Abstract
Objectives
Feline infectious peritonitis (FIP), a common disease in cats caused by feline coronavirus (FCoV), is usually fatal once clinical signs appear. Successful treatment of FIP with oral GS-441524 for 84 days was demonstrated recently by this research group. The aim of this study was to evaluate the long-term outcome in these cats.
Methods
A total of 18 successfully treated cats were followed for up to 1 year after treatment initiation (9 months after completion of the antiviral treatment). Follow-up examinations were performed at 12-week intervals, including physical examination, haematology, serum biochemistry, abdominal and thoracic ultrasound, FCoV ribonucleic acid (RNA) loads in blood and faeces by reverse transciptase-quantitative PCR and anti-FCoV antibody titres by indirect immunofluorescence assay.
Results
Follow-up data were available from 18 cats in week 24, from 15 cats in week 36 and from 14 cats in week 48 (after the start of treatment), respectively. Laboratory parameters remained stable after the end of the treatment, with undetectable blood viral loads (in all but one cat on one occasion). Recurrence of faecal FCoV shedding was detected in five cats. In four cats, an intermediate short-term rise in anti-FCoV antibody titres was detected. In total, 12 cats showed abdominal lymphadenomegaly during the follow-up period; four of them continuously during the treatment and follow-up period. Two cats developed mild neurological signs, compatible with feline hyperaesthesia syndrome, in weeks 36 and 48, respectively; however, FCoV RNA remained undetectable in blood and faeces, and no increase in anti-FCoV antibody titres was observed in these two cats, and the signs resolved.
Conclusions and relevance
Treatment with GS-441524 proved to be effective against FIP in both the short term as well as the long term, with no confirmed relapse during the 1-year follow-up period. Whether delayed neurological signs could be a long-term adverse effect of the treatment or associated with a ‘long FIP syndrome’ needs to be further evaluated.
Keywords
Introduction
Feline infectious peritonitis (FIP) is caused by feline coronavirus (FCoV), which occurs in two different biotypes (low-pathogenic FCoV and highly pathogenic FIP-associated FCoV).1–4 Low-pathogenic FCoV is endemic in cats and the prevalence of infection is up to 90%, especially in multi-cat environments, but only 4–10% of FCoV-infected cats develop FIP,5,6 a fatal immune-mediated disease characterised by granulomatous vasculitis/perivasculitis. 7 Most cats with FIP die if no effective antiviral treatment is available.3,8
However, since 2016, antiviral drugs that effectively inhibit the replication of FCoV have been developed. The protease inhibitor, GC376, which inhibits the viral 3C or 3C-like protease,9,10 necessary for the replication of FCoV, was shown to be effective in cats with experimentally induced FIP 9 and in cats with naturally occurring FIP, 10 although complete remission could not be achieved in all cats. A second antiviral drug, the nucleoside analogue GS-441524, the active form of the prodrug remdesivir, 11 showed promising results in recently published studies (Table 1). In the first controlled prospective oral treatment study, 18 cats with confirmed FIP were successfully treated with the antiviral drug Xraphconn (Mutian), containing GS-441524, for 12 weeks. 18
Overview of retrospective and prospective, experimental and field studies with number of cats with reported relapses of feline infectious peritonitis after treatment with different drugs containing the active ingredient GS-441524

Relapse: 2/10 relapse 4–6 weeks after the end of treatment, followed by a successful second treatment cycle
Relapse: 8/31 relapse (average 23 days after end of treatment), followed by a second treatment cycle: 5/8 cats with 4 mg/kg SC q24h; cats responded and were in remission at the time of publication; 3/8 cats with 2 mg/kg SC q24h: 1/3 cats relapsed again and was euthanased, 2/3 responded well, but relapsed for a second time, followed by a successful third treatment cycle
No adverse effects were mentioned in this study
Of the relapsed cats, 2.7% received a second treatment cycle
Route of administration was not indicated
FCoV = feline coronavirus; FIP = feline infectious peritonitis; IHC = immunohistochemistry; RT-qPCR = reverse transcriptase-quantitative PCR; SC = subcutaneous; SDMA = symmetric dimethylarginine
Since no data describing the detailed long-term outcome in cats with FIP treated with antiviral drugs are available so far, the aim of this study was to prospectively evaluate the outcome in the first year after the start of therapy with oral GS-441524 in the abovementioned 18 successfully treated cats, 18 evaluating clinical outcome, and laboratory and virological parameters.
Materials and methods
Study population and protocol
A total of 18 cats that successfully completed a 12-week FIP treatment protocol with the oral multicomponent drug Xraphconn containing GS-441524, PO q24h, entered long-term monitoring. All cats included in the treatment study had confirmed FIP (according to the diagnostic tool of the European Advisory Board on Cat Diseases). 19 The cats had to have clinical and clinicopathological abnormalities typical for FIP;20–22 in addition, direct detection of FCoV had to be positive either by immunohistochemistry on samples from affected organs or by detection of mutated strains of FCoV with commercial reverse transcriptase (RT)-quantitative PCR (qPCR) from effusion, blood or an organ fine-needle aspirate. Mutation PCR used targeted FCoV spike gene mutations leading to spike protein substitutions M1058L and S1060A.In the compound Xraphconn, the active ingredient GS-441524 was identified with mass spectrometry and nuclear magnetic resonance (referred to as MT-0901 in the package insert). Other ingredients mentioned were Radix scrophulariae, Platycodon grandiflorum, Phyllostachys pubescens, Forsythia suspensa and Anemarrhena asphodeloides. The tablets were film-coated and could be divided to obtain the exact dose. Besides the main ingredients, ingredients of the coating were hydroxypropyl cellulose, PEG 6000, crystalline cellulose and titanium oxide (for package insert, see the supplementary material).Two different tablets in concentrations of 2.5 mg and 10 mg, according to the manufacturer, were used. The dose of Xraphconn tablets was adjusted daily to the current weight of the cats and always administered at the same time each day on an empty stomach. Half an hour after tablet administration, the cats were offered food. From the time the cats were treated at home (after the period of hospitalisation), the owners received personalised instructions, including videos, on how to administer the tablets. The tablets could be administered with the help of a tablet feeder or wrapped in food. Owners were instructed to call in at any time in the event of problems. However, this happened only once, whereupon the owner immediately came to the clinic, where he was shown again how to administer the tablet. Cats with neurological and/or ocular signs (n = 2; cats 1 and 2) received a presumed dose of 10 mg/kg; cats without these signs (n = 16) received 5 mg/kg (according to the manufacturer’s instructions). 18 Further analysis of one tablet of the provided drug, however, suggested that one tablet of Xraphconn of the batch used for the study contained more than double the amount of the GS-441524 than officially stated by the manufacturer (a 200 mg tablet investigated by liquid chromatography–mass spectrometry contained 22.15 mg GS-441524 and not 10 mg as presumed; J Horak, unpublished data). Therefore, cats with neurological/ocular signs possibly received 22.15 mg/kg and cats without these signs possibly received 11.06 mg/kg.Initially (day 0), the enrolled cats showed laboratory changes that are considered typical for FIP (hyperproteinaemia, 6/18, 33.3%; hyperglobulinaemia, 13/18, 72.2%; hyperbilirubinaemia, 12/18, 66.7%; hypoalbuminaemia, 11/18, 61.1%; low albumin:globulin ratio, 16/18, 88.9%; anaemia, 12/18, 66.2%; and lymphopenia, 4/18, 22.2%). Sixteen of the 18 cats had abdominal and/or thoracic effusion and two cats had neurological and/or ocular signs. The median age was 7.7 months (range 4.7–56.5 months).
After the 12 weeks, all cats (18/18) had completed treatment successfully, which was defined by unremarkable clinical and neurological examinations, haematology and serum biochemistry parameters mostly in normal ranges, no signs of effusion, ocular or central nervous system disease, and no detectable viral RNA in blood. 18
Follow-up examinations were performed 168 days (24 weeks), 252 days (36 weeks) and 336 days (48 weeks) after the start of treatment, equating to 12, 24 and 36 weeks after the completion of treatment. All examined parameters are summarised in Table 2.
Overview of physical examination and laboratory parameters evaluated during the follow-up period at the individual time points

+ Examinations performed
If neurological abnormalities were seen
Abdominal ultrasound was performed in all cats; thoracic ultrasound was performed only in cats in which thoracic effusion or changes in the thorax were present before or during treatment
FCoV = feline coronavirus; RT-qPCR = reverse transcriptase-quantitative PCR; SAA = serum amyloid A; SDMA = symmetric dimethylarginine
Haematology was performed using an automatic analyser (Cell-Dyn 3500; Abbott). A differential blood count was additionally performed manually on blood smears stained with Haema-Quick Stain/Diff-Quik stain (LT-SYS; Eberhard Lehmann) when haematology parameters were abnormal. Serum biochemistry parameters were measured using an automatic analyser (Hitachi 911; Roche). The concentration of symmetric dimethylarginine (SDMA) was analysed by IDEXX Diavet AG (Bäch) using a high-throughput immunoassay, and the concentration of serum amyloid A (SAA) was determined using a latex agglutination turbidimetric immunoassay reaction (LZ Test SAA; Eiken Chemical) on a Cobas C501 clinical chemistry analyser (Roche Diagnostics). The general condition and well-being of the cats were evaluated using Karnofsky’s score (modified for cats by Hartmann and Kuffer, 23 a classification scale from 0% [dead] to 100% [normal health condition]). The reference range for the Karnofsky’s score was defined as 90–100%.
This study complied with the German guidelines for prospective studies and was approved by the Government of Upper Bavaria (reference number 55.2-2532.Vet_02-20-52) and by the ethical committee of the Centre for Clinical Veterinary Medicine of LMU Munich (reference number 288-11-10-2021). Owners gave their written informed consent for their cats to participate in the study.
FCoV viral loads and anti-FCoV antibody titres
Viral loads in blood and faeces were analysed by real-time TaqMan RT-qPCR as previously described,18,24 and anti-FCoV antibody titres in serum were determined by indirect immunofluorescence assay (IFA) as previously described,18,24 at all three follow-up examinations (weeks 24, 36 and 48).
Statistical analysis
Data were analysed using the statistical language R version 4.0.3 (R Core Team, 2020). For every single animal (served as random effects in subsequent models), repeated measurements were performed on several days; therefore, all variables were evaluated with generalised linear mixed-effects models. After model fitting, all contrasts (differences) between individual days were assessed using estimated marginal means, with a Bonferroni P value correction for multiple comparisons (P ⩽0.05).
Results
Clinical signs, laboratory values and diagnostic imaging
In week 24, 18/18 cats, in week 36, 15/18 cats and in week 48, 14/18 cats were examined. Four cats were lost to follow-up: two cats (cats 3 and 9) were not presented due to lack of owner compliance and one cat had died in a road traffic accident in week 35 (cat 1). On post-mortem examination, the third cat showed no residual FIP lesions; only a generalised lymphadenomegaly was noted. Moreover, neither FCoV RNA nor FCoV antigen was identified using RT-qPCR and immunohistochemistry, respectively, in any tissues or body fluids, including faeces of this cat. 25 One cat (cat 10) showed new onset of neurological signs resembling feline hyperaesthesia syndrome (FHS) in week 36 and was treated with a second 84-day course of GS-441524 by its owners without prior consultation with the study group and was not presented for the last follow-up examination (Table 3).
Overview of the study population available for follow-up examinations (numbers show the total number of cats available at each follow-up examination)
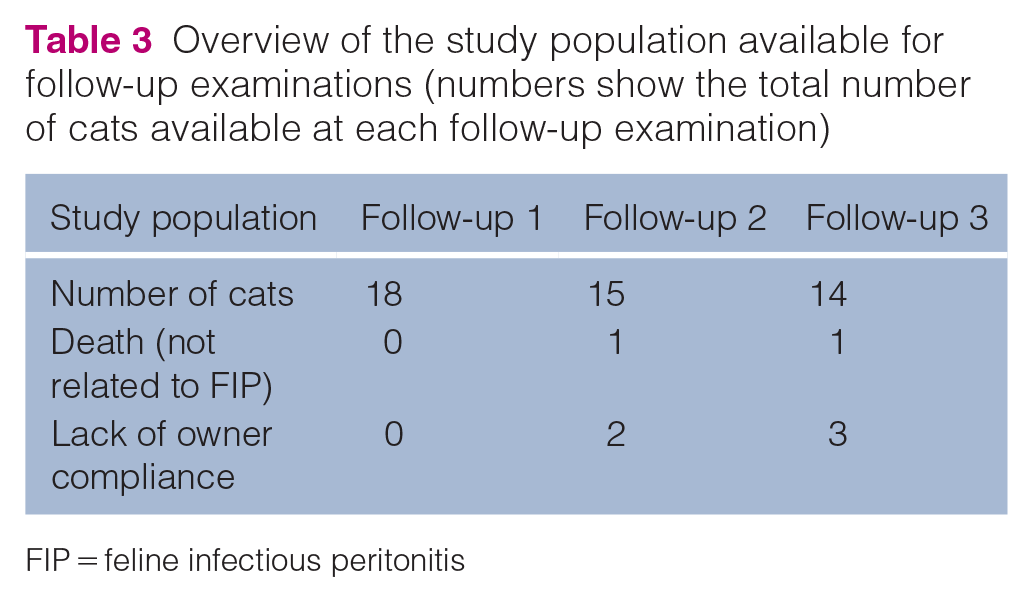
FIP = feline infectious peritonitis
All cats had achieved a modified Karnofsky score 23 of 100% at the end of the 12-week treatment period and remained healthy, with clinical and laboratory parameters mostly within reference intervals throughout the follow-up period (Table 4, Figure 1), with the exception of two cats with signs compatible with FHS (described in detail below). The physical examination parameters (such as body weight, appetite and general condition), which normalised during and towards the end of treatment, also remained consistently normal during the follow-up period. At the start of the treatment, 16 cats had abdominal lymphadenomegaly on ultrasound examination, which in 4/16 cats persisted during the entire treatment and follow-up period. During weeks 24–48, a total of 12 cats showed abdominal lymphadenomegaly (Table 4).
Selected clinical and laboratory parameters, and anti-FCoV antibody titres and viral loads of the cats

Values are given as n or median (range)
Columns are coloured according to the different study sections: orange = hospitalisation in the clinic (days 0 and 7); blue = re-checks during the treatment (days 14 and 83); green = follow-up period after the end of treatment (weeks 24, 36 and 48). Values marked in bold are outside the reference intervals
No range because all values were the same (100%) at this time point
Cat 10 developed signs compatible with FHS in week 36 and was therefore treated with a second 84-day course of GS-441524 by its owners without prior consultation with the study group and was not presented for the last follow-up examination
Number of cats that were viral RNA-positive (⩾1 copy per ml of blood/effusion and per g of faeces or per swab) at the individual time points
Within the first 3 days, overall excretion could be detected in the faeces of 11 cats
No range because all values were viral RNA-negative in blood (0 copy per ml of blood) at this time point
‘Accessible’ means that effusion could be seen on ultrasound, and it was possible to obtain a sample
Some cats showed effusion on ultrasound examination but it was not possible to obtain a sample for FCoV RNA measurement
FCoV = feline coronavirus; FHS = feline hyperaesthesia syndrome; IFA = indirect immunofluorescence assay; RI = reference interval; SAA = serum amyloid A

Timeline showing the improvement in selected clinical and laboratory parameters before, during and after treatment in predictive values and 95% confidence intervals. Values and days are coloured according to the different study sections. Orange = treatment and hospitalisation in the clinic (days 0–7); blue = treatment at home with re-checks (days 14, 28, 56 and 83); green = follow-up period after the end of treatment (days 168 [week 24], 252 [week 36] and 336 [week 48]). The grey area marks the reference interval of each parameter. (a) Karnofsky’s score (%) modified for cats by Hartmann and Kuffer (1998) (from 0%, indicating ‘dead’, to 100%, indicating ‘completely normal general condition’). 23 (b) Albumin/globulin ratio. (c) Serum amyloid A (mg/l). (d) Lymphocyte count (×109/l). Red asterisks mark significant differences (P ⩽0.05) between parameters on the different treatment days compared with day 0 (before treatment), measured with robust linear-mixed effects models
Changes in viral loads and anti-FCoV antibody titres
In blood, no FCoV RNA could be detected in any cat during the follow-up period, with one exception (cat 8). This cat was FCoV RNA-positive in blood before treatment, became negative from day 7 onwards but was positive again at the first re-check in week 24 (200 copies/ml, cycle threshold [CT] value 38.0), but tested negative again at further re-checks (Figure 2a).

Viral RNA loads of each cat in (a) blood (copies per ml) and (b) faeces (copies per g or per swab) before, during and after antiviral treatment. Blood samples were collected on days 0, 2, 4 and 7 during hospitalisation in the clinic, at the re-checks during the treatment (days 14, 28, 56 and 83) and during the follow-up period after the end of treatment (days 168 [week 24], 252 [week 36] and 336 [week 48]). Faecal samples were collected on days 0–7, days 14, 28, 56 and 83 and days 168 (week 24), 252 (week 36) and 336 (week 48). RNA loads were measured by feline coronavirus (FCoV) reverse transcriptase-quantitative PCR. (c) The anti-FCoV antibody titres of the 18 cats before, during and after antiviral treatment. The samples were collected on days 0 and 7 during hospitalisation in the clinic, at the re-checks during the treatment (days 14, 28, 56 and 83) and during the follow-up period after the end of treatment (days 168 [week 24], 252 [week 36] and 336 [week 48]); anti-FCoV antibody titre was determined by indirect immunofluorescence assay
In faeces, five other cats (cats 2, 6, 7, 11 and 16) were RT-qPCR-positive at least once during the follow-up period; 2/5 cats (cats 2 and 16) were positive at all three re-checks (with decreasing copy numbers per g faeces or per swab); 3/5 cats were positive only in week 48 (Figure 2b).
Anti-FCoV antibodies in serum were still present in all 18 cats at the first re-check (week 24), in 14/15 cats at the second and in 13/14 cats at the third re-check (Table 4). Four cats (cats 3, 8, 16 and 18; all of which had companion cats at home) had a short single time-point increase in anti-FCoV antibody titres (Figure 2c).
Signs compatible with feline hyperaesthesia syndrome
Two cats (cats 8 and 10) that had not shown neurological abnormalities before or during the treatment developed mild neurological signs compatible with FHS during follow-up. These signs were characterised by attacks of excessive licking and twitching of the skin in the lumbar region.
Cat 8 developed signs compatible with FHS in week 47, shortly before the third follow-up re-check. The episodes were characterised by frequent, uncontrolled twitching of the ears, repetitive skin twitches in the lumbar area and tail chasing, occurring several times a day (see video in the supplementary material). Usually, the cat would retreat and hide after such an episode. However, the cat did not appear dazed or disturbed during or after the episodes. In week 48, clinical and laboratory examinations were unremarkable, and no FCoV RNA could be detected in blood and faeces. The anti-FCoV antibody titre increased from 1:100 (week 24) to 1:400 (week 36) and decreased again at the last follow-up re-check (1:100) (week 48). Two weeks later (week 50) and a few days after the cat had been neutered, mild diarrhoea was reported. At that time (when the cat was readmitted), FCoV faecal shedding (CT value 32.6) was detected. Ultrasound examination at that time showed abdominal lymphadenomegaly and no evidence of free abdominal fluid. Lymph node cytology revealed reactive lymphocytes with isolated neutrophils and plasma cells consistent with reactive hyperplasia. In the lymph node, FCoV RNA could be detected at a very low load (CT value 40.0). No FCoV RNA was detected in blood. Physical, dermatological, ocular and neurological examinations were unremarkable. Palpation of the spine and tail identified no sites of pain and no lesions. Laboratory examinations, including Toxoplasma gondii antibody IgG IFA and T gondii antibody IgM IFA, were unremarkable. Further diagnostic work-up with MRI, cerebrospinal fluid (CSF) analysis and electroencephalography (EEG) was offered but declined by the owners. To exclude ectoparasite infestation as a possible cause of signs compatible with FHS, the cat was treated once with a spot-on (esafoxolaner, eprinomectin, praziquantel); however, this did not lead to improvement and episodes continued to occur. However, frequency and severity of episodes improved significantly after 6 months without any other medications and, at the time of manuscript submission, episodes occurred only very rarely.
Cat 10 developed episodes with twitching skin on the back and restlessness several times a day in week 35. The owners reported additional signs of difficulty jumping on objects, increased body temperature (40°C) and anorexia for 2 days. The same evening, the cat received supportive treatment at another hospital (a single dose of metamizole, amoxicillin/clavulanic acid and fluid therapy for 2 days). The owners reported that they had started a second 12-week course of GS-441524 treatment using a substance from an unknown source the evening of the day of discharge from that hospital. The next day, the cat was presented to the authors’ research group for the second follow-up (week 36). No further signs of reluctance to jump were reported, and appetite and body temperature were normal. Physical and neurological examinations (including mental status, posture and gait, cranial nerves, postural reactions, spinal reflexes, perineal reflex) at that time were unremarkable. A thorough palpation of the spine failed to disclose any sites of pain. Haematology and serum biochemistry revealed no abnormalities. Ultrasound examination showed abdominal lymphadenomegaly and a very small amount of free fluid (RT-qPCR-negative for FCoV RNA). No viral RNA was detected in blood and faeces, and anti-FCoV antibody titres were unchanged (1:25, since week 4 after the start of treatment). Further diagnostic work-up with MRI, CSF analysis and EEG were offered but declined by the owners. Subsequently, the owners reported that these signs resembling FHS (episodes of twitching skin on the back and restlessness several times a day) persisted throughout the second 12-week treatment course, and then disappeared a few weeks to months later. At the time of manuscript submission, the cat was clinically healthy without any further episodes.
Discussion
This study demonstrates the successful oral treatment of cats with FIP with a multicomponent drug containing GS-441524 over 12 weeks resulting in sustained remission. No cat experienced a relapse of the initial clinical and laboratory signs indicative of FIP20–22 over a 1-year follow-up period since the initiation of treatment. In a previous study by Murphy et al in 10 experimentally infected cats treated with GS-441524 (2–5 mg/kg SC q24h) for 2 weeks, 2/10 cats had a relapse 4–6 weeks after treatment. 12 The reason for these relapses might be a lower drug exposure (lower daily doses used and shorter treatment duration) compared with the present study (with 5–10 mg/kg PO q24h, as stated by the manufacturer, for 84 days). Additional analyses of the drug used in the study indicate that one tablet of the multicomponent drug Xraphconn contained more GS-441524 than officially stated by the manufacturer (J Horak, unpublished data). Although all clinicopathological parameters typically altered in cats with FIP (increased total protein, globulin and bilirubin concentrations, decreased albumin concentration, albumin:globulin ratio, haematocrit and lymphocyte count)20–22 improved significantly within a few days of treatment in the experimental study, 12 the 2-week course might have been too short for relapse-free FIP treatment. Since the optimal duration of treatment is unknown, the question arises whether 12 weeks are necessary to achieve long-term remission or whether a shorter duration could also be sufficient.The concentration of the active substance GS-441524 in one tablet was determined, which showed that the concentration differed from that stated by the manufacturer. This raises the suspicion that all tablets might contain different amounts of GS-441524 due to the unlicensed and uncontrolled production of this antiviral drug. However, it would be important to know the exact concentration of the active ingredient given to a cat as adverse effects of under- or overdosing are not known.
Within the follow-up period, all cats consistently showed a normal general condition (including cats 8 and 10, despite them developing FHS) and a constant or increasing body weight. A mild to moderate lymphocytosis was seen in 14/18 (77.7%) cats during the treatment period. 18 During the follow-up period, a very mild lymphocytosis was still present in some cats. Since the severity of initial lymphopenia is considered a negative prognostic factor in FIP, 26 lymphocytosis during treatment might indeed be a positive prognostic factor for recovery. In cats, lymphocytosis is often caused by excitement (stress after transport to the clinic and examinations performed) to which cats are very sensitive. It remains of particular interest to further determine the immunological processes and cellular immune response against FCoV before, during and after treatment. Haematocrit was very mildly decreased in only one cat at the second follow-up. SAA was minimally elevated in some cats, but as this parameter is a non-specific inflammatory marker, it might have been slightly elevated for many reasons.During treatment, there was not only a normalisation of clinicopathological parameters, but also a rapid elimination of viral RNA in blood, effusion (if still present at all) and faeces. At the end of treatment (week 12), all cats were FCoV RNA-negative in blood and remained negative during the follow-up period, with one exception. One cat (cat 8) had temporarily a very low FCoV RNA blood load of 200 copies/ml in week 24. Intestinal reinfection with FCoV and translocation into blood would seem likely as the anti-FCoV antibody titre in this cat had risen to week 36 (1:100 in week 24 and 1:400 in week 36), even though FCoV could not be detected in faeces in either week 24 or week 36. Alternatively, the virus was still present in another compartment (eg, CSF) during the entire treatment period and translocated into blood. However, the cat remained healthy.
Five cats (cats 2, 6, 7, 11 and 16) became enterically reinfected in the follow-up period as indicated by faecal FCoV shedding, compatible with the fact that all of them had companion cats at home. While three cats showed re-shedding only in week 48, two cats (cats 2 and 16) continued to shed during the entire follow-up period. No relapse of clinical signs compatible with FIP occurred in these five cats. Only 1/5 re-shedding cats (cat 16) showed a rise in anti-FCoV antibody titres during the follow-up period. Even during the treatment period, re-shedding could even be seen in one cat (cat 4) and new onset of shedding in two cats (cats 9 and 11). Reinfections caused by their partner cats are the most likely explanation, based on sequencing of FCoV RNA from faeces as shown in four cases; 24 – 8/12 partner cats were positive for FCoV in faeces, and in 4/8 of these samples, phylogenetic analysis showed common clusters with the corresponding FIP cats; 24 therefore, in the five cats shedding during follow-ups, the most likely source of reinfection were the companion cats.Thus, antiviral drugs should not be used just to stop faecal FCoV excretion, as reinfections occur continuously, even though a recently published study by Addie et al 27 reported a complete cessation of FCoV shedding in 29 healthy cats treated with oral GS-441524 (2.0–4.0 mg/kg PO q24h for 4–7 days, in 4/29 after a second round of treatment). 27 In 18/29 cats, the known duration of absence of FCoV in faeces after completion of treatment was in the range of 1–157 days. However, it must be noted that not all cats in that study underwent follow-up testing of FCoV shedding; 11/29 cats were not re-tested after treatment. Therefore, it is not known whether these cats might have become reinfected later. 27 In addition, viral resistance of FCoV strains to antiviral compounds can occur, as demonstrated in an in vitro study. 23 , 28 FCoV is able to mutate and has developed resistance to the antiviral protease inhibitor GC376; 28 it is thus, in the opinion of the authors, contraindicated and not recommended to administer GS-441524 to cats not suffering from FIP. Only cats with proven FIP after a thorough diagnostic approach should receive GS-441524. The main focus in a multi-cat household should be to reduce the risk of FCoV infection and transmission (eg, by thorough hygiene, reducing numbers of cats per group, a sufficient number of litter trays and, if possible, outdoor access). 20
A total of 12 cats showed abdominal lymphadenomegaly during the follow-up period (at any time point). This could be interpreted as a sign of an exaggerated, genetically determined reaction of the immune system comparable with a prolonged healing process or a persistent stimulus (eg, through presence of residual virus, as it is possible that FCoV was still present in certain compartments). Severe generalised follicular lymphoid hyperplasia was also found in the cured cat that died in a traffic accident. 25 The high proportion of cats with abdominal lymphadenomegaly could be a result of a recent (re)infection with FCoV, but this is not very likely as many different cats showed this change without any other signs of reinfection. Alternatively, lymphadenomegaly might be a long-term sequela of the FIP itself or possibly be a long-term adverse effect of antiviral treatment. Further clinical trials with cytological examination of the enlarged lymph nodes, especially long-term observational trials, would be interesting to understand more about the persistence of abdominal lymphadenomegaly.
The two cats (cats 8 and 10) that developed signs compatible with FHS after successful treatment of FIP with GS-441524 are the first published cases of such a syndrome after FIP treatment. However, in a round table discussion at the congress of the International Society for Companion Animal Infectious Diseases (ISCAID) (September 2022 in Glasgow, UK), other researchers reported a few similar observations. FHS is a poorly understood syndrome, first described in 1980, 29 and comprises episodes of twitching and rolling of the skin of the back and signs of discomfort, which mostly appear spontaneously, sometimes triggered by touching of the lumbar area. Various medical conditions have been reported to cause signs indicative of FHS, including dermatological, orthopaedic, neurological or behavioural diseases.29–33 In many cases, the underlying cause cannot be detected. 34 In the two cats from the present study, a neurological cause was considered the most likely as clinical, dermatological and orthopaedic examinations were unremarkable, and no spinal or tail pain was identified. Yet, meningitis cannot be excluded without further examinations. It is striking that these two cats initially had no neurological abnormalities but developed signs compatible with FHS after successful treatment. Since FCoV RNA was not detected in blood, and laboratory parameters and neurological examinations were unremarkable, these signs were not considered a relapse of FIP. This was further supported by the subsequent good clinical condition (besides the signs compatible with FHS) of both cats.
Cat 8 started faecal re-shedding of FCoV and showed an increase in anti-FCoV antibody titres from 1:100 to 1:400 (2 weeks after the third follow-up; week 50) a few weeks after the onset of signs compatible with FHS. However, the increase in anti-FCoV antibody titre likely only indicated reinfection with FCoV, and not a relapse of FIP.
In cat 10, the owner reported short-term (2 days) clinical signs that could have been suggestive of a relapse of FIP (increased body temperature, reluctance to jump, spinal hyperaesthesia) at approximately the same time as the onset of signs resembling FHS, but these other signs disappeared after 2 days. At the second follow-up, neither these nor other neurological signs indicative of multifocal neurological disease were verified.35,36 This cat was started on another 84-day treatment of presumptive GS-441524 SC by the owner, but the owner reported that the signs resembling FHS persisted throughout the second treatment and only later resolved spontaneously.
It is possible that mild neurological signs could also be a type of ‘long FIP syndrome’. In a study on children with COVID-19 and paediatric inflammatory multisystem syndrome temporarily associated with SARS-CoV-2 (PIMS-TS), 4/27 children who had been previously healthy without any SARS-CoV-2-related signs or other comorbidities developed new-onset neurological signs. The neurological situation improved in all children, with 2/4 children showing a complete recovery after 11 and 18 days, respectively. 37 In the present study, no virus could be detected in the analyses allowed by the owners, which raises the hypothesis that long-term immunological effects triggered by the virus might also contribute to the development of neurological manifestations resembling FHS.
Since neurological signs compatible with FHS appeared only a few weeks to months after the treatment, this could also be a long-term adverse effect (eg, neuropathy) of GS-441524 therapy. In the treatment of cats with GS-441524, there are no long-term data on potential adverse effects to date. Likewise, in human medicine, relatively little is known following trials on remdesivir, which is the prodrug of GS-441524, except for the occurrence of delirium in two patients in an open-label study of COVID-19 disease 38 and one possible report of neurological complications in a phase 1 trial of Ebola virus infection. 39 However, experience from previous viral pandemics suggests that the immunological response to the viruses themselves has the potential to cause neuropsychiatric manifestations, including encephalopathies and psychoses. 40 Further studies with controlled drug concentrations are needed. This is especially important since in the currently available unlicensed preparations, additives and their possible effects are not known. Despite being unlicensed, and the production of and treatment with drugs containing GS-441524 not being allowed in most countries (with the exception of the UK and Australia), these compounds are still widely used.
The main limitations of the study were that complete follow-up examinations were not available for all cats and the actual amount of the active ingredient GS-441524 each cat received was unknown. Furthermore, the owners of the two cats with signs compatible with FHS opted against further diagnostic work-up with EEG, MRI and/or CSF. Another limitation of this study was that cytology was not performed in all cats with abdominal lymphadenomegaly, which could have provided more information on the cause of the persistence of enlarged abdominal lymph nodes.
Conclusions
This is the first long-term (1-year) follow-up study of cats with a confirmed diagnosis of FIP treated with GS-441524 resulting in sustained remission. Thus, treatment with GS-441524 was effective against FIP in both the short term and the long term because all cats were still alive on the day of submission of the present manuscript (with the exception of the one cat that died in a road traffic accident), which is more than 2 years after the diagnosis, and physical, neurological and laboratory examinations remained unremarkable throughout the observation period. Two cats of this cohort developed neurological signs resembling FHS but these neurological signs did not progress and appeared to resolve spontaneously several months later. Whether mild neurological signs compatible with FHS and/or abdominal lymphadenomegaly are associated with a ‘long FIP syndrome’, are an adverse effect of treatment or reflect the presence of residual virus in these compartments, or whether signs resembling FHS are unrelated to the treatment of FIP requires further investigation (summarised in Box 1).
Key findings and questions raised by the present long-term follow-up study

Supplemental Material
File 1
Xraphconn (Mutian) package insert.
Footnotes
Acknowledgements
The oral multicomponent drug Xraphconn was kindly provided by Mutian Life Sciences. The authors thank Eva Bönzli, Julia Klaus, Martina Stirn and Evelyn Kuhlmeier from the University of Zurich for their excellent technical assistance. The FCoV RT-qPCR and anti-FCoV antibody measurements were performed using the logistics of the Center for Clinical Studies at the Vetsuisse Faculty of the University of Zurich.
Author note
This paper was presented in part at the International Society for Companion Animal Infectious Diseases (ISCAID) congress (September 2022 in Glasgow, UK) and the Internal Medicine and Clinical Laboratory Diagnostics (InnLab) conference (February 2023 in Göttingen, Germany).
Supplementary material
The following files are available as supplementary material:
File 1: Xraphconn (Mutian) package insert.
File 2: Video of a cat (cat 8) showing signs of feline hyperaesthesia syndrome.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognised high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers) for all procedure(s) undertaken (prospective or retrospective studies). For any animals or people individually identifiable within this publication, informed consent (verbal or written) for their use in the publication was obtained from the people involved.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
