Abstract
Objectives
The aim of the present study was to assess the effect of gabapentin on blood pressure (BP) in cats with and without chronic kidney disease (CKD).
Methods
A randomized, blinded, placebo-controlled crossover study was performed. A total of 29 cats were included: 13 cats with stable CKD (IRIS stage 2–4) and 16 apparently healthy cats (serum creatinine <1.6 mg/dl and urine specific gravity >1.035). The cats were evaluated twice, approximately 1 week apart, and BP (Doppler sphygmomanometry) was obtained 3 h after cats received either a single dose of gabapentin 10mg/kg PO or placebo. For each cat, BP readings were obtained at each visit using the same Doppler and sphygmomanometer unit, and the same cat holder and Doppler operator, in the same location.
Results
After administration of a single dose of gabapentin (10 mg/kg PO), BP was significantly lower (median 122 mmHg, range 82–170) than after administration of the placebo (median 150 mmHg, range 102–191; P = 0.001). In the CKD subgroup, BP was significantly lower after administration of gabapentin (median 129 mmHg, range 96–170) than after administration of the placebo (median 155 mmHg, range 102–191; P = 0.008). In the healthy cat subgroup, BP was significantly lower after administration of gabapentin (median 121 mmHg, range 82–139) than after administration of the placebo (median 137 mmHg, range 102–177; P = 0.002). The median change in BP was −12 mmHg (range −95 to 10) for healthy cats and −12 mmHg (range −43 to 21) for cats with CKD (no significant difference between subgroups).
Conclusions and relevance
Gabapentin may decrease arterial BP in cats with and without CKD and these findings should be taken into account when gabapentin is administered to patients in which measurement of BP is needed.
Introduction
Visits to the veterinary clinic can be a source of stress for both the feline patient and the caregiver. This can impact the welfare of the pet and the human–animal bond, and result in hesitancy to attend routine visits.1,2 Palliating the patient’s stress level is important to make an accurate assessment of the patient’s health and perform procedures safely and effectively during veterinary interactions. One routinely measured and notable change in physiologic parameters that results from stress is situational hypertension: anxiety or excitement-induced high blood pressure (BP) that does not accurately reflect the animal’s baseline BP. 3 Yet, measurement of arterial BP in feline patients is critical to a full medical assessment, and accurate readings can be challenging. Specifically, chronic kidney disease (CKD) is highly prevalent in feline patients, and the measurement of BP is an important aspect of evaluation in this disease.
In conjunction with cat friendly interactions to reduce protective behaviors, anti-anxiety medications can be helpful to limit the challenges associated with stress in the clinic environment. 2 Gabapentin has been shown to decrease visit-associated stress in feline patients in a variety of clinical scenarios, including the measurement of BP, and in general is considered to be a relatively safe medication with minimal clinically significant effects on physiologic parameters.4 –13 However, special consideration should be given when using gabapentin in patients with compromised kidney function. Gabapentin is eliminated almost entirely through renal excretion, and decreased renal function significantly influences the pharmacokinetics of gabapentin in humans. 14 While doses of gabapentin in the range of 50–150 mg/cat have been used in normal cats,4,5,11 it should be noted that higher doses may be unsuitable for cats with CKD. 15 In a study where cats with CKD received a single oral dose of 10 mg/kg, dose-normalized serum gabapentin concentrations were significantly higher in cats with CKD in comparison with normal cats. 15 In addition, dose-normalized serum gabapentin concentrations were positively correlated with serum creatinine, indicating that greater caution is needed in more advanced kidney disease. 15
Previous studies evaluating the effect of gabapentin on BP have been performed in normal feline patients,11,12 but the effect of gabapentin on BP in cats with CKD has not been assessed. Anecdotal incidents of hypotension associated with higher doses of gabapentin (particularly in cats with CKD) have been reported. An effect on BP is perhaps unsurprising given both the anxiolytic and potential direct cardiovascular effects of gabapentin.16,17 This has important implications for its use in patients with CKD, which merits further investigation.
The aim of this study was to assess the effect of gabapentin on BP in cats with and without CKD. We hypothesized that gabapentin would result in a significantly lower BP in comparison with a placebo. A secondary hypothesis was that this effect would be more pronounced in cats with CKD. These findings will help characterize the physiologic effects of gabapentin and assist veterinarians in making an educated interpretation of BP when a patient has received gabapentin.
Materials and methods
Cats
A randomized, blinded, placebo-controlled crossover study was performed. Cats were evaluated twice, approximately 1 week apart, and BP, compliance and sedation assessments were obtained 3 h after cats received either a single dose of gabapentin 10 mg/kg PO or placebo (Figure 1). The study was approved by the Institutional Animal Care and Use Committee (IACUC-2021A00000096) at The Ohio State University. The treatment order for each cat was randomized for each group of cats (healthy cats and cats with CKD). Randomization procedures were completed using an online random assignment research randomizer (randomizer.org). A power calculation (biomath.info/power/prt.htm, paired t-test) was performed using pilot data from the first eight cats in each group. For the normal group, a mean ± SD difference in BP of 29 ± 32 mmHg between treatment arms resulted in a calculation of 12 cats to achieve 80% power (alpha 0.05). For the CKD group, a mean difference of 21 ± 23 mmHg resulted in a calculation of 15 cats to achieve the desired power. Therefore, the enrollment goal was 15 cats per group and 30 cats in total.

Study design timeline
Healthy cats
Client-owned presumably healthy cats with normal BP, serum biochemistry profile, complete blood count (CBC), urinalysis and serum total thyroxine (TT4) measurement were enrolled. Healthy control cats were defined as those with no historical or clinical abnormalities, normal physical examination and creatinine <1.6 mg/dl with urine specific gravity >1.035.
Cats with CKD
Cats with stable International Renal Interest Society (IRIS) stage 2–4 CKD were enrolled. BP, serum biochemistry profile, CBC, urinalysis and serum TT4 measurement were performed before enrollment. The exclusion criteria included a history of exhibiting repelling protective behaviors in the veterinary clinic, other uncontrolled systemic illnesses, known or suspected enteropathies, complications of CKD such as pyelonephritis or ureteral obstruction, decompensation of CKD requiring hospitalization, and intravenous fluid or current gabapentin therapy. Concurrent therapies for CKD, such as dietary management, potassium supplementation, phosphorus binders, antihypertensive medications and subcutaneous fluids, were acceptable as long as no changes were made between the two consecutive study visits.
Gabapentin capsule preparation
Capsules were compounded in 5 mg increments (eg, 20 mg, 25 mg, 30 mg) by The Ohio State University Veterinary Medical Center Pharmacy following compounding standards set by US Pharmacopeia (USP) chapters 795 and 1163. Gabapentin capsules were filled with the same active pharmaceutical ingredient (API): gabapentin, bulk USP Powder (lot DR0198; Attix Pharma). The excipient for all of the capsules was Lactose NF Powder (lot 151894A; Medisca). The capsules were filled using a manually operated capsule-filling machine (ProFiller 100; Torpac). The API and excipient were weighed according to the required quantity for each capsule strength and then thoroughly blended before being used to fill empty gelatin capsules. Capsules were prepared in batches of 10 for each desired concentration. Capsules from each batch were weighed after preparation to verify the percentage error range. No capsule had an error >5%; the average capsule weight was within 99.4% of the projected final weight. Placebo capsules were compounded using Lactose NF Powder, and were identical in weight, size and color as the study capsules. The gabapentin and placebo capsules were designated ‘A’ or ‘B’ for the purposes of blinding and this designation was kept confidential from study personnel until the completion of the study.
Gabapentin administration and initial compliance assessment
Compliance and sedation scores were obtained before administering gabapentin or placebo. The same clinician assessed and graded compliance and sedation at time 0 and 3 h, and at both consecutive visits. The compliance score (3 = no resistance to restraint; 2 = minimally resistant to restraint; 1 = struggling, difficult to restrain; 0 = extreme struggling with or without urination or defecation) was assigned by the attending clinician for two interactions. These included palpation and isolation of the bladder (as if for cystocentesis in lateral recumbency), and placement of the cat in dorsal recumbency in a padded ultrasound trough (as if for ultrasound-guided cystocentesis). The compliance scores from the activities were summed to reflect that higher summed scores represent a more compliant patient. Reflecting on the entire interaction, sedation was scored (4 = fast asleep/no response to a hand clap; 3 = can lift its head; 2 = in sternal recumbency; 1 = can stand but is wobbly; 0 = no sedation) and assigned by the attending clinician before the administration of gabapentin. Then, a single dose of either gabapentin 10 mg/kg (rounded to the nearest 5 mg) or placebo was administered orally followed by 3 ml of water.
Blood pressure assessment
Three hours after administration of either gabapentin 10 mg/kg or placebo, and before sample collection and compliance assessment, BP measurements were obtained using Doppler sphygmomanometry (Parks Doppler, Model 811-BL; Parks Medical) and headphones. The BP measurement was obtained by the same operator at each visit using a Doppler unit and headphones specifically designated for the study. The cat holder, cuff size and location, cat position (standing, ‘cat loaf’, ie, sitting in a sternal position with the paws tucked under the body, or lateral, as chosen by the patient) and location in the clinic were kept consistent for both visits. Cuff size was determined based on the cuff width equal to 30–40% of the limb circumference. Five BP measurements were obtained and averaged. Sphygmomanometers were evaluated against a mercury manometer (Diagnostix 922 Desktop Mercurial Sphygmomanometer; American Diagnostic Corp) at three pressures over a range of 50–200 mmHg once every 2 months throughout the time frame they were utilized for the study.
Sample collection and 3 h compliance assessment
Three hours after administration of either gabapentin 10 mg/kg or placebo, and after BP measurement, a blood sample was collected via the medial saphenous vein, and compliance and sedation were reassessed. The compliance score was assigned by the attending clinician for four interactions at the 3 h time point. These included obtaining a BP measurement, venipuncture for sample collection, palpation and isolation of the bladder in lateral recumbency, and placement of the cat in dorsal recumbency in a padded ultrasound trough. The compliance scores from the four activities were summed to reflect that higher summed scores represent a more compliant patient. Sedation was scored as previously described.
Gabapentin serum concentration analysis
Sample preparation and determination of gabapentin serum concentrations were conducted using a previously published liquid chromatography-tandem mass spectrometry (LC/MS/MS) assay. 18 A partial validation was performed using feline serum as a matrix. Gabapentin calibration curves (0.5–30,000 ng/ml) and negative control samples were prepared fresh for each quantitative assay. In addition, quality control samples (serum fortified with analyte at three concentrations within the standard curve) were included as an additional check for accuracy.
The precision and accuracy of the assay were determined by assaying quality control samples in replicates (n = 6). Accuracy was reported as percent nominal concentration and precision as percent relative standard deviation. For gabapentin, accuracy was 92% for 1.5 ng/ml, 100% for 10 ng/ml and 105% for 10,000 ng/ml. Precision was 4% for 1.5 ng/ml, 10% for 10 ng/ml and 11% for 10,000 ng/ml. The technique was optimized to provide a limit of quantitation of 0.5 ng/ml and a limit of detection of approximately 0.25 ng/ml for gabapentin.
Gabapentin capsule analysis
Eight gabapentin capsules were randomly selected for analysis (20, 30, 30, 45, 45, 55, 55 and 60 mg). Gabapentin working solutions were prepared by diluting the stock solution with methanol to concentrations of 10 and 100 ng/µl. Calibrators were prepared by diluting the working standard solutions with 5% acetonitrile in water, with 0.2% formic acid, to concentrations in the range of 0.05–1.5 ng/ml. Before the analysis, gabapentin capsules were dissolved in 100 ml acetone:water (1:1 v:v), diluted further 100-fold in acetone:water (1:1 v:v). The samples then underwent a final 10-fold dilution in 5% acetonitrile in water with 0.2% formic acid, and 10 µl was injected into the LC/MS/MS system. The analytical method used for the LC/MS/MS analysis was as described for the blood samples.
Statistical analysis
Data were assessed for normality using the D’Agostino and Pearson test and visual inspection of QQ plots, and analyzed as parametric or non-parametric data accordingly. A paired t-test was used to assess differences in BP measurements between the gabapentin and placebo visits. A Wilcoxon sign rank test was used to assess differences in: BP between the gabapentin and placebo visits for the CKD and healthy cat subgroups; compliance scores between the gabapentin and placebo visits for all cats and the CKD and healthy cat subgroups; compliance scores between baseline and the 3 h time point for all cats and the CKD and healthy cat subgroups; and sedation scores between the gabapentin and placebo visits for all cats and the CKD and healthy cat subgroups. A Mann–Whitney U-test was used to compare sedation scores between cats with CKD and healthy cats that had received gabapentin, and to compare the change in BP between the cats with CKD and the healthy cats. Spearman’s rank was used to assess the correlations between: serum gabapentin concentration and compliance scores for all cats and the CKD and healthy cat subgroups; serum gabapentin concentration and change in BP for all cats and the CKD and healthy cat subgroups; serum gabapentin concentration and serum creatinine concentration; change in BP and compliance scores for all cats and the CKD and healthy cat subgroups; and change in BP and serum creatinine concentration in the cats with CKD.
Results
Cats
A total of 37 client-owned cats (16 with CKD and 21 apparently healthy) were screened for study participation. Five apparently healthy cats failed the health screening and one cat with CKD was excluded for a urinary tract infection. In total, 16 apparently healthy cats and 15 cats with CKD were randomized and completed the study. After completion of the study, two cats with CKD were excluded from the analysis: one due to protocol deviation (BP was not measured at the same time of day and clinic location at both visits) and the other due to a post-study diagnosis of gastrointestinal neoplasia. Of the 13 cats with CKD that were included in the data analysis, six were IRIS stage 2, six were IRIS stage 3 and one was IRIS stage 4. Cats with CKD had a median age of 12 years (range 8–18), a median serum creatinine concentration of 2.8 mg/dl (range 1.6–4.3) and consisted of nine domestic shorthairs (DSHs), three domestic longhairs (DLHs) and one Tonkinese (four castrated males and nine spayed females). The 16 apparently healthy cats included in the analysis had a median age of 6.7 years (range 2.5–12), a median serum creatinine concentration of 1.4 mg/dl (range 0.9–1.5) and consisted of 11 DSHs and five DLHs (seven castrated males and nine spayed females).
Blood pressure
After administration of a single dose of gabapentin 10 mg/kg PO, BP was significantly lower (median 122 mmHg, range 82–170) than after administration of the placebo (median 150 mmHg, range 102–191; P = 0.001) (Table 1, Figure 2a). In the CKD subgroup, BP was significantly lower after administration of gabapentin (median 129mmHg, range 96–170) than after administration of the placebo (median 155 mmHg, range 102–191; P = 0.008) (Table 1, Figure 2b). In the healthy cat subgroup, BP was significantly lower after administration of gabapentin (median 121 mmHg, range 82–139) than after administration of the placebo (median 137 mmHg, range 102–177; P = 0.002) (Table 1, Figure 2c). The median change in BP was −12 mmHg (range −95 to 10) for healthy cats when the BP at the gabapentin visit was compared with the placebo visit, and −12 mmHg (range −43 to 21) for cats with CKD. No significant difference was found in the degree of change in BP between the subgroups. Two cats had BP <100 mmHg measured (confirmed on a second measurement) after the administration of gabapentin: one in the healthy cat group (82 mmHg after gabapentin vs 177 mmHg after placebo) and one in the CKD group (96 mmHg after gabapentin vs 133 mmHg after placebo).
Doppler BP in cats with and without CKD 3 h after administration of a single dose of gabapentin 10 mg/kg PO or placebo

Data are median (range) unless otherwise indicated
BP = blood pressure; CKD = chronic kidney disease

(a) Administration of a single dose of gabapentin 10 mg/kg PO to all enrolled cats (n = 29) resulted in a significantly lower blood pressure (BP) when compared with placebo (P = 0.001). (b) In the chronic kidney disease (CKD) subgroup (n = 13), gabapentin resulted in a significantly lower BP when compared with placebo (P = 0.008). (c) In the healthy cat subgroup (n = 16), gabapentin resulted in a significantly lower BP when compared with placebo (P = 0.002). Data are displayed as box and whisker dot plots, illustrating median, range and interquartile range
Sphygmomanometer evaluation
When the two dedicated sphygmomanometers were evaluated throughout the course of the study against a mercury manometer, both devices performed accurately with no deviation, with one exception. At the last checkpoint at the end of the study, one of the sphygmomanometers read 2 mmHg higher than the mercury manometer at the highest point evaluated.
Compliance and sedation scores
When a single dose of gabapentin 10 mg/kg PO was administered to all enrolled cats, sedation scores at the 3 h time point were significantly higher at the gabapentin visit in comparison with the placebo visit (P = 0.004) (Figure 3a). This effect was primarily due to the significantly higher sedation scores in cats with CKD in comparison with healthy cats (P = 0.03) (Figure 3b). The healthy cat subgroup did not have significantly higher sedation scores in comparison with the placebo visit (P = 0.5) whereas the CKD cat subgroup did (P = 0.02). When a single dose of gabapentin 10 mg/kg PO was administered to all enrolled cats, sedation scores at the 3 h time point were also significantly higher when compared with the baseline of the same visit (P = 0.004). The healthy cat subgroup did not have significantly higher sedation scores at the 3 h time point in comparison with baseline (P = 0.5), whereas the CKD subgroup did (P = 0.02). No significant difference in sedation score was observed between the 3 h time point and baseline after administration of placebo.
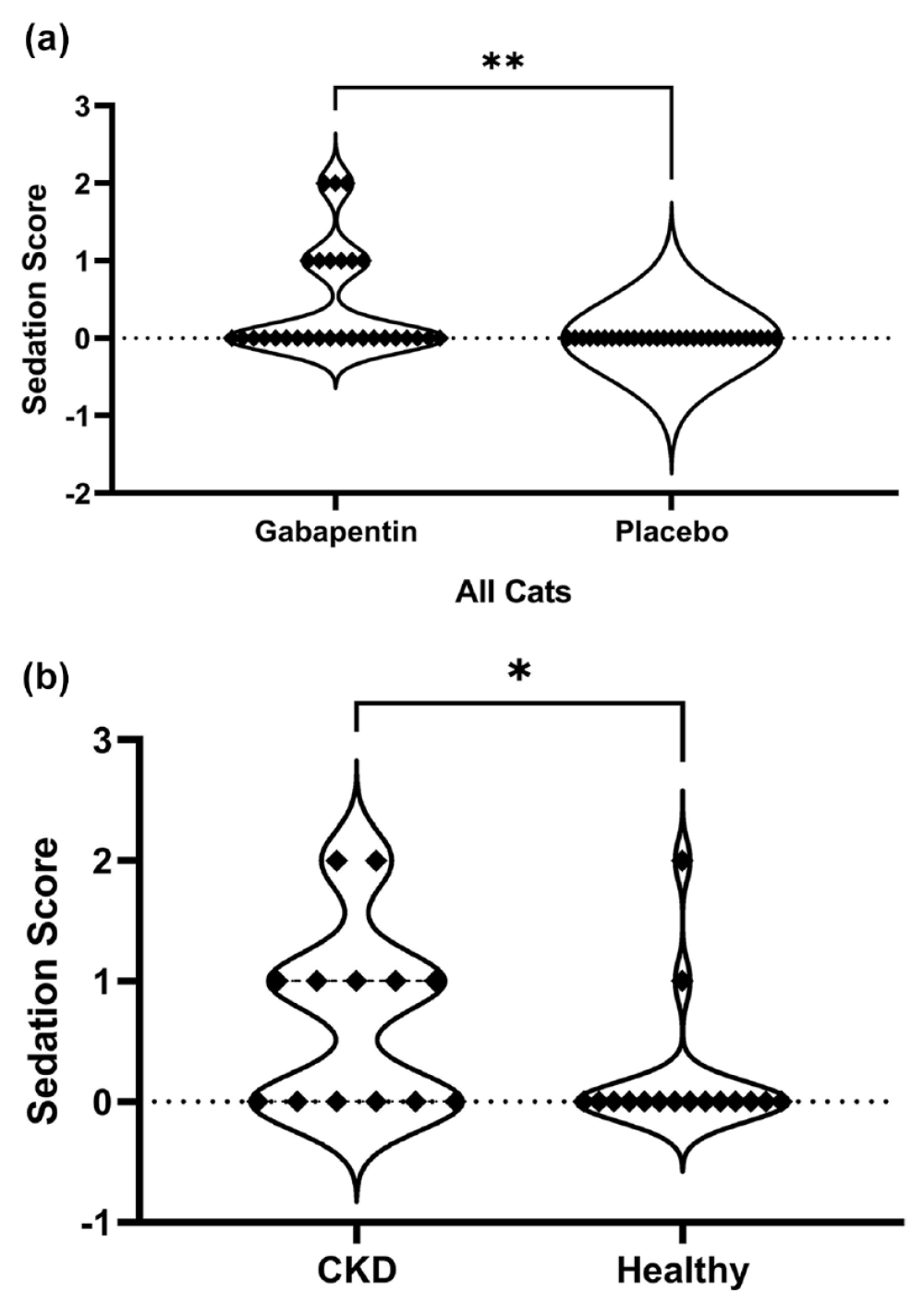
(a) Sedation scores were significantly higher at the gabapentin visit in comparison with the placebo visit (n = 29; P = 0.004). (b) This effect was primarily due to the significantly higher sedation scores in cats with CKD (n = 13) in comparison with healthy cats (n=16) (P = 0.03). Data are displayed as violin plots, illustrating the distribution and density of the data
Compliance scores at the 3 h time point were significantly higher in all cats at the gabapentin visit in comparison with the placebo visit (P <0.0001) (Figure 4a). Significantly higher compliance scores were also observed when the CKD (P = 0.03) (Figure 4b) and healthy cat (P = 0.001) (Figure 4c) subgroups were assessed individually. There was no significant correlation between the change in compliance score and the change in BP (P = 0.06) in all cats. After gabapentin administration, compliance scores at the 3 h time point were significantly higher in all cats in comparison with baseline (P = 0.0004). The CKD subgroup did not have significantly higher compliance scores at the 3 h time point in comparison with baseline (P = 0.19), whereas the healthy cat subgroup did (P = 0.001). No significant difference in compliance score was observed between the 3 h time point and baseline after administration of placebo.

(a) Compliance scores were significantly higher in all cats (n = 29) at the gabapentin visit in comparison to the placebo visit (P <0.0001). (b, -c) Significantly higher compliance scores were also observed when the (b) chronic kidney disease (CKD) (n = 13) (P = 0.03) and (c) healthy cat (n = 16; P = 0.001) subgroups were assessed individually. Data are displayed as violin plots, illustrating the distribution and density of the data
Serum gabapentin concentrations
Serum gabapentin concentrations were analyzed for 11/13 cats with CKD and 15/16 healthy cats. The median serum gabapentin concentration 3 h after administration was 10,330 ng/ml (range 1469–15,010) in healthy cats and 11,559 ng/ml (range 3709–22,050) in cats with CKD. There was no significant correlation between serum gabapentin concentration and serum creatinine concentration (P = 0.055) when all cats were considered. In cats with CKD, serum gabapentin concentrations were correlated with the degree of change in BP between the placebo visit and the gabapentin visit (P = 0.02) (Figure 5), with cats that showed a larger decrease in BP having a higher serum gabapentin concentration. This correlation was not appreciated when all cats were assessed together or in the healthy cat subgroup alone.

In cats with chronic kidney disease (n = 11), serum gabapentin concentrations were correlated with the degree of change in blood pressure between the placebo visit and gabapentin visit (P = 0.02)
Gabapentin capsule analysis
When eight capsules of gabapentin that were compounded for the study were randomly selected and analyzed for gabapentin concentration, the median dose was 115% (range 89–123) of the target dose.
Discussion
In comparison with placebo, administration of a single dose of gabapentin 10 mg/kg PO resulted in a significantly lower BP in both normal cats and cats with CKD in this study. This finding is in contrast to previous data regarding the effect of gabapentin on BP.4,11,12 There are several potential explanations for why previous data might differ, including timing and methodology of measurement, as well as study design and statistical power.
In a placebo-controlled, masked crossover study in 10 normal cats undergoing echocardiography and BP assessment, gabapentin (21–36 mg/kg, ie, 100 or 150 mg/cat) was administered 30 mins before leaving home. 11 BP was measured using Doppler sphygmomanometry on the left rear limb 120 mins after administration of gabapentin. It was concluded that gabapentin was appropriate as a mild sedative for an echocardiogram and had a modest but not clinically significant effect on echocardiographic measurements. Subjectively, mean BP was lower (122.2 ± 20.3 mmHg) after the administration of gabapentin as opposed to placebo (130.9 ± 16.2 mmHg) but this difference did not reach statistical significance. Given the small number of cats, the study may have been underpowered to detect a difference in BP as a primary outcome variable.
In a placebo-controlled, masked crossover study of 20 cats with a history of repelling protective behaviors, gabapentin (13–29 mg/kg, ie, 100 mg/cat) was administered 90 mins before placing the patient in the carrier for transport. 4 It was concluded that gabapentin was effective in decreasing cat stress scores and improving compliance scores. An oscillometric BP was measured on the tail approximately 120 mins after administration of gabapentin or the placebo and no significant difference in BP was appreciated. This difference in methodology and timing is not comparable with the current study.
In a placebo-controlled, masked study in which 40 cats were randomized to receive either gabapentin 100 mg (20 cats) or placebo (20 cats), echocardiography and BP were performed and no significant effect of gabapentin was found. 12 BP was measured using Doppler sphygmomanometry on the left rear limb 90 mins after gabapentin administration. No values for BP readings were included for evaluation. Given that this clinical trial was not designed as a crossover study, this could have affected the ability to determine an effect of gabapentin on BP if the effect is small or variable between individuals, which is possible given the data collected in our study.
Two of the cats in our study had repeatable BP measurements <100 mmHg after receiving gabapentin, which could clinically be considered a concern for hypotension. The difference in BP measurements between the gabapentin and placebo visits, and the degree to which BP was lowered in these two patients, provides support that the significantly lower BP associated with gabapentin administration may not be simply from a reduction in stress and a concomitant reduction in situational hypertension. Given the mechanism of action of gabapentin, it seems entirely plausible that a direct cardiovascular effect on BP can be observed in feline patients. Gabapentin is a ligand of the auxiliary α2δ subunit of the L-type voltage-dependent calcium channel. 16 These ligands decrease surface expression of channels in arterial myocytes. Acute administration of gabapentin lowered BP in conscious spontaneously hypertensive rats, and this effect was not observed with chronic administration. 16 It was concluded that gabapentin induced changes in sympathetic nerve transmission, which has effects on hemodynamic mechanisms. In a 5/6 nephrectomy rat model of CKD and hypertension, the administration of gabapentin resulted in significantly decreased BP in comparison with placebo. 19 Recent work in rat models has theorized that gabapentin may modulate BP and heart rate centrally through nitric oxide synthase in the nucleus tractus solitarii. 17
In humans, premedication with gabapentin decreases the hypertensive response to laryngoscopy and endotracheal intubation in people both with and without pre-existing hypertension.20,21 When gabapentin was administered to patients with pre-existing hypertension 2 h before routine anesthesia, or both the night before and 2 h before anesthesia, the mean arterial BP during anesthesia was decreased in comparison with the BP of patients who received placebo. 20 In another study, preoperative gabapentin administered to human patients undergoing endoscopic sinus surgery resulted in reduced sodium nitroprusside requirements to maintain BP within the desired range. 22 Hypotension as an adverse event in human patients seems rare, occurring in <1% of patients receiving gabapentin. 23 Intraoperative hypotension attributed to preoperative gabapentin has been described in a case report of a young adult. 23
Amlodipine is commonly prescribed for the management of hypertension in cats with CKD, and this drug is also an L-type voltage-dependent calcium channel blocker (α2ς subunit). Little information exists regarding whether amlodipine and gabapentin might have synergistic effects on BP; however, it is known that amlodipine potentiates the anticonvulsant action of gabapentin. 24 Two cats in the CKD group were receiving amlodipine for hypertension. These two cats did not have any subjectively greater deviation in BP than cats not receiving amlodipine (difference in BP between visits of 4 and 43, respectively).
Doppler sphygmomanometry was purposely utilized in this study as the methodology to assess BP with the goal of documenting a phenomenon we felt we were seeing quite regularly in our clinical practice. As Doppler is commonly used to assess BP in feline patients, our results provide a clinically applicable perspective. Previous studies have documented that Doppler methodology has inconsistent agreement with direct methods of BP measurement.25 –27 However, implantation of telemetry for the gold-standard determination of BP was considered too invasive in this patient population, given the number of cats needed to achieve statistical power. In addition, the clinical question was not related to the ‘actual’ BP measurement, but rather to determine whether a difference in BP was observed between treatment groups as measured by this particular commonly used device. These results may not be translatable to other methodologies for measuring BP. A study assessing the effects of gabapentin in a small number of research cats (n = 5) with surgically placed telemetry devices did not find an effect of gabapentin on systolic BP measured by direct methodology. 25 Interestingly, a subjective difference between the direct and indirect systolic BP (with Doppler measuring lower BP after gabapentin administration) was observed, although given the small number of cats, this was not statistically significant. This piece of information raises the question of whether there might be a perceived effect on BP specific to the Doppler technology, perhaps related to the mechanism of peripheral vasodilation and altered arterial myogenic tone. 23 Anecdotally, it has been noted by veterinary staff that peripheral venipuncture sometimes seems more difficult due to poor blood flow in cats that have received gabapentin.
The data collected in this study did not support the secondary hypothesis that the median change in BP as a result of gabapentin administration would be greater in cats with CKD. However, only in cats with CKD was the degree of change in BP between the placebo and gabapentin visits correlated with serum gabapentin concentrations, with cats that experienced a larger decrease in BP having a higher serum concentration of gabapentin. In addition, cats with CKD had significantly higher sedation scores than normal cats. Given the renal elimination of gabapentin and its altered pharmacokinetics in CKD, 15 together these data support cautious use at the lowest necessary dose in this patient population.
Previous studies about the use of gabapentin to relieve visit-associated stress have recommended 20 mg/kg in healthy cats.4,6 In the current study, significantly higher compliance was observed after administration of just a 10 mg/kg dose in healthy cats. It should be noted that cats with a history of exhibiting repelling behaviors were excluded from our study, and thus the patient population was different. However, the finding that a smaller dose could also be effective is similar to a previous study, where community cats received a single dose of gabapentin 100 mg (mean 35.3 mg/kg, range 23.1–47.6) or 50 mg (mean 16.3 mg/kg, range 9.2–24.4) during trapping. 5 Community cats that received the dose of 50 mg still had a significant decrease in cat stress scores 2–3 h after administration. In some cats without CKD, dose titration to optimal effect is likely possible based on these findings.
A limitation of this study was that gabapentin had to be compounded to provide a standard dose of 10 mg/kg. However, the capsule analysis indicated that despite the methodology, the measured dose did not fall within 10% of the target dose in some capsules. Therefore, it was beneficial that gabapentin serum concentrations were also measured to obtain more accurate information about drug exposure. This study focused only on a single dose of gabapentin as the investigators did not feel comfortable administering higher doses of gabapentin to cats with CKD and wanted to standardize the dose between cat subgroups. Therefore, 10 mg/kg was chosen, and additional research would be needed to assess the effects of lower doses of gabapentin in cats with CKD and higher doses in normal cats. Another limitation was that the effects of gabapentin administration on BP were assessed at only a single time point and thus it was not possible to determine whether the effects observed were at their maximum or for how long the effects lasted. In addition, a baseline BP before the administration of gabapentin or the placebo was not measured, which would have provided further insight in describing the effect of gabapentin on BP. The effects of gabapentin were not studied in a population of cats with uncontrolled hypertension so conclusions cannot be drawn on what to expect for those patients.
Conclusions
Gabapentin has an effect on arterial BP in cats with and without CKD and this effect appears to be more than just the result of stress reduction. These findings should be taken into account when gabapentin is administered to patients for which measurement of BP is needed.
Footnotes
Acknowledgements
The authors wish to thank John Freitag-Engstrom, Jessi Braatz, Nicole Stephens and Tessa Ross for technical support, Rikki Horne, PharmD, for assistance with drug compounding, and Heather Knych DVM, PhD, of the UC Davis School of Veterinary Medicine Pharmacology Lab.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The study was funded by the EveryCat Health Foundation (previously Winn Feline Foundation) and in part by Buttons Fund for Feline CKD Research at The Ohio State University.
Author note
The results of this study were presented in part, as a research report, at the American College of Veterinary Internal Medicine Annual Forum 2023.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals and procedures that differed from established internationally recognized high standards (‘best practice’) of veterinary clinical care for the individual patient. The study therefore had prior ethical approval from an established (or ad hoc) committee as stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers) for all procedure(s) undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
