Abstract
Ultrasonographic assessment of fetal growth to estimate fetal weight has been widely used in clinical obstetrics but not in laboratory mice. Even though it is important to assess fetal growth abnormalities for gene-targeting studies using mice, there have been no reports of accurately estimated fetal weight using fetal biometric parameters in mice. The aim of this study was to establish an accurate mouse formula using fetal biometric parameters under ultrasound imaging. Using a high-frequency ultrasound system with a 40 MHz transducer, we measured 293 fetuses of biparietal diameter and mean abdominal diameter from day 12.5 postcoitus (p.c.) until day 18.5 p.c every day. Thirteen algorithms for humans based on head and/or abdominal measurements were assessed. We established an accurate formula based on measurement of the abdomen in Jcl:ICR mice to investigate gestational complications, such as intrauterine growth restriction.
Developmental processes prior to the implantation stage are similar in all animals. However, after that there is a variety of differences between species. Nonetheless, mice are very useful and are widely used for understanding the physiology and pathophysiology of pregnancy-related conditions and complications, especially for gene-targeting study. Sometimes gene-targeting studies show embryonic lethality. The assessment of a fetus in utero in realtime would provide important information for phenotypic development in gene-targeting studies. Moreover, it is important to assess intrauterine growth restriction (IUGR), which is one of the most common human gestational complications. It is reported that IUGR is a risk factor for cardiovascular disease, type 2 diabetes mellitus, and obesity in adulthood. 1 It could be important to assess fetal growth in utero to understand not only the human gestational complication model, but also other disease models. Many studies using mice to study gestational complications assessed the outcome of pregnancy by measuring actual fetal weight and number of live pups. 2 However, it does not provide realtime and sequential information. Realtime imaging using ultrasonography can reduce the number of animals for research and it may provide more information about sequential growth patterns of the fetus during the course of the pregnancy.
Ultrasonography was introduced in 1958 and since then ultrasonographic assessment of human fetal growth has been widely used in daily practice. However, it is still not used as widely in laboratory animals as it is in humans. The problem of applying ultrasonography in such small animals as mice is that it requires a much higher frequency imaging system to assess such small body parts. It is difficult to get accurate images using the same system as used in human subjects, because of the depth of the target. Recently such technologies are being applied to animal-based research as higher frequency imaging has recently become available, making accurate imaging of small animals possible. In utero ultrasound imaging has been reported in a variety of animal models including sheep, 3,4 rats 5 and dogs. 6 Furthermore, high-frequency ultrasound has been used in many murine studies of pregnancy and development. 7–17
More recently, the processes from implantation through growth and placentation in the mouse have been imaged in a qualitative manner. 11,17–19 But there was no report of accurately estimated fetal weight by fetal biometric parameters. Several formulas for human fetal weight estimation have been proposed over the past 30 years, which use different combinations of standardized fetal biometric parameters, such as measurements of the head, abdomen and femur. 20 In this study, we used a high-frequency ultrasound system with a 40 MHz transducer to assess mouse fetal growth in utero. We propose here an algorism for mouse estimating fetal weight derived from the accumulated data of human studies.
Materials and methods
Animals
Specific pathogen-free Jcl:ICR mice purchased from a commercial breeder (SLC, Shizuoka, Japan) were used in this study. The animals were maintained under controlled conditions (temperature, 23 ± 1.5°C; relative humidity, 45 ± 15%; 12 times of 100% fresh air changes/hour; maximum ammonia concentration, 20 ppm). The light cycle was 12/12 h: lights were turned off at 20:00 h and turned on at 08:00 h (fluorescent lighting 150–300 Lux at 40–85 cm above floor). The mice were acclimatized for at least one week before breeding. Female Jcl:ICR mice, aged 8–10 weeks, were bred with male Jcl:ICR mice at the rate of 1:1. The morning when a vaginal plugging was first observed was designated as day 0.5 postcoitus (p.c.). Ethical reviews were undertaken by the Animal Ethics Committees of Osaka University Graduate School of Medicine. All animal experiments were performed according to the appropriate guidelines for animal use approved by the Institutional Animal Care and Use Committee of Osaka University Graduate School of Medicine.
Anaesthesia protocol
Pregnant female mice were anaesthetized with isoflurane (Forane®, Abbott Japan Co, Ltd, Tokyo, Japan) delivered at 5% during induction and at 1–2% via a facemask during imaging and heart rate was monitored (450–550 beats/min). Body temperature was monitored via a rectal thermometer and maintained at 36–38°C using a heating pad. The anaesthesia time for one scanning was approximately 35 min for each dam.
Ultrasound imaging
The abdomens of the dams were shaved and coupling gel was applied. Ultrasound scanning was conducted on fetuses in utero, using the mother's bladder as a landmark. Ultrasound was performed with a Vevo2100 (VisualSonics Inc, Ontario, Canada) using a MS-550S transducer (40 MHz frequency). Biparietal diameter (BPD) was recorded at the level of the cavum septum pellucidum, and thalamus with falx in the midline. Callipers were placed ‘leading edge to leading edge’ of the skull. When the fetal position made this image plane unobtainable, an axial image through the orbits and cavum septum pellucidum was used and BPD measured at the widest diameter (Figure 1c). The mean abdominal diameter (MAD) was measured lengthwise and crosswise at the level of the mid third of the umbilical vein (Figure 1d). For the 293 fetuses, BPD and MAD were collected from day 12.5 p.c. until day 18.5 p.c. Immediately after Caesarean section was performed on each day, the actual fetal weights were measured.
Gestation sac, fetus and placental primordium at day 8.0 p.c. (a). Limb buds at day 10.5 p.c. (b). Measurement of biparietal diameter (c), mean abdominal diameter (d) at day 14.5 p.c. Eyes, pinna of ears and tail at day 14.5 p.c. (e–g)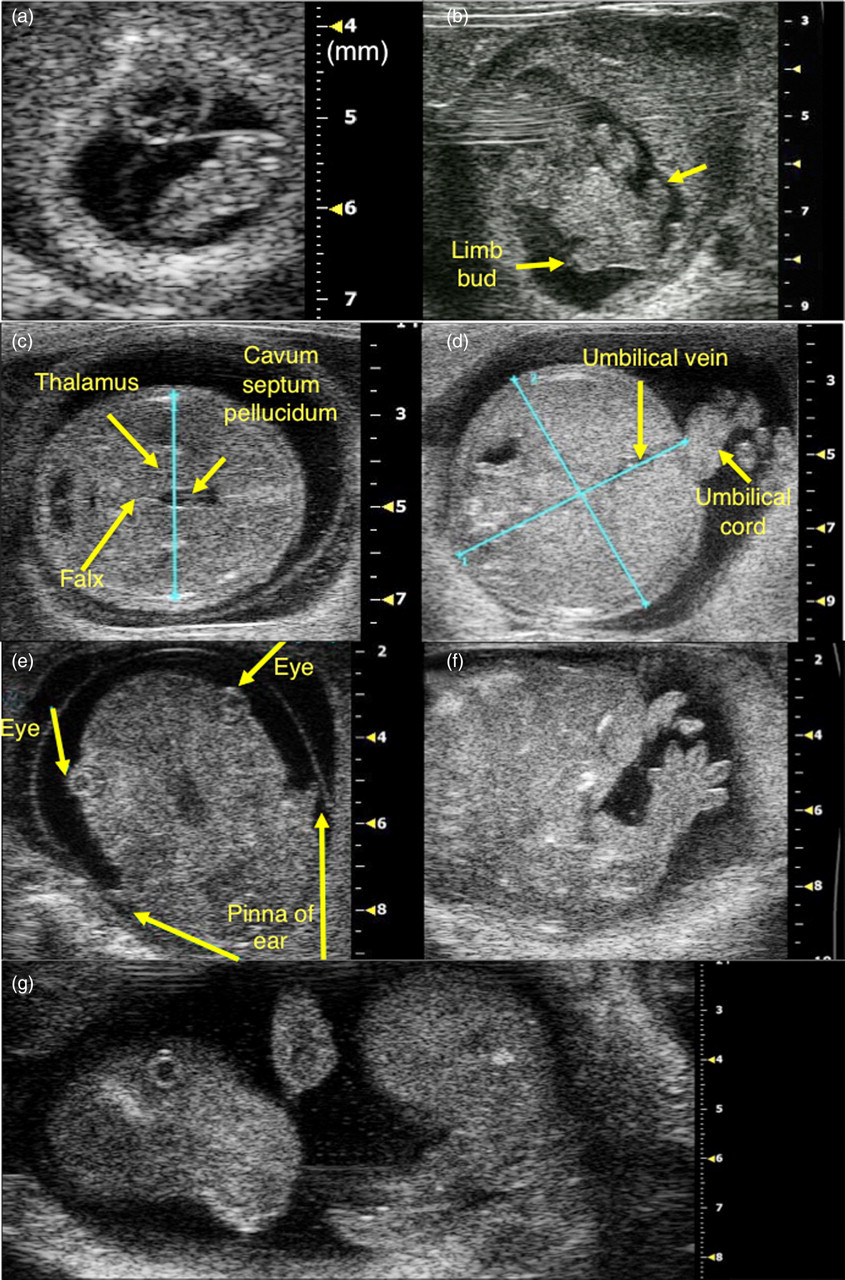
Data analysis and statistical evaluation of results
Assessment of the ultrasonic equations for estimation of human fetal weight categorized by fetal biometric measurements used for mice
R: multiple correlation coefficient; R 2: adjusted coefficient of determination; H: head; A: abdomen; EFW: estimated fetal weight; BPD: biparietal diameter; AC: abdominal circumference
Some of the formulas for human subjects to estimate fetal weight use abdominal circumference (AC). However, the equipment used did not have a program to measure the circumference. Instead of measuring the AC under these conditions, MAD was used in this study by measuring lengthwise and crosswise at the level of the mid third of the umbilical vein. It approximated a circular shape. The formulas which rely on measurement of the abdomen were calculated using MAD as AC = MAD × π.
The accuracy of all formulas was evaluated as the absolute value of the ‘potential error’ of the estimated weight proposed by Edwards et al.,
32
and adopted by Anderson et al.,
5
who highlighted that ultrasonographic estimation represents clinical management, using the following formula: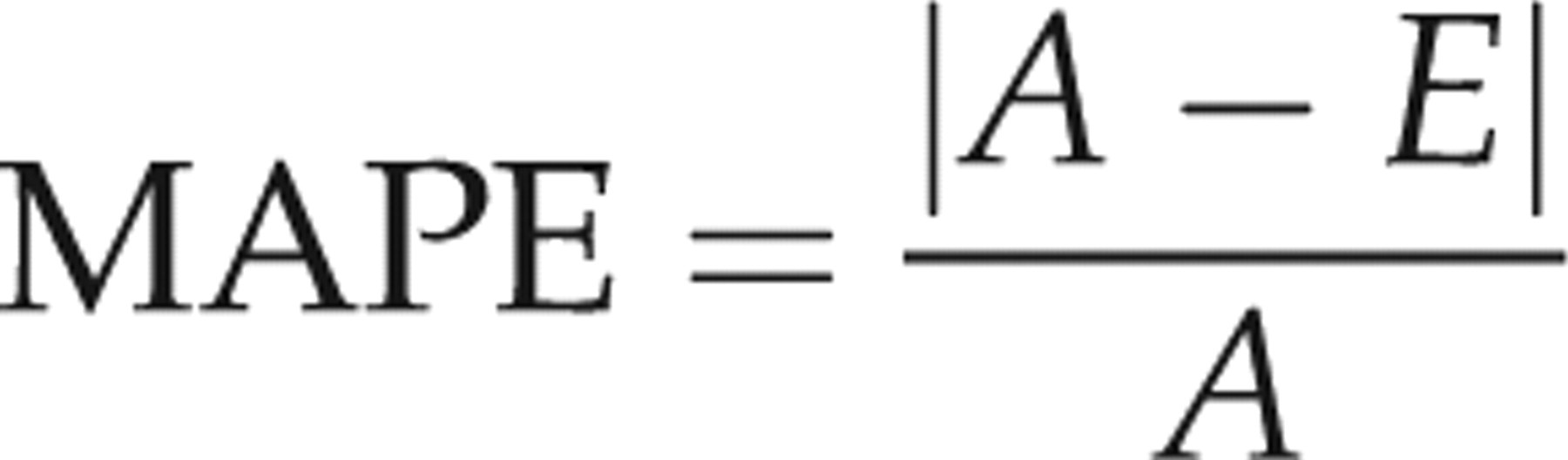
Results
At the day 8.0 p.c., the gestation sac, fetus and placental primordium were recognized under ultrasound (Figure 1a). The limb buds were first apparent at day 10.5 p.c. (Figure 1b). Then on day 14.5 p.c., eyes, pinna of ears and tail were clearly recognized (Figures 1e–g).
Measurement of BPD and MAD
From days 12.5 to 18.5 p.c., a total of 293 fetuses of BPD and MAD were measured every morning as the standardized fetal biometric parameters (Figure 2). Female mice had an average of 13.6 fetuses in their uterine horns. An average of 9.5 fetuses were measured in one dam. It was difficult to measure BPD and/or MAD in some fetuses because of the position. An average of 41.9 fetuses were measured on each day of pregnancy (n = 33 on day 12.5 p.c., n = 36 on day 13.5 p.c., n = 54 on day 14.5 p.c., n = 34 on day 15.5 p.c., n = 34 on day 16.5 p.c., n = 58 on day 17.5 p.c., n = 44 on day 18.5 p.c.).
Measurement of biparietal diameter (BPD) and mean abdominal diameter (MAD) from days 12.5 to 18.5 p.c. (n = 33–57)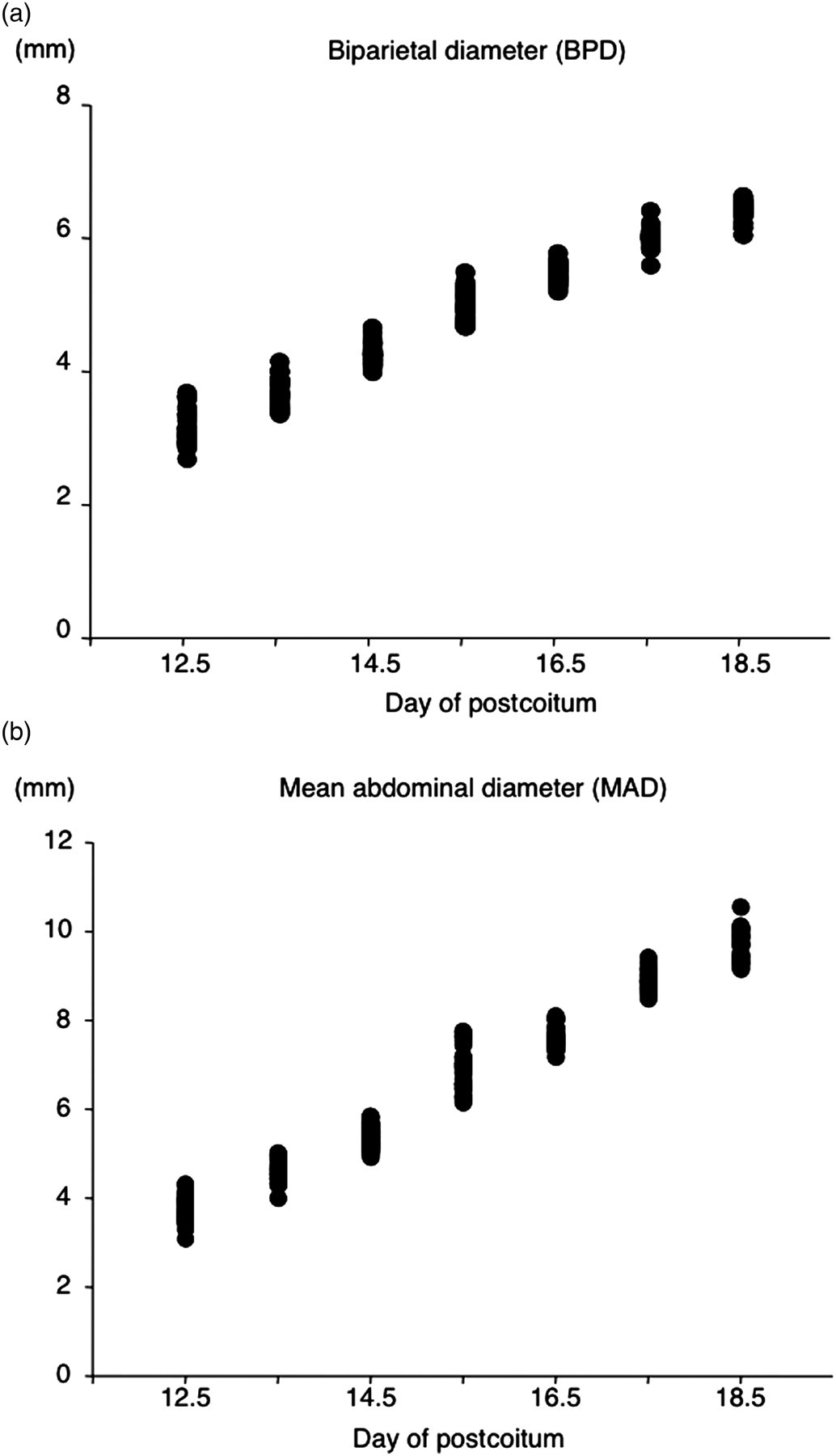
The 95% confidence interval (CI) of BPD means was from 0.0271 to 0.0899 and the 95% CI of MAD means was from 0.0653 to 0.140. Two experienced sonographers independently measured BPD and AC each gestational day. There were no significant differences between the data recorded by the two sonographers (data not shown).
Assessment of algorithms for humans
Thirteen algorithms for human subjects based on BPD and AC were assessed in this study (Table 1). MAPEs of all 13 algorisms were shown in Figure 3. The formula of Warsof 1
27
was the most accurate of these 13 formulas (30.516 ± 3.792%). All the formulas failed to provide accurate estimates of birth weight (that was defined as 10% or less of mean absolute error
33
) and were excluded from subsequent analysis.
Mean absolute errors (±1 SD) in estimated fetal weight using 13 different original human formulas that incorporated a combination of biometrical parameters shown on the left-hand side: H: head (biparietal diameter); A: abdomen (abdominal circumference as AC = MAD × π)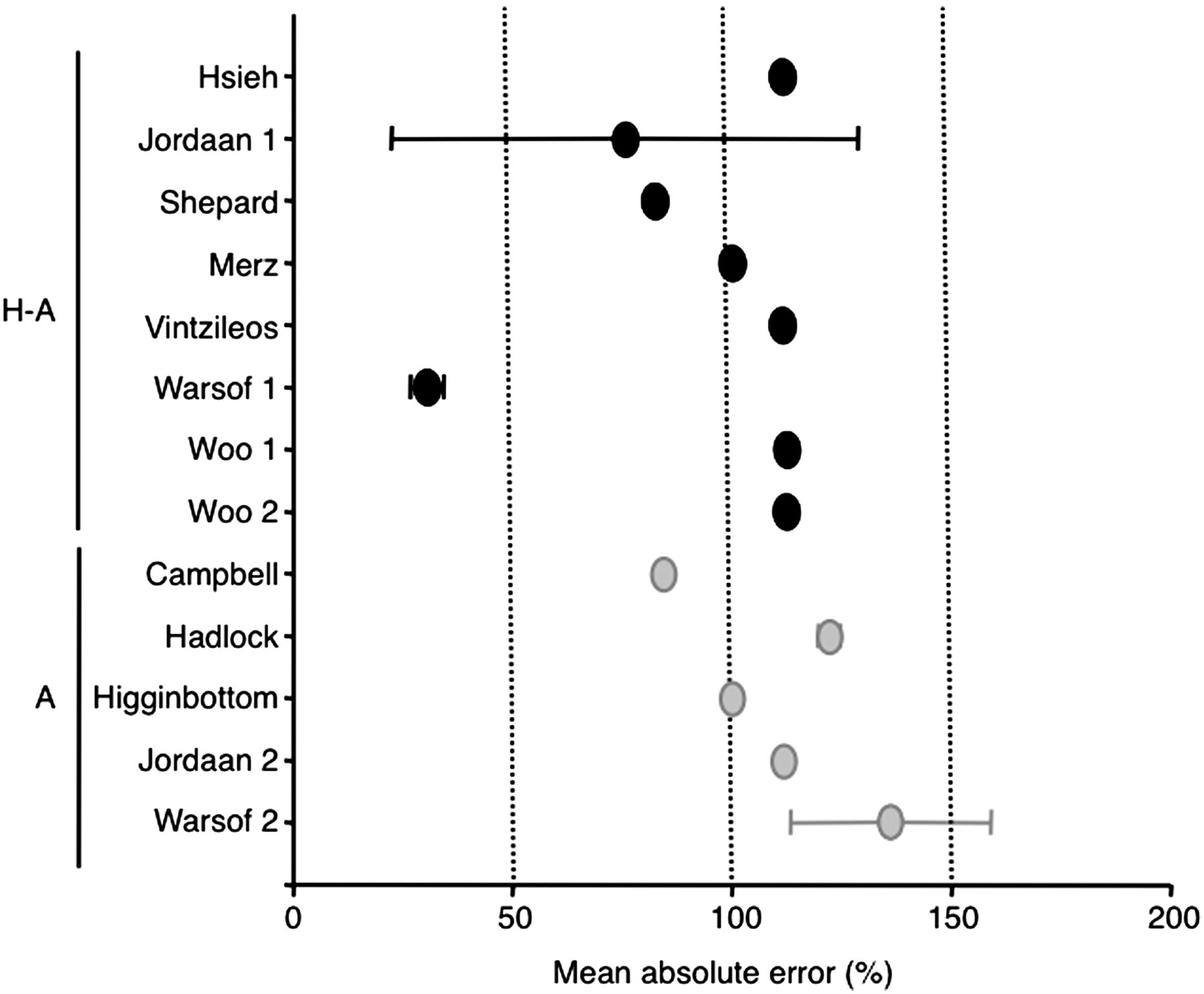
Algorism for mouse estimating fetal weight
Multiple regression analyses using actual mouse fetal weight and fetal biometric measurements
R: multiple correlation coefficient; R 2: adjusted coefficient of determination; EFW: estimated fetal weight; BPD: biparietal diameter; AC: abdominal circumference
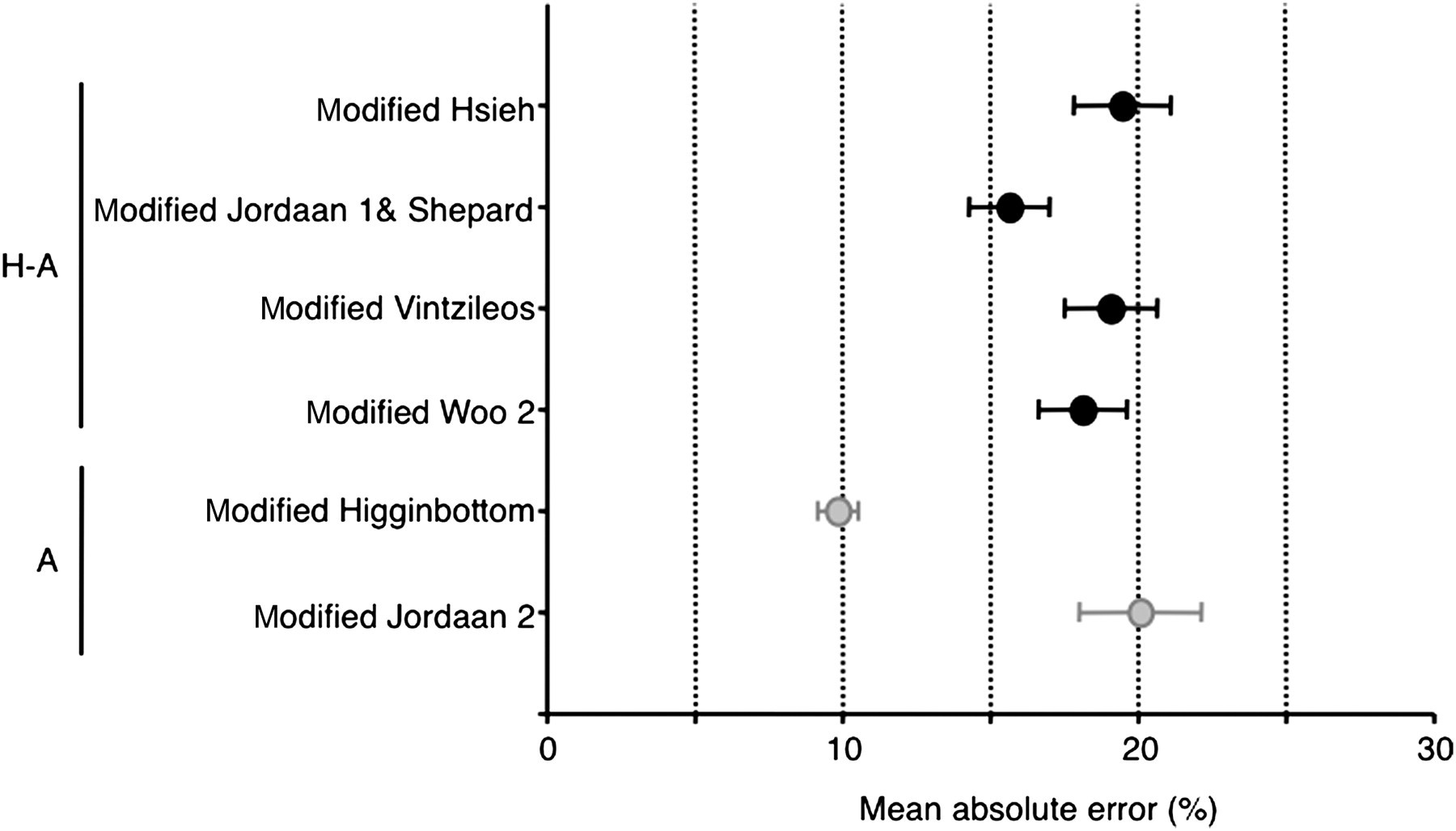
Mean absolute errors (±1 SD) in estimated fetal weight using six different modified formulas that incorporated a combination of biometrical parameters shown on the left-hand side: H: head (biparietal diameter); A: abdomen (abdominal circumference as AC = MAD × π)
Discussion
Ultrasonographic assessment of fetal growth to human estimate fetal weight has been used in daily practice for more than 30 years. In this study, we proposed an accurate formula based on abdomen measurement to estimate mouse fetal weight derived from the accumulated data of human studies.
For the estimation of fetal weights in human subjects, among the categorized algorithms, formulas based on head–abdomen–femur measurements are most common. However, using this system, it was difficult to measure femur length accurately in mice. Error due to the equations could be the largest source of disagreement between predictions and actual birth weight. Thus, we decided to use BPD and MAD as the standardized fetal biometric parameters to estimate fetal weight. In this study we measured murine fetal BPD and MAD from day 12.5 p.c. to day 18.5 p.c. as the standardized fetal biometric parameter to estimate fetal weight. An average of 41.9 fetuses each day of pregnancy, about 70% of fetuses per dam were measured for BPD and MAD. Some of them were in a difficult position to measure BPD and/or MAD accurately. Using these data we assessed the algorisms for mice using human formulas based on head and/or abdomen measurements. Only the formula based on abdomen, modified Higginbottom 31 (EFW = 0.0000541 AC3), provided accurate estimation of fetal weight.
The most common human formulas use head–abdomen–femur measurements. Recently, it has been considered sufficient to just use abdominal measurement for estimation of fetal weight in human subjects, because the differences in weight must be explained primarily by variations in girth. AC is among the least predictive measures of fetal age but the most predictive of fetal growth. 34 The results from this study using mice reach the same conclusions as human studies.
There have been some reports already published about murine in utero ultrasound imaging using a 15 MHz transducer. At first, we tried using the 15 MHz and 18 MHz transducers. However, it was too difficult for us to identify the edge of the organs. In this study, using a 40 MHz transducer showed imaging as clear as in human subjects in a clinical setting. We were concerned about the effect of the physical energy from the high-frequency transducer upon such a small fetus. After these sequential measurements, there were no intrauterine fetal deaths and it did not affect the number of live pups (data not shown).
This is the first report to propose an accurate formula to estimate mouse fetal weight. It would also help in understanding the mechanisms of gestational complications and development. Moreover, it would help to identify a phenotype, fetal growth, which has been missed in the past in gene-targeting studies. It might also provide some feedback applicable to further clinical situations.
Footnotes
ACKNOWLEDGEMENTS
We thank Tsuyoshi Ono, Yoshiharu Tsuru (Primetech Co, Japan), Ryota Tsuda (Nihon Koden Co, Japan), Hiroshi Shigeta and Hideki Suzuki (Aloka Co, Japan) for organizing the ultrasonography machine. We are also grateful to the animal care staff of Osaka University Graduate School of Medicine for their implementation of the standard operating procedures that provide biosecurity to these animals during use of the ultrasonography. This study was supported by Grants-in-Aid for Scientific Research (Nos. 20890120, 20659258, 21390453 and 21791553) from the Ministry of Education, Science and Culture of Japan (Tokyo, Japan).
