Abstract
Objectives
Diaphragmatic dysfunction (DD) is often underdiagnosed in veterinary medicine. Various ultrasonographic diaphragmatic parameters, including diaphragmatic excursion (DE), diaphragmatic thickness (Tdi) and diaphragmatic thickening fraction (DTF), can be used to identify DD. We aimed to establish normal reference intervals in healthy cats, examine the effects of intrinsic factors on diaphragmatic parameters and evaluate the efficacy of these parameters in identifying DD in healthy and diseased cats.
Methods
This prospective observational study was conducted on 22 healthy cats and 12 diseased cats using ultrasonographic techniques to evaluate DE, Tdi and DTF. The correlation between diaphragmatic parameters and intrinsic factors, including sex, age, body weight and body condition score, was analysed using Pearson correlation analysis. Cut-off values for healthy and diseased cats were established using receiver operating characteristic curve analysis.
Results
The mean DEs of the left and right hemidiaphragms were 0.66 ± 0.16 and 0.64 ± 0.18 cm, respectively. The mean Tdis in the end-inspiration phase were 0.13 ± 0.03 and 0.12 ± 0.03 cm for the left and right hemidiaphragms, respectively, whereas in the end-expiration phase the mean Tdi was 0.07 ± 0.03 cm for both hemidiaphragms. The DTF ranges for the left and right hemidiaphragms were 23.90–122.1% and 38.80–107%, respectively. Intrinsic factors had no significant impact on DE, Tdi and DTF. The DE measurements for the left hemidiaphragm were more accurate for evaluating diaphragmatic function. To distinguish between healthy and diseased cats, the cut-off value, sensitivity and specificity were 0.458 cm (area under the curve [AUC] 0.846), 75.00% and 86.36%, respectively, for the left hemidiaphragm and 0.423 cm (AUC 0.704), 41.67% and 100%, respectively, for the right hemidiaphragm.
Conclusions and relevance
Normal reference values for DE, Tdi and DTF were established for healthy cats. DE, particularly in the left hemidiaphragm, obtained using routine ultrasonography, improves the efficacy of identifying DD and assists clinicians in the diagnosis and treatment of cats with cardiorespiratory disease.
Introduction
The diaphragm is a primary muscle essential for breathing and respiratory function. Impairment of respiratory function, known as diaphragmatic dysfunction (DD), can significantly affect breathing. Diaphragmatic excursion (DE), diaphragmatic thickness (Tdi) and diaphragmatic thickening fraction (DTF) are essential parameters reflecting diaphragmatic function. These parameters are critical to evaluating diaphragmatic function in cases of various respiratory diseases, particularly in humans, including detecting diaphragmatic paralysis caused by phrenic nerve injury,1,2 assessing cardiorespiratory diseases,1,2 monitoring critically ill patients on ventilators3–5 and guiding diaphragmatic pacing protocols.1,6,7
The diagnosis of DD typically involves a combination of clinical examination – pulmonary function tests, including spirometry,8,9 phrenic nerve stimulation tests 8 and electromyography 8 – and imaging techniques such as radiography,8–10 fluoroscopy,8–10 CT, 10 MRI 10 and ultrasonography (US).8–10 Among these methods, US stands out because it is cost-effective, easy to use, radiation-free and offers high reproducibility and repeatability.1,2 Using US for diaphragmatic assessment provides critical insights into muscle characteristics, including thickness and movement, and enables quantitative and qualitative evaluations. DD has been reported in various animal species, such as llamas, 11 alpacas, 12 ponies, 13 dogs14,15 and cats. 16 However, in confirmed cases of DD, diagnostic imaging primarily relies on DE.14–16 To better understand diaphragmatic function in an animal using DE, Tdi and DTF, various factors, including location and side of the diaphragm, sex, age, body weight (BW) and body condition score (BCS), must be considered. Therefore, understanding how these factors influence DE, Tdi and DTF in healthy cats would ensure their proper use as diagnostic and monitoring parameters in clinical practice.
Cats are susceptible to cardiorespiratory diseases that can lead to DD, such as hypertrophic cardiomyopathy, mediastinal lymphoma, feline infectious peritonitis with pleural effusion and feline asthma, which are all highly prevalent in the feline population.17–20 There is limited information on DD in healthy and diseased cats; therefore, the aims of this study were to: (1) establish the normal reference values for DD-related parameters, including DE, Tdi and DTF, in healthy cats; (2) examine the effect of BW, BCS, sex, age and diaphragmatic side on DE, Tdi and DTF values in healthy cats; and (3) explore the efficacy of DE, Tdi and DTF in distinguishing diaphragmatic function between healthy and diseased cats observed using US techniques. We hypothesised that intrinsic factors would significantly affect DE, Tdi and DTF values and that DE, Tdi and DTF would demonstrate consistent efficacy in distinguishing diaphragmatic function between healthy and diseased cats.
Materials and methods
Animals
This prospective, observational study was approved by the Institutional Animal Care and Use Committee of the Faculty of Veterinary Science at Chulalongkorn University (approval number 2231059). Written informed consent for participation was obtained from all cat owners. Adult cats aged between 8 months and 15 years with a BCS of 6–7/9 were recruited from those that visited the Diagnostic Imaging Unit at The Small Animal Hospital, Faculty of Veterinary Science, Chulalongkorn University between November 2022 and June 2023. There were no restrictions regarding the sex or gonadal status of cats. The cats were subsequently divided into healthy and diseased groups. The healthy group included cats clinically confirmed as healthy based on complete physical examinations encompassing general appearance, mental status, temperature, heart and lung sounds, mucous membrane colour, capillary refill time, complete blood count and chemistry profile, thoracic radiography and abdominal US. The diseased group included cats with pulmonary parenchymal abnormalities, such as interstitial or alveolar lung patterns, pleural effusion and cardiovascular diseases. Cats were excluded if they had a history of diseases that could interfere with diaphragmatic structure or neuromuscular function, such as diaphragmatic rupture, cervical intervertebral disease, history of diaphragmatic or cervical surgery, rib fractures and intra-abdominal abnormalities causing diaphragmatic compression, including intra-abdominal organomegaly, pregnancy, peritoneal effusion or paradoxical movements of the diaphragm.
Diaphragmatic parameter measurements
US assessments of DE, Tdi and DTF were performed by a diplomate of the Thai Board of Veterinary Surgery, a well-trained sonologist (PS) experienced in diaphragmatic examinations. All cats were fasted (no food or water) for 8 h before the examination and remained fully conscious and without sedation during the procedure. A general examination of the entire abdomen was performed in the supine position using a low-frequency microconvex transducer (7 MHz) equipped with real-time brightness (B)-mode US (Logiq P6; GE Healthcare). To obtain accurate DE, Tdi and DTF measurements, each cat was positioned until it was calm and relaxed. Cats that vocalised, exhibited aggressive behaviour or showed paradoxical diaphragmatic movements were excluded from the study because these conditions could yield unreliable results. The transducer was then positioned craniodorsally to the paramedian of each costal arch in the sagittal plane, as described by Saisawart et al 21 (Figure 1). The right hemidiaphragmatic window was established using the liver as a reference point, whereas the stomach or spleen was used for the contralateral left hemidiaphragmatic window. Subsequently, the US setting was switched from B-mode to motion (M)-mode. The M-mode plane was adjusted to be perpendicular to the diaphragmatic line to facilitate accurate evaluation of diaphragmatic movement. Normal diaphragmatic movement is characterised by motion towards the transducer during inspiration and outward motion during expiration. For the quantification of DE, a digital calliper was used to measure the difference between the peaks of inspiration and expiration on an M-mode US image (Figure 2). Three consecutive DE measurements were obtained systematically and averaged. Other parameters, including inspiration time (s), diaphragmatic contraction velocity (cm/s) and total respiratory cycle time (s), were also measured and calculated. The inspiration time was determined using M-mode US and delineated as the time interval from the onset of diaphragmatic contraction to the maximum amplitude of the inspiratory phase (Figure 2). Finally, the diaphragmatic contraction velocity was calculated by dividing the DE by the inspiration time. Tdi was evaluated using B-mode US images, obtained using a high-frequency linear array transducer (11 MHz). Data on Tdi in the end-inspiration phase (Tdi [ins]) and in the end-expiration phase (Tdi [exp]) were collected. The diaphragm was defined as the line between two parallel echogenic lines indicating the pleural and peritoneal membranes, and the distance between these lines was measured only in the hypoechoic layer (Figure 3). DTF was also evaluated and calculated as (inspiratory thickness – expiratory thickness)/end-expiratory thickness and the result was expressed as a percentage. To ensure the accuracy and repeatability of these measurements, the values were derived from at least three different breathing cycles and subsequently averaged. The same measurement technique was used to evaluate diaphragmatic parameters in both the healthy and diseased groups.
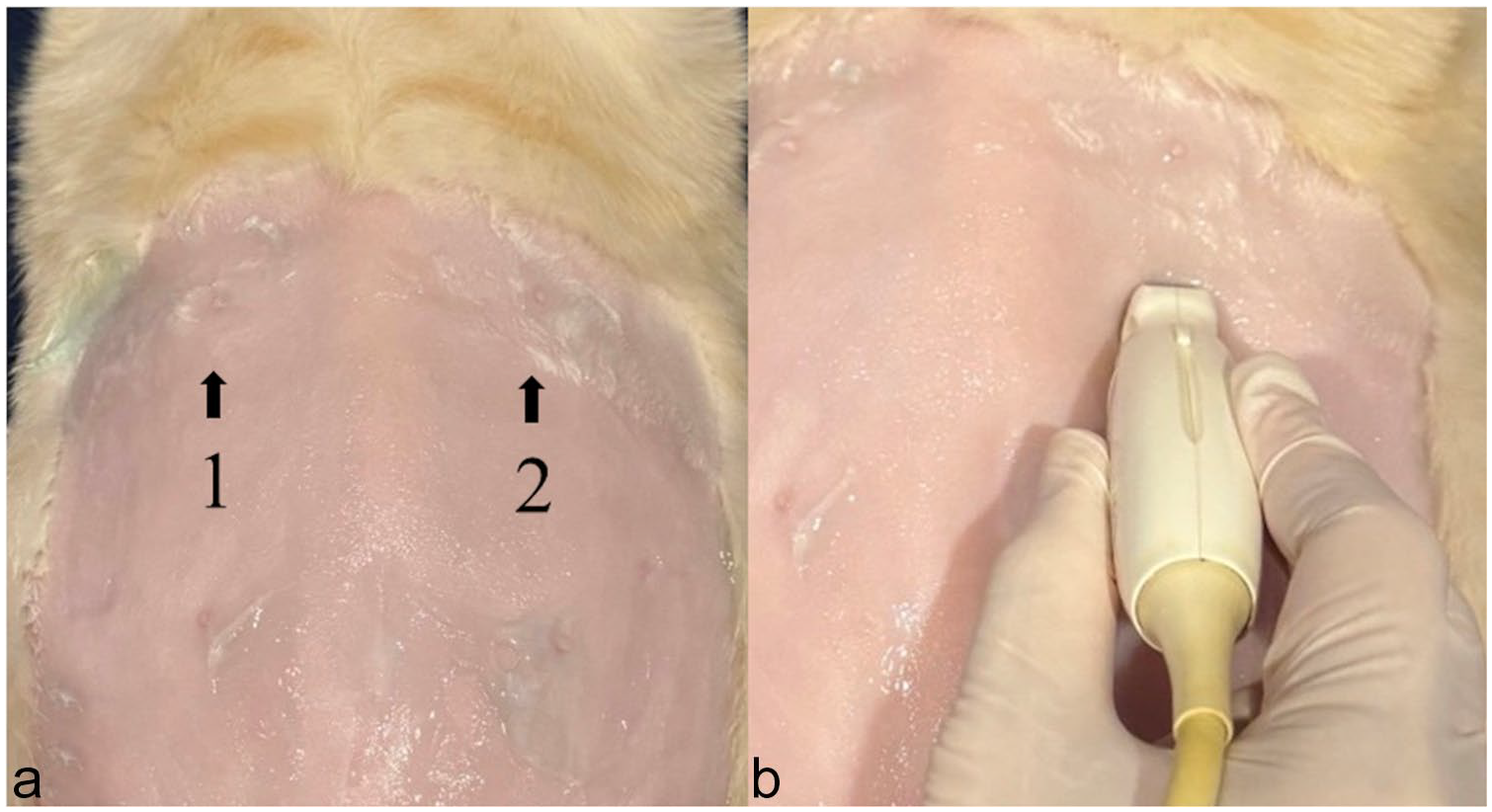
(a) Diaphragmatic location for detecting diaphragmatic excursion: (1) right diaphragmatic crus; (2) left diaphragmatic crus. (b) Location of the probe and direction for measuring diaphragmatic excursion

Measurement method for diaphragmatic excursion (d), inspiratory time (t), total inspiratory time (Ttot) and contraction velocity (Ve). The contraction velocity (cm/s) can be calculated from formula: Ve = d/t

(a) Diaphragmatic location for detecting diaphragmatic thickness: (1) the right diaphragm; (2) the left diaphragm. (b) Location and direction of the probe for measuring diaphragmatic thickness and (c) measurement method for measuring diaphragmatic thickness; 1 = diaphragmatic thickness during expiration, 2 = diaphragmatic thickness during inspiration. Diaphragmatic thickening fraction (DTF) can be calculated using the formula: DTF (%) = (inspiratory thickness − expiratory thickness)/end-expiratory thickness = (2 − 1)/1
Statistical analysis
All data were analysed using Prism8 (GraphPad Software). The normality of each data set was analysed using the Shapiro–Wilk test. The clinical data of the healthy and diseased cats were expressed using descriptive statistics, with results either presented as mean ± SD or as median (range), depending on the data distribution. The effects of age, BW and BCS on the left and right DE, Tdi and DTF values in healthy cats were assessed using Pearson’s correlation analysis. The Student’s t-test was used for comparisons between sexes and between groups. Receiver operating characteristic (ROC) curves for the healthy and diseased groups were used to calculate the cut-off values and the Youden index was used to select the best cut-off value. The 95% confidence interval (CI) and area under the ROC curve (AUC) were calculated. Statistical significance was set as P <0.05.
Results
Clinical demographic data
A total of 34 cats met the inclusion criteria and were divided into two groups: healthy (n = 22) and diseased (n = 12). Patient demographic characteristics are presented in Table 1. Diseased cats were significantly older than healthy cats (P = 0.0015). However, BW was comparable between the groups (P = 0.4708). When examining the data based on sex, diseased male cats were significantly older than healthy male cats (P = 0.0007), whereas the age of female cats was not significantly different between the groups (P = 0.1227). The healthy group comprised 11 domestic shorthair cats, five Scottish Folds and one each of the following breeds: British Shorthair, domestic Lynx, Himalayan, Persian, Sphynx and Siamese. The diseased group comprised nine domestic shorthair cats, two British Shorthairs and one American Shorthair. Diseased cats had asthma (n = 7), lung collapse (n = 2) and lung fibrosis (n = 4).
Demographic information for the 22 healthy cats and 12 diseased cats used in this study
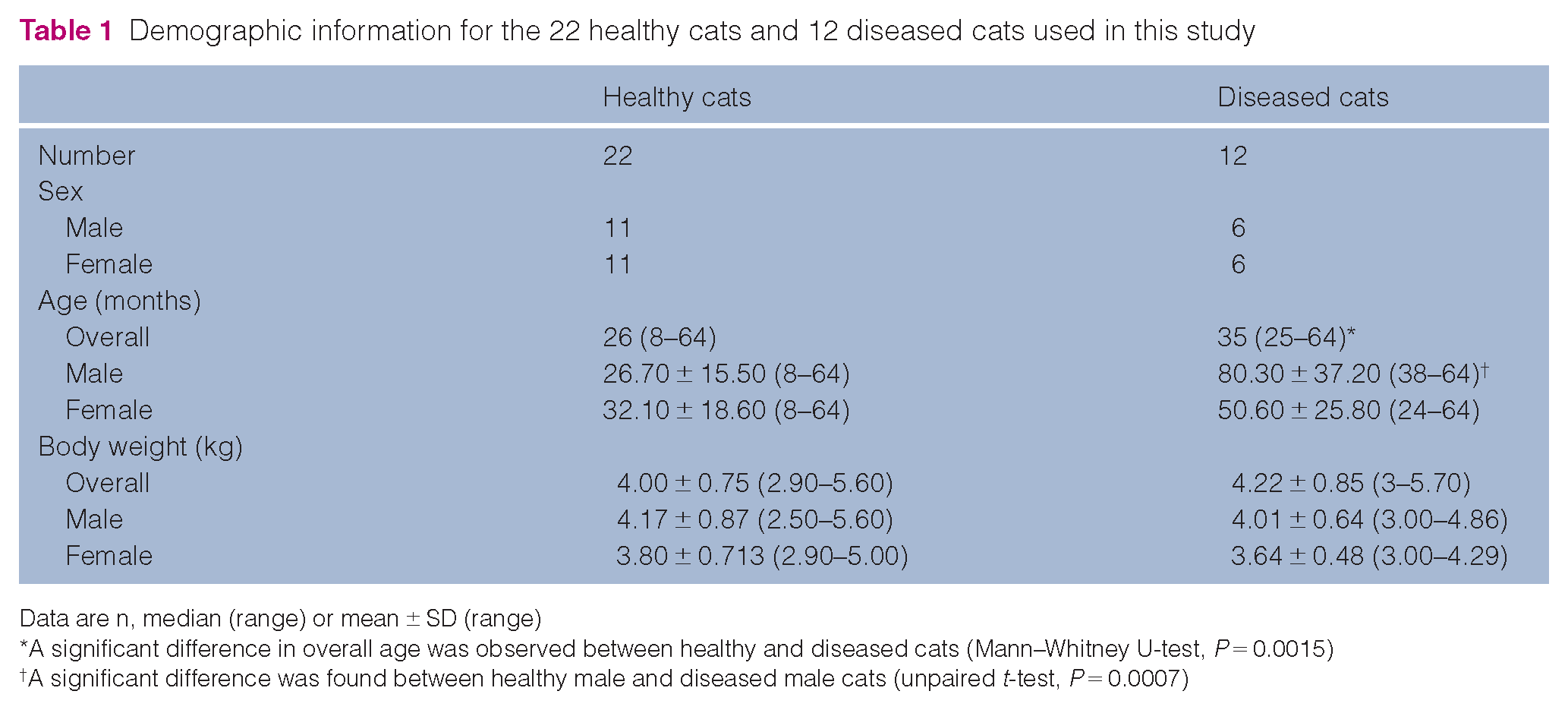
Data are n, median (range) or mean ± SD (range)
A significant difference in overall age was observed between healthy and diseased cats (Mann–Whitney U-test, P = 0.0015)
A significant difference was found between healthy male and diseased male cats (unpaired t-test, P = 0.0007)
Comparison of DE, Tdi and DTF between healthy and diseased cats
The diaphragmatic parameters of the healthy and diseased cats are presented in Table 2. DEs of the left and right hemidiaphragms were significantly higher in the healthy group than in the diseased group (P = 0.0006 and P = 0.025, respectively). There were no significant differences in the other assessed parameters. In the healthy group, intrinsic factors such as sex, age, BW and BCS were not significantly correlated with DE, Tdi and DTF. The correlation coefficients and P values are presented in Table 3
Diaphragmatic parameters for healthy and diseased cats

Data are mean ± SD (95% confidence interval)
A significant difference in DE was found between healthy and diseased cats (unpaired t-test, P = 0.025 for the right hemidiaphragm)
A significant difference in DE was found between healthy and diseased cats (unpaired t-test, P = 0.0006 for the left hemidiaphragm)
DE = diaphragmatic excursion; DTF = diaphragmatic thickening fraction; exp = expiration phase; ins = inspiration phase; Tdi = diaphragmatic thickness
Comparison of diaphragmatic correlations between the diaphragmatic parameters and sex, age, BW and BCS of healthy cats

BCS = body condition score; BW = body weight; DE = diaphragmatic excursion; DTF = diaphragmatic thickening fraction; exp = expiration phase; ins = inspiration phase; L = left; R = right; Tdi = diaphragmatic thickness
The ROC curve was used to evaluate only the DEs to differentiate the cut-off value of DE between the healthy and diseased groups. The result indicated that the cut-off values of DE between the healthy and diseased groups were 0.458 cm for the left hemidiaphragm (AUC 0.846, 95% CI 0.710–0.982, P = 0.001, sensitivity 75%, specificity 86.36%) and 0.423 cm for the right hemidiaphragm (AUC 0.704, 95% CI 0.509–0.899, P = 0.05, sensitivity 41.67%, specificity 100%) (Figure 4).

ROC curves showing the AUC and P value of significance for using diaphragmatic excursion in the diagnosis of diaphragmatic dysfunction in (a) the right hemidiaphragm and (b) left hemidiaphragm. AUC = area under the ROC curve; ROC = receiver operating characteristic
Discussion
Respiration primarily relies on the diaphragm, a vital muscle involved in respiratory processes. Diseases that impair diaphragmatic mobility can also induce respiratory dysfunction. Prolonged impairment of diaphragmatic activity results in diaphragmatic atrophy. 22 In human medicine, increasing attention has been paid to assessing DE, Tdi and DTF because of their significant role in identifying diaphragmatic abnormalities, assessing disease severity and monitoring disease progression.1,23 These evaluations are crucial and should be performed accurately using suitable techniques. These characteristics refer to the evaluation of diaphragmatic function using DE, Tdi and TDF, which should be performed accurately with appropriate techniques for measuring the diaphragmatic movements to ensure accurate interpretation. Notably, there has been limited research on DE, Tdi and DTF in veterinary medicine, particularly in cats. Conducting an in-depth study of these aspects in animals, utilising appropriate methods and accounting for individual differences could offer valuable information. Diaphragmatic information can enhance our understanding of the diaphragmatic function of cats, particularly those with cardiorespiratory diseases.
In the present study, we demonstrated the effectiveness of US in evaluating DE, Tdi and DTF. The study offers valuable insights that can be applied to further investigations in veterinary medicine. In humans, DE, Tdi and DTF are valuable parameters for assessing diaphragmatic performance, with each parameter offering unique advantages. DE is commonly used to measure the distance of diaphragmatic movement and determine its direction. Unexpected or paradoxical diaphragmatic movements can occur frequently in cases of diaphragmatic paralysis and can serve as an indicator of abnormalities. 9 Tdi is a valuable diagnostic tool for chronic cases of diaphragmatic paralysis, when the diaphragm presents as consistently thin with minimal changes over time. 24 However, in acute cases of diaphragmatic paralysis, Tdi measurements may not be sufficiently sensitive to detect immediate changes. 24 In such situations, DTF can be a more reliable indicator. DTF reflects the diaphragmatic inspiratory activity and can provide valuable anatomical and functional insights into various acute respiratory diseases. 24
When comparing the DE, Tdi and DTF parameters, DE was the most straightforward for evaluating diaphragmatic function in cats. The DE measurement method showed clear visibility of the hyperechoic line between the inspiration and expiration phases. Conversely, the Tdi measurement was more challenging owing to the characteristically thin hypoechoic line between the pleural and peritoneal lines. DTF was calculated based on the percentage difference between Tdi (inspiration) and Tdi (expiration). Therefore, measuring these parameters may also be challenging. We suggest that DE is most generally applicable to assess the diaphragm. In the present study, in evaluating diaphragmatic performance to distinguish between healthy and diseased cats, DE was the only reliable parameter for assessing DD in cats with cardiorespiratory disease. This was due to the AUC values obtained for the DE measurements, which were 0.846 and 0.704 for the left and right hemidiaphragms, respectively, indicating the diagnostic potential of DE. In particular, the left hemidiaphragm had a high AUC value, indicating the accurate identification of diaphragmatic function in cats. This level of diagnostic accuracy is crucial in detecting abnormalities associated with cardiorespiratory diseases. However, the reason the left hemidiaphragm had a higher AUC than the right hemidiaphragm remains unclear but may be due to the unique anatomical and physiological variations between species. Furthermore, the high specificity (100%) for the right hemidiaphragm indicates that this test is excellent for identifying healthy individuals; however, the test’s lower sensitivity (41.67%) suggests that it may miss many diseased cases. Conversely, the left hemidiaphragm showed a balance between sensitivity (75%) and specificity (86.36%), making it a more reliable indicator of diaphragmatic disease. These findings suggest DE has potential clinical applications in diagnosing respiratory disease-induced DD.
Diseased cats had a lower DE than healthy cats. This could be attributed to several mechanisms. Interstitial lung disease, including inflammation, scarring and fibrosis, increases lung elastic recoil and decreases diaphragmatic compliance. Therefore, cats require greater diaphragmatic effort to achieve lung expansion, which increases their diaphragmatic workload. 25 In addition, potential systemic inflammation and hypoxic conditions can result in the weakening and atrophy of the diaphragm muscle. 25 Subsequently, the diaphragm works harder to overcome increased airway resistance and handle the increased inspiratory load. This can be seen in severe cases of asthma, an obstructive airway disease that causes lung hyperinflation. Hyperinflation interrupts the ability of the diaphragm to generate efficient subatmospheric pressure, thereby increasing the workload of the respiratory muscles. 26 Furthermore, inflammatory markers, such as reactive oxygen species, in the case of asthma can impair diaphragmatic contractility by promoting oxidative damage to muscular proteins, thereby compromising the ability of the respiratory muscles to maintain adequate ventilation.27,28
The onset of DD after cardiorespiratory conditions remains unclear, even in humans. A previous study indicated that diaphragmatic weakness could emerge within 24 h of initiating mechanical ventilation in approximately 53% of patients who experienced a 10% reduction in diaphragm function.29,30 In addition, Zambon et al 31 suggested that the daily decrease in Tdi during mechanical ventilation was approximately 7.5%. This variability has also been observed in animal models, with DD manifesting differently in baboons and rats under prolonged mechanical ventilation.32,33 These findings are necessary to develop effective diagnostic methods to detect and monitor DD, enhance treatment efficacy and prevent the deterioration of respiratory function.
In the present study, we also reported the normal values for DE, Tdi and DTF in healthy cats. DE, Tdi and DTF were comparable between the left and right hemidiaphragms, reflecting the synchronised functioning of each hemidiaphragm. Notably, these values in cats were not affected by intrinsic factors such as age, sex, BW and BCS, highlighting the unique physiological characteristics of the species. However, humans have a wide normal reference interval (RI) for diaphragmatic parameters. For example, the normal RI for DE during tidal breathing in humans is 1–2.5 cm,8,34,35 whereas that for Tdi (expiration) during quiet breathing is 0.11–0.3 cm.2,8,36 This difference can be explained by the larger body size and distinct thoracic dimensions in humans, which can affect the normal RI for diaphragmatic parameters. The broader thoracic cavity in humans allows for a greater DE during tidal breathing, whereas cats’ smaller body size and specific thoracic architecture lead to comparatively lower DE. 37 The compact thoracic structure in cats likely influences their diaphragmatic movement, contributing to the differences in DE, Tdi and DTF measurements. Furthermore, variations in the thoracic shape and musculature between species result in different Tdi and DTF values.37,38 Notably, diaphragmatic parameters in humans are influenced by intrinsic factors such as age, 39 sex,2,24,36,40,41 body composition,24,36,39 scanning position 2 and inspiration phase.34,35,40,41 These findings indicate that body size and chest architecture play crucial roles in diaphragmatic function in different species.37,38
This study has some limitations. First, the included cats had similar BW and BCS; therefore, it may be inappropriate to conclude that BW and BCS exert no effect on diaphragmatic parameters. Further study is needed on a larger scale involving a more diverse group of cats, considering factors such as age, sex, BW, BCS, breed and health status to ensure more reliable and accurate findings. Second, this study had a very limited range of diseases represented in the diseased group of cats, with no cases of pleural effusions, thoracic masses or cardiac disease. Further study should include a broader range of diseases to enhance the robustness and applicability of the findings. Third, this study lacked histopathological examination for clinical research because this would have required invasive procedures. Finally, blood gas analysis was not performed to assess respiratory function in healthy and diseased cats. Future studies incorporating blood gas analysis will enrich our understanding of respiratory function in cats.
Conclusions
US effectively assessed diaphragmatic function and indicated that intrinsic factors such as sex, age, BW and BCS did not affect DE, Tdi and DTF. DE is a valuable and efficient diagnostic tool for DD, particularly in the left hemidiaphragm, and improves the detection of various respiratory diseases, thereby enhancing diagnostic accuracy in veterinary medicine. However, it is important to recognise that this is a preliminary study and the sample size was limited. Further research with larger sample sizes is necessary to validate these findings and ensure their generalisability to a wider population.
Footnotes
Acknowledgements
The authors would like to thank all staff at Chulalongkorn University and the cat owners who participated in this study.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: a scholarship from the Graduate School, Chulalongkorn University to commemorate the 72nd anniversary of His Majesty King Bhumibol Aduladej and the 90th anniversary of the Chulalongkorn University Fund (Ratchadaphiseksomphot Endowment Fund) (grant number GCUGR1125661061D,061).
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals and procedures that differed from established internationally recognised high standards (‘best practice’) of veterinary clinical care for the individual patient. The study therefore had prior ethical approval from an established (or ad hoc) committee as stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental animals or non-experimental animals, including cadavers, tissues and samples) for all procedure(s) undertaken (prospective studies or retrospective studies). No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
