Abstract
Mast cell infiltration occurs in malignant, inflammatory (eg, allergic, infectious), and idiopathic disease processes in humans and animals. Here, we describe the clinical and histological features of a unique proliferative conjunctivitis occurring in 15 cats. Ocular specimens were examined histologically, and polymerase chain reaction (PCR) for feline herpesvirus 1 (FHV-1) was performed on ocular tissues obtained from 10 cats. Cats had a median age of 8 years (range: 7 months–17.5 years). The known median duration of ocular lesions prior to biopsy was 4 months (range: 1 week–3 years). Ocular disease was unilateral in 12 cats, and 9 cats had coexisting corneal disease. Clinically and histologically, proliferative or nodular conjunctival lesions were noted in 13 cats. The nictitating membrane was affected in 10 cats. Histologically, lesions were characterized by mixed inflammatory infiltrates with an abundance of Giemsa-positive and toluidine blue–positive intraepithelial and subepithelial mast cells, marked edema, and papillary epithelial hyperplasia. Feline herpesvirus 1 was demonstrated by PCR in 1 of 10 cats tested. Follow-up information was available for 14 cats: 8 had no recurrence during a median follow-up period of 17.5 months (range: 4.5–30 months), 2 underwent orbital exenteration, 3 had recurrence that was medically managed, and 1 cat had diffuse conjunctivitis at the time of biopsy and recurrence was deemed irrelevant. Various ocular medications were administered before and after surgical biopsy. This condition was designated as feline epitheliotropic mastocytic conjunctivitis, with intraepithelial mast cells being an essential feature and papillary epithelial proliferation being characteristic but not diagnostic alone. The condition appears to be uncommon and benign. Although the cause is unknown, an allergic component is possible.
Keywords
Tissue infiltration by mast cells occurs in cats with mast cell tumors, 2,13,15 eosinophilic keratitis, 3 eosinophilic conjunctivitis, 1,19 and papular mastocytic dermatitis. 17 Papillary conjunctivitis with intraepithelial mast cells has been described in people with vernal 11 or giant papillary 10 conjunctivitis but not with allergic or atopic conjunctivitis. While vernal conjunctivitis is presumed to be a seasonal allergic condition, giant papillary conjunctivitis is mostly associated with contact lens wear. 10,11
Mast cell epitheliotropism is uncommon but has been documented in 3 dogs with an unusual cutaneous mast cell tumor, 2 of which were located in mucocutaneous tissues, 18 and in 2 cats with mast cell tumors, 1 of which was located in mucocutaneous tissue. 2,12 Formation of conjunctival papillae with mast cells was described in 3 of 5 cats with eosinophilic conjunctivitis. 19 Intraepithelial mast cells were also described in keratectomy samples from cats diagnosed with feline eosinophilic keratitis. 21 In captive North American cheetahs, plaque-like edematous swellings affecting the medial conjunctiva were associated with the presence of herpesvirus 1 nuclear inclusions and with concurrent ulcerative keratitis and facial dermatitis. 16
At the Comparative Ocular Pathology Laboratory of Wisconsin, we recognized a unique proliferative conjunctivitis associated with mast cell epitheliotropism in cats. The purpose of this study was to report the signalment, clinical presentation, and outcome of cats with these lesions and to describe their histopathological features.
Materials and Methods
The Comparative Ocular Pathology Laboratory of Wisconsin holds approximately 10 000 formalin-fixed, paraffin-embedded feline ocular tissues. A search of the database for the terms “feline” and “conjunctiva” and “mastocytosis” or “mast cell” yielded 24 records from 23 cats. Fifteen cats were diagnosed with feline epitheliotropic mastocytic conjunctivitis, which was previously recognized in the laboratory as conjunctival papillary mastocytosis. 4 The remaining 8 cats had a conjunctival mast cell tumor and were excluded from this study. Signalment and information regarding clinical presentation were available for all cats. Diagnostic tests performed by commercial laboratory services included complete blood count (7 cats) and serum biochemistry panels (6 cats); polymerase chain reaction (PCR) testing for feline calicivirus, Chlamydia felis, feline herpesvirus 1 (FHV-1), Bordetella bronchiseptica, Mycoplasma haemofelis, and H1N1 influenza virus (2 cats); PCR testing of whole blood and nasal swab for FHV-1 (1 cat); PCR testing for C. felis, Mycoplasma spp., and FHV-1 (1 cat); serology for Bartonella spp. (1 cat); and corneal culture and sensitivity (1 cat). Conjunctival samples had been previously submitted to other laboratories for histopathologic (4 cats) or cytologic (2 cats) evaluation; in 4 cats cytologic evaluation was performed by the veterinary ophthalmologist. Histological sections were routinely prepared and stained with hematoxylin and eosin as well as Giemsa or toluidine blue. Two board-certified veterinary pathologists (R.R.D. and L.B.C.T.) examined and recorded histologic features for each histological specimen. Follow-up information and medical records were available for 14 of 15 cats.
Polymerase Chain Reaction for FHV-1
Five 20-µm, formalin-fixed, paraffin-embedded tissue scrolls were cut from each of the paraffin blocks of ocular tissues from 10 cats. A new microtome blade was used for each specimen to prevent cross contamination. Scrolls were submitted to a laboratory (Real-time PCR Research and Diagnostics Core Facility, University of California–Davis, Davis, California) for PCR amplification of FHV-1 DNA (Supplemental methods).
Results
Study Population
Fifteen cats met all inclusion criteria. Their median age was 8 years (range: 7 months–17.5 years). Eleven cats were of domestic breeds and the remaining 4 included one each of the following breeds: Maine coon, Siamese, Himalayan, and Donskoy. Seven cats were neutered males and 8 were spayed females. Tissues were submitted from 8 geographically dispersed states of the USA, 1 tissue was submitted from Canada, and 1 from Sweden.
Clinical and Clinicopathologic Features
Submitting veterinarians described proliferative conjunctival lesions in 3 cats (Figs. 1–2), a solitary nodular conjunctival lesion in 10 cats (Fig. 3), or diffuse variably severe keratoconjunctivitis in 2 cats (Fig. 4). Information regarding lesion duration prior to biopsy was available for 12 cats; median reported duration was 4 months (range: 1 week–3 years). Conjunctival disease was unilateral in 12 of 15 cats. The conjunctiva of the nictitating membrane was affected in 10 cats; in 2 of these cats there was involvement of both the nictitating membrane and either the palpebral or the bulbar conjunctiva. In 1 cat only the palpebral conjunctiva was affected, in 2 cats only the bulbar conjunctiva was affected, and in 2 cats both the palpebral and bulbar conjunctiva were affected. Sixteen ocular specimens were submitted from 15 cats. Tissues submitted included 10 incisional biopsies of the lesion, 2 exenterated orbital contents (including globe, nictitating membrane, and extraocular tissue), and 4 resected nictitating membranes (1 partial and 3 complete). Surgical biopsies were performed with local anesthesia, sedation, or general anesthesia.
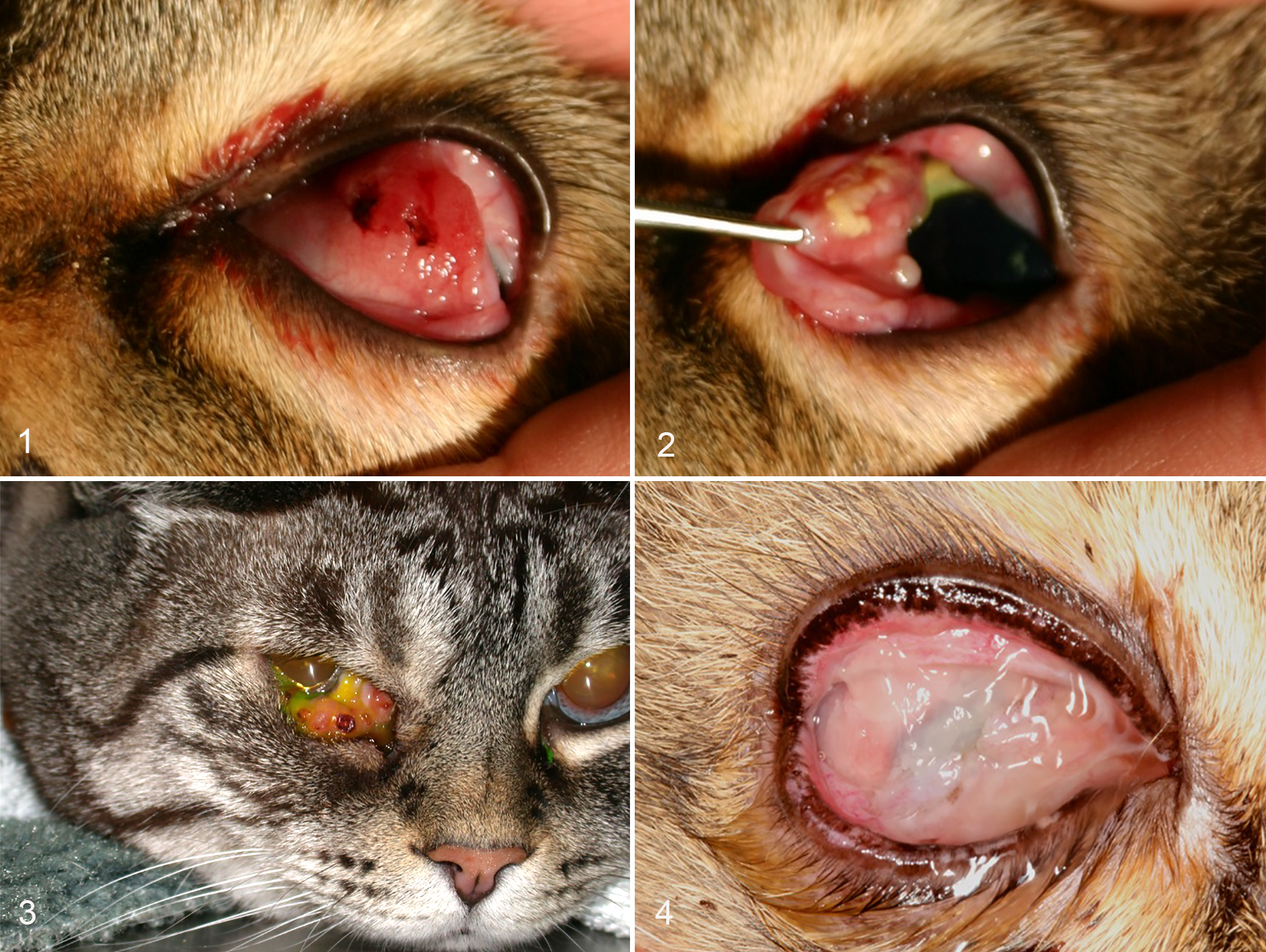
Coexisting corneal lesions were present in 10 of 15 cats; most were suspected by the clinician to be associated with FHV-1. Three cats had suspected coexisting eosinophilic keratitis, and in 3 cats the facial skin or eyelids were involved (Supplemental Table 1).
Complete blood count results were unremarkable (5 cats) or revealed a stress leukogram (2 cats). Abnormalities in serum biochemistry included hyperglycemia (1 cat, previously diagnosed with diabetes mellitus) or mildly elevated serum globulins (1 cat); serum biochemistry results were unremarkable in 4 cats. The PCR results for a feline calicivirus, C. felis, FHV-1, B. bronchiseptica, M. haemofelis, and H1N1 influenza virus panel (2 cats); FHV-1 performed on whole blood and nasal swab (1 cat); and a C. felis, Mycoplasma spp., and FHV-1 panel (1 cat) were all negative. Serology for Bartonella spp. was strongly positive (1 cat). Corneal culture and sensitivity from a melting corneal ulcer grew Mycoplasma spp. in 1 cat (Supplemental Table 1).
Before and following surgical biopsy, various medications were administered including topically applied antibiotics, corticosteroids, tacrolimus, nalbuphine, antiviral medication, antihistamines, and orally administered famciclovir and
Histopathologic diagnoses for the 4 previous biopsies submitted to another pathologist were suspected of carcinoma in situ (1 cat), allergic conjunctivitis with reactive mast cells (1 cat), eosinophilic conjunctivitis (1 cat), and eosinophilic and neutrophilic conjunctivitis (1 cat). The cytologic diagnosis for 2 samples submitted to a laboratory included marked eosinophilic and neutrophilic inflammation (1 cat) and possible early squamous cell carcinoma (1 cat). Four cytologic assessments performed by the ophthalmologist included neutrophils and monocytes (1 cat); white blood cells, erythrocytes, and eosinophils (1 cat); eosinophils and round cells (1 cat); and large amount of mast cells and epithelial cells (1 cat).
Follow-up information was available for 14 cats. Of these, no recurrence was reported in 8 cats, with a median follow-up period of 11.5 months (range: 4.5–30 months). The 2 cats that underwent orbital exenteration at the time of surgical biopsy had no evidence of recurrence 2 weeks and 21 months following surgery. In 2 cats, recurrence occurred within 1 and 2 months and required long-term treatment with topical administration of tacrolimus or corticosteroid to control recrudescence. One cat had recurrence following 8 months which was treated with further surgical debulking and topical steroids; no additional follow-up information was available for this cat. One cat (Fig. 4) had diffuse conjunctivitis, and recurrence was deemed irrelevant following an incisional biopsy; this cat exhibited significant clinical improvement after 1 month of treatment with tacrolimus. Two cats had similar lesions in the contralateral eye 1 and 2 years following initial diagnosis. Recurrence was not noted in 4 cats which underwent adjunctive cryotherapy, despite incomplete surgical margins in 3 of 4 of these cats. Three cats with no recurrence of the conjunctival mass lesions continued to experience chronic corneal disease, despite medical management (Supplemental Table 1).
Microscopic Features
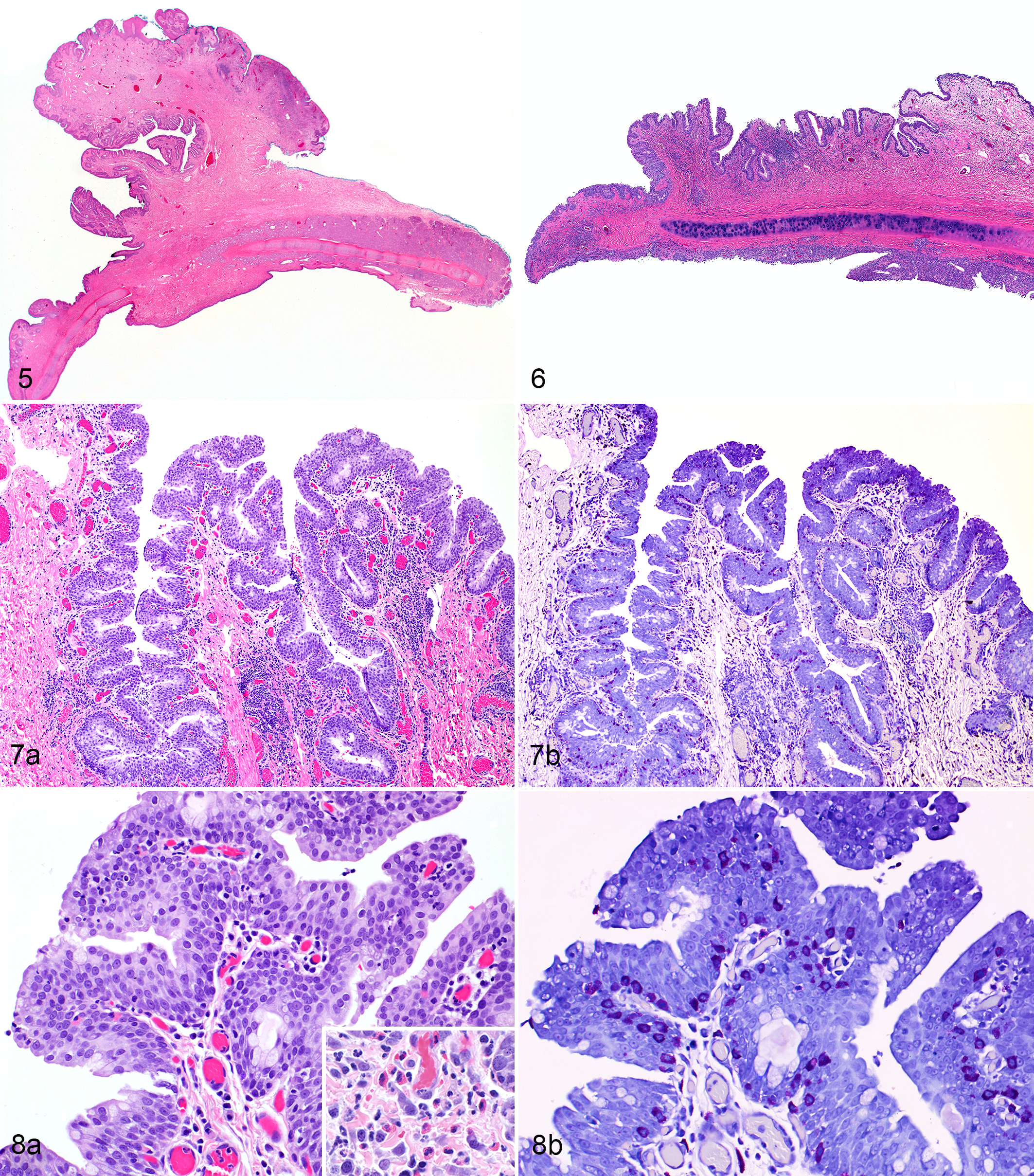
In all 15 cats, the lesions were histologically characterized by moderate to severe hyperplasia of the conjunctival epithelium with irregular proliferative projections; the projections were exophytic and papilliferous in 11 cats (Fig. 5) and sessile in 4 cats (Fig. 6). In all 15 cats, the conjunctival substantia propria demonstrated moderate to marked edema and was infiltrated by lymphocytes, plasma cells, eosinophils, and fewer neutrophils (Figs. 7–8). In all cats, there were large numbers of well-differentiated mast cells dispersed throughout the substantia propria and within the conjunctival epithelium (Figs. 7–8). Mast cells appeared as a well-differentiated population of round cells with basophilic to amphophilic granular cytoplasm, with metachromatic granules that were stained by toluidine blue, Giemsa, or both (Figs. 7b, 8b). At times, intraepithelial mast cells were visualized only following staining with toluidine blue or Giemsa. No histological evidence of malignancy was present in the mast cell population. Five biopsies were submitted with complete margins, including the 2 orbital exenterations and the 3 complete nictitating membrane resections. All incisional biopsies and 1 partial nictitating membrane resection had incomplete margins.
Polymerase Chain Reaction
Of the formalin-fixed, paraffin-embedded tissue scrolls submitted from 10 cats for FHV-1 PCR testing, 9 had sufficient DNA for PCR testing. Feline herpesvirus 1 DNA was demonstrated in 1 of 9 cats.
Discussion
The present retrospective series describes for the first time a condition with distinct clinical and histopathological appearance that we recommend be called feline epitheliotropic mastocytic conjunctivitis. Although the etiology of this condition is unknown, it has several similarities to vernal conjunctivitis in humans: Both conditions affect the conjunctiva and frequently present with papilliferous regions; moreover, both conditions commonly occur in conjunction with corneal ulceration or keratitis. 11 The etiology of vernal keratitis remains unclear; however, seasonal allergies and genetics appear to play crucial roles. 11 The frequent involvement of the nictitating membrane in cats of the present study could suggest that exposure to allergens or foreign material is important in the pathogenesis, as has been suggested in vernal conjunctivitis or giant papillary conjunctivitis. 10,11 The shape and location of the nictitating membrane in the cat may allow it to trap foreign material and allergens and could potentially prolong exposure to such substances.
The majority of cats in the current study had proliferative conjunctival inflammation which, clinically, was believed to be associated with FHV-1 infection. In addition, 3 cats had concurrent eosinophilic keratoconjunctivitis which has also been associated with FHV-1. 3 Likewise, FHV-1 has been implicated in eosinophilic and mastocytic facial dermatitis in cats 6,20 and cheetahs, 16 and these have sometimes involved the eye. However, it remains unclear in these patients whether the virus and eosinophilic infiltrate are causally associated, and, if so, which is the inciting cause. Regardless of whether FHV-1 is present, mast cell degranulation and subsequent self-trauma likely play a role in the pathogenesis of eosinophilic conjunctival inflammation and concurrent corneal disease. The presence of FHV-1 DNA was assessed using PCR assays conducted on blood or nasal swabs (4 tests in 2 cats) or on formalin-fixed biopsy tissue (10 cats). By contrast, in previous studies, FHV-1 DNA was detected in 10%–27% of cats with conjunctivitis. 7 Therefore, detection of FHV-1 DNA in only 1 of 10 cats tested on 14 occasions in the present study does not strongly support a role for FHV-1 in feline epitheliotropic mastocytic conjunctivitis. However, it is possible that negative test results could be due to previously administered antiviral medications, intermittent shedding, testing at a more chronic phase of disease, or poor nucleic acid quality in the formalin-fixed, paraffin-embedded tissue samples.
Despite some similarities with eosinophilic keratoconjunctivitis of cats and eosinophilic and mastocytic facial dermatitis in cats 6,20 and cheetahs, 16 the morphological and histopathological appearance of feline epitheliotropic mastocytic conjunctivitis differs notably from these previously described conditions. While the diagnosis of feline eosinophilic keratoconjunctivitis relies on the presence of an eosinophilic infiltrate, the patients presented herein had variable eosinophilic inflammatory infiltrates. Moreover, feline epitheliotropic mastocytic conjunctivitis is characterized by the presence of mast cells within the conjunctival epithelium, a feature that has not been described previously in association with eosinophilic keratoconjunctivitis or eosinophilic conjunctivitis. 1,3 Nonetheless, mast cells have been described within the corneal epithelium of cats with feline eosinophilic keratitis. 21
Cytologic evaluation from these patients does not seem to be sufficient for the diagnosis of feline epitheliotropic mastocytic conjunctivitis, as similar findings are seen in feline eosinophilic conjunctivitis or keratitis, conjunctival mast cell tumors, or other causes of feline conjunctivitis (eg, C. felis, M. haemofelis). 1,3,9 Due to cytologic similarities between feline eosinophilic conjunctivitis and feline epitheliotropic mastocytic conjunctivitis, the authors considered the possibility that feline epitheliotropic mastocytic conjunctivitis may be an uncommonly severe variant of feline eosinophilic conjunctivitis. In humans, vernal conjunctivitis varies in severity and may range from mild hyperemia and chemosis, to cobblestone-like papillae, or to large papillary conjunctival lesions in severe cases. 8,10,11 Moreover, both eosinophilic conjunctivitis and feline epitheliotropic mastocytic conjunctivitis appear to respond to topical anti-inflammatory treatment and do not have a clear underlying cause. 1
Although follow-up information was variable for the study population, it appears that in most cases prognosis for survival was excellent and that recurrence was uncommon with combined surgical and medical treatment. The wide array of treatments prescribed makes it difficult to comment upon preferred protocols; however, recurrences in 2 cats were reasonably controlled with long-term topical therapy with tacrolimus or corticosteroid. Immunomodulating therapy (eg, cyclosporine, tacrolimus) is also an effective way of managing human vernal conjunctivitis 8,11 and proliferative or eosinophilic keratitis in cats 22 and is typically not associated with the adverse effects seen with long-term topical treatment with corticosteroids. Moreover, corticosteroids are often contraindicated when FHV-1 infection is suspected. 22 It is possible that tacrolimus or cyclosporine could be effective as sole therapies rather than as an adjunct to surgical excision for feline epitheliotropic mastocytic conjunctivitis; however, further data would be needed to evaluate medical management alone. Nonetheless, a diagnostic biopsy is necessary in order to differentiate this condition from neoplasia.
Conjunctival mast cell tumors in dogs 5 and periocular mast cell tumors in cats 15 have a relatively good prognosis and low recurrence rates even when excision is incomplete. Mast cell tumors in general are less aggressive in cats compared to dogs, with affected cats usually having excellent survival rates. 14 Mast cells examined in specimens from the present series showed no evidence of malignancy, and, in general, the lesions did not resemble solitary mast cell tumors. Therefore, we believe that feline epitheliotropic mastocytic conjunctivitis is likely a benign condition of unknown etiology, which should not be confused with mast cell tumors or other neoplasms. Both medical and surgical management appear to be effective means of care in this condition.
Footnotes
Acknowledgements
The authors thank Dr. Barbara Easton, Dr. Matthew Chandler, and Dr. Matthew Fife for providing the clinical images shown in this case series.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
