectious ophthalmic and respiratory disease: a prospective, randomized, masked, placebo-controlled trial in 373 kittens
Appendix B: Ophthalmology examination scoring chart.
Footnotes
Acknowledgements
The authors thank the students of the Orphan Kitten Project, staff of the Yolo County Society for the Protection of Cruelty to Animals as well as their kitten caregivers, staff at the UC Davis Community Surgery, Neurology/Neurosurgery, Emergency and Critical Care, Pharmacy and Ophthalmology Services for their collaborative care of kittens, Dr Chrisoula Toupadakis Skouritakis for her expert assistance with figure preparation and Dr Kate Hurley for her thoughtful comments on the manuscript. The authors also acknowledge the support of Nestlé Purina for providing the kitten food used in this study.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was financially supported by The Center for Companion Animal Health, School of Veterinary Medicine, UC Davis, Maddie’s Fund, The Orphan Kitten Club and National Institutes of Health (P30 EY12576 and R01 EY016134 to SMT).
Supplementary material
The following files are available as supplementary material:
Table 1: Dose of famciclovir administered to kittens per body weight.
Table 2: Dose of doxycycline administered to kittens per body weight.
Appendix A: Foster parent daily scoring chart.
Appendix B: Ophthalmology examination scoring chart.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognised high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers, tissues and samples) for all procedure(s) undertaken (prospective or retrospective studies). For animals or people individually identifiable within this publication, informed consent (either verbal or written) for their use in the publication was obtained from the people involved.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
