Abstract
Objectives
The present study aimed to establish reference intervals (RIs) for ultrasonographically derived caudal vena cava (CVC) parameters in healthy cats.
Methods
A prospective, observational, experimental single-centre study involving 110 cats was conducted. Two trained operators scanned all cats in a randomised order. Two sets of cine loops per cat were conducted by each operator from the subxiphoid view with cats in lateral recumbency. Two blinded raters measured the narrowest inspiratory (CVCinsp) and widest expiratory (CVCexp) diameter over two separate respiratory cycles for each operator (four measurements in total for each rater) at the point the CVC crosses the diaphragm. The CVC collapsibility index (CVCCI) was calculated. Reference intervals and limits were calculated for CVC parameters with outliers removed to include the central 95% of CVC values and the non-parametric 90% confidence intervals (90% CIs). The CVCCI interval included the central 95th (2.5th and 97.5th) centiles bounded by the 90% CI. Intra- and inter-rater and intra- and inter-operator reliabilities were calculated for each parameter using a two-way random-effects model. A Bland–Altman plot was used to visualise the CVCCI between the two raters.
Results
In total, 880 measurements were included for each diameter and 440 CVCCI values were calculated. The CVC diameters were normally distributed, whereas the CVCCI was not. The calculated RIs were 1.4–6.1 mm for CVCinsp and 2.2–6.7 mm for CVCexp. The median (range) CVCCI was 23% (11–36%) with 2.5th and 97.5th centiles of 2% and 67%, respectively. There was a moderate to good intra-rater and intra-operator reliability for CVC diameter. The intra-rater reliability for the CVCCI was moderate. The inter-rater and inter-operator reliabilities for the CVCCI were poor.
Conclusions and relevance
The CVC RIs determined from the subxiphoid site in cats appear to be wider than those reported in dogs, and rater and operator variability is present. Whether precise measurements of CVC parameters are suitable to guide clinical decisions remains to be determined.
Keywords
Introduction
Assessing intravascular volume status (IVVS) is an essential task when developing a diagnostic and therapeutic plan for a sick cat. Hypovolemia and fluid overload (FO) can be detrimental, as suggested by current evidence in people and animals.1–5
Perfusion parameters, such as mentation, heart rate, pulse quality, mucous membrane colour, capillary refill time and temperature, as well as mean arterial blood pressure and lactate concentrations, are often used to identify veterinary patients with hypovolemia. However, these parameters have been shown to be unreliable. This can contribute to either underdosing or, more often, overprescription of intravenous fluids (IVFs) and subsequent FO, especially in patients with pre-existing risk.6 –8
Currently, FO is defined as a pathological accumulation of extravascular body water with diffuse interstitial oedema. 2 Diagnosis remains challenging, because clinical signs other than weight gain lack sensitivity, manifesting when significant morbidity is already present. Cats are particularly predisposed to FO owing to their relatively small blood volume, 9 decreased rate of fluid elimination 10 and risk of occult cardiomyopathies. 11 Anaemia, which also predisposes to FO, is a common finding in sick cats.12,13 Moreover, cats in circulatory shock often present in a hypothermic state, which could contribute to FO during and after stabilisation.14,15
Studies have demonstrated that even patients with signs of hypovolemia may not respond to IVF administration. In a study in critically ill people, only 50% of subjects were considered fluid responders. 16 By contrast, studies suggest that 80–90% of dogs may be fluid-responsive. 17 Interestingly, recent studies in hypovolemic and hypotensive cats have demonstrated only 30–40% to be fluid responders.18,19 The low proportion of fluid responders in addition to the predisposition to FO establishes the need for a readily available tool to assess IVVS in cats.
The caudal vena cava (CVC) is a highly elastic vessel. During the respiratory cycle, intrathoracic pressure change, affecting the volume within the thoracic and abdominal portions of the CVC. This creates a variation in CVC diameter, which is exacerbated in hypovolemic states and mitigated in hypervolemic states.20,21 With the use of point-of-care ultrasound (POCUS), this change in diameter can be monitored and used as a non-invasive tool for assessment of IVVS.20,22 In the past decade, the use of POCUS to assess IVVS and to guide IVF administration has become a standard of care in critically ill people. 20 The feasibility of obtaining ultrasonographically derived CVC parameters has been established in cats, foals, calves and dogs, with reference intervals (RIs) established in dogs.23 –26 To determine whether CVC parameters can be used as a tool to assess IVVS and guide IVF administration in sick cats, RIs from healthy cats are needed.
The present study thus aimed to establish RIs for ultrasonographically derived CVC parameters during inspiration and expiration, as well as the CVC collapsibility index (CVCCI), in healthy, lightly sedated cats obtained from the subxiphoid view. A second objective was to assess reliability between operators and raters.
Materials and methods
Animals
The present study was a prospective, observational, experimental single-centre study. The study protocol was approved by the Massey University Animal Ethics Committee (protocol 19/136). One-hundred and ten healthy research-purposed cats from the Centre for Feline Nutrition at Massey University, New Zealand, were enrolled. Cats were deemed healthy based on physical examination, complete blood count and biochemistry analysis. Additionally, all cats were screened for occult cardiomyopathy using the 2-min screening echocardiogram for cats as described by Loughran et al. 27
Devices and operators
The study was performed using a Mindray ultrasound system (model M7-vet; BCF), with a medium-frequency (7 MHz) microconvex transducer with depth set at 5–7 cm. Without clipping, the fur was parted and a combination of alcohol and ultrasonographic gel was used as the coupling agent. The two operators (both board-certified emergency and critical-care specialists) were extensively trained in abdominal, pleural space, lung and cardiovascular POCUS.
Study protocol
Approximately 10–30 mins before examination, each cat was sedated using butorphanol (Butorgesic 10 mg/ml; Troy Laboratories) at a dose of 0.3 mg/kg SC and gabapentin (Neuropentin 100 mg; Sinokem Pharmaceuticals Ltd) at a dose of 100 mg per cat PO. Low-stress handling, provided by a certified research assistant, was used for all cats throughout each POCUS evaluation. Cats were placed on a table and scanned in right lateral recumbency for both operators.
Operators were blinded to each others’ sonographic image acquisition. The two operators scanned each cat in a randomised order. For all scans, the probe was placed on midline just caudal to the xiphoid cartilage in the longitudinal axis, at approximately a 45° angle to the spine and with the marker pointing cranially (Figure 1). The liver and diaphragm were identified, and the probe was then gently fanned to the right of the midline until a longitudinal view of the CVC was obtained, visualised as two horizontal parallel lines (walls of the CVC) crossing the diaphragm (Figure 2). The probe was then slowly fanned through all longitudinal planes across the CVC to identify its luminal centre, subjectively identified as the largest distance between the dorsal and ventral wall of the CVC. Multiple 5-10 s cine loops were recorded for each cat and operator for post-hoc measurement and analysis of the CVC parameters.

A schematic illustration of the position of a cat, an ultrasound probe and caudal vena cava (blue vessel). All cats were scanned in right lateral recumbency

Schematic illustration of the caudal vena cava (CVC) imaged from the subxiphoid view. The CVC is identified as two parallel lines crossing the diaphragm. The CVC was measured on the inside of the lumen at the most caudal point where the diaphragm was still in contact with the dorsal wall of the vessel (blue line)
Measurements
Three to four cine loops were recorded for each cat by each operator. The two raters (both emergency and critical care residents) then assessed the cine loops for each cat and operator blinded and in a randomised order. Rater 1 determined which two cine loops, from each cat, should be assessed (based on image quality and motion artefacts) and which frames within the same respiratory cycle should be used to measure inspiratory and expiratory CVC diameters. The narrowest diameter during inspiration (CVCinsp) and the widest diameter during expiration (CVCexp) were then measured for each cat, from the two cine loops described above, by each operator, resulting in a total of eight measurements per cat. Measurement of the CVC by each rater was standardised. For this, the intraluminal CVC diameter was measured at the most caudal point where the diaphragm could be visualised as being in contact with the dorsal wall of the CVC (Figure 2). The measurement was then obtained at a 90° angle from the visualised ventral wall to the dorsal wall of the CVC. A fully collapsed CVC was recorded as 0. The CVCCI was then calculated using (CVCexp – CVCinsp)/CVCexp × 100.
Statistical analysis
Continuous data were described using the mean (SD) if normally distributed, or the median and interquartile range (IQR) if non-normally distributed. The range was included for all continuous variables. Normality was assessed using histograms.
The CVCCI was calculated as described above. Cine loops resulting in a CVCCI of <0.01 were removed from analysis because a negative CVCCI is not biologically plausible in a spontaneously breathing patient.
Intra- and inter-rater or operator reliability
The intra-rater reliability was assessed using a two-way mixed-effects model, using the absolute measure of agreement. 28 The same approach was used for determining intra-operator reliability. The two intra-rater measurements were defined as intra-rater for comparison within raters and intra-operator for comparison within operators.
Inter-rater reliability was assessed using a two-way random-effects model to enable generalisation of the RI to raters who possess the same characteristics as the rater in the present study. 29
The intraclass correlation coefficient (ICC) random-effects model assumes one measurement per animal per rater. 28 In the present study, each rater contributed four measurements per animal. To account for this and maintain the ICC random-effects model structure, a variable combining cat (1–110) and observation number (1–4) was generated, so that each measurement by each rater was considered within the model. Similarly, a variable combining cat and operator measurement (1–4 for each rater) was generated. Rather than absolute agreement, the consistency of agreement between raters was considered for this model based on the (3, k) model proposed by Shrout and Fleiss. 28 The two inter-rater measurements were defined as inter-rater for comparison between raters and inter-operator for comparison between operators.
The agreement of the operators performing the measurements, or the reliability between measurements within raters, was considered poor if the ICC values were less than 0.50, moderate if between 0.50 and 0.75, good if between 0.75 and 0.90 and excellent if greater than 0.90. 29
Bland–Altman plots were used to visualise relationships between the differences and the averages of the CVCCI of the two raters, evaluating any systematic biases and identifying possible outliers. 30 Kendall’s tau (τ) was calculated as a measure of the concordance component of agreement between the two raters. The bias parameters for the sample were estimated using the mean sign of the difference between the two raters.
Reference intervals
To remove right skew, variables were log-transformed and outliers determined in a stepwise process using Tukey’s interquartile fences. 31 The criteria for outlier exclusion were values exceeding IQR fences set at Q1–1.5 × IQR and Q3 + 1.5 × IQR. At each step, outliers were identified and removed from the working data set, with descriptive statistics generated including IQR, Q1 and Q3 values. New interquartile fences were generated and values excluded until no further outlying values were identified and the descriptive statistics no longer varied. Outliers were identified and presented on the data from both raters. Data with outliers removed were presented as medians, IQR and ranges.
The RIs were calculated using the data set with the outliers removed, to encompass the central 95% of the reference values, and to be bounded by the upper and lower reference limits. 30 For inspiration and expiration, non-parametric 90% confidence intervals (90% CIs) based on centiles were generated for each reference limit, based on the study having a sample size greater than 120 values.
CVCCI interval
Because the CVCCI is given as a percentage, and is therefore bounded by 0 and 100, the methods for calculating the RIs outlined above are inappropriate as the assumptions were violated. Instead, the 2.5th and 97.5th centiles for the CVCCI and the corresponding 90% CIs based on this centile were generated for the upper and lower limits. Despite not conforming to the definition and assumptions of the RIs, the CVCCI interval is henceforth referred to as RIs. All statistical analyses were conducted using Stata/SE, version 17 (Stata). P <0.05 was considered to be statistically significant.
Results
Descriptive statistics
One hundred and ten cats with a total of 880 measurements were included in the data set. The mean (range) age was 5.2 (1–14) years and the mean (range) body weight was 3.8 (2.1–6.3) kg. Twenty-six cats were entire females, 26 were spayed females and 58 were castrated males. The mean (SD; range) CVC at inspiration was 3.33 (1.32; 0.48–8.01) mm (n = 843). The mean (SD; range) CVC at expiration was 4.14 (1.21; 1.08–7.87) mm (n = 850). There was a positive correlation between CVC inspiration and expiration measurements (Figure 3).

Scatterplot of the caudal vena cava (CVC) inspiratory and expiratory diameters in millimetres, stratified by rater (n = 880 measurements)
Reference intervals and limits
The descriptive results for CVC parameters with outliers excluded are presented in Table 1. In total, 36 (4%) and 19 (2%) measurements were removed from the inspiratory and expiratory measurements, respectively. Of the calculated CVCCI values, 159 (18%) had to be removed owing to not being biologically plausible (CVCCI < 0%). Established RIs are presented in Table 2.
Descriptive statistics for caudal vena cava (CVC) diameter (mm) during inspiration and expiration and CVC collapsibility index (CVCCI) for each rater and combined (n = 880 measurements, with outliers removed)
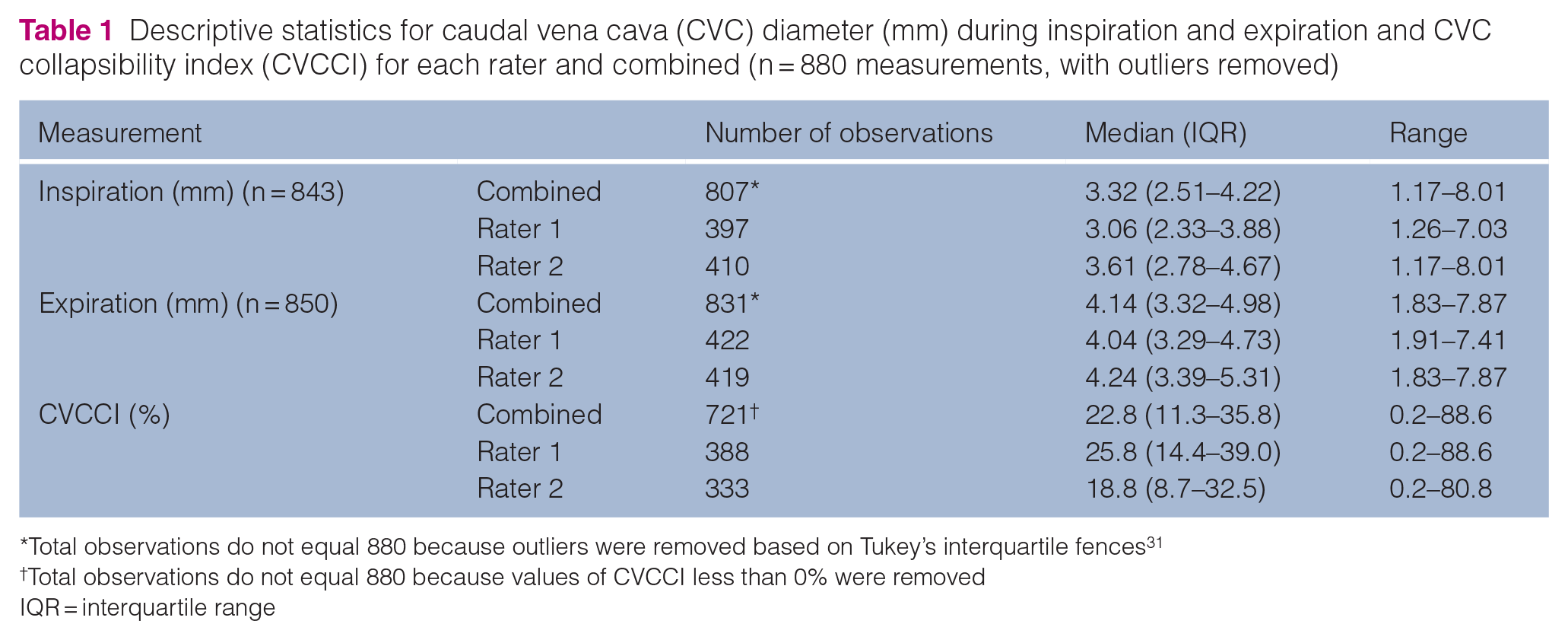
Total observations do not equal 880 because outliers were removed based on Tukey’s interquartile fences 31
Total observations do not equal 880 because values of CVCCI less than 0% were removed
IQR = interquartile range
Reference intervals for caudal vena cava (CVC) parameters obtained at the subxiphoid view in lightly sedated cats

The interval presented for CVCCI was based on the 2.5th and 97.5th centiles
CI = confidence interval; CVCinsp = CVC diameter during inspiration where the CVC crosses the diaphragm; CVCexp = CVC diameter during expiration where the CVC crosses the diaphragm; CVCCI = CVC collapsibility index
Intra- and inter-rater reliability
The ICC for intra- and inter-rater reliability is shown in Table 3. The reliability for CVC diameter was considered moderate to good within raters, whereas reliability between raters was considered poor to moderate. For the CVCCI, the reliability within raters was considered moderate and poor between raters.
Intra-rater and inter-rater reliability measures for caudal vena cava (CVC) parameters as calculated by a two-way mixed-effects model and intraclass correlation coefficient random-effects model for intra-rater and inter-rater, respectively

CVCinsp = CVC diameter during inspiration where the CVC crosses the diaphragm; CVCexp = CVC diameter during expiration where the CVC crosses the diaphragm; CVCCI = CVC collapsibility index
In total, 312 CVCCI values could be compared between raters. The Bland–Altman plot identified 14 (4.5%) observations outside the 95% limit of agreement of –24.9% and 38.9% (Figure 4). The mean difference for the CVCCI between raters was 7.0%, with mean values for both raters between 1.0% and 79.8%. Kendall’s τ was 0.36 (95% CI = 0.27–0.43, P <0.001). The bias parameter was 0.33 (95% CI = 0.23–0.43), indicating that rater 1 was observing higher CVCCIs compared with rater 2.

Bland–Altman plot of the caudal vena cava collapsibility index of rater 1 and rater 2 (n = 312 paired observations)
Intra- and inter-operator reliability
The ICC for intra- and inter-operator reliability is shown in Table 4. The reliability for the CVC diameter was good within operators but poor between operators. The reliability for the CVCCI was poor between operators and moderate to good within operators.
Intra- and inter-operator reliability for caudal vena cava (CVC) parameters as calculated by a two-way mixed-effects model and intraclass correlation coefficient random-effects model for intra-rater and inter-rater, respectively

CVCinsp = CVC diameter during inspiration where the CVC crosses the diaphragm; CVCexp = CVC diameter during expiration where the CVC crosses the diaphragm; CVCCI = CVC collapsibility index
Discussion
The present study has established RIs for ultrasonographically derived CVC parameters from 110 healthy, lightly sedated cats. The RIs established are 1.4–6.1 mm for inspiratory CVC diameter and 2.2–6.7 mm for expiratory CVC diameter. The median (range) CVCCI is 23% (11–36%) with 2.5th and 97.5th centiles of 2% and 67%, respectively. The strengths of the present study include the prospectively collected data and the large sample size. Moreover, the robustness of the data was further increased with two operators and two raters, resulting in 880 measurements for inspiratory diameter and expiratory diameter, respectively. The study showed varying results for reliability between and within raters and operators.
Overall, the reliability for the CVCCI was considered poor to moderate within and between raters and operators. Multiple studies have previously assessed inter-rater and intra-rater reliability for the CVCCI in human and veterinary patients and showed similar results.19,22,25,32 –35 One recent study evaluating CVC parameters in cats during blood donation concluded that the CVCCI is highly operator-dependent. 34 Another study, assessing fluid responsiveness in cats using CVC parameters, also demonstrated a poor intra- and inter-rater reliability for the CVCCI. 19 Similarly, a recent pilot study in 20 cats evaluating CVC parameters in lateral and standing positions showed a poor inter-operator reliability for both CVC diameter and CVCCI. 24 In dogs, one study establishing RIs for CVC parameters also reported poor intra-rater reliability for the CVCCI obtained from the subxiphoid window. 25 A likely reason for the poor reliability is that the CVCCI is a generated variable, and multiplicative augmentation in diameter differences will lower the reliability compared with individual diameter measurements. Considering the small nature of the feline CVC (2–7 mm), as little as a 1 mm difference in either inspiratory or expiratory diameter for one rater, and not the other, could cause a difference in the CVCCI of 25%. Even though a measuring protocol was established before the study and both raters measured the same frame for each respiratory cycle, factors such as image quality or the subjective assessment of where the diaphragm ends is likely to affect the measurement within the range of 1 mm. Another reason for the varying reliability within raters and operators might be a consequence of respiratory movement of the liver, diaphragm and CVC that occurs during spontaneous breathing, making it difficult to measure the diameter at the same site along the CVC during both inspiration and expiration. Moreover, the cats in the present study were only lightly sedated, and motion artefacts, shallow breathing and purring could have influenced the results.
In spontaneously breathing cats, the abdominal portion of the CVC collapses during inspiration.24,34 This is owing to increased negative intrathoracic pressure during inspiration and a consequent decrease of right atrial and right ventricular pressure. Concurrently, inspiration also increases the abdominal pressure, creating a gradient that shifts blood from the abdominal portion of the CVC into the thoracic portion, augmenting venous return to the heart. The extent of this gradient is conditioned by the absolute level of right atrial pressure and compliance of the CVC. 21 Based on this premises, the degree of CVC collapse has been shown to be increased in hypovolemic patients where the right atrial pressure is decreased and compliance increased and, reversibly, CVC collapse, decreased in FO patients. 36
However, hypovolemia is not the only condition that can exacerbate CVC collapse. Conditions that have been demonstrated to contribute to an increase in CVCCI (and underestimation of intravascular volume) include increased respiratory effort, intrabdominal hypertension or a measuring error, such as ‘off-centre scan’ (cylinder tangent effect). By contrast, conditions that have been described to mitigate CVC collapse and contribute to a decrease in the CVCCI (and overestimation of the intravascular volume) include cardiac tamponade, decreased inspiratory effort/shallow breathing or severe pulmonary thromboembolism. 21
According to the results of the present study, the ranges for CVC diameter and CVCCI in cats appear to be wider than those in dogs and people, especially for the CVCCI. There are limited studies on CVC parameters in cats; however, the majority show similar results to those of the present study. One small study, investigating CVC parameters in cats during blood donation, showed a median (range) CVCCI of 19.4% (5–55%) prior to blood donation. The results from Sänger et al 34 are similar to those of the present study, with measurements of the CVC diameters in the range of 2.2–5.6 mm and 2.8–7.1 mm during inspiration and expiration, respectively. One recent study investigating whether CVC parameters (from the right transhepatic view) can be used to predict fluid responsiveness in cats, demonstrated a median (range) CVCCI of 47% (31–72%) for fluid responders and 15% (5–73%) for non-responders. That study recommended an optimal cut-off at >31% for fluid responders, which coincides with the range for the CVCCI from our study. 17 Moreover, a recent study investigating CVCCI in 12 cats found the mean CVCCI to be 28 ± 3%. 37
Multiple studies have demonstrated a significant variation in CVCCI in cats and other species. Considering this and the non-IVVS-related causes for changes in CVC collapsibility, it is possible that absolute CVC values at end expiration or comparing CVC diameter to a less collapsible vessel (eg, the aorta) in sites where collapsibility is minimal, may have greater clinical utility. Indeed, studies in animals and people have demonstrated that the CVC-to-aorta ratio may be a reliable method for IVVS assessment.22,25,33,38–42 Further studies are needed to establish the feasibility and reliability of this ratio in cats. Furthermore, research focusing on the subjective classification of the CVC as small, normal or enlarged may prove to be more transferable to the clinical setting.
The limitations described above should not preclude the use of the CVCCI in cats. In people, the greatest agreement between inferior vena cava collapsibility index and central venous pressure is seen when measurements in patients fall outside of the RIs. 20 Therefore, in a clinical context, identification of cats with CVC parameters that fall outside of the RI could potentially permit differentiation of a cat benefiting from IVF administration from one in which it could potentially be harmful, especially when combined with other tools for the assessment of IVVS.
Another limitation of this study is the relatively high number of CVCCIs that were excluded owing to biological implausibility (CVCCI <0%). However, all cats had at least one plausible CVCCI calculation from the multiple cine loops that were recorded for each cat. The high number of negative CVCCI calculations in the current study is likely owing to movement of the probe or cat that occurred within a respiratory cycle, resulting in an ‘off-centre scan’ and an expiratory diameter appearing smaller than the inspiratory diameter. If this occurred in a clinical scenario, the clinician is likely to scan the CVC over multiple respiratory cycles and subjectively assess the CVCCI, which was not undertaken in the present study. Based on all cats contributing at least one CVCCI calculation, we consider that the RIs calculated for this value is representative of this population of healthy cats.
Conclusions
The present study has established RIs for CVC parameters in healthy cats. It is shown that, while intra-rater and operator reliability was acceptable, inter-rater and operator reliability was not. The next step in evaluating CVC parameter utility in cats is to determine whether the established RIs can be used to assess cats with changes in IVVS.
Footnotes
Acknowledgements
We thank Dr Janelle Wierenga, DVM, MPH, DACVECC, PhD and Ms Rebecca Owen, BSc for their immense help with image acquisition and data processing.
Author note
Preliminary results from this study were presented as an abstract (poster) at the European Veterinary Emergency and Critical Care Society congress in Ghent, Belgium, 2022.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This project received funding from the Massey University Wenzel Research Fund (RM22764).
Ethical approval
The work described in this manuscript involved the use of experimental animals and the study therefore had prior ethical approval from an established (or ad hoc) committee as stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers) for all procedure(s) undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
