Abstract
Objectives
The aim of this study was to determine famciclovir content (strength) in compounded formulations and to determine if potency changed over time.
Methods
Four concentrations of oral oil suspension in three distinct flavors, three concentrations of oral paste, three chew treats and 62.5 mg tablets from one compounding pharmacy were evaluated for famciclovir content. Specific sample preparation procedures were used for each drug formulation prior to determination of famciclovir content through mass spectrometry tandem liquid chromatography. Analysis was performed on arrival from the compounder and on days 7, 14, 28, 56 and 120. Samples were run in triplicate and concentration determined by comparison with a standard curve. Content was considered appropriate if within 90–110% of the labeled concentration.
Results
On arrival from the compounding pharmacy, 5/12 oral oil suspensions of varying concentrations were <90% of the labeled concentration and 3/3 oral pastes were >110%. Famciclovir content in oil suspensions ranged from 72% to 118% of the label value while oral pastes ranged from 95% to 202% of the label concentration over the 120 study days, and all concentrations varied in an unpredictable fashion. Tablets contained 90–110% of the labeled value throughout the study period. Chew treats could not be successfully analyzed.
Conclusions and relevance
This study found substantial variation in famciclovir content in the compounded products evaluated, which, in turn, raises concerns that substandard dosing could result in lack of efficacy or a failed treatment trial. Drug toxicity might also be encountered. Veterinarians must be aware that while compounded medications can improve compliance, they might not deliver the drug dose expected.
Introduction
Feline herpesvirus-1 (FHV-1) is a pathogen with worldwide significance in the cat population. Clinical conditions associated with this virus include acute, chronic and recurrent ulcerative keratoconjunctivitis, blinding corneal disease, acute rhinitis, symblepharon, ulcerative dermatitis and, potentially, corneal sequestrum, uveitis and eosinophilic keratoconjunctivitis in domestic and exotic felids.1–4 Limited mortality is reported except in shelter populations where chronically affected cats are deemed unadoptable; however, morbidity is substantial owing to viral persistence in the population associated with viral latency and recrudescence of infection.
Famciclovir is an antiviral medication that has revolutionized control of ocular and dermatologic diseases associated with FHV-1 infection. Pharmacokinetic studies have demonstrated complex metabolism in the cat,5–8 and a multitude of dosing regimens have been used clinically.9,10 Most recently a dose of 90 mg/kg PO q12h has been recommended to ensure appropriate concentrations in the tears and plasma. 8 While commercially available tablet sizes (125 mg, 250 mg and 500 mg) are somewhat appropriate for use in adult cats of typical body weights, smaller cats and kittens are more difficult to dose. Because of the difficulties in giving medications to cats, famciclovir is often compounded into differing doses or concentrations in a multitude of formulations, such as tablets, capsules, oral suspensions, chewable treats and transdermal pastes. While it is a requirement that these products are made from the US Food and Drug Administration (FDA)-approved form of the drug, these compounded preparations are not evaluated by the FDA for efficacy, content or stability. In addition, compounded drugs have been demonstrated to differ in potency and stability when compared with commercially available medications,11–14 which can result in toxicity or lack of efficacy.
Drug content in compounded medications is a primary consideration, and the acceptable range is considered to be 90–110% of the labeled concentration. 15 Another concern is drug stability. A 100 mg/ml oral suspension of famciclovir in a 1:1 mixture of Ora-Plus and Ora-Sweet SF (Perrigo Laboratories) was reportedly stable for 3 months when maintained at 2–8°C or room temperature. 16 However, commercially available oral oil suspensions range in concentration from 15 to 400 mg/ml and are supplied in a variety of flavored vehicles. Oral pastes have a similar wide range in concentrations and formulations. This study was performed to assess famciclovir content in various compounded formulations on delivery from a single compounding pharmacy. We also repeatedly assessed drug content over a 4-month time frame. We hypothesized that famciclovir content in formulations compounded into oral oil suspension, oral paste, tablets and chew treats would be between 90% and 110% of the labeled concentration on receipt and would remain stable over the 4-month test period.
Materials and methods
Drug formulations
Famciclovir was purchased for office use as legally allowed by the state of California. Multiple concentrations and formulations were obtained from one of the top four commercial 503a compounding pharmacies used by our hospital services. The pharmacy is in compliance with the US Pharmacopoeia (USP) 795 and 797 standards for non-sterile and sterile compounding, has earned the Seal of Accreditation from the Pharmacy Compounding Accreditation Board (PCAB) and purchases pharmaceutical-grade chemicals from FDA-registered suppliers. Accreditation from PCAB requires an application fee, on-site review of education, training, policies, procedures, licensure and quality assurance, and pre- and post-survey evaluation with submission to an accreditation committee for approval.The study of drug levels was not discussed with the pharmacy a priori, in order to obtain the same product that would typically be supplied to owners. Oral oil suspensions of 25, 125, 200 and 400 mg/ml concentrations were purchased in unflavored, anchovy flavored and tutti frutti forms. Fish flavored oral paste was obtained in 125, 250 and 400 mg/ml concentrations. Unflavored tablets (62.5 mg) and chew treats (31.25, 75, 100 and 250 mg/treat) were also purchased.Immediately upon arrival, temperature within the package was assessed for each shipment to ensure compliance with recommended shipping temperature, and the package and drug container were inspected for any overt damage. Upon arrival at the laboratory, 1 ml of each concentration and each flavor of the oil suspensions was aliquoted into six separate vials (for analysis on day 0 to day 120). Each vial was sealed and stored in a dark drawer at room temperature until analysis (as recommended by the compounding pharmacy). Other formulations (paste and tablets) were also stored and handled according to the directions provided by the compounding pharmacy. The concentration of each compound was tested in triplicate as detailed below on the day of arrival (day 0), and on days 7, 14, 28, 56 and 120 after receipt.
Concentration determination
Famciclovir working solutions were prepared by dilution of a famciclovir stock solution (AK Scientific) with methanol to a concentration of 100 ng/µl and calibrators prepared by dilution of the working standard solution to concentrations ranging between 0.5 and 6 ng/µl. A USP-certified analytical standard was not used in this study; however, the Certificate of Analysis on the product used confirmed that the product was 98.3% pure, based on high performance liquid chromatography. Calibration curves were prepared fresh for each quantitative assay. In addition, quality control samples (samples containing analyte at a concentration within the standard curve) were included with each sample set as an additional check of accuracy. The sample preparation protocol for each formulation was as described below.
Oil suspensions
On each day of analysis, sample preparation was performed on a single aliquot of each flavor and concentration. Each aliquot was prepared by serially diluting the oil suspension three times. The first two dilutions were with acetone and the final with 1:1 acetone:water.
Pastes
On the day of analysis, 1 ml was removed from each tube and serially diluted three times. The first dilution was with methylene chloride, the second with acetone and the final with 1:1 acetone:water.
Unflavored tablets
Tablets (62.5 mg) were prepared for analysis by crushing the tablet in 10 ml 1:1 acetone:water, followed by rotation for 15 mins (40 rpm), sonication at room temperature for 15 mins, and an additional 15 mins of rotation and centrifugation to sediment any particulates. The tablets were then serially diluted twice with 1:1 acetone:water. No particulate matter was visible after sample preparation.
Chew treats
Multiple sample preparation methods were attempted without success. The initial protocol involved dissolving a chew treat in 10 ml of 100% methanol and crushing the chew with a metal rod. The tube was rotated for 10 mins, sonicated for 5 mins at room temperature and then centrifuged. The protocol was subsequently altered to include longer rotation, sonication and centrifugation, use of different solvents and incorporation of two freeze–thaw cycles. Finally, to investigate a possible reason for the failure of these experiments, blank treats containing no famciclovir were obtained from the compounding pharmacy, and known concentrations of famciclovir were added prior to sample preparation and analysis. A second method involved placing a macerated blank chew treat in a tube, adding 0.1 ml of drug and mixing it until dried.
Liquid chromatography tandem-mass spectrometry
The concentration of famciclovir was measured in the oil suspension, paste and tablet formulations by liquid chromatography tandem–mass spectrometry (LC-MS/MS). For each formulation, 10 µl of the final diluents were injected into the LC-MS/MS system. Quantitative analysis was performed on a TSQ Vantage triple quadrupole mass spectrometer (Thermo Scientific) coupled with a turbulent flow chromatography system (Thermo Scientific) having 1100 series liquid chromatography systems (Agilent Technologies) and operated in laminar flow mode. The spray voltage was 3501 V, the vaporizer temperature was 300°C, and the sheath and auxiliary gas were 45 and 30 arbitrary units, respectively. Product masses and collision energies of each analyte were optimized by infusing the standards into the mass spectrometer. Chromatography employed an ACE 3C18 column (Mac-Mod Analytical) and a linear gradient of acetonitrile in water with a constant 0.2% formic acid at a flow rate of 0.4 ml/min. The initial acetonitrile concentration was held at 5% for 0.67 mins, ramped to 90% over 6 mins, and held at that concentration for 0.16 mins before re-equilibrating for 4 mins at initial conditions. Detection and quantification were conducted using selective reaction monitoring of the initial precursor ion for famciclovir (mass to charge ratio 322 [m/z]). The responses for the product ions for famciclovir (136, 262 and 280 m/z) were plotted and peaks at the proper retention time integrated using Quanbrowser software (Thermo Scientific). Quanbrowser software was used to generate calibration curves and quantitate famciclovir in all samples by linear regression analysis. A weighting factor of 1/x was used for all calibration curves. The LC-MS/MS instrument response for famciclovir was linear and gave correlation coefficients of 0.99 or better.
Famciclovir concentrations were determined in triplicate by LC-MS/MS. The average of the three values was compared with the concentration listed on the compounding pharmacy’s label to determine the percentage of the nominal concentration in each formulation. Per USP standards, 15 famciclovir concentrations of 90–110% of the labeled values were considered within the acceptable range.
Results
All packages were shipped by standard US mail without ice packs, and median temperature on arrival was 22.5°C (range 22–26°C). For all formulations, containers were sealed on arrival and there was no visual or olfactory evidence of leakage of material from any container.
Three flavors (unflavored, anchovy, tutti frutti) of four concentrations of famciclovir in oral oil suspension were obtained. Liquid chromatography/mass spectrometry yielded a range of results for famciclovir content in each concentration and flavor on each day of assessment (Figure 1). On arrival from the compounding pharmacy, 3/3 oral oil suspensions of famciclovir in the 25 mg/ml concentration and 2/3 oral oil suspensions in the 200 mg/ml concentration were <90% of the labelled concentration. All 125 mg/ml oral suspensions were within an acceptable concentration range, as were 2/3 oil suspensions of 400 mg/ml.

Famciclovir content as determined by liquid chromatography/mass spectrometry in oil suspensions of (a) 25 mg/ml, (b) 125 mg/ml, (c) 200 mg/ml and (d) 400 mg/ml for unflavored (black), anchovy (white) and tutti frutti (gray) flavors on days 0, 7, 14, 28, 56 and 120 of experimentation. Data are presented as the percent famciclovir in the compounded product compared with the label concentration
On days 7 and 14, 4/12 (33%) suspensions of all concentrations and flavors were below 90% of the acceptable level and on day 28, 8/12 (67%) were <90% of the labeled concentration (Table 1). Overall, throughout the 4-month testing period, measurement of famciclovir content in the 25 mg/ml and the 200 mg/ml oral oil suspensions had the highest number (10 and 12/18 samples, respectively) below the acceptable concentration range, while the 400 mg/ml oral oil suspension had the highest number of values within the acceptable range when comparing measured-to-labeled concentrations (n = 15/18; 83%).
Proportion of samples in which measured famciclovir content fell outside 90–110% of stated concentration for 25, 125, 200 and 400 mg/ml famciclovir oil suspension (without flavoring, or flavored with anchovy or tutti frutti), as assessed on receipt and on specific days after arrival

Data are n (%)
Paste formulations demonstrated a broad range of measured relative to labeled famciclovir concentrations at each time point. On arrival, 2/3 concentrations were approximately double the labeled value (Figure 2). On subsequent days, the drug formulation within the tube was mixed by massaging the contents back and forth prior to aliquoting for analysis. However, on days 7 and 28, all three paste concentrations still exceeded the stated content of famciclovir. One of three concentrations (400 mg/ml) was >110% of the label content on day 56, and the 250 mg/ml paste was 112% of the labelled concentration at 4 months after receipt.
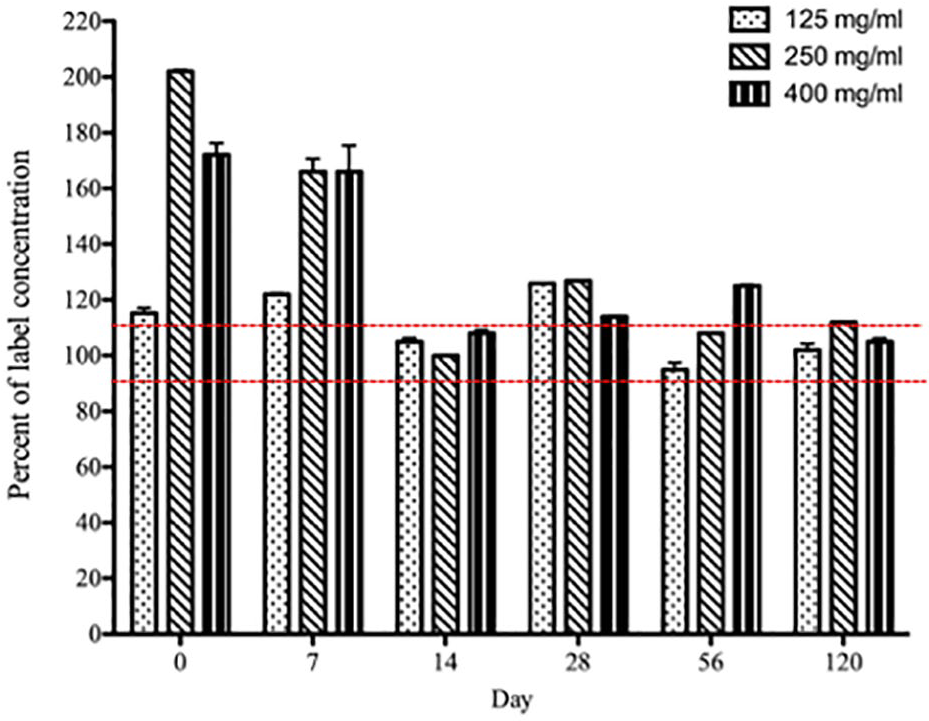
Famciclovir content as determined by liquid chromatography/mass spectrometry in compounded paste formulations of 125 (open bars), 250 (cross hatched bars) and 400 (black bars) mg/ml famciclovir on experimental days 0, 7, 14, 28, 56 and 120. Data are presented as the percent famciclovir in the compounded product vs the label concentration
Famciclovir content in 62.5 mg compounded tablets remained stable and within 90–110% of the labeled concentration throughout the study (Figure 3).
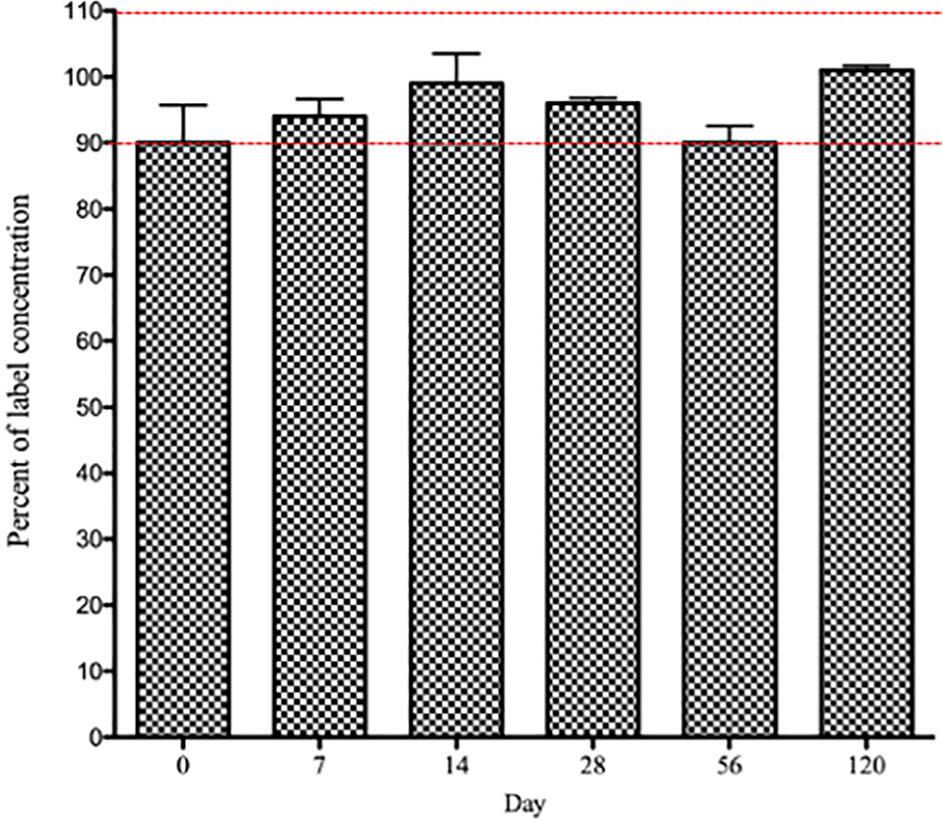
Measured famciclovir content in compounded famciclovir tablets was 90–110% of the reported concentration of 62.5 mg at all time points
The original protocol for sample preparation and analysis of famciclovir in chew treats yielded a content of each chew calculated to be <5 mg in tablets labeled with drug amounts ranging from 31.25 to 100 mg. Analysis of blank chew treats to which a stock solution of famciclovir was added achieved a maximum of 72% recovery.
Discussion
Results of this study demonstrated out-of-range variation in famciclovir content on arrival for 9/16 formulations (56%) of compounded medications. Compounded drugs were obtained from a single pharmacy and comprised four concentrations of oral oil suspensions in three different flavors (12 total oil suspensions), three paste formulations and one tablet. Sample preparation and analysis of famciclovir from three strengths of chew treats proved problematic and further evaluation is needed to determine the clinical utility of this product. Compounded tablets (62.5 mg) had the most reliable drug concentrations, with famciclovir content consistently between 90% and 110% of the labeled concentrations from arrival to the end of the study. Day 0 famciclovir content in oral oil suspensions of 25 mg/ml and 200 mg/ml were below the acceptable concentration range in 5/6 assays and provided the least predictable drug concentrations over the 4-month study period, with over half of the values below the acceptable limit. The lower concentration suspension (25 mg/ml) is likely targeted for use in kittens aged 1–2 months, or approximately 0.5–1 kg; 17 however, that concentration is too low for ease in administration at the recommended dose of 30–40 mg/kg q8h or 90 mg/kg q12h. 8 Instead, the 62.5 mg tablets had stable concentrations of famciclovir detected and would be more suitable for use in kittens of this size or age.
The compounding pharmacy from which famciclovir was obtained for this study is one of the more commonly used in our area, and it shares similar description of methodology with many other large-scale compounders. We elected not to identify the specific compounding pharmacy used in this study because it was considered imprudent to potentially enhance or impugn the reputation of a pharmacy based on a single study. We understand that the source of our medication could be of interest to practicing veterinarians; however, we suspect that many of the results obtained here might be found in medications from other compounding pharmacies, despite adherence to best practices. Comparative studies involving other products are lacking and the point of this study was to highlight potential pitfalls in the use of compounded medications in general. Compounding pharmacies do not typically reveal specific manufacturing processes and it would be difficult to determine whether the results of our study were related to the methodology of compounding or, perhaps, due to receipt of a substandard drug used in the compounding process. Clearly, caution is warranted whenever using a product that has not been FDA approved.
There are many features involved in compounding that can result in lower than normal drug concentrations in a formulation, including contamination by microorganisms, degradation by hydrolysis or oxidation, and interactions with additives used in the compounding process. 18 Use of invalidated methods or failure to adhere to USP standards for compounding could also contribute. Causes for higher than expected drug concentrations are more difficult to explain. Traditional compounding pharmacies (503a pharmacies), which compound prescriptions for specific patients for at-home use, have little oversight from the FDA and the level of state oversight varies greatly. These pharmacies are largely expected to self-regulate with internal quality control measures and standard operating procedures. However, 503a pharmacies are not required to test each batch of final product prior to releasing it. This can result in significant batch-to-batch variation for compounded products vs FDA-approved products or those compounded by 503b pharmacies, which are permitted to make large batches of drugs for subsequent distribution. The 503b pharmacies prepare drugs in a method that aligns with manufacturing and requires verification of the strength of the drug prior to releasing a batch, whereas 503a pharmacies are not held to that standard. Therefore, both drug failures and intoxications can occur with compounded medications.11,13,14,19
Cats are particularly challenging to medicate and can be more accepting of liquids, pills, pastes or suspensions depending on the individual; however, it is critical that information is available on the reliability of drug content in such formulations. Doxycycline concentration in a compounded suspension markedly decreased from 90–100% to only 20% 14 days after formulation, 12 which would result in ineffective plasma concentrations These concerns could be amplified when two or more drugs are compounded into a single product. Doxycycline compounded with famciclovir for the management of feline upper respiratory tract disease could be particularly problematic owing to instability of doxycycline, 12 as well as potential concerns about the antiviral component described here.
There are few studies validating the multiplicity of compounded products available on the market.12,20,21 The flavorings evaluated in this report did not result in a statistically evident impact on famciclovir content; however, there are virtually endless combinations of flavors, drug concentrations and formulations available for purchase. This study examined products from a single pharmacy and, undoubtedly, given the multiplicity of compounding pharmacies that can be accessed by veterinarians, there are endless concentration studies that could be considered. Analysis of the oral paste obtained from the compounding pharmacy yielded very high famciclovir concentrations initially, and subsequent experiments suggested separation of the drug from the vehicle. Further evaluation of this phenomenon would be helpful, including sampling the ends of the tube to determine if a gradient of concentrations might be present within the delivery device. While mixing of the paste seemed to modulate a portion of the out-of-range concentrations encountered, use of this formulation in the clinical setting could lead to misinterpretation of clinical results.
Variations in famciclovir drug content could be particularly problematic in clinical practice because the diagnosis of FHV-1-related disease is often based on treatment response to an antiviral medication, with famciclovir reported as the most effective treatment available.4,9,10 Placebo-controlled clinical trials often must use a modification of commercially available medications, and this is particularly true when studies involve variably sized and aged animals. Fortunately, commercially available famciclovir is readily pulverized for use in clinical trials; however, knowledge of the stability of famciclovir in compounded formulations is critical for interpreting therapeutic trials. Despite using an FDA-approved product prepared contemporaneously in individual gel caps, trials addressing respiratory disease in cats have failed to demonstrate a clinical effect in individual 22 or shelter animals, 23 although reduced viral shedding was documented in the shelter study. In addition, the pharmacokinetics of famciclovir in the cat are non-linear,5,7 and cats demonstrate incomplete absorption of the drug, which is another contributor to possible treatment failures with any drug. 24 Taken together, these studies indicate that a compounded drug formulation should not be used in the initial treatment trial or in a clinical trial, and that absorption studies of compounded products would be of value. In addition, evaluation of compounded famciclovir in a well-designed clinical trial would enhance our understanding of the utility of such drugs, which is of obvious importance due to the potential to enhance ease of administration, and thus owner and patient compliance.
Conclusions
Compounded drugs provide major benefits in the management of disease in cats by improving compliance and acceptance of drug administration; however, veterinarians need to be aware of possible pitfalls. This is particularly important in the case of famciclovir, which is invaluable in the treatment of FHV-1 yet is difficult to administer because of the dose and pill size available commercially, leading to veterinarians commonly employing compounded medications.In this analysis, substantial variation was found in diverse formulations of famciclovir obtained from a single commonly used pharmacy, with content outside the acceptable range in the majority of cases. Our results raise concerns that substandard dosing could result in lack of efficacy or a failed treatment trial, and drug toxicity might also occur. Additional investigations are needed to establish the accuracy and stability of compounded famciclovir from this and other pharmacies, and to investigate other forms of these drugs.Further, the impact of compounding on drug absorption, metabolism and pharmacodynamic responses requires additional study, given the complex metabolism of famciclovir in the cat. Oil formulations are often employed by compounding pharmacies due to the long beyond-use-date default. However, with famciclovir, as well as many other medications, there is concern about the bioavailability of medications compounded in oil formulations. Therefore, follow-up studies of famciclovir bioavailability in various formulations would add valuable details to available clinical information. Determining the bioequivalence of varied drug formulations is critical.
Footnotes
Acknowledgements
We would like to acknowledge Dr David Maggs for his review of and invaluable comments on the manuscript. We would also like to thank Ms Sheri Pendergraft, Mr Daniel McKemie and Ms Sandy Yim for their technical assistance.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors would like to thank the Student Training in Advanced Research program and the Bailey Wrigley Fund at the University of California, Davis, for funding this project.
Ethical approval
This work did not involve the use of animals and therefore ethical approval was not necessarily required.
Informed consent
This work did not involve the use of animals and therefore informed consent was not required. No animals or humans are identifiable within this publication, and therefore additional informed consent for publication was not required.
