Abstract
Recent advances in image technology, including significant gains in spatial resolution, have made realtime sequential ovarian evaluations possible in small rodents, allowing longitudinal (continued) studies of the ovarian cycle and reducing the required number of experimental animals. The aim of this study was to evaluate exogenous stimulated follicular growth in mice using high-resolution ultrasound technology. Female mice (n = 15) received a 5 IU intraperitoneal injection of equine chorionic gonadotropin (eCG) and 48 h later a 5 IU injection of human chorionic gonadotropin (hCG), and were allowed to mate thereafter. In experiment 1, animals (n = 7) were evaluated every 6 h, from 3 to 51 h after eCG injection, with an ultrasound biomicroscopy (UBM) equipped with a realtime 45 MHz microvisualization probe (RMV 707b). The ovaries were identified and follicular population quantified, and follicles were classified according to the diameter as small (≤449 μm) or large (≥450 μm). A significant change in the distribution of follicle population according to category was observed only 45 h after eCG injection (P < 0.05). In experiment 2, animals (n = 8) were evaluated every 2 h, from 2 h to 10 h after hCG treatment. The largest follicles reached a maximum size (596.7 ± 106.0 μxm) 5.8 ± 2.3 h after hCG injection. As expected, the population of large follicles decreased thereafter, indicating the progress of ovulations, but large follicles were still detected late after treatment (10.1 ± 1.1 h). In conclusion, UBM can be used to evaluate follicle dynamics in superstimulated mice (C57BL/6 and BALB/c); significant changes in follicle distribution only occur at later stages after eCG stimulation; and hCG-induced ovulations may not occur synchronously in mice.
Mice are widely used in reproductive biology studies due to their particular characteristics, such as short life cycle, prolificacy and ease of manipulation of their oestrous cycle. The mechanisms of follicle recruitment, selection and ovulation in mice, however, are less understood when compared with human 1 and other domestic animals such as cattle 2 and goats. 3 The recent advances in the knowledge of follicular dynamics in mammals were strongly related to the development of image technologies that allowed daily in vivo monitoring of ovarian structures. Most ultrasound devices available, however, have an image resolution limit of about 2–3 mm, and are therefore not suitable for small rodents. The earliest studies in these species were based only on histological evaluations, and the assessment of follicular growth during the oestrous cycle could only be performed after euthanasia of the animals under study. Consequently, a reliable sequential evaluation of ovarian changes during the oestrous cycle was not feasible.4–6
The development of ultrasound biomicroscopy (UBM) provided a new alternative for the study of follicular growth in mice, since it allows the evaluation of high-resolution images of organs and systems through ultra-high-frequency ultrasound waves (30–55 MHz7). UBM has been used to assess ovarian follicles, corpora lutea and corpora haemorrhagica in rats and mice. 8 The accuracy of UBM in quantifying and measuring ovarian follicles greater than 300 μm has also been demonstrated. 9
Most of the experiments with follicular growth analysis and embryo production in mice use exogenous hormone stimulation protocols. 10 Although UBM was used to describe changes in follicular population during the oestrous cycle, 11 to our knowledge no study applied this technology to sequentially evaluate ovarian follicle dynamics in response to superovulation protocols. The aim of this study was to evaluate exogenous stimulated follicular growth and ovulation in mice using high-resolution ultrasound technology.
Materials and Methods
Animals
Adult female mice, BALB/cAnNCrl (n = 7) and C57BL/6J (n = 8), 50 days of age, were used in experiments 1 and 2, respectively. These animals were produced and maintained in animal facilities at the Gonçalo Moniz Research Center, FIOCRUZ, Brazil. The animals were acclimatized for 15 days before the experiments, during which they were handled three times a day to reduce the effect of handling stress on ovarian cyclicity. During the entire period, animals were kept under a photoperiod of 12 h of light/12 h of darkness and a temperature of 21°C. Food and water were available ad libitum. All procedures and handling of the animals used in this study were in accordance with the ethical principles of the Brazilian Council of Animal Experimentation.
Ultrasound biomicroscopy
Ultrasonographic evaluations were performed using the high-resolution UBM (Vevo 660, Visual Sonics, Toronto, ON, Canada) equipped with a transducer for Micro Realtime visualization (RMV 707b), with broadband frequency of 45 MHz, axial resolution of 55 μm, lateral resolution of 115 μm, focal length of 12.7 mm and field of view of 20 mm. The equipment was connected to a personal computer allowing the control of image quality and resolution. Representative images were recorded in .jpeg format.
Ovarian imaging technique
In each ultrasonographic evaluation, mice were anaesthetized with 0.17 × 10-3 g xylazine (Anasedan®, Industry Sespo Ltda, São Paulo, Brazil) associated with 1.5 × 10-3 g ketamine (Ketamine®, Pharmaceuticals and Chemicals Products Cristália, São Paulo, Brazil) diluted in 200 μL of physiological solution (7.9 and 69.8 mg/kg, respectively). Throughout experiments 1 and 2 mice were anaesthetized eight and five times, respectively. The dosage of ketamine/xylazine was previously optimized for the anaesthetic effect to last for the shortest time possible (about 10 min).
The dorsolateral body hair coat was shaved and the mice were then transferred to the platform of the equipment, which was kept warm at 37°C. Contact ultrasound gel Mercur® (Mercur SA, Porto Alegre, Brazil) was applied on the shaved region. The transducer was positioned and, once in contact with the skin, the platform was moved cranially and caudally until the kidney was identified. Each ovary was visualized caudally to the ipsilateral kidney, and cross-section images were obtained. The follicles visualized were quantified, measured and classified according to diameter as small (≤449 μm) or large follicles (≥450 μm).
Experiment 1
BALB/c female mice (n = 7) were superovulated with 5 IU equine chorionic gonadotropin (eCG; Novormon 5000®, Syntex SA, Pharmaceutical Industry and Biochemistry, Buenos Aires, Argentina), and 48 h later received 5 IU human chorionic gonadotropin (hCG; Vetecor®, Calier Brasil Laboratories Ltda, São Paulo, Brazil), as previously described.12,13 Immediately after the hCG injection, females were allowed to mate with fertile F1 males (C57BL/6 × BALB/c). The male:female ratio used was 1:1. Ultrasonographic evaluations were performed (as described above) at 6 h intervals, beginning 3 h after eCG injection, in a total of eight exams during the period. Because of the longer examination period of this experiment, the body hair coat was re-shaved at 27 and 45 h after eCG injection.
Experiment 2
C57BL/6 female mice (n = 8) were superovulated with 5 IU eCG followed 48 h later by 5 IU hCG, as described in experiment 1, and were allowed to mate with fertile F1 males (C57BL/6 × BALB/c) at the rate of 1:1 (male:female). In this experiment, ultrasonographic ovarian assessments were performed every 2 h, beginning 2 h after hCG injection.
Statistical analyses
The number of follicles in each category (size) and the mean follicle size for each evaluation in experiments 1 and 2 were tested for normality and homoscedasticity of variances by Lilliefors and Bartlett tests, respectively. Data were analysed by analysis of variance and differences among means compared using Tukey's test. The efficiency of UBM in follicle detection was compared between experiments by the chi-square test. All analyses were performed using the GraphPad Prism® software version 5 (GraphPad™ Software, San Diego, CA, USA), and SAEG software version 9.0 (SAEG Federal University of Viçosa, MG, Brazil). A probability of P < 0.05 indicated that the difference was significant. The results are presented as means ± SEM.
Results
The average weight of these animals was 21.5 g. Ovaries and follicles could be identified in 93.5% (232 of 248) of the ultrasound exams (Figure 1). The efficiency of examinations was not different between experiments 1 and 2 (96.3 versus 92.6%, respectively; P > 0.05), i.e. it was possible to evaluate both C57BL/6 and BALB/c mice.
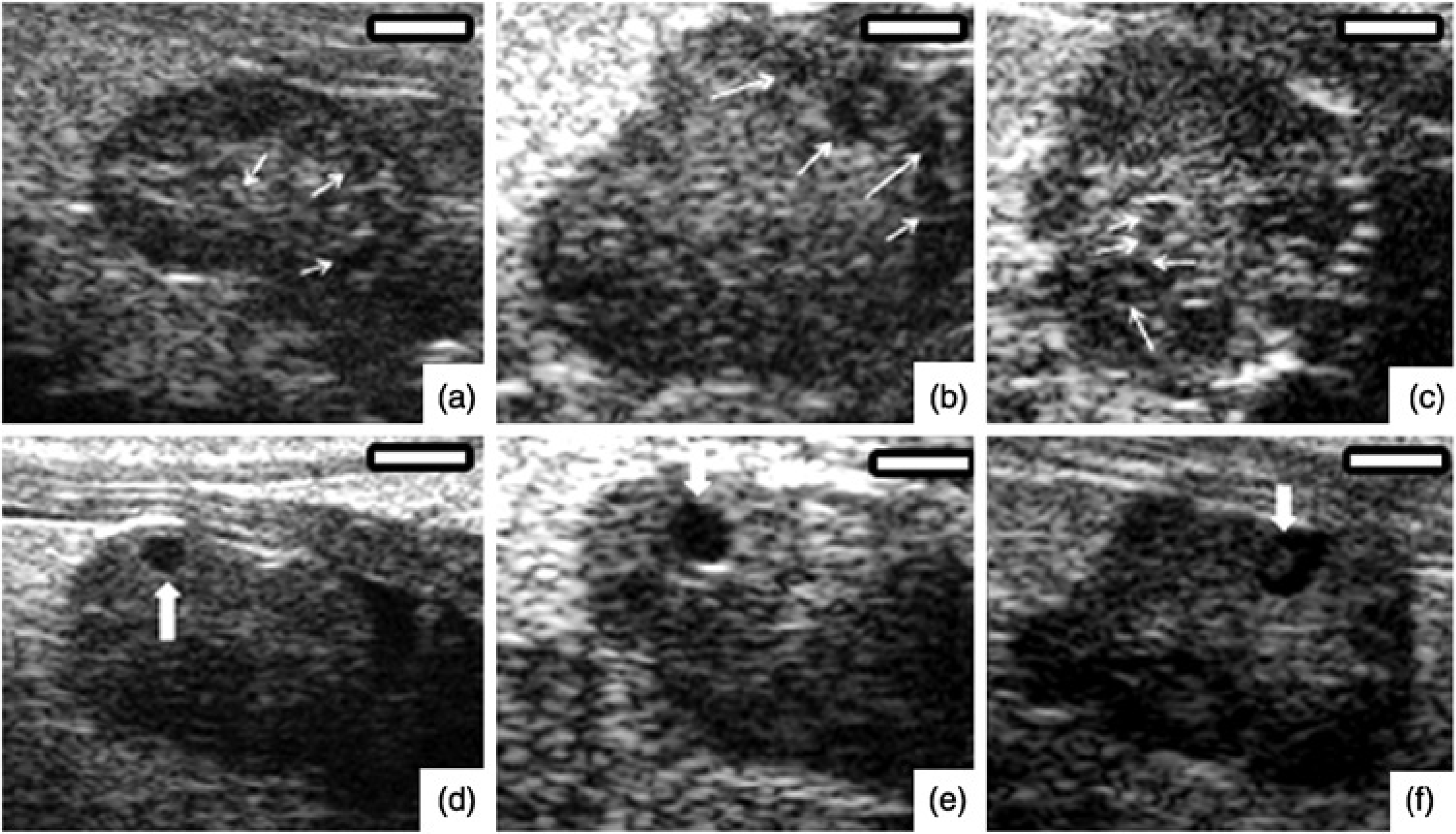
Ultrasound images of mouse ovaries from experiment 1 (a, b, c) and 2 (d, e, f). (a, b, c) show growing small follicles (≤449 μm, white arrows). (d, e, f) show large preovulatory follicles (≥450 μm, white arrows). Bars = 1000 μm
In experiment 1, ovarian follicular population was characterized by a greater number of small follicles (≤449 μm), and a lower number of large follicles (≥450 μm; Figure 2). As expected, eCG-induced follicle growth resulted in the recruitment of small follicles, but a significant increase (P < 0.05) in the number of large follicles was only observed 45 h after the eCG injection.
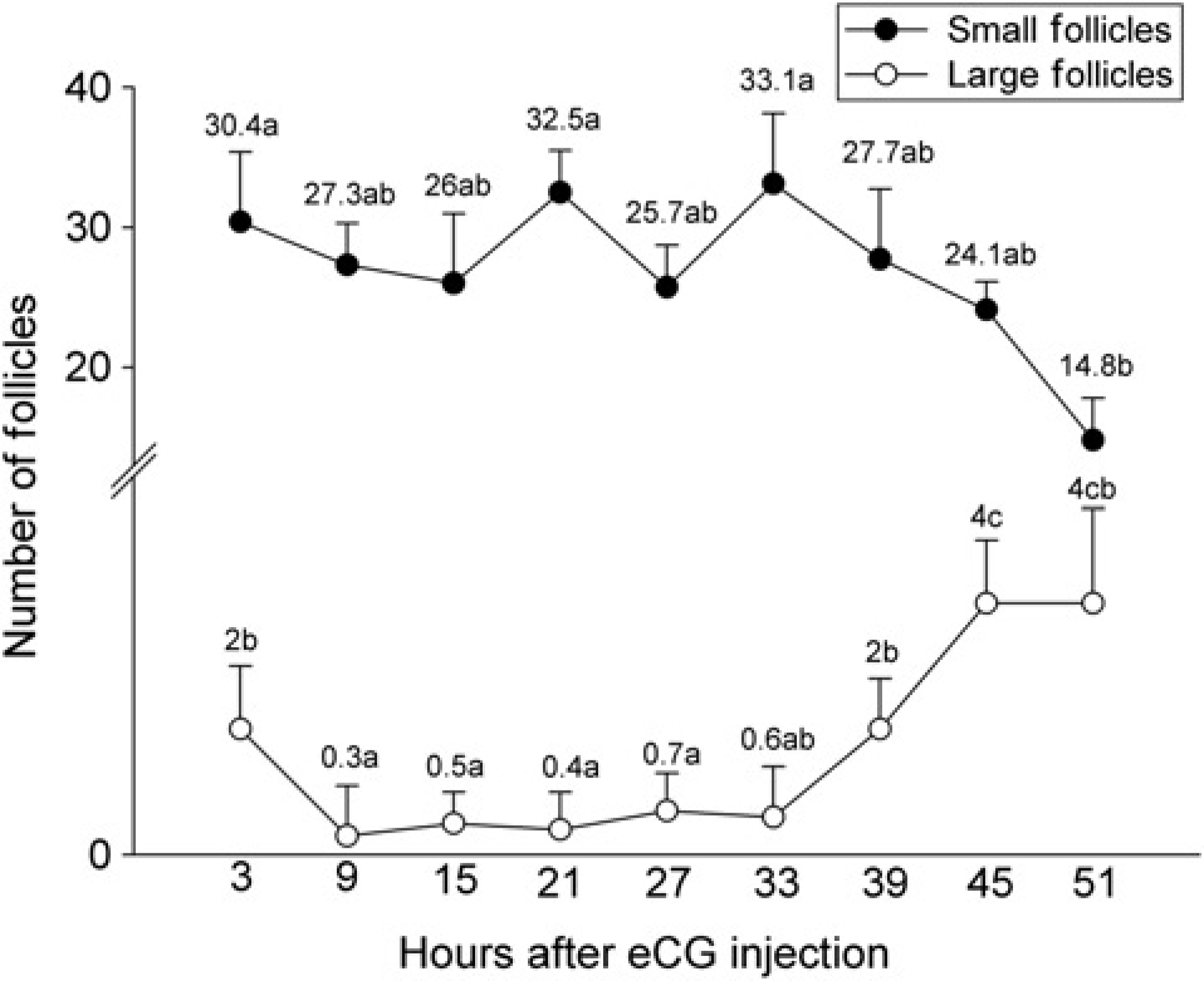
Small (≤449 μm) and large (≥450 μm) follicle numbers in BALB/c mice after eCG injection (experiment 1). Different letters in each follicle category denote significant differences (P < 0.05)
In experiment 2, follicle population of both size categories progressively decreased after hCG treatment (P < 0.05; Figure 3). Follicles reached their maximum size (596.7 ± 106.0 μm) 5.8 ± 2.3 h after hCG injection. In 62.5% (5 of 8) of the animals, however, large follicles (550.0 ± 103.5 μm) were detected later after the disappearance of the previous large follicles (ovulation), on average 10.1 ± 1.1 h from hCG treatment.
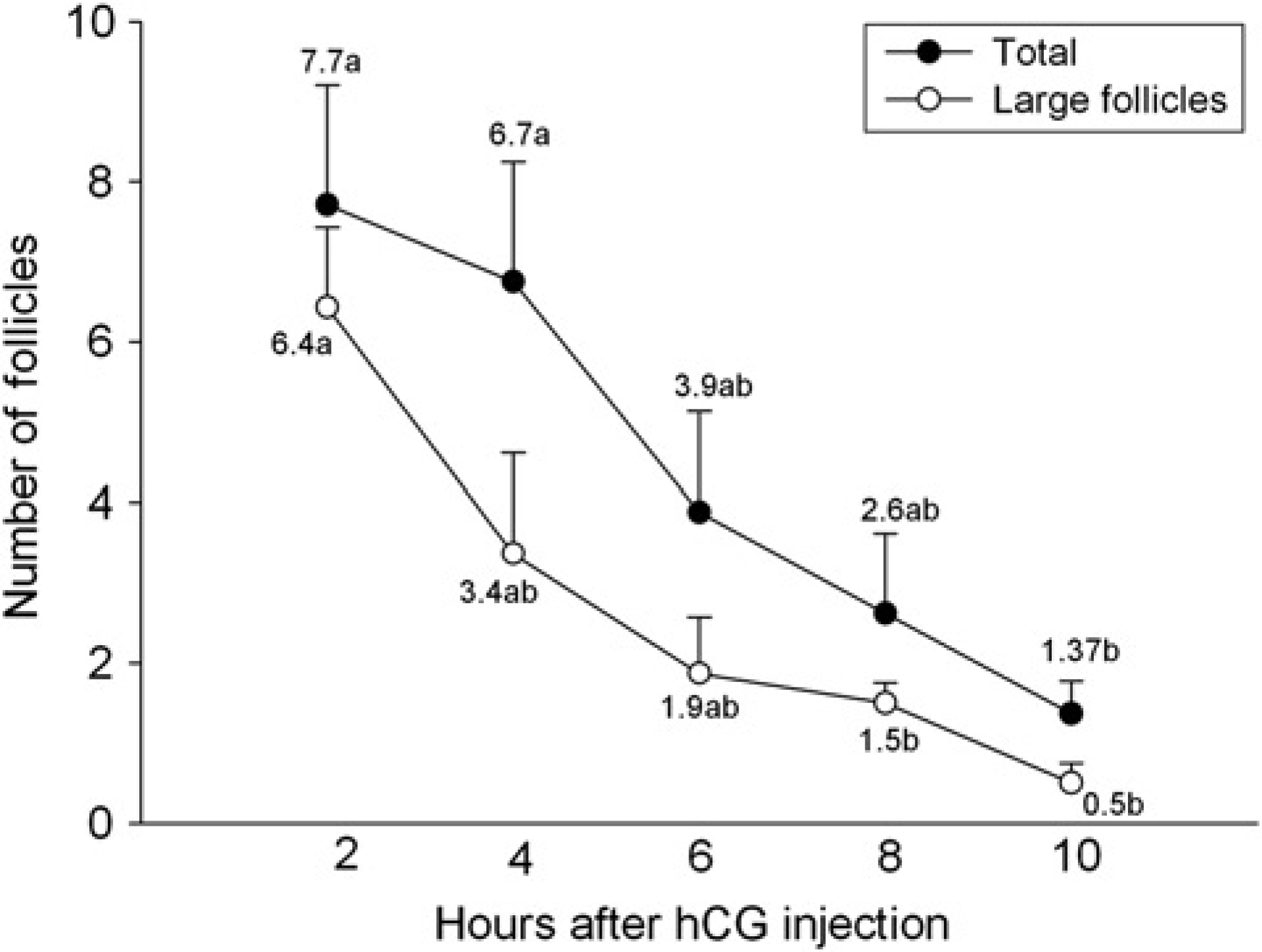
Total follicle population and number of large follicles (≥450 μm) in C57BL/6 mice after hCG injection (experiment 2). Different letters in each follicle category denote significant differences (P < 0.05)
Discussion
The improvement in imaging technologies, including the development of high-frequency ultrasound waves (30–55 MHz), made high-resolution imaging of structures smaller than 1 mm possible. In the last few years, studies have been conducted to evaluate the potential of UBM as a tool to assess ovarian follicular growth in laboratory animals.7,8 Nevertheless, several possible applications of UBM for studying ovarian physiology remain to be explored. The present experiments were designed to evaluate one of these possibilities, i.e. the study of follicle dynamics after exogenous stimulation of follicular growth and ovulation in adult mice.
In both experiments, UBM was considered an effective tool to visualize ovaries and follicles larger than 200 μm, since these structures could be seen in 93.5% of the 248 ultrasonographic exams performed. Although this effectiveness was not affected by the strain used, it shall be considered that both BALB/c and C57BL/6 mice had similar body characteristics, and therefore other mouse strains should be evaluated in order to determine possible influences of genetic background in ovary size or follicle population characteristics.
The present study was performed using mice with induced follicular growth and ovulation. Consequently, follicle population characteristics at the beginning of the treatment period, such as the total number of follicles and the eventual presence of follicles larger than 450 μm, were probably determined by the phase of the oestrous cycle when treatment began. Previous histological analysis of the ovaries showed evidences that the number and diameter of antral follicles change during the mouse oestrous cycle, and large follicles may be observed at the end of the pro-oestrus. 14 Coherently, follicle population distribution among size categories at the beginning of the eCG treatment in the present study was similar to that previously reported in mice by Jaiswal et al., 11 using the same technique (UBM). The comparison of results from this and from the present study, however, shall be taken carefully, once different anaesthesia procedures were used.
Jaiswal et al. 11 used isoflurane for anaesthesia and demonstrated the inability to perform the ultrasound assessments without anaesthetizing the animals, and there was also no observed effect of the anaesthetic procedure on ovarian cyclicity. The isoflurane not only provides a faster induction and recovery of anaesthesia, but also requires specific equipment and training, along with special care to avoid accidental inhalation by the operators in such repeated use. The ketamine/xylazine protocol is much simpler to use and are standardized for mice in laboratory procedures. The longer recovery interval (approximately 10 min, in our anaesthetic protocol) was not a major problem, considering the time needed to prepare animals and scan both ovaries.
Even though the change in follicle distribution among size classes was consistent with the expected eCG-induced follicle growth, the increase in the number of large follicles was only significant a few hours before ovulation induction (performed 48 h after eCG). This fact may be related to the progress of follicular growth in mice. Follicle re-distribution among size classes occurs progressively in response to superovulation in cattle, 15 but stimulation protocols in other species are also longer than that used for mice, and comparisons should be taken carefully. The follicle growth rate observed in mice in the current study was higher than the rate observed in large animals, in which follicles take days to double their diameter. 16 Hence, considering the maximum size of follicles among species and similarity of mechanisms associated with antrum formation, 17 the final growth of mouse follicles would be expected to occur in a matter of hours. Induced follicle mobilization to a larger size class in the present experiment was associated with a reduction in the population of small follicles by the end of the treatment, consistently with that observed at the end of natural pro-oestrus in mice.4–6,18
The lack of technology for more detailed studies on the growth of antral follicles in mice resulted in a large gap in the knowledge regarding the mechanisms of follicle dynamics in this species. The feasibility of longitudinal studies and realtime follicular antral monitoring with UBM can lead to a significant advance in the understanding of antral follicle growth mechanisms, as has been achieved for large animals. 2 Results of the current study suggest a short period of follicle development at high rates of growth; however, evaluation periods shorter than those used in this study and also in previous studies 11 may be required if an individual follicle tracking is desired. The need of anaesthesia may be a challenge for further UBM investigations, because the required recovery period may restrict the number of sequential exams. In the present study, separated evaluation periods for follicle growth (experiment 1) and follicle ovulation (experiment 2) were needed to overcome such limitations. A previous study reported that anaesthesia did not affect oestrous cycle characteristics, but a longer exam interval was used, 11 which may be insufficient for accurate follicle tracking.
The decrease in the follicle population after hCG treatment was consistent with the reduction in the number of large follicles, as a consequence of their ovulation. Follicle visualization by ultrasound is possible because of the antrum lower echogenicity and follicular rupture during ovulation causes follicles to disappear from the ultrasonographic image. 17 The maximum size of follicles observed in the present study suggests that ovulation in the mouse occurs when a follicle reaches a size of 400–600 μm, which is, proportionally, a large size if ovary diameter (about 2 mm) is taken into account. In most cases (75%; 6 of 8), large follicles disappeared within 6 h after hCG Interestingly, in 62.5% of the eight animals enrolled in experiment 2, large follicles were detected later, and sometimes in opposite ovaries, suggesting the possibility of successive waves of follicular growth after ovulation induction. In large animals, ovulation is preceded by a plateau phase, with no significant follicular growth after the luteinizing hormone (LH) surge. 16 It is hypothesized that the limited space available within the ovary and the relatively large diameter of preovulatory follicles, which is about one-fourth of the entire mouse ovary, explains the need for a later period of follicular growth even after hCG treatment. Further investigation will be required to confirm this hypothesis.
Additionally, this study demonstrated the efficiency of UBM for longitudinal studies with short lapses of time between examinations, making it possible to evaluate the follicular growth in mice, decreasing the number of animals needed for this same type of analysis when compared with histology, and avoiding euthanasia. UBM is an efficient tool to assess follicular growth after exogenous stimulation in mice. The stimulated follicular growth is characterized by a late presence of preovulatory follicles and some asynchrony among ovulations after the LH surge.
Footnotes
Acknowledgements
This research received financial support from FAPEMIG (Minas Gerais State Research Support Foundation) and CNPq (Brazilian National Research Council). The authors thank the Embrapa Dairy Cattle Research Center for technical support.
