Abstract
Objectives
The aim of this study was to describe the technical success and outcomes of ureteral and urethral decompression using the subcutaneous ureteral bypass (SUB) device and transurethral self-expanding metallic stents (SEMS) as a palliative treatment option for feline malignant urinary outflow tract obstructions.
Methods
A retrospective study was conducted of 14 cats with ureteral and/or urethral obstructions secondary to diagnosed or suspected transitional cell carcinoma (TCC). In all cats, a SUB device and/or a SEMS was placed to relieve the obstruction(s). Group 1 consisted of cats with ureteral obstructions, with or without concurrent urethral obstructions, and group 2 consisted of cats with only urethral obstructions.
Results
Eight cats were included in group 1 (seven with concurrent urethral obstructions) and six cats were included in group 2. TCC was confirmed in 8/14 cats. Repeat urethral obstruction due to tumor in growth occurred in 6/13 (46%) cats with a SEMS, and no cats developed recurrent ureteral obstructions after placement of the SUB device. Three cats had additional covered stents placed after urethral re-obstruction. The median survival time (MST) from the time of device placement was 52 days in group 1 (mean 92; range 14–349) and 80 days in group 2 (mean 96; range 7–209). The MST from the time of mass identification of the cats that did and did not receive adjunctive therapy was 349 days (mean 358; range 124–602) and 43 days (mean 113; range 14–423), respectively.
Conclusions and relevance
The use of bypass devices for feline malignant urinary outflow tract obstructions is a viable option to provide immediate renal and urinary bladder decompression. TCC in cats is locally aggressive and can result in urethral re-obstruction, prompting consideration for placing a covered urethral stent. The use of adjunctive therapies for malignant neoplasia should be considered to improve overall survival once the obstruction has been relieved.
Keywords
Introduction
In many institutions, the subcutaneous ureteral bypass (SUB) device has become the preferred option for renal decompression for benign feline ureteral obstruction(s). Placement of the SUB device carries a good to excellent prognosis with decreased perioperative morbidity and mortality compared with traditional surgery.1–4 The majority of feline ureteral obstructions are benign, commonly caused by ureterolithiasis and strictures; however, obstructions secondary to neoplasia, blood stones, purulent plugs and trauma have also been reported.5–10 There are limited reports documenting treatment options for feline malignant ureteral obstructions, with a single case report of a SUB device used to address bilateral obstructions secondary to a retroperitoneal paraganglioma in a cat. 7
Although neoplasia of the lower urinary tract is uncommon in cats, the urinary bladder is the second most prevalent site of feline urinary tract neoplasia, after renal lymphoma. 11 Surgical excision of the tumor is a treatment option if complete resection is considered possible. However, given the invasive nature of TCC in veterinary species and the common trigonal location, excision often requires radical resection and re-routing procedures. Therefore, multimodal therapy, including radiation, chemotherapy and non-steroidal anti-inflammatory drugs (NSAIDs), is often used. For malignant obstructive disease in dogs, minimally invasive urethral and ureteral stenting are effective palliative treatment options.12–14
Although less is known about feline TCC, there have been two retrospective studies evaluating its presentation and outcomes. These studies have documented relatively low rates of secondary ureteral and urethral obstructions, at 11.9% and 15%, respectively.8,15 The survival time of cats with urethral obstructions, which were not addressed with stents, was limited, in the range of 0–38 days. 15 Favorable outcomes for the placement of urethral stents for malignant feline urethral obstructions have been documented; however, literature evaluating the use of SUB devices for malignant feline ureteral obstructions is lacking.16–18 The prognosis for feline TCC has been reported as guarded, with clinical decline frequently attributable to local disease. 8 Therefore, further investigation into palliative options for obstructive disease is needed.
The purpose of this study was to describe the technical success and short- and long-term outcomes of SUB device and/or urethral stent placement as a palliative treatment option for feline malignant ureteral and/or urethral obstructions.
Materials and methods
Case selection
Medical records from the Schwarzman Animal Medical Center between September 2012 and September 2022 were reviewed to identify cats diagnosed with a ureteral and/or urethral obstruction secondary to a urinary outflow tract tumor. Cats were included if their obstruction was treated with a SUB device(s) and/or a urethral stent. Diagnosis of a ureteral obstruction was based on ultrasonographic evidence of hydronephrosis/hydroureter to the level of an obstructive mass and was confirmed with antegrade ureteropyelography. Diagnosis of a urethral obstruction was based on clinical presentation and confirmed with cystourethrography. Cats were excluded if methods of renal decompression other than SUB placement were employed. Not all cats had sampling of the mass performed because of the risk of tumor seeding or bleeding. In cases where biopsy was attempted, it was performed endoscopically or retrograde via an introducer sheath with fluoroscopic guidance, although biopsy via these methods was occasionally non-diagnostic because of the small sample sizes obtained. All patients were managed consistently by the authors (AB and/or CW); however, their oncologic protocol was not standardized because of the retrospective nature of the study.
Cats were divided into two groups. Group 1 included all cats with a ureteral obstruction treated with a SUB device(s), with or without a concurrent urethral stent (Figure 1). Group 2 included cats with only a urethral obstruction addressed with a urethral stent.

Intraoperative ventrodorsal radiographic image obtained with a digital subtraction technique during placement of bilateral subcutaneous ureteral bypass devices and a urethral stent in a cat from group 1
Medical record review
Review of the records included signalment, history, physical examination, diagnostic imaging, laboratory results, urinalysis and bacterial culture, and surgical and anesthesia reports. Pyuria was defined as >5 white blood cells per high-power field in the urine sediment. Hospitalization times and complications were recorded. Cytologic and histopathologic reports of the urinary masses were reviewed. Adjunctive oncologic treatments were recorded, along with outcomes and causes of death.
Diagnostic imaging
All available images were reviewed, including preoperative abdominal ultrasound images, antegrade ureteropyelography, retrograde cystourethrography, and thoracic and abdominal radiographs. Ultrasound examinations were performed by a board-certified radiologist or resident. Renal pelvis (in transverse plane) and ureteral diameters were recorded.
Surgical intervention
Placement of the SUB device (Norfolk Vet) was performed routinely, as described by the Norfolk Vet surgical guide.19,20 Either a SUB 2.0 (2012–2019), with a three-way subcutaneous shunting port, or a SUB 3.0 (2019–2022), with an X-piece connector, was used. In cases of urethral obstruction, a transurethral self-expanding metallic stent (SEMS) (Infiniti Medical) was placed, as previously reported. 16 In female cats, or those with a previous perineal urethrostomy, this was done retrograde through the urethra. In male cats, this was done antegrade through the bladder apex at the insertion site for the cystostomy tube of the SUB or through a surgical approach.
Statistical analysis
Because of the small sample size, descriptive statistics were performed, including means, medians and ranges where appropriate.
Results
Signalment
A total of 15 cats met the inclusion criteria; however, one cat was excluded because of placement of a ureteral stent rather than a SUB (Table 1). Breeds included domestic shorthair (10/14) and one each of domestic longhair, American Curl, Bengal and Siberian. Eight cats were spayed females and six were castrated males. Eight had ureteral obstructions, seven of which had concurrent urethral obstructions, which were included in group 1. Six had only a urethral obstruction and were included in group 2.
Summary data of signalment, diagnosis, interventions, complications and outcomes of the 14 included cats
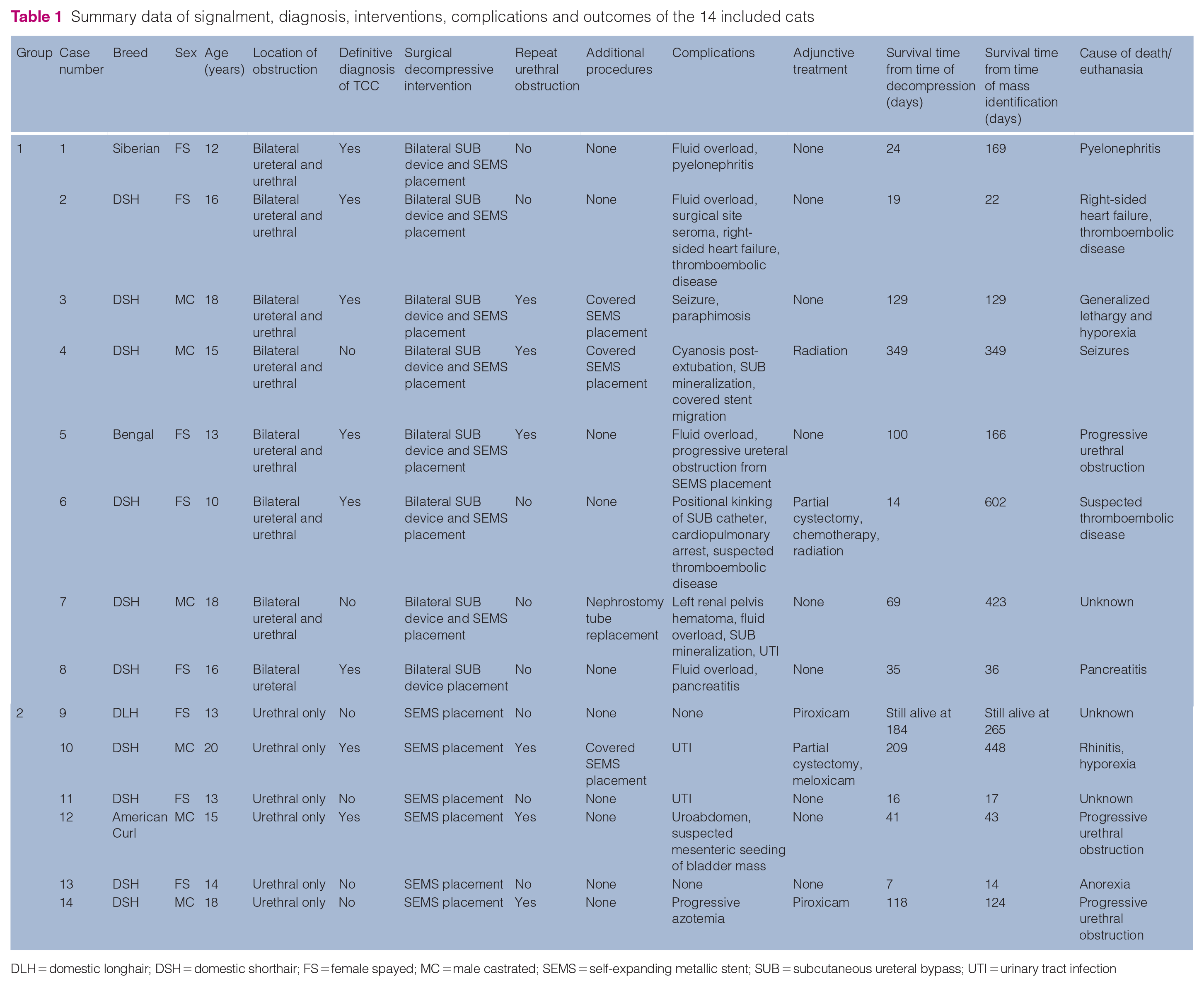
DLH = domestic longhair; DSH = domestic shorthair; FS = female spayed; MC = male castrated; SEMS = self-expanding metallic stent; SUB = subcutaneous ureteral bypass; UTI = urinary tract infection
Clinical signs and physical examination
Presenting clinical signs are summarized in Table 2. Physical examination findings at presentation were available for 13/14 cats, with findings unavailable for one cat in group 2. The most common findings included abnormal renal palpation (6/13), dehydration (6/13), heart murmur (6/13) and an enlarged, firm bladder (5/13). Three cats in group 1 and all cats in group 2 were assessed as having either a partial or a complete urethral obstruction based on history and examination.
Clinical signs at presentation of group 1 (n = 8) and group 2 (n = 6) cats
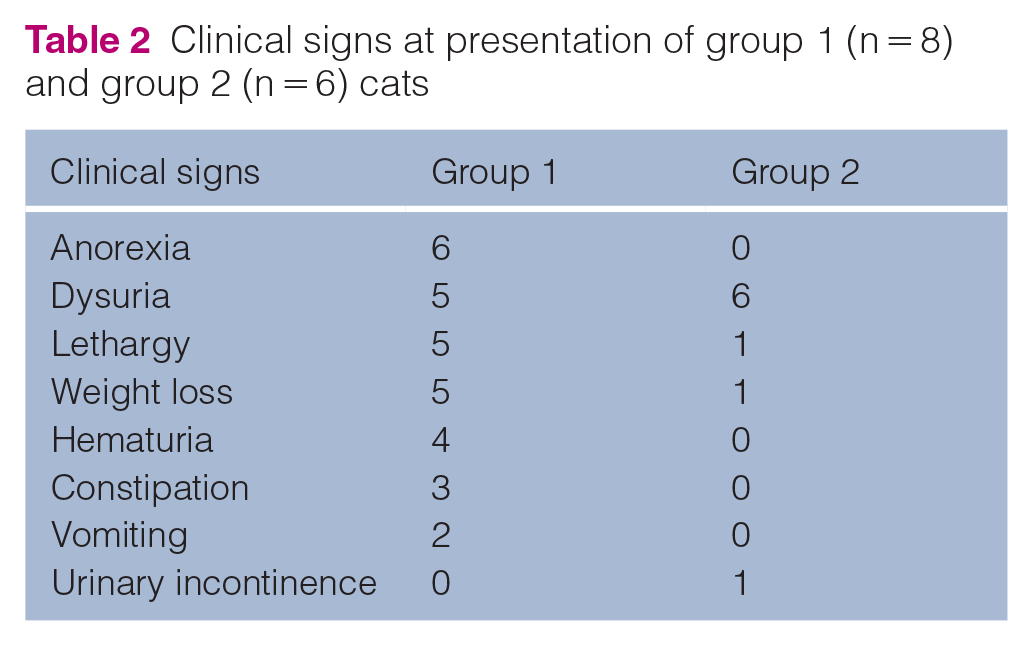
Clinicopathologic data
The median hematocrit and total solids for all cats at the time of presentation was 26.5% (range 19–37.7; reference interval [RI] 29–45) and 7.4 g/dl (range 5.8–9.1; RI 5.9–8.5), respectively. All cats, except for one in group 2, were azotemic on presentation (Table 3). One cat from group 1 was hyperkalemic, with an overall median potassium of 4.2 mmol/l (range 3.4–7.2; RI 4.0–5.4). Urinalyses from within 3 days of admission were available for nine cats. Inadequately concentrated urine was noted in all nine samples (range 1.008–1.018), although eight samples were collected from cats that were on fluids. Two cats had pyuria, although their urine cultures were negative. Of 12 cats, three had a positive urine culture and were treated based on susceptibility results.
Renal values at presentation and discharge of cats in groups 1 and 2
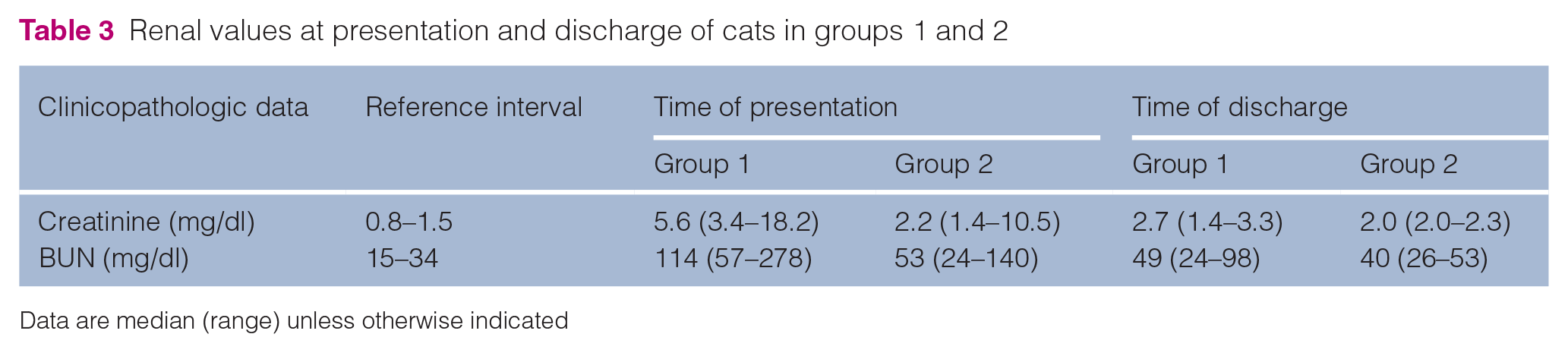
Data are median (range) unless otherwise indicated
Diagnostic imaging
Preoperative thoracic radiographs were performed in all cats in group 1 and five cats in group 2, and abdominal radiographs were performed in three cats in group 1 and four cats in group 2, with no evidence of metastatic disease. On abdominal ultrasonography, all eight cats in group 1 had evidence of trigonal involvement of a urethral or bladder mass with bilateral renal pelvic dilation and ureteral dilation to the level of the mass. The median renal pelvic diameter was 0.83 cm (range 0.2–1.32). Ureteral diameters were documented in 7/8 cats with a median of 0.4 cm (range 0.2–0.6). Five cats in group 2 had an abdominal ultrasound examination performed that revealed a urinary bladder mass or irregular thickening involving the trigone.
Diagnosis
TCC was definitively diagnosed in 8/14 cats, six in group 1 and two in group 2. Seven were diagnosed via histopathology and one via cytology. In the remaining cats, TCC was presumed based on ultrasonographic features of the mass and the invasive, irregular nature documented at surgery. In three cats with a presumptive diagnosis, sampling was attempted but non-diagnostic. Two of the non-diagnostic samples were fine-needle aspirates and one was a urethral mass biopsy sample obtained in a retrograde manner with fluoroscopic guidance.
Preoperative management
Preoperative treatments included intravenous fluids (12/14), opioid analgesia (7/14), antibiotics (4/14), steroids (2/14) and furosemide because of concern for fluid overload (1/14).
Surgical intervention
An antegrade ureteropyelogram was performed in all cats in group 1, confirming bilateral ureteral obstructions before placement of the SUB device. One cat was noted to have bilateral circumcaval ureters, although no evidence of associated strictures at the level of the circumcaval findings was seen on the ureterogram. All cats had bilateral SUB devices placed. Six cats had a SUB 2.0 and two had a SUB 3.0. The duration of the procedure was available for seven cats, with a median of 80 mins (range 58–135), including placement of a SEMS in 6/8 cats. All cats in group 1 had an esophagostomy tube placed at the time of the SUB device placement.
A urethral obstruction secondary to the bladder and/or urethral mass was documented via positive contrast cystourethrography in seven cats in group 1 and all cats in group 2. In group 1, six cats had a urethral stent placed concurrently with the SUB devices, and one had it placed the day before the SUB device placement. The median duration of the procedure in group 2 was 66 mins (range 40–125). Both antegrade (6/13) and retrograde (7/13) urethral access techniques were used. 16 All urethral stents were uncovered at the time of original stent placement.
Complications
Information on intraoperative complications was available for all cats. Intraoperative complications in group 1 occurred in two cats, including a left renal pelvis hematoma (1/8) and cyanosis after extubation (1/8), which resolved with re-intubation and extubation. The renal pelvis hematoma obstructed the SUB device in the short-term period and did not improve with infusion of tissue plasminogen activator, necessitating replacement of the nephrostomy tube. One cat in group 2 had a uroabdomen from leakage around the percutaneous cystolithotomy port in the bladder, but this access point was closed routinely.
Information regarding perioperative complications (1–7 days postoperatively) was available for all cats. Perioperative complications occurred in seven cats in group 1, including fluid overload (5/8), seizure (1/8), paraphimosis (1/8), positional kinking of the SUB device (1/8), surgical site seroma (1/8), pancreatitis (1/8) and cardiopulmonary arrest (1/8). The cause of arrest was not evident and a return of spontaneous circulation occurred. One cat developed a ureteral obstruction after SEMS placement. This cat was included in group 1 because of the SUB device placement a day later. A packed red blood cell transfusion was needed in 6/8 cats in group 1. In group 2, one cat had a perioperative urinary tract infection (UTI).
Information regarding short-term complications (8–30 days postoperatively) was available for all cats in group 1 and four cats in group 2. Short-term complications occurred in four cats in group 1, including suspected thromboembolic disease (2/8), UTI (2/8), one of which had pyelonephritis, and right-sided heart failure (1/8). One cat in group 2 had suspected mesenteric seeding of the bladder mass identified on ultrasound examination, which was not associated with any clinical consequences.
Long-term complications (>30 days postoperatively) occurred in 2/5 surviving cats in group 1, both involving SUB device mineralization at 35 (non-obstructive) and 48 (obstructive) days. Long-term complications occurred in 2/4 surviving cats in group 2, including progressive azotemia suspected to be due to piroxicam administration (1/4) and a UTI (1/4).
Adjunctive therapy
Out of 14 cats, five received adjunctive treatment for TCC either before and/or after SUB device and/or SEMS placement. Adjunctive treatments included NSAIDs (3/5), partial cystectomy (2/5), radiation (2/5) and chemotherapy (1/5).
Follow-up
All 14 cats survived to discharge, although one cat in group 1 died the day after discharge due to suspected thromboembolic disease. This cat was not included in the post-discharge follow-up. The median duration of hospitalization was 7.5 days (range 3–17) and 3 days (range 1–4) for groups 1 and 2, respectively. Cats were generally discharged with antibiotics, analgesia and, occasionally, alpha-adrenergic blockers. Seven cats in group 1 and three cats in group 2 had bloodwork at the time of discharge, and all had improvement in their azotemia (Table 3).
Seven cats in group 1 had a reduction in renal pelvic and ureteral diameters on ultrasound examination after placement of the SUB device. One cat had persistent left pyelectasia 48 days postoperatively, with slight mineralization of the nephrostomy tube and an improving azotemia. Tube replacement was declined.
Postoperative clinical signs in group 1 included dysuria (5/8), hematuria (4/8) and urinary incontinence (3/8). Two cats with postoperative dysuria also had preoperative dysuria related to their tumors. All cats in group 2 had postoperative dysuria, although all also had preoperative dysuria, and two had mild urinary incontinence. Overall, 2/11 cats with preoperative dysuria had resolved dysuria after SEMS placement, but those with persistent dysuria were improved in severity based on the owner’s impression.
Five cats, two in group 1 and three in group 2, had at least one positive postoperative urine culture: one was asymptomatic, and the remainder were associated with dysuria or pyelonephritis. Three of these cats had a positive preoperative urine culture. Antibiotic therapy cleared the infection in one cat and two had chronic UTIs. The remaining two were either euthanized shortly thereafter (unrelated to the positive culture) or lost to follow-up.
Of 13 cats with urethral obstruction, six experienced repeat urethral obstruction due to tumor growth. Two re-obstructed in the short-term and four in the long-term period. Of those that re-obstructed, the obstruction was located within the existing urethral stent (1/6), beyond the margins of the urethral stent (1/6), both within and beyond the stent (2/6) or not specified (2/6). Three cats had additional covered urethral stents placed, and repeat stenting was declined in the others. Covered stent migration occurred in one cat, requiring its replacement with a larger, covered stent (Figure 2).

Intraoperative lateral radiographic image from a cat in group 1 with bilateral subcutaneous ureteral bypass devices and migration of a covered urethral stent
The median survival time (MST) in group 1 from the time of SUB device placement was 52 days (mean 92 days; range 14–349), and 168 days (mean 237 days; range 22–602) from the time of mass identification. The MST in group 2 was 80 days (mean 96 days; range 7–209) from the time of stent placement and 84 days (mean 152 days; range 14–448) from the time of mass identification. The MST from the time of initial decompression for cats that had repeat urethral stenting was 209 days (mean 229 days; range 129–349). Three cats were euthanized because of repeat urethral obstruction from tumor growth at 41, 100 and 118 days after SEMS placement and repeat stenting was declined. The remaining deaths were not directly related to recurrent obstructions or device complications (Table 1). The MST from the time of mass identification of cats that did and did not receive adjunctive therapy was 349 days (mean 358 days; range 124–602) and 43 days (mean 113 days; range 14–423), respectively.
Discussion
Lower urinary outflow tract obstruction can be a life-threatening consequence of urothelial neoplasia in both dogs and cats, and presents with a need for urgent decompression. This study suggests that SUB device and urethral stent placement are viable options for urinary tract decompression in cases of feline malignant ureteral and urethral obstructions. In addition, adjunctive therapy may improve survival from the time of diagnosis of the urinary tumor, with obstruction no longer being the cause of death if decompression is pursued when needed.
In the present study, all ureteral obstructions were bilateral, which is a greater frequency than that reported in dogs. 12 This finding could be explained by the more distal ureterovesicular junction location in cats, which sits in the proximal urethra, where the orifices are only millimeters apart. 16 Furthermore, since TCC is less common in cats, it may be more likely to be diagnosed later in the disease course, allowing the mass more time to grow and affect both orifices.
This study supports a higher frequency (85%) of concurrent urethral obstructions in cases of feline malignant ureteral obstructions than previously considered. 8 However, it must be noted that our study selected for the presence of urinary tract obstruction, biasing the population towards those with obstruction. Partial urethral obstructions can present with a small bladder due to frequent small urinations, so care should be taken to evaluate the degree of dysuria required to empty the bladder, knowing that decompression improves these signs.
Complications directly related to the SUB device or urethral stent were rare, with the most significant complications including a renal pelvis hematoma and a ureteral obstruction secondary to occlusion of the ureterovesicular junctions after SEMS placement. This cat likely had a partial ureteral obstruction before stent placement, but the owners had declined assessment for SUB placement. After the development of anuria after the SEMS placement, SUB devices were placed successfully. Iatrogenic ureteral obstruction has been previously observed in a cat that had a urethral stent placed in the proximal urethra and is an important potential sequela when placing urethral stents in cats, in contrast to dogs, in which it is uncommon. 16
Dysuria was common pre- and postoperatively, occurring in approximately 78% of all cats, occasionally associated with tumor growth or urethral stent migration. Postoperative dysuria is relatively uncommon in cats with urethral stents and SUB devices placed for benign obstructions.1,16 Although the frequency of pre- and postoperative dysuria in this study is similar, the severity improved after urethral stent placement. Cats in this study likely experienced a higher frequency of persistent dysuria due to the continued presence of their urethral tumor, not because of the device itself, yet the signs of obstructive dysuria resolved.
The frequency of any positive urine culture after placement of a SUB device has been reported to be in the range of 24–37%, with chronic infections being under 10% in most of the largest studies to date.1,2,21,22 This is in line with the frequency of positive urine cultures observed in cats with TCC.8,15 In this cohort, 21% of cats had a preoperative positive urine culture, and 36% had at least one positive postoperative urine culture, of which 20% were not associated with clinical signs, likely representing subclinical bacteriuria. Postoperative UTIs did not occur more commonly in cats with SUB devices than those with just urethral stents.
As demonstrated by the postoperative improvement in hydronephrosis and azotemia seen in almost all cats, SUB device and urethral stent placement are feasible options to address feline malignant ureteral and urethral obstructions. However, the duration of their benefit depends on the progression of the underlying tumor. There was a relatively high rate of recurrent urethral obstruction (46%) and need for a covered stent to address tumor in growth. All three cats that had repeat urethral stenting had a longer MST from the time of initial decompression than those that had a urethral re-obstruction but whose owners did not opt for a repeat urethral stent. Importantly, no cat with SUB devices developed recurrent malignant ureteral obstruction, since the device is placed at the bladder apex, which is not where local tumor progression typically occurs. Given the high urethral re-obstruction rate, placement of a covered urethral stent at the outset in cats with malignant urethral obstructions should be considered to prevent the need for re-intervention.
This study highlights the potential importance of adjunctive therapy to delay tumor progression. The MST from the time of mass identification of cats that underwent multimodal therapy was 349 days compared with a shorter MST of 43 days for cats that did not receive adjunctive treatment. Two cats that received adjunctive treatment also required repeat urethral stenting with covered stents, which likely contributed to their longer survival. However, these findings could also support the utility of multimodal treatments for feline TCC, although further investigation is needed.
The main limitations of the study are its retrospective nature and lack of adjunctive therapeutic standardization. The follow-up diagnostics were variable, and the small sample size limits any statistical power of the study. An additional limitation is the lack a definitive diagnosis of TCC in some cases as a result of the risk and challenge associated with biopsy in this region. Although considered unlikely based on the appearance and location of the masses, it is possible that in cases without a definitive diagnosis, the masses were inflammatory lesions or neoplasia of a different etiology, and alternative adjunctive treatments may have been indicated.
Conclusions
Placement of a SUB device and/or a urethral stent for feline malignant urinary outflow tract obstructions is a viable option to provide palliative urinary tract decompression. TCC in cats is locally aggressive and can result in urethral re-obstruction, prompting consideration of placing covered urethral stents to avoid re-obstruction through the interstices of a bare metal stent. Prospective assessment of multimodal therapy for feline TCC is needed to understand how to maximize survival benefit, as these data support that adjunctive therapy can improve overall survival.
Footnotes
Conflict of interest
Allyson Berent and Chick Weisse are consultants for Norfolk Vet and Infiniti Medical, which are the companies that supply the SUB device and urethral stents for veterinary patients.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognized high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers, tissues and samples) for all procedure(s) undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
