Abstract
Case series summary
Four confirmed cases of xanthinuria in cats, and one suspected case based on pedigree analysis, were identified. Clinical presentations varied and included haematuria, pollakiuria, dysuria, and urethral and ureteral obstruction. All cats had upper urinary tract uroliths. Diagnosis was obtained through infrared mass spectrometry of uroliths or urine. Clinical signs commenced at 3–8 months of age and reduced in all cats in the medium to long term after the introduction of a protein-restricted diet. Four cats were castrated males and one was a spayed female. Cases consisted of four Munchkin pedigree cats and one unrelated domestic shorthair cat. All four affected Munchkin pedigree cats were related, with three cases full siblings and the fourth case a half-sibling. No connection to the Munchkin pedigree could be established for the domestic shorthair cat. A candidate causative genetic variant (XDH p.A681V) proposed for this cat was excluded in the Munchkin family.
Relevance and novel information
All affected cats presented diagnostic challenges and routine urinalysis was insufficient to obtain a diagnosis. Cases of feline xanthinuria may be underdiagnosed due to situations where uroliths cannot be retrieved for analysis and there is an inability to make a diagnosis using crystal morphology alone on routine urinalysis. Metabolic screening of urine may provide an effective mechanism to confirm xanthinuria in suspected cases where uroliths are inaccessible or absent. In this case series, male cats were more common. Their anatomy may increase the risk of lower urinary tract signs and urethral obstruction developing secondary to xanthine urolithiasis. A protein-restricted diet appears to reduce clinical signs as part of long-term management.
Plain language summary
Four closely related Munchkin cats and one domestic shorthair cat were found with a suspected genetic disease causing high levels of xanthine in their urine. The case series looks at similarities and differences in their clinical signs, as well as difficulties experienced in obtaining a correct diagnosis. All cats had upper urinary tract stones and required metabolic testing of the stones or urine to diagnose. All cats were young when their clinical signs started and were on a high-protein diet. Four cats were desexed males and one was a desexed female. A genetic variant that may have caused the disease in the domestic shorthair cat was ruled out in the Munchkin family. Cases of high xanthine levels in feline urine may be underdiagnosed as the stones may not be accessed for testing. In this case series, male cats were more common. Their anatomy may increase the risk of lower urinary tract signs. A protein-restricted diet appears to reduce clinical signs as part of long-term management.
Keywords
Introduction
Xanthine uroliths account for 0.1–0.5% of feline calculi.1–3 Xanthinuria occurs when defective purine metabolism prevents the conversion of xanthine to uric acid (Figure 1). Xanthine accumulates in plasma, is excreted unchanged in urine, and due to poor solubility, can precipitate into uroliths.4,5 While large uroliths can occur,6,7 other presentations include profuse small, amorphous, birefringent crystalluria or gravel-like urolithiasis.4,8,9 Xanthinuria can also be subclinical, further complicating diagnosis.5,10–13
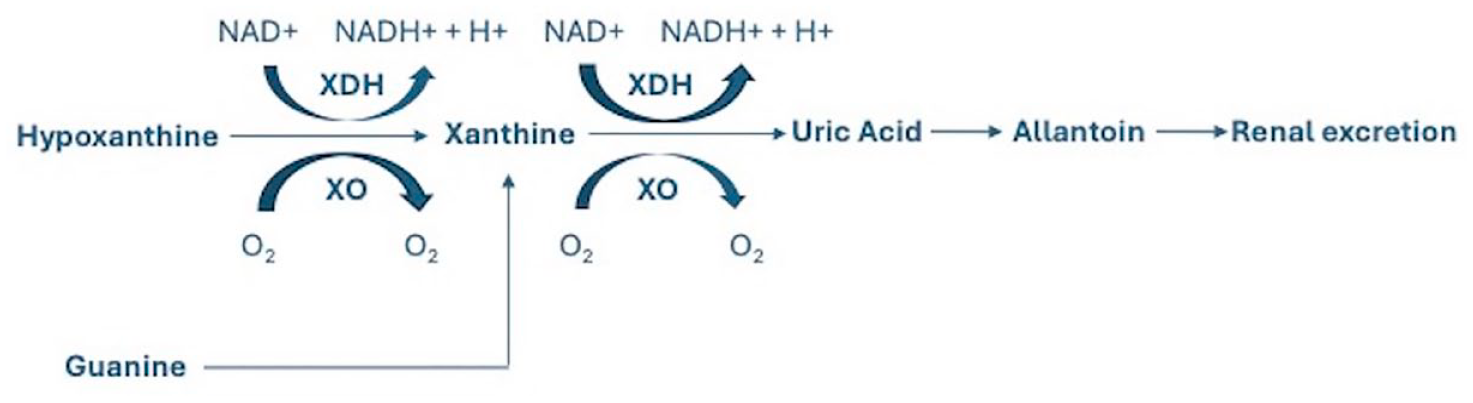
Final steps of purine metabolism showing the role of xanthine oxidase/dehydrogenase (XO/XDH) in the conversion of hypoxanthine to xanthine and uric acid in normal purine metabolism in animals. Hypoxanthine can be converted to inosine monophosphate as part of purine salvage, preventing the accumulation of hypoxanthine in the absence of XO/XDH activity. However, once xanthine is produced from hypoxanthine or guanine, the process becomes irreversible, resulting in accumulation of xanthine in the absence of XO/XDH activity. NAD = nicotinamide adenine dinucleotide; NADH = nicotinamide adenine dinucleotide + hydrogen
In the absence of xanthine dehydrogenase (XDH) inhibitory therapy, xanthinuria is typically hereditary, with an autosomal recessive mode of inheritance, and results from variants affecting the function of XDH (type I xanthinuria) or its cofactor molybdenum cofactor sulfurase (MOCOS) (type II xanthinuria). Causative genetic variants associated with xanthinuria have been identified in humans, dogs, goats and cattle.5,9,11–29 In cats, the condition is rarely reported, and to date, one exonic candidate variant affecting XDH has been proposed in this species (case 1 of this case series). 30
In humans and dogs, purine urolithiasis is effectively managed with a protein-reduced, purine-restricted diet.9,31–34 The condition is potentially more difficult to manage in cats due to their innate protein requirements as obligate carnivores.
Case series description
Four confirmed cases and one suspected case of xanthinuria in cats were identified. The first cat was a domestic shorthair adopted from a rescue organisation as a kitten and no relationship could be established with the other cases. The four other cases were related Munchkin pedigree cats, three full-siblings and one half-sibling (Figure 2). Breeders mate short-legged phenotype Munchkins with domestic cats (non-dwarf phenotype) to avoid producing homozygous kittens because homozygosity for the causal variant is lethal. 35 In this family, all four affected Munchkin pedigree cats were offspring of the same tom, an Australian domestic shorthair. Cases 2, 4 and 5 were the offspring of the same queen, a short-legged Munchkin imported from France and with no familial relationship to the tom. Case 3 was the granddaughter of the same queen. No cats had received treatment with an XDH inhibitor such as allopurinol. Upper urinary tract uroliths were present in all five cases and crystalluria in four cases.

Pedigree analysis of the Munchkin family showing the Munchkin/domestic shorthair (DSH) phenotype, affected status for xanthinuria and case number
Case 1
An 8-month-old castrated male domestic shorthair cat presented with 24 h of pollakiuria and dysuria. The cat had been acquired 2 months earlier and was fed a routine commercial diet (ZIWI Peak Mackerel & Lamb Wet Cat Food, 55.1% protein as % dry matter basis [DMB]; ZIWI Peak Venison Wet Cat Food, 54.2% protein DMB [https://ziwipets.com/en-au/pages/typical-analysis, accessed 10 January 2024]). Urinalysis (cystocentesis) showed a urine specific gravity (USG) of 1.016, pH 5.0 and 4+ blood. Marked crystalluria and >10 small (1 mm diameter) uroliths were grossly visible in the urine. Urine culture was negative. Haematology, biochemistry and abdominal radiographs were unremarkable. Treatment included meloxicam and diazepam.
One month later, urinalysis revealed cloudy urine, USG of 1.027, pH 6.0, 1+ protein, 4+ blood and round fan-like crystals. Abdominal ultrasound identified bilateral nephroliths that appeared larger within a dilated left renal pelvis. Due to potential risks of left ureteral obstruction, and to retrieve nephroliths for analysis, a left-sided nephrotomy was performed. Hundreds of small nephroliths were removed. Infrared spectrometry of the nephroliths confirmed xanthine (Minnesota Urolith Center, College of Veterinary Medicine, St Boyd, MN, USA). Dietary management was commenced with a veterinary nutritionist-formulated diet (Veterinary Nutrition Group, Sydney, NSW, Australia – 33.80–35.3% protein dry matter basis [DMB], purine-restricted, 75–80% moisture, target urine pH 7.1–7.5) and a commercial renal diet (Hills k/d Kidney Care with Chicken Wet Cat Food, 29% protein DMB [www.hillspet.com/cat-food/pd-kd-feline-with-chicken-canned], Hills k/d Kidney Care with Chicken Dry Cat Food, 28.9% protein DMB [www.hillspet.com/cat-food/pd-kd-feline-dry].
The cat had one further episode of dysuria and renal parameters were static at follow-up to 24 months later (creatinine 130 µmol/l, reference interval [RI] 89–200; urea 6.5 mmol/l, RI 5.0–15.0; symmetric dimethylarginine [SDMA] 15 µg/dl, RI 0–14). Case 1 was unrelated to the other four cases and was acquired from a rescue shelter and had unknown ancestry. Whole-genome sequencing results for this case have previously been reported and revealed homozygosity for a candidate causal variant for xanthinuria affecting XDH (p.A681V). 30
Case 2
A 7-month-old castrated male Munchkin cat presented with 12 h of dysuria and subsequent urethral obstruction. The cat was fed varied commercial kitten diets (protein content in the range of 44–47% DMB) and raw meat (chicken, broiler or fryers, breast, skinless, boneless, meat only, raw, 86.2% protein DMB; chicken, ground, raw, 64.9% protein DMB; fish, tuna, light, canned in water, without salt, drained solids, 90.9% protein DMB [as analysed by the Veterinary Nutrition Group]). Haematology and biochemistry revealed creatinine 198 µmol/l (RI 71–212), urea 21.2 mmol/l (RI 5.7–12.9) and phosphate 2.24 mmol/l (RI 1–2.42). Under general anaesthesia, retropulsion relieved the obstruction and a urethral catheter with a closed urine collection system was placed. Urinalysis identified a USG of 1.022, pH 6.5, trace blood and amorphous crystals. Treatment included intravenous fluid therapy, prazosin, gabapentin and buprenorphine. During hospitalisation, the urinary catheter was repeatedly obstructed with large volumes of pasty, yellow, sand-like material.
Abdominal radiographs and a retrograde urethrogram performed under general anaesthesia were unremarkable. Abdominal ultrasound showed multiple small nephroliths in both renal pelvises. The bladder wall was thickened and there was dependent hyperechoic material within the lumen and urethra (Figures 3 and 4). The urinary catheter was removed after 36 h and the cat was discharged after 4 days. The cat re-presented 24 h later with recurrence of pollakiuria, and dantrolene was commenced. External laboratory urinalysis identified yellow, polarising, birefringent amorphous crystals suspected to be urate (Figure 5). Pre- and postprandial bile acids were within normal limits. Xanthinuria was confirmed by urine infrared mass spectrometry (Victorian Clinical Genetics Service, The Royal Children’s Hospital, Parkville, VIC, Australia).

Case 2. Sagittal view ultrasound of hyperechoic sediment (white arrows) with post-acoustic shadowing in the urinary bladder

Case 2. Longitudinal view ultrasound showing multiple hyperechogenicities (white arrows) within the urethral lumen

Case 2. Yellow, polarising, birefringent amorphous crystals identified on urinalysis
One month later, the cat re-presented with marked azotaemia (creatinine 700 µmol/l) and bilateral ureteral obstruction was diagnosed using ultrasound. The left kidney was 3.2 cm long with a renal pelvic dilation of 0.53 cm, and the right was 3.4 cm long with a renal pelvic dilation of 0.82 cm (Figure 6); both proximal ureters were dilated with hyperechoic ureteroliths identified. Bilateral subcutaneous ureteral bypass devices (SUBs; Norfolk Vet Products) were placed. The azotaemia reduced and stabilised at International Renal Interest Society (IRIS) stage 3 chronic kidney disease. The cat was managed with a veterinary nutritionist-formulated diet (Veterinary Nutrition Group, Sydney, NSW, Australia - 33.80–35.3% protein dry matter basis [DMB], purine-restricted, 75–80% moisture, target urine pH 7.1–7.5) and a commercial renal diet (Hills k/d Kidney Care with Chicken Wet Cat Food, 29% protein DMB [https://www.hillspet.com/cat-food/pd-kd-feline-with-chicken-canned, accessed 10 January 2024]; Hills k/d Kidney Care with Chicken Dry Cat Food, 28.9% protein DMB [https://www.hillspet.com/cat-food/pd-kd-feline-dry, accessed 10 January 2024]).

Case 2. Sagittal plane view ultrasound of the right kidney showing hydronephrosis
The SUBs were monitored using ultrasound-guided saline flushes every 1–3 months. Over time, the SUBs intermittently appeared to be partially or completely obstructed at the nephrostomy catheter end based on an inability to document saline flow into the renal pelvis. Renal parameters, urinalysis and culture were monitored. At 1073 days after original presentation, the cat was clinically well, had a stable body weight, body condition score of 4–5/9 and had IRIS stage 3 chronic kidney disease (creatinine 292 µmol/l, RI 68–167; urea 15.6 mmol/l, RI 4.3–11.7). The genetic variant XDH p.A681V 30 was excluded by PCR for this case.
Case 3
A 5-month-old spayed female Munchkin cat was referred for persistent haematuria and intermittent periuria since acquisition at the age of 3 months. The cat was fed a commercial kitten food (Royal Canin Kitten Wet Cat Food, 54.5% protein DMB [calculated from analysis listed at https://www.royalcanin.com/au/cats/products/retail-products/kitten–chunks-in-gravy-4058, accessed 10 January 2024]; Royal Canin Kitten Dry Cat Food, 38.7% protein DMB [calculated from analysis listed at https://www.royalcanin.com/au/cats/products/retail-products/kitten-2522, accessed 10 January 2024]) and raw meat (chicken, broiler or fryers, breast, skinless, boneless, meat only, raw, 86.2% protein DMB; chicken, ground, raw, 64.9% protein DMB; fish, tuna, light, canned in water, without salt, drained solids, 90.9% protein DMB [Veterinary Nutrition Group, 2024]). Idiopathic cystitis was initially suspected and was non-responsive to treatment with meloxicam, decapeptide alpha-casozepine (Zylkene; Vetoquinol), synthetic feline facial pheromone (Feliway; Ceva) and a commercial urinary diet (Royal Canin Urinary S/O Wet Cat Food, 58.8% protein DMB [calculated from analysis listed at https://www.royalcanin.com/au/cats/products/vet-products/urinary-so-1254, accessed 10 January 2024]; Royal Canin Urinary Care Dry Cat Food, 35% protein DMB [calculated from analysis listed at https://www.royalcanin.com/au/cats/products/vet-products/urinary-so-3901, accessed 10 January 2024]). Haematology was unremarkable. Biochemistry identified SDMA 23 µg/dl (RI 0–14), urea 16.9 mmol/l (RI 5.0–15.0) and phosphate 3.2 mmol/l (RI 1.0–2.3). Urinalysis revealed a USG of 1.024, pH 6.5, 4+ blood and 3+ protein without crystalluria. Urine culture was negative.
Urinary tract ultrasound identified hyperechoic urinary bladder sediment, marked bilateral hydronephrosis (measurements not available) and small hyperechoic nephroliths. Ureteral dilation was not present. A repeat urinalysis identified small, round, yellow-tan-coloured crystals (Figure 7). Xanthinuria was confirmed by tandem mass spectrometry (Victorian Clinical Genetics Service, The Royal Children’s Hospital, Parkville, VIC, Australia). The cat was managed with a veterinary nutritionist-formulated diet (Veterinary Nutrition Group, Sydney, NSW, Australia - 33.80–35.3% protein dry matter basis [DMB], purine-restricted, 75–80% moisture, target urine pH 7.1–7.5). Three years later, the cat remained clinically well, with creatinine of 200 µmol/l (RI 80–200), urea 17.5 mmol/l (RI 5.0–15.0), SDMA 16 µg/dl (RI 0–14) and USG 1.021. The genetic variant XDH p.A681V 30 was excluded by PCR for this case.
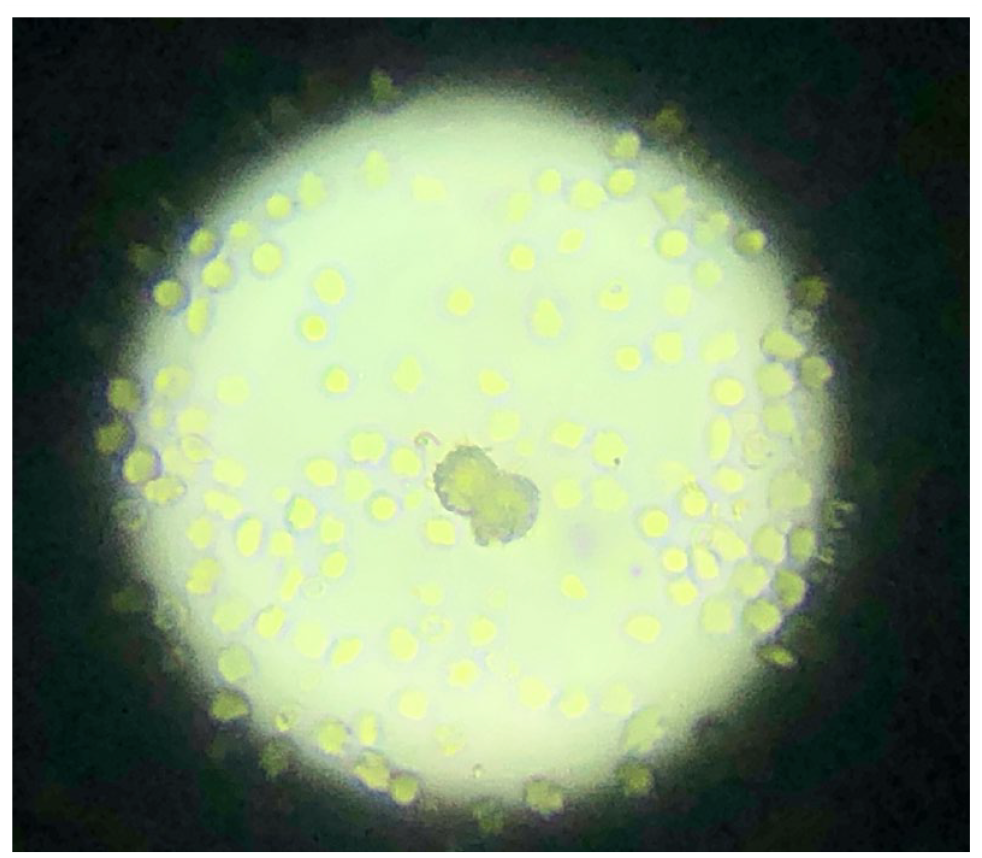
Case 3. Yellow-tan round crystals identified on urinalysis
Case 4
An 18-month-old castrated male domestic shorthair cat, the offspring of a Munchkin queen, was referred after recurrent intermittent haematuria from 4 months of age. The cat had been managed with a commercial urinary diet (Hills c/d Multicare Wet Cat Food, 41.4–44.1% protein DMB [https://www.hillspet.com/cat-food/pd-cd-multicare-feline-with-chicken-canned, accessed 10 January 2024]; Hills c/d Multicare Dry Cat Food, 33.8% protein DMB [https://www.hillspet.com/cat-food/pd-cd-multicare-feline-stress-dry, accessed 10 January 2024]). Urinalysis (cystocentesis) revealed cloudy urine, USG 1.028, pH 6.5, protein 2+, blood 3+ and increased white cell count (60 ×106/l, RI 0–20). Large amounts of amorphous crystals were present, which were originally identified as sulfonamide crystals by an external laboratory. Urine culture was negative. Haematology and biochemistry showed urea 15.1 mmol/l (RI 5.36–12.14) and creatinine 156 µmol/l (RI 88–194). Abdominal ultrasound identified a smaller left kidney (left 3.42 cm and right 4.02 cm in length), bilateral small nephroliths and non-shadowing dependent bladder sediment. Abdominal radiographs were unremarkable.
Treatment included meloxicam, dantrolene and prazosin, and the cat subsequently urinated approximately 20 small uroliths. Voiding urohydropropulsion was then performed under general anaesthesia and >200 small (<0.5 mm diameter) yellow-brown uroliths were retrieved and identified as xanthine via infrared spectrometry (Minnesota Urolith Center, College of Veterinary Medicine, St Boyd, MN, USA). A protein-restricted diet (commercial renal diet, then veterinary nutritionist-formulated diet [Veterinary Nutrition Group, Sydney, NSW, Australia - 33.80–35.3% protein dry matter basis [DMB], purine restricted, 75–80% moisture, target urine pH 7.1–7.5]) was commenced. Due to persistently low urine pH of 6.0–6.5, potassium citrate (75 mg/kg q24h PO) was used to alkalinise the urine to reduce xanthine precipitation. Renal parameters and USG remained static, and episodes of haematuria reduced in frequency over 3.5 years of follow-up. The genetic variant XDH p.A681V 30 was excluded by PCR for this case.
Case 5
A 6-month-old castrated male Munchkin cat presented for acute vomiting. The cat was fed both grain-free (Wellness Core Grain Free Dry Kitten Food, 50% protein DMB [calculated from analysis listed at https://wellnesspetfood.com.au/cat-wellness/natural-cat-food/core-grain-free-dry-cat/kitten-turkey/, accessed 10 January 2024]) and routine commercial diets (Whiskas Wet Kitten Food, 50% protein DMB [calculated from analysis listed at https://www.whiskas.co.uk/cat-food/wet-cat-food/classics/whiskas-kitten-2-12-months-poultry-feasts-jelly-wet-kitten-food-pouches-12-x-85g, accessed 10 January 2024]). The cat was small for its age, weighing 1.5 kg. Bilateral renomegaly was detected on physical examination and radiographs (Figures 8 and 9). Haematology and biochemistry revealed urea 16 mmol/l (RI 3.6–10.7), creatinine 136 µmol/l (RI 27–186) and potassium 2.6 mmol/l (RI 3.7–5.8). Treatment included buprenorphine and intravenous fluid therapy. Clinical signs resolved over 24 h.

Case 5. Right lateral abdominal radiograph showing marked bilateral renomegaly. Two small radiopaque renal mineralisations (black arrows) are visible

Case 5. Ventrodorsal abdominal radiograph showing marked bilateral renomegaly. Two small radiopaque renal mineralisations (black arrows) are visible
Three days later at a different hospital, the cat was well but renomegaly was still apparent. Biochemistry revealed urea 14 mmol/l (RI 5.4–10.7), creatinine 99 µmol/l (RI 70–160) and potassium 5.2 mmol/l (RI 3.5–5.8). Abdominal ultrasound showed numerous small (1–2 mm) uroliths within the urinary bladder and bilateral renomegaly (left kidney 3.35 cm and right 3.87 cm in length). Bilateral pyelectasia was present (width of renal pelvis: left = 0.5 cm and right = 0.59 cm). Both kidneys had mildly increased echogenicity and reduced corticomedullary distinction. Both renal pelvises and diverticula contained numerous small hyperechoic nephroliths, with the largest up to 3.8 mm in the left kidney and 1.9 mm in the right kidney. A 1.5 mm hyperechoic focus was identified in the right distal ureter and was documented to be moving along the ureter. No ureteral dilation was present. A repeat ultrasound 24 h later showed that the ureterolith was no longer present. Urinalysis performed after fluid therapy showed a USG of 1.015, pH 6.0 and 3+ blood without crystalluria. Bacterial culture (cystocentesis and pyelocentesis) was negative.
The cat was suspected to have calcium oxalate calculi (based on urine pH and location within the proximal urinary tract) and was commenced on a commercial urinary diet (Hills c/d Multicare Wet Cat Food, 41.4–44.1% protein DMB [https://www.hillspet.com/cat-food/pd-cd-multicare-feline-with-chicken-canned, accessed 10 January 2024]; Hills c/d Multicare Dry Cat Food, 33.8% protein DMB [https://www.hillspet.com/cat-food/pd-cd-multicare-feline-stress-dry, accessed 10 January 2024]) for suspected calcium oxalate calculi (based on urine pH and location within the proximal urinary tract) was commenced. Two months later, the cat was well and had gained weight (2.1 kg). Urinalysis (cystocentesis) revealed a USG of 1.016, pH 6.0 and 4+ blood. Ultrasound showed that both kidneys were smaller (left 3.1 cm and right 3.4 cm in length). The left renal pelvis was dilated (0.66 cm wide) with a subjective increased amount of dependent hyperechoic sediment. The right renal pelvis was within normal size limits and contained hyperechoic sediment. No ureteral dilation was present. The urinary bladder contained a marked amount of dependent hyperechoic content with acoustic shadowing, including a urolith measuring 1.7 mm. The cat was euthanased 10 months later after two episodes of urethral obstruction. Xanthinuria was not confirmed; however, pedigree analysis identified the cat as the littermate of case 4.
Discussion
Clinical presentations were varied and included haematuria, pollakiuria, dysuria, and urethral and ureteral obstruction. However, all affected cats had upper urinary tract uroliths present on imaging, and four cases had crystalluria characterised as amorphous. The presence of these clinical signs should trigger further investigation for possible xanthinuria.
In all identified cases, clinical signs commenced at a young age (3–8 months). Clinical signs of recurrent lower urinary tract disease, or nephro-/ureterolithiasis in the absence of another definitive diagnosis, may be indications for further investigation for possible xanthinuria in young cats.
The affected cats were all fed a moderate to high protein diet at the time clinical signs emerged. After a range of acute interventions, the affected cats were placed onto a protein-restricted diet that was continued for the duration of the follow-up (24–42 months). Although repeat imaging was not carried out in all cases, so reduced urolithiasis cannot be confirmed, all the cats had reduced clinical signs (haematuria, pollakiuria, urinary tract obstruction events) and stable renal biochemistry for the duration of the follow-up. While further research is required to confirm the efficacy of dietary management, a moderate protein, purine-reduced diet appears to reduce overt clinical signs.
In the four confirmed cases, a diagnosis of xanthinuria was obtained after infrared mass spectrometry of uroliths or urine. Routine urinalysis was insufficient to confirm the crystal type and uroliths were retrieved for testing in only two cases, one after nephrotomy. Metabolic screening of urine may provide a non-invasive mechanism to confirm a diagnosis of xanthinuria where uroliths are not readily accessible.
In one case, xanthinuria was not confirmed; however, the cat was a littermate of an affected cat and the clinical history was suggestive. Where available, a pedigree analysis may assist in identifying targets for metabolic screening.
In dogs and humans, xanthinuria does not necessarily result in the precipitation of xanthine crystals or uroliths.5,10–13 While all cases in this series had clinical signs, urine metabolite testing was not carried out on clinically normal related cats, and genetic analysis is currently in progress. It is unknown if additional clinically normal cats with xanthinuria are present in this family. The prevalence of xanthinuria in cats may therefore be underestimated. Where there is a strong index of suspicion and in the absence of a known causative genetic variant, urine mass or infrared spectrometry should be considered, even in the absence of crystalluria.
In humans, two-thirds of classical xanthinuria cases are male 36 and in dogs, xanthinuria has been reported in males in 62.5% of cases. 11 In cats, reported cases of xanthinuria are infrequent and show an even sex distribution.4,6–8 In this case series, however, 4/5 cases were male. Anatomical features of the male urinary tract make them more likely to present with urethral obstruction secondary to urolith formation.
The candidate genetic variant affecting XDH proposed for case 1 (XDH p.A681V) 30 was excluded in the Munchkin family. Based on inheritance patterns in other species, an autosomal recessive mode of inheritance is suspected in this Munchkin cat family; however, other modes of inheritance or multiple cumulative variants cannot be excluded. Additional genetic analysis is in progress for the Munchkin family and a domestic cat population prevalence study is planned.
Conclusions
An autosomal recessive mode of inheritance for xanthinuria in the Munchkin pedigree cats in this case series is suspected. The prevalence of a causative genetic variant within the wider Munchkin and domestic cat populations is unknown. Broader screening of Munchkin and non-Munchkin cats for xanthinuria is desirable to ascertain the prevalence among both populations. Line breeding of cats from Munchkin pedigrees may increase the risk of producing homozygous affected offspring.
Feline xanthinuria can lead to serious clinical disease and presents notable diagnostic and treatment challenges. Regardless of breed, young cats presenting with unidentifiable crystalluria and upper urinary tract urolithiasis should be assessed for xanthinuria to facilitate optimal treatment including, where appropriate, dietary management. Metabolic screening of urine should be considered in the absence of accessible uroliths.
Footnotes
Acknowledgements
The authors thank Seddon Vet Hospital and Ringwood Veterinary Clinic for follow-up case information.
Author note
This case series was presented as an oral abstract at the 2023 Science Week conference of the Australian and New Zealand College of Veterinary Scientists.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognised high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers) for all procedure(s) undertaken (prospective or retrospective studies). For any animals or people individually identifiable within this publication, informed consent (verbal or written) for their use in the publication was obtained from the people involved.
