Abstract
Objectives
The aim of this retrospective study was to describe the neoplastic and non-neoplastic lesions seen on histopathological examination of cat testes in Hong Kong between 2018 and 2024.
Methods
A total of 26 single or dual testes samples were collected from 18 cats by veterinarians at 14 veterinary clinics and submitted for histopathological examination. Laboratory records, including signalment, lesion location, age, breed and histopathological findings, were reviewed for each cat.
Results
Neoplastic testicular lesions were seen in three older cats (median age 8.5 years; range 3–17) compared with 18 non-neoplastic lesions in 15 cats (median age 1 year; range 0.5–3). The most common non-neoplastic lesions included inflammation (in the testes, epididymis, tunics and ductus deferens), cryptorchidism, and one case each of polyorchidism and epididymal cyst formation. Two of the testes with inflammation were identified on immunohistochemical staining as feline coronavirus-infected and one pair of testes was associated with the presence of extracellular Gram-negative bacteria at the lesion site. Three different neoplastic lesions were identified, one each of Sertoli cell tumour, leiomyoma and fibrosarcoma.
Conclusions and relevance
Non-neoplastic testicular lesions were most common, including inflammation, cryptorchidism, polyorchidism and epididymal cysts. To our knowledge, leiomyoma and fibrosarcoma have not been reported in cat testes before and represent important differential diagnoses for testicular lesions.
Plain language summary
Most of the testicles examined from castrated cats because of disease noted by the owner had evidence of inflammation. Others were poorly developed because they had not descended correctly into the scrotum. Three cases of testicular cancer were noted and two of the types of cancer had not been reported in cats before but have been seen in humans.
Introduction
Testes pathology in cats includes non-neoplastic and neoplastic lesions. Non-neoplastic testicular lesions reported in cats include cryptorchidism, 1 polyorchidism, 2 epididymal cysts, 3 epididymitis, orchitis, periorchitis and vasitis, 4 with some cases of inflammation attributed to feline coronavirus (FCoV) infection in cats with feline infectious peritonitis (FIP).5,6
Testicular tumours are rare in cats and reports on them are limited.7 –10 Tumours can occur in both castrated and uncastrated cats, within the intra-scrotal testes, ectopic testes and extra testicular tissue. Sertoli cell tumours have been reported in uncastrated cats, 11 while interstitial cell tumours have been reported in both uncastrated12,13 and castrated cats.14,15 A case of a Sertoli cell and an interstitial cell tumour in a single uncastrated testis has been reported 16 and there are also reports of teratomas from cryptorchid testes.17,18
Keeping domestic cats is popular in Hong Kong despite limited living space of 13.6 m2 per person in a densely urban environment. 19 In the 2021 census, 4% of Hong Kong households kept a total of 184,000 cats and 77.2% of the households indicated the cats had been neutered 20 compared with 88% in New Zealand 21 and 83.6% in Australia, where early (<1 year) neutering is encouraged. 22
The aim of the present study was to review the aetiology and prevalence of neoplastic and non-neoplastic lesions recorded in cat testes from samples submitted over a 6-year period to CityU Veterinary Diagnostic Laboratory (CVDL) in Hong Kong.
Materials and methods
Case selection
The final histopathology reports for cat testes submitted to CVDL between May 2018 and January 2024 were identified by a keyword search of the laboratory information management system database using the terms ‘feline’, ‘testicle’ and ‘histopathology’. Cases where testes from cats had been submitted by veterinarians for histopathological examination were included in the submissions reviewed. Data collected included the histopathological diagnosis, age and breed as well as details about submitted testes, including location (scrotal, inguinal or abdominal), whether a single right or left testis, or both testes. Lesions were categorised as neoplastic or non-neoplastic using histopathological examination.
Tissue samples were fixed in 10% neutral buffered formalin at the veterinary clinics where the surgery was performed and then submitted to CVDL for histopathological examination. After fixation, a veterinary board-certified pathologist examined all the samples macroscopically, and representative sections were cut into blocks for routine processing and embedded in paraffin wax. The 4 µm-thick sections were stained with haematoxylin and eosin.
If inflammation was seen or bacterial infection suspected, selected slides were stained with Gram stain for bacteria. To investigate if collagen was produced, Masson’s trichrome stain was applied to selected slides.
For immunohistochemical (IHC) analysis, selected paraffin sections, along with positive and negative control tissue sections, were cut at 4 µm and attached to charged slides (Leica BOND Plus Slides; Leica Microsystems Limited). The antibodies used were anti-alpha-smooth muscle actin (α-SMA) (1:200, mouse polyclonal antibody; Dako), anti-desmin (1:100; mouse monoclonal antibody; Dako), anti-DOG1 (1:300, rabbit monoclonal antibody; Cell Marque), anti-FCoV (1:1000, polyclonal mouse antibody; BioRad) and anti-vimentin (1:100, polyclonal mouse antibody; Dako). Tissue sections with the primary antibody replaced by antibody diluent (Leica Biosystems Newcastle) served as negative controls. Normal feline lymph node (FCoV), gastrointestinal tract (α-SMA), skeletal muscle (desmin), caecum (DOG1) and skin (vimentin) were used as positive controls.
All stained histopathology slides were reviewed by specialist veterinary pathologists.
Data analysis
Cases were entered into an Excel spreadsheet (Microsoft) and imported into STATA 18 (StataCorp) for statistical analysis. The difference between neoplastic and non-neoplastic groups for age was evaluated using descriptive statistics.
Results
Sample population
Between 2018 and 2024, 26 testes were received from 18 cats for histopathological examination, collected by veterinarians at 14 veterinary clinics. The age range of the cats was 0.5–17 years (median age 1 year).
Nine different cat breeds were represented in the study population (Table 1), including domestic shorthair (n = 6, 33.3%) and British Shorthair (n = 5, 27.7%). The remaining breeds (American Shorthair, Chinchilla, Exotic Longhair, Exotic Shorthair, Ragdoll, Scottish Fold and Turkish Angora) were all represented by one (5.5%) cat.
Age, breed, lesion location and lesion type in 26 testes from 18 cats
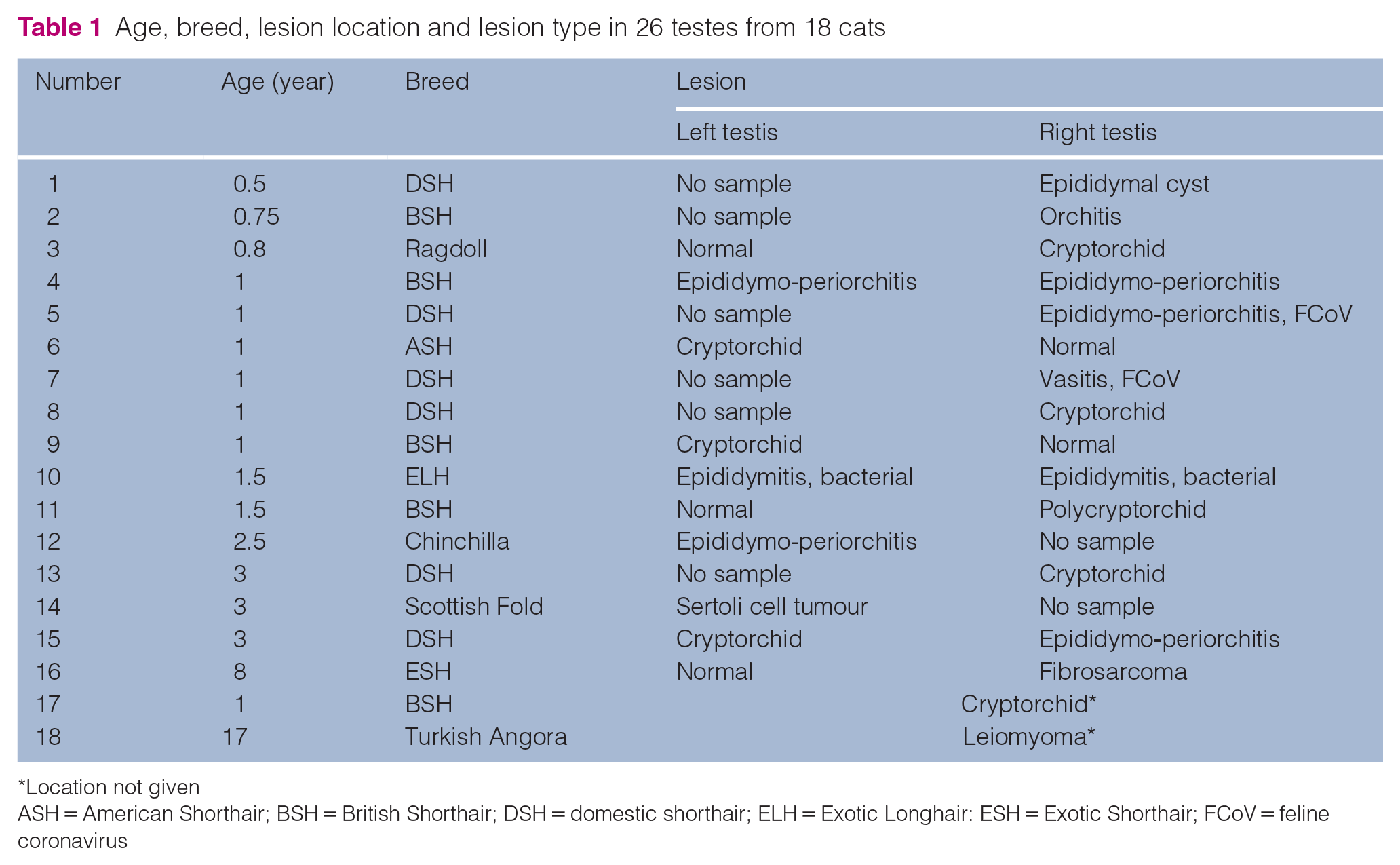
Location not given
ASH = American Shorthair; BSH = British Shorthair; DSH = domestic shorthair; ELH = Exotic Longhair: ESH = Exotic Shorthair; FCoV = feline coronavirus
The samples included eight pairs of testes, two single left testes, six single right testes and two in which the location was not specified (Table 1). Eight testes were from an inguinal or abdominal cryptorchid location, including three from the left, four from the right and one not specified.
The median age of cats with neoplastic lesions in the testes was 8 years (range 3–17), while non-neoplastic testicular lesions occurred more frequently in younger cats with a median age of 1 year (range 0.5–3).
There were five testes (three right and two left) that had no histopathological lesions detected submitted together with a diseased, contralateral testicle.
Non-neoplastic lesions
Non-neoplastic testicular lesions were most common, occurring in 15/18 (83.3%) cats. Seven cats had testes inflammation, five cats had only one testis submitted (four right testes, one left testis) and two cats had bilateral testes inflammation. Testes inflammation primarily involved the epididymis (epididymitis, 5/7, 71%), followed by the testicular tunic (periorchitis, 4/7, 57%), testis (orchitis, 3/7, 42.8%) and ductus deferens (vasitis, 1/7, 14.3%).
There were three cats with vasitis, epididymitis or orchitis, two cats with epididymitis and periorchitis, and two cats with orchitis, periorchitis and epididymitis. Gram staining was performed on five cats with testicular inflammation and was positive in one cat only with bilateral epididymitis where rare, Gram-negative, short, bacterial rods were seen. Bacterial culture was not requested or undertaken on any of the samples of testes with inflammation. Immunohistochemical staining for FCoV was performed in four cats with testicular pyogranulomatous vascular inflammation suggestive of FCoV infection and was positive in two cases, a cat with vasitis (Figure 1) and a cat with periorchitis.
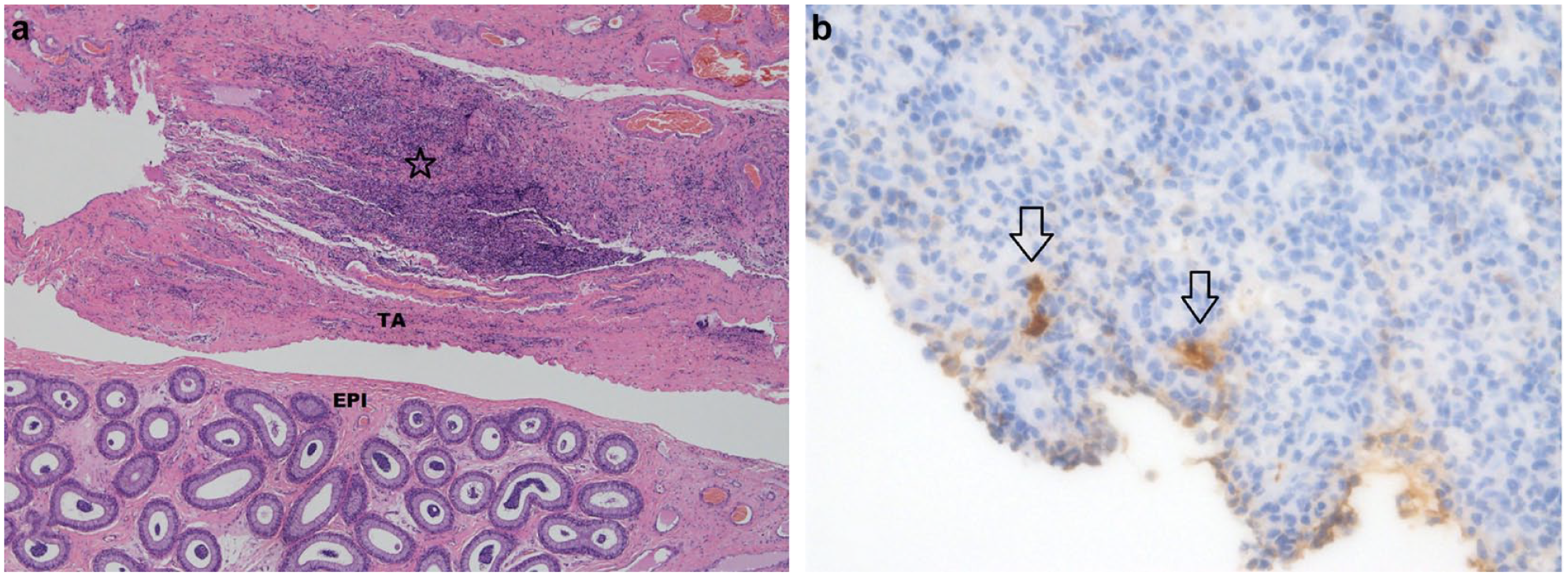
(a) A section of the testis including the epididymis (EPI) and tunica albuginea (TA) with perivascular inflammation in the vas deferens (star). Haematoxylin and eosin stain, 4×. (b) Feline coronavirus (FCoV) within the area of inflammation highlighted by FCoV immunohistochemical staining (arrows) 40×
There were eight atrophic cryptorchid testes (five right testes, three left testes, one unknown location), one epididymal cyst and one cryptorchid polyorchid testicle. A histopathological examination of the cryptorchid testes revealed a lack of spermatogonia and lack of development of the seminiferous tubules. The mean diameter of 10 seminiferous tubules of a cryptorchid testis (101 µm) and a non-cryptorchid testis (241 µm) from the same cat revealed a difference of 2.3 times, consistent with atrophy of the cryptorchid seminiferous tubules.
The polyorchid testis was found in the right abdominal cavity and consisted of three oval, 8, 10 and 11 mm, firm, distinct masses contained within a 40 mm long piece of adipose and connective tissue. Each of the three masses contained seminiferous tubules and ductus deferens (Figure 2), confirming a triorchid, cryptorchid testis. The left testis from this cat was normal and located in the scrotum.
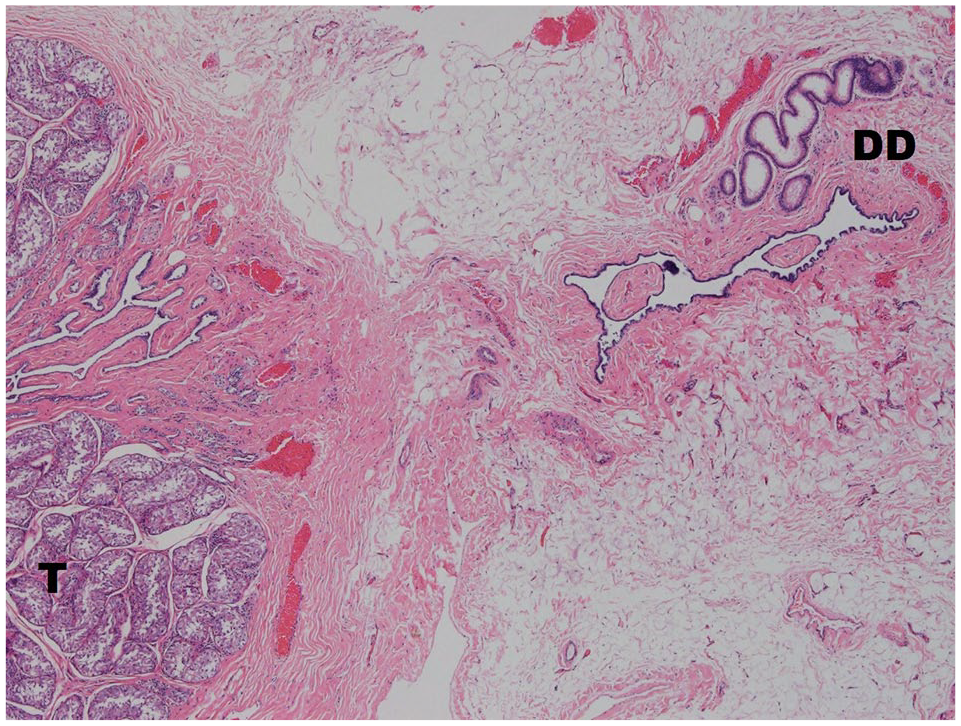
Polyorchidism; testis stroma (T) and ductus deferens (DD) in the abdominal cryptorchid triorchid testicle. Haematoxylin and eosin stain, 4 ×
One right testis had an epididymal cyst located within the epididymis with the cystic space lined by a single layer of ciliated cuboidal epithelial cells (Figure 3).
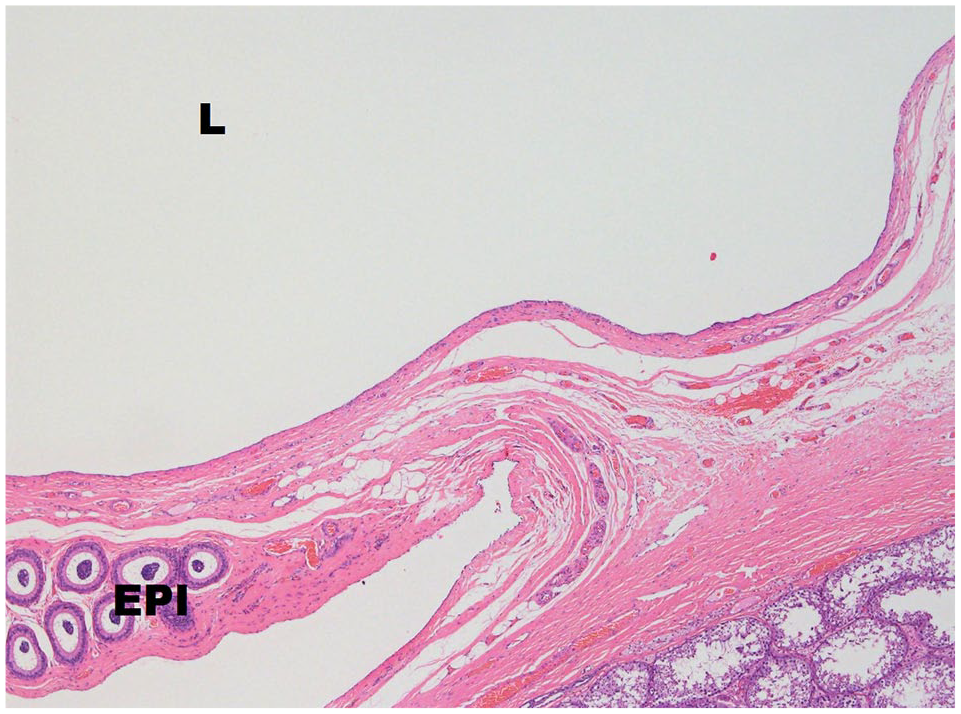
Epididymal cyst including the lumen (L) lined by cuboidal epithelium adjacent to the epididymis (EPI). Haematoxylin and eosin stain,4×
Neoplastic lesions
Neoplastic lesions were detected in 3/18 (16.7%) cats, including one each of Sertoli cell tumour, fibrosarcoma and leiomyoma. The Sertoli cell tumour was present in a diffusely enlarged left testis measuring 60 mm long and 20 mm in diameter. The right testis was not received from this cat. The lesion consisted of an expansile tan mass replacing most of the testis. The normal testis parenchyma was replaced by palisading, elongated, columnar, pleomorphic neoplastic Sertoli cells supported by a prominent fibrous stroma. The neoplastic cells had variably distinct cell margins, moderate amounts of eosinophilic to vacuolated cytoplasm and centrally located round to oval nuclei with vesicular chromatin and one to two distinct, magenta nucleoli. Five mitotic figures were seen per 10 high powered fields (hpf), representing an area examined of 2.37 mm2.
The fibrosarcoma was located in an enlarged right testis measuring 40 × 25 × 20 mm, while the smaller, normal, left testis measured 10 × 15 × 35 mm. The tumour contained within the tunica albuginea was poorly demarcated and infiltrative, consisting of neoplastic spindle cells forming interlacing bundles and fascicles on a fine fibrovascular stroma with peripheral small lymphocyte aggregates. The neoplastic cells were polygonal to spindle shaped with indistinct cell margins, moderate amounts of pale, sometimes vacuolated, eosinophilic cytoplasm, a round to oval nucleus with finely clumped chromatin and none to two indistinct magenta nucleoli. There was moderate anisocytosis and anisokaryosis (Figure 4a). Four mitotic figures were seen per 10 hpf (2.37 mm2). Seminiferous tubules in the adjacent testis were lined by vacuolated Sertoli cells and were devoid of spermatogonia. The neoplastic cells demonstrated uniform immunoreactivity with antibodies directed against vimentin (Figure 4b) and Masson’s trichrome stains highlighted collagen laid down between the tumour cells (Figure 4c), confirming a tumour of mesenchymal origin consistent with a fibrosarcoma.
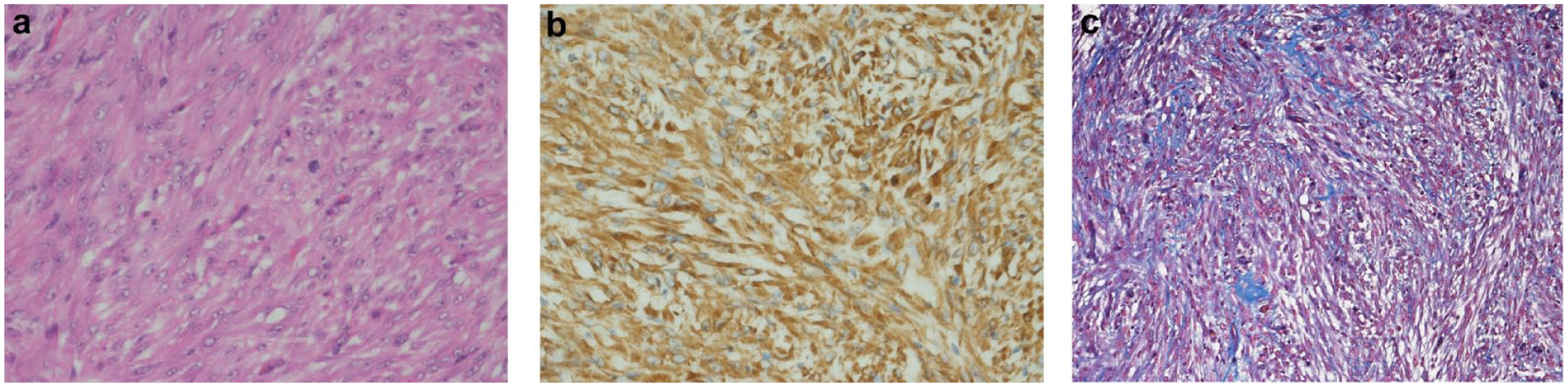
(a) Tunica albuginea fibrosarcoma; pleomorphic population of neoplastic spindle cells with frequent irregular mitotic figures. Haematoxylin and eosin stain, 40×. (b) Vimentin immunohistochemical staining highlights mesenchymal cells, 40×. (c) Collagen produced by the neoplastic cells stains blue. Masson’s trichrome, 40×
The leiomyoma was present in a single, diffusely enlarged testis (location not given) measuring 30 × 40 × 45 mm. The tumour was located within the tunica albuginea adjacent to the compressed and atrophic testis, epididymis and ductus deferens characterised by regular proliferation of short fascicles of plump, spindle-shaped interwoven cells. Individual cells had abundant eosinophilic cytoplasm and discernible cell borders. The nuclei varied from oval and elongated to cigar shaped (Figure 5a). Mitotic figures were rarely observed. The neoplastic cells demonstrated uniform immunoreactivity with antibodies directed against α-SMA (Figure 5b) but not desmin or DOG1.
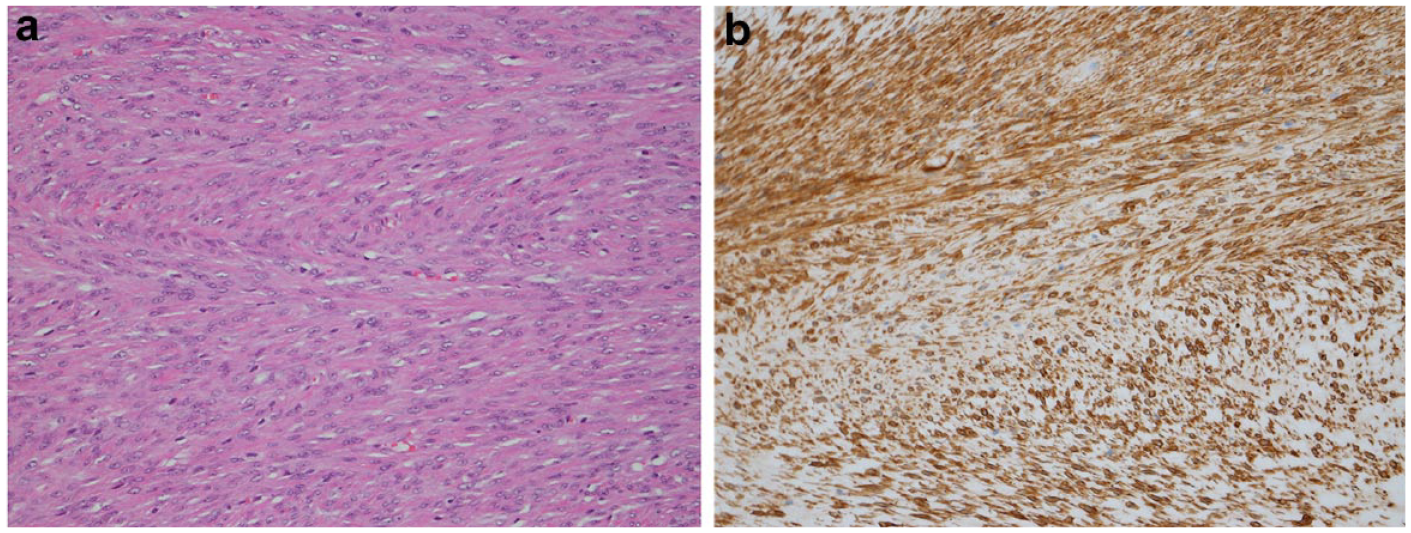
(a) Testis leiomyoma; uniform population of neoplastic spindle cells with abundant eosinophilic cytoplasm and centrally located ovoid nuclei. Haematoxylin and eosin stain, 20×. (b) Alpha-smooth muscle actin immunohistochemical staining highlights smooth muscle cells, 40×
Discussion
Non-neoplastic lesions were the most common finding in testes samples collected from domestic cats in Hong Kong undergoing histopathological examination. Most of the non-neoplastic lesions were inflammatory, including one associated with the presence of Gram-negative bacteria and two associated with FIP.
Cryptorchidism in cats has a reported incidence of 1.0–1.3%. 23 Although the location of the cryptorchid testis was not always described in the histopathology submission forms, a previous study found they were mostly located in the inguinal canal, while abdominally located testes were less common. 23 Incompletely descended testes have an increased risk of testicular torsion and neoplasia development, hence cryptorchidectomy is recommended. 24 The histopathological findings of cryptorchid testes in our study are similar to those previously reported, including atrophy, a decrease in seminiferous tubule diameter and absent spermatogonia, compared with non-cryptorchid cat testes. 25
We identified one case of multiple testes in a cat with a right-sided, abdominal, triorchid testis. Polyorchidism is a rare finding in cats and previous reports found abdominally located polyorchid testes.2,26,27
Another unusual non-neoplastic lesion in our study was the epididymal cyst within the right epididymis of a 5-month-old cat. Epididymal cysts are rare in cats and have been previously reported only once in an 8-month-old cat in the head of the right epididymis. 3 They are most frequently developmental in origin owing to abnormalities of the rete testis but can also be associated with trauma in humans. 28
Two of the neoplastic lesions in our study, a fibrosarcoma and a leiomyoma, have not previously been described in cats. Testicular leiomyoma is a rare tumour in animals and has been described in a ram, 29 dog 30 and horses.31,32 Leiomyomas occur most commonly in the reproductive and gastrointestinal tracts containing abundant smooth muscle but can also arise from smooth muscle in the skin and subcutis, spleen, respiratory tract and blood vessels. 33 A testicular leiomyoma could arise from the smooth musculature of the testicular blood vessels or the contractile smooth muscle cells of the tunica albuginea thought to be associated with the motility of semen transport. 34
Our study found the leiomyoma stained strongly with α-SMA but did not stain with desmin or DOG1. This has been previously reported in another study, where the recommended IHC staining panel to identify non-visceral leiomyoma or leiomyosarcoma included the application of α-SMA, while desmin was not found to be useful. 35 DOG1 should be applied to rule out a gastrointestinal stromal tumour (GIST) in all visceral organs including the gastrointestinal tract, genital tract, urinary tract, liver and spleen. 35
Testicular fibrosarcoma is also a rare tumour not previously described in domestic pets or farm animals 7 nor in previous surveys of testicular tumours in dogs 36 but has been described in humans. 37
A Sertoli cell tumour was diagnosed in the left testis of one cat and this tumour has been described in cats many times.11,15,16
Testicular neoplasia is rarely reported in cats. 8 A lack of diagnostic reports may be due to early castration before lesions have time to develop, low neoplasia incidence or testes with lesions are not submitted for histopathological examination. Based on the authors’ experience, pet ownership of older uncastrated cats is more common in Hong Kong than in other regions where testicular pathology has been reported in cats. Few data are available, but a survey of neoplasia in 3266 cats in Portugal 9 and urogenital lesions in eight cats in Poland did not identify any male genital tumours. 38
Conclusions
Our retrospective survey (although based on a limited number of cases) describes two testicular neoplastic lesions not previously reported in cats, including a fibrosarcoma in an 8-year-old cat and a leiomyoma in a 17-year-old cat. Both tumours were located in the tunica albuginea. Other rare findings included a single case of abdominal triorchidism and a single case of epididymal cyst formation. Submission of castrated testes with lesions is recommended for histopathological examination in cats of all ages to confirm the diagnosis and provide prognostic information.
Footnotes
Acknowledgements
We thank our contributing veterinarians for providing biopsies for analysis. We thank the histology technologists for processing the samples.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognised high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers) for all procedure(s) undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
