Abstract
Feline xanthinuria is a rare autosomal recessive disorder of purine metabolism due to genetic mutations in the xanthine dehydrogenase (XDH) gene. It is characterised by excessive excretion and accumulation of xanthine in the urine, which can lead to the formation of xanthine uroliths. Xanthine uroliths may be present in both the upper and lower urinary tracts, causing clinical signs associated with renal disease and feline lower urinary tract disorders (FLUTDs). Hallmark diagnostic findings of xanthinuria are elevated xanthine and hypoxanthine, and reduced uric acid concentrations in serum and urine. Uroliths can be submitted for compositional analysis to confirm the presence of xanthine and definitive diagnosis for xanthinuria. Management involves dietary modification to purine-restricted diets and increased fluid intake. Commercially available renal diets are preferred over urinary diets because of their lower protein composition, and consulting a veterinary nutritionist is strongly recommended. Urinary alkalisation is not considered an effective method for the dissolution of xanthine uroliths owing to their poor solubility. Despite these interventions, recurrence of xanthine urolithiasis is possible. Given the limited treatment options and risk of recurrence, feline xanthinuria is a lifelong condition that requires ongoing management and monitoring to mitigate complications. This review will provide an overview of the current understanding of the pathophysiological, metabolic and genetic aspects of the disorder and discuss current diagnostic approaches, management strategies and clinical expectations of feline xanthinuria. Findings from this review highlight the need for greater recognition of feline xanthine urolithiasis as a cause of FLUTD, given current gaps in diagnostic methods and treatment options. A deeper understanding of the condition will help veterinarians accurately differentiate it from other causes of FLUTD and support further research aimed at improving the detection, prevention and management of xanthinuria.
Plain language summary
Feline xanthinuria is a rare inherited condition caused by mutations in the xanthine dehydrogenase (XDH) gene, leading to high levels of xanthine in the urine. This can result in the formation of xanthine stones (uroliths) in the urinary tract causing signs of kidney disease or feline lower urinary tract disorder (FLUTD). Diagnosis involves detecting high levels of xanthine and hypoxanthine, and low levels of uric acid in blood and urine. Urinary stone samples can be submitted for laboratory analysis to confirm xanthine composition of the stone. Managing xanthinuria requires a low-purine diet and increased water intake. Renal diets are preferred over urinary diets because of their lower protein content; seeking advice from a veterinary nutritionist is strongly recommended. Unlike other types of stones, xanthine uroliths do not dissolve easily; therefore, altering the pH of urine is not effective. Even with treatment, stones may recur, making lifelong monitoring essential. This review outlines the current understanding of the genetic, metabolic and clinical aspects of feline xanthinuria. It also highlights the need for greater awareness of xanthine uroliths as a cause of FLUTD and calls for improved diagnostic tools and treatment options. Better recognition of this condition will help veterinarians distinguish it from other urinary disorders and support future research to enhance care for affected cats.
Keywords
Introduction
Hereditary xanthinuria is a rare autosomal recessive disorder reported in humans and animals. The first documented case of xanthinuria – described as an ‘inborn error of purine metabolism’ – was reported in a human patient in 1951, laying the groundwork for the current understanding of the disease across various mammalian species.1,2 To date, feline xanthinuria has been identified in various breeds including the domestic shorthair, Siamese, Himalayan and Munchkin cats, and is an emerging condition of the feline lower urinary tract disorder (FLUTD) complex.3 –5 Xanthinuria is defined by excess accumulation of xanthine – an extremely insoluble purine metabolite – in the urine, resulting from disruptions in purine metabolism.6 –9 In highly saturated urine, xanthine can precipitate to form xanthine uroliths, causing clinical signs associated with FLUTDs, including stranguria, pollakiuria, dysuria, periuria and haematuria, as well as life-threatening urethral obstructions.4,8 –12 In the absence of xanthine urolithiasis, cats with hereditary xanthinuria may remain subclinical. 4
The limited diagnostic tools and treatment options together present ongoing challenges for the management and resolution of the disorder. 13 Xanthinuria cannot be easily diagnosed with routine in-house urinalysis or biochemistry tests, as these methods do not allow for the quantification of hypoxanthine and xanthine. The current recommendation for managing feline xanthinuria is dietary protein restriction while encouraging increased fluid intake; however, owner compliance with dietary restriction is a common challenge in veterinary medicine.14 –17 To date, three causative variants for feline xanthinuria have been proposed, yet none have been functionally validated.18,19 Although genetic panels for hereditary feline xanthinuria may be commercially available, their application should be regarded as investigational rather than diagnostic, owing to the absence of functionally confirmed variants.
Xanthinuria is a lifelong metabolic disorder, with the clinical outcome dependent on the presence, severity and recurrence of xanthine urolithiasis, underlying comorbidities and compliance in following up with treatment recommendations. This review will provide an overview of the current understanding of the pathophysiological, metabolic and genetic aspects of the disorder, and outlines the available diagnostic approaches, management strategies and clinical expectations of feline xanthinuria. By fostering a deeper understanding, ongoing research can focus on the development of accessible diagnostic tools and reliable treatment protocols to enhance detection, prevention and management of feline xanthinuria. Further studies investigating the prevalence of xanthinuria will also help clarify the significance of the condition in the feline population.
Pathophysiology of xanthinuria
Disruptions in normal purine metabolism occur in the absence or deficiency of the xanthine dehydrogenase enzyme (XDH), resulting in the accumulation of hypoxanthine and xanthine in both plasma and urine.12,16 Based on the human model, purine metabolism is maintained by two primary pathways: (1) the salvage pathway, which occurs under normal physiological conditions; and (2) the de novo pathway, which occurs with increased intracellular purine demand (Figure 1).20 –26 Both pathways share inosine 5’-monophosphate (IMP), which is converted into hypoxanthine, a fundamental metabolite in the terminal steps of purine metabolism (Figure 1). Hypoxanthine is metabolised by the rate-limiting enzyme, xanthine oxidoreductase (XOR). 27 Mammalian XOR takes two forms: XDH and xanthine oxidase (XO).28 –30 XOR exists predominantly as XDH in mammalian tissue but can be readily and reversibly converted into XO during hypoxemic or ischaemic conditions.29,31 –34 Both XDH and XO are responsible for the terminal steps of purine metabolism, by catalysing hypoxanthine into xanthine, and xanthine into uric acid (Figure 2).30,35 Uric acid can be further metabolised into allantoin via urate oxidase (also known as uricase), which is an enzyme present in mammalian species other than humans and higher primates.23,36 Although there is a dual accumulation of hypoxanthine and xanthine in the urine, hypoxanthine contributes to the pathophysiological process of xanthinuria to a lesser extent in comparison with xanthine. This is attributed to the characteristics of hypoxanthine, which has higher solubility in urine, and can be recycled back into the salvage pathway of purine metabolism via IMP.8,13,37 Thus, the predominant role of xanthine in the pathophysiological process of the disorder is highlighted by the terminology: ‘xanthinuria’.

The purine metabolism pathway. The de novo pathway (blue) involves 10 steps to convert phosphoribosylpyrophosphate (PRPP) into inosine monophosphate (IMP). The enzymes involved in this pathway include phosphoribosylpyrophosphate amidotransferase (PPAT), glycinamide ribonucleotide synthetase (GARS), glycinamide ribonucleotide transformylase (GAR Tfase), formylglycinamidine ribonucleotide synthetase (FGAMS), aminoimidazole ribonucleotide synthetase (AIRS), carboxyaminoimidazole ribonucleotide synthetase (CAIRS), succinyl aminoimidazole carboxamide ribonucleotide synthetase (SAICARS), adenylosuccinate lyase (ADSL), 5-amino-4-imidazolecarboxamide ribonucleotide transformylase (AICAR Tfase) and inosine monophosphate cyclohydrolase (IMPCH). The purine salvage pathway (yellow) can also regenerate IMP by recycling partially degraded purine bases and metabolites (adenine, guanine and hypoxanthine). In the downstream catabolic pathway, IMP is further degraded into hypoxanthine. Xanthine oxidoreductase (XOR) catalyses the conversion of hypoxanthine to xanthine and xanthine into uric acid (green). Uric acid is the terminal product of purine metabolism in humans and higher primates but can be further oxidised into allantoin via urate oxidase enzyme in other mammals. ADSS = adenylosuccinate synthetase; AICAR = 5-amino-4-imidazolecarboxamide ribonucleotide; AIR = aminoimidazole ribonucleotide; AMP = adenosine monophosphate; AMPD = adenosine monophosphate deaminase; AMPS = adenylosuccinate monophosphate; APRT = adenine phosphorybosyltransferase; CAIR = carboxyaminoimidazole ribonucleotide; FAICAR = formylaminoimidazole-4-carboxamide ribonucleotide; FGAM = formylglycinamidine ribonucleotide; FGAR = formylglycinamide ribonucleotide; GAR = glycinaminde ribonucleotide; GMP = guanosine monophosphate; GMPR = guanosine monophosphate reductase; GMPS = guanosine monophosphate synthetase; HPRT = hypoxanthine phosphorybosyltransferase; IMPDH = inosine monophosphate dehydrogenase; PRA = ribosylamine-5-phosphate; SAICAR = succinyl aminoimidazole carboxamide ribonucleotide; XMP = xanthosine monophosphate

Schematic diagram of the mammalian xanthine oxidoreductase (XOR) structure and function. XOR has three functional domains: (1) the 20kDa N-terminal domain (red), which contains two distinct iron-sulfur cluster (2[Fe/S]) binding sites, (2) the 40kDa intermediate domain (yellow), which contains the flavin adenine dinucleotide (FAD) cofactor, and (3) the largest 85kDa C-terminal molybdopterin cofactor (Moco), which contains the molybdenum atom (Mo). Each domain is connected by unstructured hinge regions (asterisk). The electron (e⁻) donated by xanthine oxidises at the Moco site first, and propagates through to the flavin adenine dinucleotide (FAD) site via the 2[Fe/S] redox centres, which reduces the electron acceptor (nicotinamide adenine dinucleotide [NAD+] or O2). XOR takes two forms: xanthine dehydrogenase (XDH), which reduces NAD+ to produce NADH (nicotinamide adenine dinucleotide [reduced form]), and xanthine oxidase (XO), which reduces O2 to produce reactive oxygen species (ROS) (H2O2 and O2−). Xanthine oxidises and forms uric acid in the process
Genetic basis of feline xanthinuria
Xanthinuria follows an autosomal recessive mode of inheritance, which has been supported by population studies and pedigree analysis across various mammalian species with xanthinuria, including cats.12,38,39 XDH deficiency is caused by variants in the XDH or molybdenum cofactor sulfurase (MOCOS) gene, resulting in type I and type II xanthinuria, respectively (Table 1).6,7,9,18,19,38 –59 In humans, multiple single nucleotide polymorphisms (SNPs) causing xanthinuria have been identified across the three functional domains of the XDH gene. A correlation between the domain-specific location of the SNP and the level of enzymatic expression has been suggested, with some SNPs identified as benign and others causing only partial loss of enzyme activity, resulting in subclinical cases.8,45,60 The XDH gene in cats, as in humans, consists of 36 exons encoding the three functional domains of the XDH enzyme, and multiple SNPs have been identified throughout the gene. 18 Based on these structural similarities, it is possible that certain SNPs in cats could also result in partial enzyme deficiency and subclinical phenotypes. Although candidate causal variants for xanthinuria have been identified in cats, they have so far been found exclusively in XDH and are yet to be validated.16,23 Additional variants identified in the feline XDH and MOCOS genes were neither predicted to affect enzyme activity, nor consistent with the expected mode of inheritance, suggesting they were unlikely to be causal for hereditary xanthinuria.18,61
Summary of causative and putative causal variants of type I and type II xanthinuria reported in humans and animals*
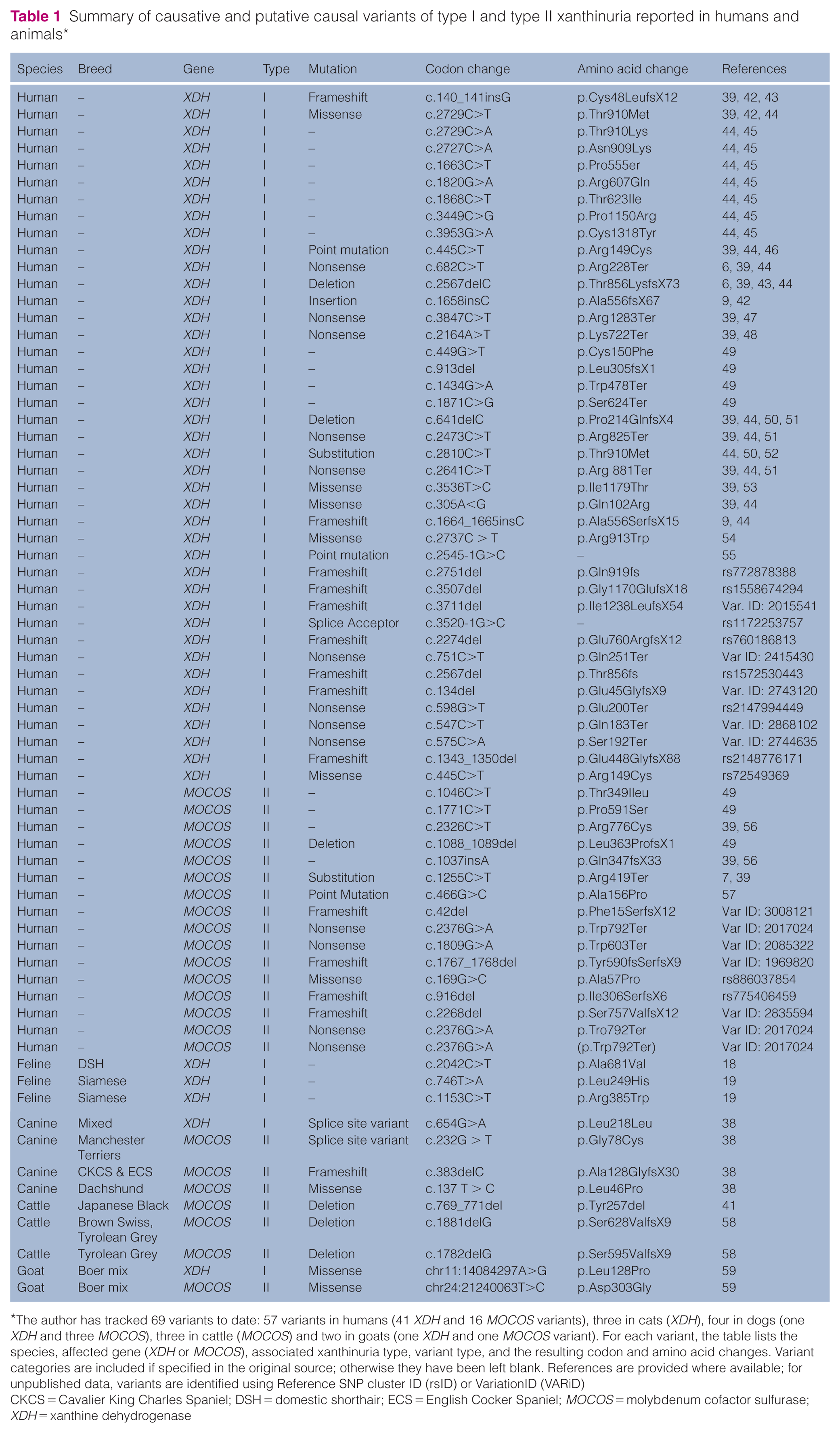
The author has tracked 69 variants to date: 57 variants in humans (41 XDH and 16 MOCOS variants), three in cats (XDH), four in dogs (one XDH and three MOCOS), three in cattle (MOCOS) and two in goats (one XDH and one MOCOS variant). For each variant, the table lists the species, affected gene (XDH or MOCOS), associated xanthinuria type, variant type, and the resulting codon and amino acid changes. Variant categories are included if specified in the original source; otherwise they have been left blank. References are provided where available; for unpublished data, variants are identified using Reference SNP cluster ID (rsID) or VariationID (VARiD)
CKCS = Cavalier King Charles Spaniel; DSH = domestic shorthair; ECS = English Cocker Spaniel; MOCOS = molybdenum cofactor sulfurase; XDH = xanthine dehydrogenase
Epidemiology of feline xanthinuria
In cats, urolithiasis accounts for approximately 15–23% of FLUTDs.3,14,62 Epidemiological studies of feline uroliths conducted between 1998 and 2020 across various geographical regions have reported that pure xanthine uroliths account for less than 1% of all submitted stones.5,63 –71 One study identified the mean age of affected cats as 3 years (range 3–176 months), with 94 (55%) of those male castrated, 17 (10%) intact males, 57 (33%) spayed females and two (1%) intact females. 5 Given the small sample size and limited data, it is difficult to draw conclusions about the prevalence of xanthine urolithiasis with respect to sex, neuter status or geographical distribution.63,67,71 As xanthinuria is an autosomal genetic disorder, no inherent sex predisposition is expected; however, male cats may be overrepresented in case reports because their longer, narrower urethra increases the risk of obstruction, and subsequent diagnosis. Although current evidence remains limited owing to the small number of reported cases, it is possible that certain cat breeds may be predisposed to the disease given the hereditary nature of the condition. To date, feline xanthinuria has been identified in multiple breeds, including the domestic shorthair, domestic mediumhair, domestic longhair, European Shorthair, Siamese, Himalayan and Munchkin cats, suggesting both pedigree and non-pedigree cats are affected.3 –5,12,14,16,18,19,61,72 Although the prevalence of xanthine urolithiasis in cats remains low, the prevalence of xanthinuria itself is still unknown and represents a research gap. Gaining a better understanding of this condition will guide responsible breeding practices and future preventative strategies.
Clinical classification and manifestations of xanthinuria in cats
In humans, xanthinuria cases are categorised as follows: (1) by their clinical signs: ‘classical xanthinuria’ (type I and type II) or type III xanthinuria; or (2) by the type of enzymatic deficiency: type I (XDH deficiency), type II (dual XDH and aldehyde oxidase [AO] deficiency) or type III (triple XDH, AO and sulfite oxidase [SO] deficiency).6 –9 The classification of xanthinuria in veterinary medicine follows the nomenclature of human xanthinuria. Type I and type II xanthinuria are clinically indistinguishable, with urinary tract disorders representing the hallmark features of classical xanthinuria reported in various mammalian species.41,58,73 –75 This is because the physiological function of AO is yet to be defined and no clinical signs have been specifically linked to the lack of AO.8,76 –78 In comparison, type III xanthinuria is clinically distinguishable from type I and type II, and occurs because of a triple deficiency of XDH, AO and SO. 21 Deficiencies in SO result in the accumulation of sulfite – a toxic by-product of sulfur-containing amino acid – in the brain, leading to severe neurological signs, lens dislocation, dysmorphism and early death in addition to urinary tract disorders.8,27,79 To date, only classical xanthinuria has been observed in cats; type III has not been reported in veterinary medicine. 18
Clinical signs of reported cases of feline xanthinuria reflect the presence and severity of urolithiasis.4,13,14 Cats with xanthine urolithiasis present with clinical signs associated with FLUTDs, including dysuria, pollakiuria, stranguria, haematuria and periuria, which may be obstructive and recurring.3,4,12,14,16 Any urinary tract obstruction in cats is considered a medical emergency as it is potentially life-threatening and can rapidly lead to critical electrolyte, metabolic and cardiovascular derangements.18,80 Xanthine uroliths may present as ‘sand-like’ deposits or crystalluria, which often precede the formation of discrete stones; however, these findings can be incidental, particularly in subclinical cases.4,14,16 As a result of the high renal clearance of xanthine, both nephroliths and ureterolithis have been observed in xanthinuric patients.4,12,14,16 Cats with upper urinary tract uroliths may present with additional clinical signs, such as asthenia, anorexia, emesis and abdominal pain, often associated with acute kidney injury. 4 Furthermore, prolonged presence of nephroliths can cause direct damage to the kidneys, tubular obstruction and subsequent tubular damage, resulting in the development of chronc kidney injury.14,16 The clinical signs reported with feline xanthinuria are based on the small number of case reports and insights from xanthinuria in other species such as humans and dogs. In clinical practice, xanthinuria is most often recognised in cats presenting with lower urinary tract signs, leading to the observation of crystalluria or urolithiasis. Further diagnostic testing, such as ultrasound, may then reveal additional urolithiasis or pathological changes in the upper urinary tract (Table 2).4,12,14,16 Although xanthine urolithiasis may affect both the upper and lower urinary tracts, clinical presentations are often inconsistent. Therefore, a thorough evaluation of the entire urinary tract is essential during diagnosis.
Summary of feline xanthinuria cases

DSH = domestic shorthair; FLUTD = feline lower urinary tract disorder; F = female; FE = female entire; FS = female spayed; HPLC = high-performance liquid chromatography; HPLC-UV = ultraviolet high-performance liquid chromatography; MC = male castrated; mo = months old; SDMA = symmetric dimethylarginine; SUB = subcutaneous ureteral bypass device; yo = years old
Some rarer, non-urinary tract-related clinical signs of xanthinuria reported in humans include myopathy, arthropathy and duodenal ulcers.1,13,40,81 Myopathy can manifest as muscle pain and cramps in association with xanthine deposition in soft tissues, which can be exacerbated by strenuous exercise.1,40,82 –84 Arthropathy is speculated to be the accumulation of xanthine microcrystals that induce arthritis, resulting in migratory or rheumatoid arthritis.1,50 Duodenal ulcers have been reported in xanthinuric patients, with the literature suggesting that a low level of uric acid, a scavenger of free radicals, may contribute to ulcer formation. 85 However, it remains unclear whether the association between these clinical signs reflects correlation or causation. Although these clinical signs have not been reported in cats, their occurrences in cats cannot be ruled out, and veterinarians should consider the possibility of these manifestations when evaluating cats with xanthinuria.
Diagnostic approaches to feline xanthinuria
Two diagnostic methods have been used in published studies for the diagnosis of xanthinuria in cats: (1) analysis of hypoxanthine, xanthine and uric acid concentrations in serum and urine; and (2) stone composition analysis if uroliths are present and obtainable.12,14,16,18,61
As a result of the limited XDH activity to catalyse the conversion of hypoxanthine and xanthine into uric acid, uric acid is observed at lower concentrations than normal. 13 As a result, xanthinuria is characterised by low to undetectable levels of uric acid with high concentrations of hypoxanthine and xanthine in serum and urine.1,13,40,86 High concentrations of xanthine may be detected in urine in comparison with hypoxanthine, and this preferential accumulation results from the higher solubility of hypoxanthine in the urine and the recovery of hypoxanthine back into the salvage pathway.8,13 To diagnose xanthinuria, both hypouricosuria and hypouricaemia should be observed and interpreted with xanthine and hypoxanthine concentrations.13,86 Low uric acid alone is neither a specific nor definitive indicator of xanthinuria as it is observed in other disorders of purine metabolism in humans. 86 In cats, disorders of purine metabolism have not been characterised as extensively as in humans, limiting the use of uric acid levels alone as a diagnostic marker for xanthinuria. Xanthine and hypoxanthine concentrations in urine and serum can be measured using enzymatic colorimetric or fluorometric assays or more advanced chromatographic and mass spectrometry-based techniques including high-performance liquid chromatography, reverse phase high-performance liquid chromatography-ultraviolet, gas chromatography-mass spectrometry, liquid chromatography-tandem mass spectrometry and infrared mass spectrometry.3,4,12,14,61,87 –90 However, the assays for quantifying plasma and urine concentrations of xanthine and hypoxanthine, and the reference intervals, have not yet been validated and established for cats, and are not available as a commercial veterinary diagnostic tool. As these methods often require specialised equipment and expertise that is not commercially available for veterinary use, published case reports of feline xanthinuria have relied on non-veterinary services.4,12,61 As a result, meaningful interpretation of purine concentrations requires direct comparison between affected and unaffected animals (Table 3).4,14,16,61,91 –93
Comparative serum and urine concentrations of hypoxanthine, xanthine and uric acid in companion animals (cats, dogs, cattle) with xanthinuria vs healthy controls
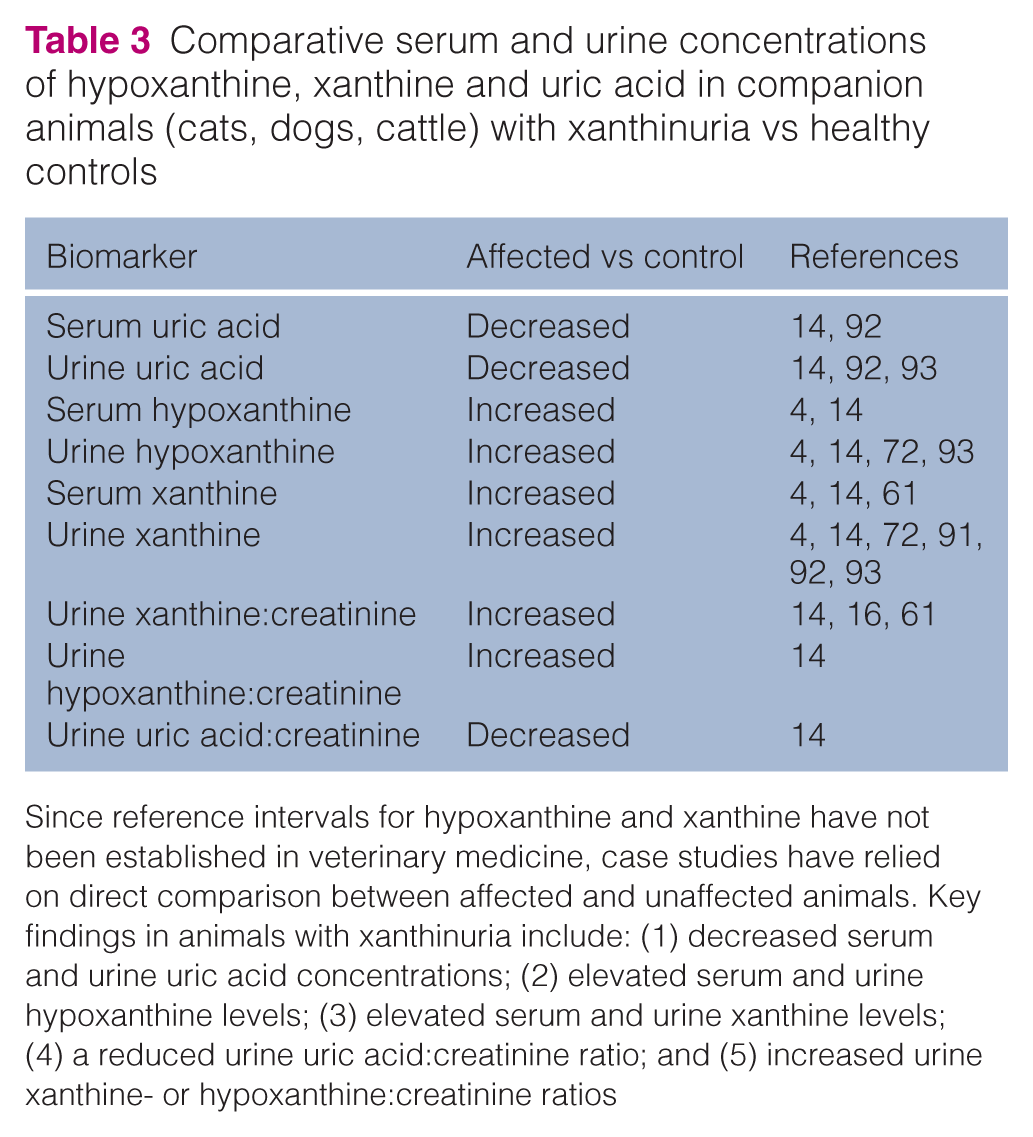
Since reference intervals for hypoxanthine and xanthine have not been established in veterinary medicine, case studies have relied on direct comparison between affected and unaffected animals. Key findings in animals with xanthinuria include: (1) decreased serum and urine uric acid concentrations; (2) elevated serum and urine hypoxanthine levels; (3) elevated serum and urine xanthine levels; (4) a reduced urine uric acid:creatinine ratio; and (5) increased urine xanthine- or hypoxanthine:creatinine ratios
Quantitative analysis of xanthine uroliths can be made using infrared or mass spectroscopy to provide definitive diagnosis of xanthinuria. 5 Xanthinuric cats are reported to have a urolith composition of 95–100% xanthine.3,14,16,61,72 Xanthine uroliths are often solid with a yellowish-brown colour, which can resemble ammonium urate uroliths.3,5,12,61 Depending on the composition of the stones, their surface may be smooth and ovoid or rough.3,5,61 As xanthine uroliths do not present with consistent shapes, an accurate diagnosis cannot be made by observation alone. Veterinarians can submit urinary urolith samples (eg, uroliths, urinary stones, urinary calculi, crystalline plugs) to Minnesota Urolith Center (College of Veterinary Medicine, University of Minnesota) for quantitative mineral analysis. Xanthine sediments and crystals are generally not accepted as they are too small to be interpreted. However, they may be observed microscopically. Xanthine crystals in urine can be yellowish-brown in colour, with varying sizes and shapes, sometimes appearing spheroid, needle-shaped or amorphous.3–5,12–15,58,61,94 They may also be indistinguishable from ammonium urates or amorphous urates, sharing similar colouration, urinary environments and amorphous shapes, as they do not have distinct, consistent, crystal lattice shapes like struvite or calcium oxalates. 5 This presents diagnostic challenges, as the misidentification of xanthine crystalluria as ammonium urate crystalluria potentially leads to the diagnostic work-up for hepatic disorders instead.
Uroliths can be detected using radiography or ultrasonography; however, these imaging modalities lack the specificity needed to determine stone composition. This is particularly challenging on radiographs, where xanthine uroliths share similar radiodensity with ammonium urate uroliths. 5 Some xanthine uroliths may also appear radiolucent and become difficult to discern from surrounding soft tissue, complicating diagnosis using imaging alone. 5 In such situations, contrast studies can be employed to assess for lower urinary tract uroliths. A double-contrast cystogram has been successfully utilised in a feline xanthinuria case revealing the presence of cystoliths, which were later confirmed to be composed of xanthine. 72 Where detection of xanthine uroliths by radiography is limited, ultrasound may be more valuable, especially in the upper urinary tract and for assessing pathological changes.3,4 Ultrasound-guided changes observed in cats with xanthine urolithiasis include thickening of the bladder wall, smaller kidney sizes, hypoechogenic cortices, hydronephrosis, renomegaly, renal pelvis dilation, hydroureter and dilated proximal ureters, suggesting various degrees of kidney disease (Table 2).12,14,61 The most sensitive modality for detecting uroliths is CT. 95 Although its use has not been reported for detecting feline xanthine uroliths, a human study demonstrated that xanthine stones are readily detected on CT. 95 Based on these findings, ultrasound should be the first-line imaging modality, with CT reserved for cases where ultrasound findings are inconclusive or insufficient.
Routine genetic testing for xanthinuria is currently not available for cats, but there is a premise for its development and clinical application in the future. Three candidate causal variants on the XDH gene have been proposed to date: one from a domestic shorthair (p.A681V) and two from a Siamese cat (p.Leu249His, p.Arg385Trp) (Figure 3).18,19 The functional impact of these three variants is yet to be confirmed. The presence of multiple causative SNPs in humans and dogs suggests a strong likelihood that feline xanthinuria can similarly be due to different causative variants. Consequently, if functionally validated variants are identified in cats, a single-target genetic test would likely be insufficient, and comprehensive approaches using multi-variant panels would be necessary to identify the causative variants.

Schematic representation of the candidate causative variants associated with feline xanthinuria mapped onto the XDH protein. The positions and location of residues (p.Leu249His [red], p.Arg385Trp [green] and p.Ala681Val [yellow]) are illustrated across the three functional domains of the xanthine dehydrogenase protein (iron sulfur cluster (2Fe/S), flavin adenine dinucleotide [FAD] and molybdenum cofactor [MOCO])
Treatment and management for feline xanthinuria
There is no curative treatment of hereditary xanthinuria.1,13,40 In cases of FLUTD with suspected xanthinuria, patient stabilisation through necessary supportive care should be prioritised before initiating long-term management. Low-purine diets and high fluid intake has been recommended for the long-term management of xanthinuria in humans, dogs and cats.1,12 –16,38,40,75,93 This is because consumption of purine-rich diets including meats increases upstream substrates of purine metabolism.96,97 As obligate carnivores, the innate protein requirements of cats means that dietary management of xanthinuria may be only partially successful and that it is difficult to manage. 18 When considering commercially available diets for its management, it is important to recognise that xanthine urolithiasis should not be managed with commercially available urinary diets, despite xanthinuria being a urinary tract disease. Although urinary diets typically contain moderately reduced protein compared with standard feline diets, their protein reduction is not as stringent as that of renal diets and does not adequately limit purine intake. Instead, commercially available specialised veterinary renal diets appear to be the better option, as they generally have a lower protein content compared with urinary diets. When recommending commercially available diets, veterinarians should consider and compare factors that may influence protein content, such as the formulation and source of protein, and consult a veterinary nutritionist for guidance in formulating individualised protein or purine-restricted diets. 12 Poor owner compliance has been reported in cats with xanthinuria, suggesting that long-term adherence to dietary therapy can be challenging and may compromise management efficacy.14,16
Urinary pH plays a critical role in the formation and dissolution of common feline uroliths, particularly calcium oxalate and struvite stones. Regulating urinary pH has become a key component in both the prevention and treatment of uroliths in cats.98,99 Xanthine is more soluble in alkaline than in acidic solutions, with solubility increasing as pH rises.92,96,100 Accordingly, it has been observed that an acidic urinary environment may promote the formation of xanthine uroliths.92,100 Theoretically, xanthine uroliths should dissolve in more alkaline urine; however, both in vitro studies and clinical observations have shown that urine alkalisation is not always effective because of the inherently poor solubility of xanthine.13,15,100 Alkalinising urine may be taken into consideration as a preventative measure, but should not be used as a sole and primary treatment method for xanthinuria. 15 Potassium citrate (75 mg/kg PO q24h) can be used if urinary pH is consistently less than 6.5 to alkalinise and maintain urinary pH. 12 Aggressive hydration is recommended to encourage frequent urination, maintain urinary xanthine concentration below its saturation point to reduce urolith formation, and achieve a urine specific gravity (USG) less than 1.030.15,100,101 High moisture diets or wet formulations have been shown to significantly increase urine volume and decrease both USG and relative supersaturation of calcium oxalate in cats, suggesting benefits of increased fluid intake in the management of uroliths. 102 Thus, high-moisture diets are recommended for cats when increasing voluntary water intake is challenging.
Quantification of hypoxanthine and xanthine is currently not available using standard in-house veterinary diagnostic machines; to date, there are no validated assays for measuring the concentration of these metabolites in feline urine, serum or plasma. Quantification of hypoxanthine and xanthine could be a useful parameter for monitoring xanthinuria cases and therapeutic response; however, this is an area that requires further research. Despite these parameters being influenced by dietary composition, it is uncertain if dietary management alone is enough to bring these parameters within the normal range. 14 The development and validation of assays capable of accurately quantifying hypoxanthine and xanthine in feline urine, serum or plasma, along with the determination of feline-specific reference intervals, would aid in the diagnosis, monitoring and research of feline xanthinuria.
Although challenging, dietary management has been reported to keep cats free from clinical signs of xanthine urolithiasis for extended periods (Table 2). 12 Although the recurrence of xanthinuria may not be inevitable, it remains a clinical concern because of limitations in current management. Key factors contributing to recurrence of xanthinuria include (1) poor client compliance with protein-restricted diets for their cats, and (2) follow-up consultations that often rely on history-taking and physical examination, which may overlook subclinical cases.14,16 These limitations highlight the importance of proactive monitoring to reduce the risk of recurrence of xanthine urolithiasis and allow timely intervention before significant complications arise. As the time frame for potential reprecipitation of xanthine uroliths in the feline urinary tract is difficult to predict, close monitoring of clinical signs and frequent follow-up consultations are pivotal in managing xanthinuric cats. Not only would this allow the subtle signs of FLUTD to be closely monitored and recorded, but it would also enable clinicians to touch base with clients to ensure dietary requirements are strictly adhered to. Biochemistry analysis should be repeated for monitoring changes in renal parameters, and urinalysis should be performed for monitoring crystalluria and USG, at the minimum. As part of the gold-standard approach, imaging should be performed every 3–6 months to track and detect uroliths, especially in patients without clinical signs of xanthine urolithiasis. Follow-up consultations should be conducted frequently after the diagnosis and resolution of FLUTD signs secondary to xanthine urolithiasis. However, the frequency of these follow-up consultations may be gradually reduced at the clinician’s discretion once the cat remains clinically stable over time.
It is important to understand that the current management options for xanthinuria are limited and, unfortunately, not entirely effective. A high recurrence rate of xanthine urolithiasis is expected. Feline xanthinuria is a lifelong condition, which can be further limited by comorbidities if patients present with urinary obstructions and/or chronic kidney disease secondary to xanthine urolithiasis. In severe clinical cases, or frequent recurrences, the welfare of the cat should be discussed, and humane euthanasia considered if deemed necessary.
Conclusions
The prevalence of inherited xanthinuria in the wider feline population remains difficult to determine as many affected cats may not exhibit clinical signs of xanthine urolithiasis, and standard diagnostic tests are often insufficient for definitive diagnosis. Given the limited treatment options and high recurrence rates, feline xanthinuria is a lifelong condition that requires ongoing management and monitoring to mitigate complications. Xanthinuria is becoming a more widely recognised condition of FLUTD, likely due to increasing clinical awareness. It should be considered in cats presenting with crystalluria and/or urolithiasis, given its significant impact on feline health and welfare. Validating the candidate causative variants of xanthinuria is crucial, as it will assist veterinarians to diagnose affected cats and aid breeders in identifying clinically silent carriers, facilitating early intervention and improving management strategies. Establishing reference parameters for hypoxanthine and xanthine levels in urine and serum will also greatly support the diagnosis of xanthinuria. Ongoing research should aim to increase the development of accessible diagnostic tools for general veterinary practice, improving the detection, prevention and management of xanthinuria in cats.
Footnotes
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
This work did not involve the use of animals and therefore ethical approval was not specifically required for publication in JFMS.
Informed consent
This work did not involve the use of animals (including cadavers) and therefore informed consent was not required. No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
