Abstract
Objectives
Ultrasonography of the caudal vena cava (CVC) has been previously established to assess fluid status in dogs but not in cats. The aim of this study was to determine CVC diameter changes during feline blood donation.
Methods
Inter- and intra-observer variability were assessed in 11 client-owned cats. Minimal and maximal CVC diameters were assessed longitudinally in the subxiphoid view (SV) and right paralumbar view (PV), and transversely in the right hepatic intercostal view (HV). Eighteen client-owned, healthy, anaesthetised cats were evaluated during 21 blood donation procedures of 10 ml/kg in the same anatomical locations before (T0) and after (T1) blood donation, and after volume resuscitation with 30 ml/kg lactated Ringer’s solution (T2). The CVC index was calculated.
Results
Intra-observer variability was acceptable for all probe positions, except for the HV, whereas inter-observer variability was considered unacceptable for all probe positions. Complete measurements were obtained during 21 blood donations at T0, T1 and T2 at the SV, during 18/21 blood donations at the HV and during 16/21 blood donations at the PV. At the SV, the minimal CVC diameter between T1 and T2 (P <0.001), and the maximal CVC diameter between T0 and T1 and between T1 and T2 (P <0.001) were significantly different. At the HV, the minimal vertical diameter, maximal vertical diameter and minimal horizontal diameter were different between all timepoints (P <0.001). The maximal horizontal diameter was different between T1 and T2 (P = 0.002). At the PV, both diameters were different between all timepoints (P <0.001). The CVC index was not different between timepoints.
Conclusion and relevance
Significant probe position dependent CVC diameter changes with marked overlap were observed before and after blood donation, and after fluid bolus. No absolute CVC diameter could be used to indicate hypovolaemia. Ultrasonographic assessment of the feline CVC is highly operator-dependent. The CVC index is not useful in cats.
Introduction
To guide appropriate therapeutic interventions, clinical estimation of the true intravascular volume status is critical in feline medicine. 1 In human medicine, accurate assessment of volume status is essential to optimise fluid resuscitation and avoid hypo- and hypervolaemia in hypovolaemic patients. 2 Subjective and insensitive parameters, such as physical examination findings, biochemical markers and the results of non-invasive blood pressure measurements, are commonly used as measures to assess the volume status in these patients. However, no single specific parameter has been established.3,4 Central venous pressure is considered a weak parameter for the estimation of volume status in humans. 5 Other invasive or technically sophisticated methods, such as the pulmonary artery occlusion pressure measurement and transoesophageal or transthoracic echocardiography, are riskier, more invasive and more technically challenging, and therefore are inappropriate for emergency patients. 6
For several years, assessment of the diameter of the inferior vena cava (IVC) has been a frequently used technique in hypovolaemic human patients for monitoring volume status. 7 It is a non-invasive bedside technique that can be rapidly performed immediately after the patient is admitted to the emergency room. The aortic diameter, IVC collapsibility index and IVC:aorta ratio are also considered appropriate parameters for assessing volume status. In humans, the aortic diameter does not vary significantly with increasing or decreasing intravascular volume due to the aorta having a thicker and more rigid vessel wall than the vena cava. Therefore, the IVC:aorta ratio is currently considered to be a useful parameter for assessing volume status. 8
Multiple studies in dogs have evaluated the use of ultrasonographic measurements of the caudal vena cava (CVC) diameter, aorta and CVC:aorta ratio for assessing volume status.9–11 In one study, the aortic diameter did not change significantly with decreasing intravascular volume, whereas the CVC and CVC:aorta ratio were significantly reduced after blood donation. 9 The CVC diameter also decreased after the repeated administration of furosemide owing to fluid loss and concomitant hypovolaemia in initially normovolaemic dogs. 12 Reference intervals for ultrasonographically measured CVC and aortic diameters using different ultrasonograhic views in dogs of different body weights have been established. 13 However, the standardisation and training of this technique are critical. A comparison between one cardiologist and two non-cardiologists after a 6 h training session showed high inter-observer variability for measurements of the CVC diameter at different sites. 14 To date, no studies evaluating this technique in cats have been published.
Therefore, the aims of this study were to evaluate inter- and intra-observer variability of the ultrasonographic CVC diameter assessed in cats and the CVC diameter assessed at different ultrasonic probe positions during blood donation in cats.
Materials and methods
Ethical approval
This study was approved by the ethical committee of the Center of Clinical Veterinary Medicine of the Ludwig-Maximilians-University Munich (number 115-06-03-2018). Cat owners signed an informed consent form before participating in the study.
Animals
Inter- and intra-observer variability
For the assessment of inter- and intra-observer variability, client-owned hospitalised cats without evidence of hypovolaemia, based on history and physical examination findings, were evaluated. Patients were hospitalised for blood donation purposes or because of illness.
Blood donation and CVC diameter assessment
The study population consisted of client-owned healthy cats that were presented for blood donation. The inclusion criteria for the cats were defined as being between 1 and 10 years old and having a body weight >3.5 kg. Only clinically healthy cats without a history of chronic illness were included. Health status was assessed by physical examination, a complete blood count and a biochemistry panel.
All healthy blood donor cats were anaesthetised intravenously (VasoVet, 22 G; B Braun Vet Care) with butorphanol 0.2 mg/kg (Butorgesic; CP-Pharma) and alfaxalon (Alfaxan; Dechra) to effect, followed by orotracheal intubation and inhalation anaesthesia with isoflurane in 100% oxygen (Isofluran CP; CP-Pharma). The cats were breathing spontaneously. Vital parameters were monitored with a multiparameter monitor (Intelli Vue MP50 Neonatal; Philips). Blood donation of 10 ml/kg was performed using the right or left jugular vein with a commercial blood collection system (Feline Blood Collection Bag; Alvedia). After blood donation, cats received 30 ml/kg of lactated Ringer’s solution (Ringer-Lactat; B Braun Vet Care) intravenously over a 15 min period for volume resuscitation. In cases of hypothermia (<37.0°C), cats received temperature support with forced air (Bair Hugger; Arizant) or with heating pads.
Ultrasonographic measurements
Point-of-care ultrasound examination (POCUS) of the CVC was performed with a Loqig P 6 ultrasound machine (GE Healthcare) and an 8 MHz microconvex probe without clipping the fur. Isopropyl alcohol (Softasept Iso 70%; B Braun Vet Care) was used to provide adequate contact of the probe to the skin. Cats were placed in left lateral recumbency. The CVC was identified and measured at three sites: the paralumbar view (PV), the hepatic view (HV) and the subxiphoid view (SV; Table 1 and Figure 1). Identification of the CVC was confirmed based on its anatomical location, its anatomical appearance on ultrasound and the colour Doppler for each location. The minimal and maximal vertical diameters and the minimal and maximal horizontal diameters of the CVC at the HV site were measured in B-mode on multiple still images (Figure 2). On the HV, the horizontal diameter of the CVC was assessed at the largest diameter, and the vertical diameter was assessed perpendicular to this diameter.
Description of the ultrasound probe positions for identification and measurement of the caudal vena cava (CVC) diameter


(a,b) Paralumbar view (PV): the right kidney was identified in a longitudinal view. The transducer was moved backwards and fanned medially until the caudal vena cava (CVC) was identified. Hepatic view (HV): the transducer was placed on the right side, transverse, perpendicular in the ninth or tenth intercostal space ventral to the spinal column. Subxiphoid view (SV): the transducer was placed longitudinally under the xiphoid; the probe was fanned from right to left until the CVC was identified before passing the diaphragm

Measurement of the caudal vena cava (CVC) from the hepatic view in the horizontal and vertical diameters. The horizontal (hor) diameter was assessed at the largest diameter, and the vertical (vert) diameter was assessed perpendicular to it. The CVC is marked in red, and the portal vein is marked in blue
Inter- and intra-observer variability
For the assessment of inter- and intra-observer variability, ultrasonographic measurements were performed on 11 client-owned non-sedated cats by two independent observers. Measurements were performed by observer 1, followed by observer 2, who was blinded to the measurements of observer 1. Thereafter, observer 1 repeated the measurements to assess intra-observer variability. Observer 1 was an emergency clinician trained in POCUS. Observer 2 was an ECVECC diplomate trained and experienced in POCUS and ultrasonography. Inter- and intra-observer variability with a bias >0.2 mm and a 95% limit of agreement (LOA) of >2 mm were defined as unacceptable in this study, as these values correspond to the acceptable percentage of variation compared with the vessel size in humans.
Blood donation and CVC diameter assessment
Measurements during blood donation were performed by observer 1 before blood donation (T0), immediately after blood donation (T1) but before the fluid bolus was given over a 15 min period, and immediately after volume resuscitation (T2). The minimal and maximal CVC diameters at each probe position were documented throughout the breathing cycle. The minimal CVC diameter was measured at inspiration, and the maximal CVC diameter was measured at expiration. The CVC index was calculated with the formula [collapsibility (%) = (maximal diameter – minimal diameter)/maximal diameter × 100%], as described in human studies. 15
Statistical analysis
The sample size was determined using an open-source program (http://powerandsamplesize.com). The required sample size for the blood donation group to detect a CVC diameter difference of 20% was 16 with a power of 80% and an alpha error of 5%
Statistical analysis was performed with commercial software (Prism 5 for Windows; GraphPad). Data were evaluated for normality with the D’Agostino and Pearson normality test. Normally distributed, parametric data are reported as mean ± SD. Non-normally distributed, non-parametric data are reported as median (range). Intra- and inter-observer variability were analysed by Bland–Altman analysis and reported as the bias and 95% LOAs. The CVC diameters at different timepoints were analysed with the Friedman test and the post-hoc Dunn’s multiple comparison test. A P value of <0.05 was considered significant.
Results
Inter- and intra-observer variability
For the assessment of inter- and intra-observer variability, 11 cats were included. Four cats were castrated males and seven were spayed females. There were nine domestic shorthairs, one Russian Blue and one British Shorthair. The mean age was 10.6 ± 5.9 years, and the mean weight was 3.9 ± 0.9 kg. Patients were hospitalised with a diagnosis of seizures, chronic kidney disease, mastocytoma or lymphoma, and were included after rehydration. The intra-observer variability bias for CVC measurement was within the acceptable range at all probe positions, except for the maximal horizontal diameter at the HV, and the 95% LOAs were within the acceptable range at all probe positions, except for the maximal and minimal horizontal diameter at the HV (Table 2). The inter-observer variability bias for CVC measurement was within the acceptable range for the maximal vertical diameter at the SV, and the 95% LOAs were within the acceptable range for the maximal and minimal vertical diameter at the HV and for maximal and minimal vertical diameter at the PV (Table 3). Overall, the inter-observer variability for CVC measurement was poor even if the intra-observer variability was within the acceptable range.
Intra-observer variability of ultrasonographic caudal vena cava (CVC) diameter measurements in 11 cats at different anatomical locations analysed by Bland–Altman analysis
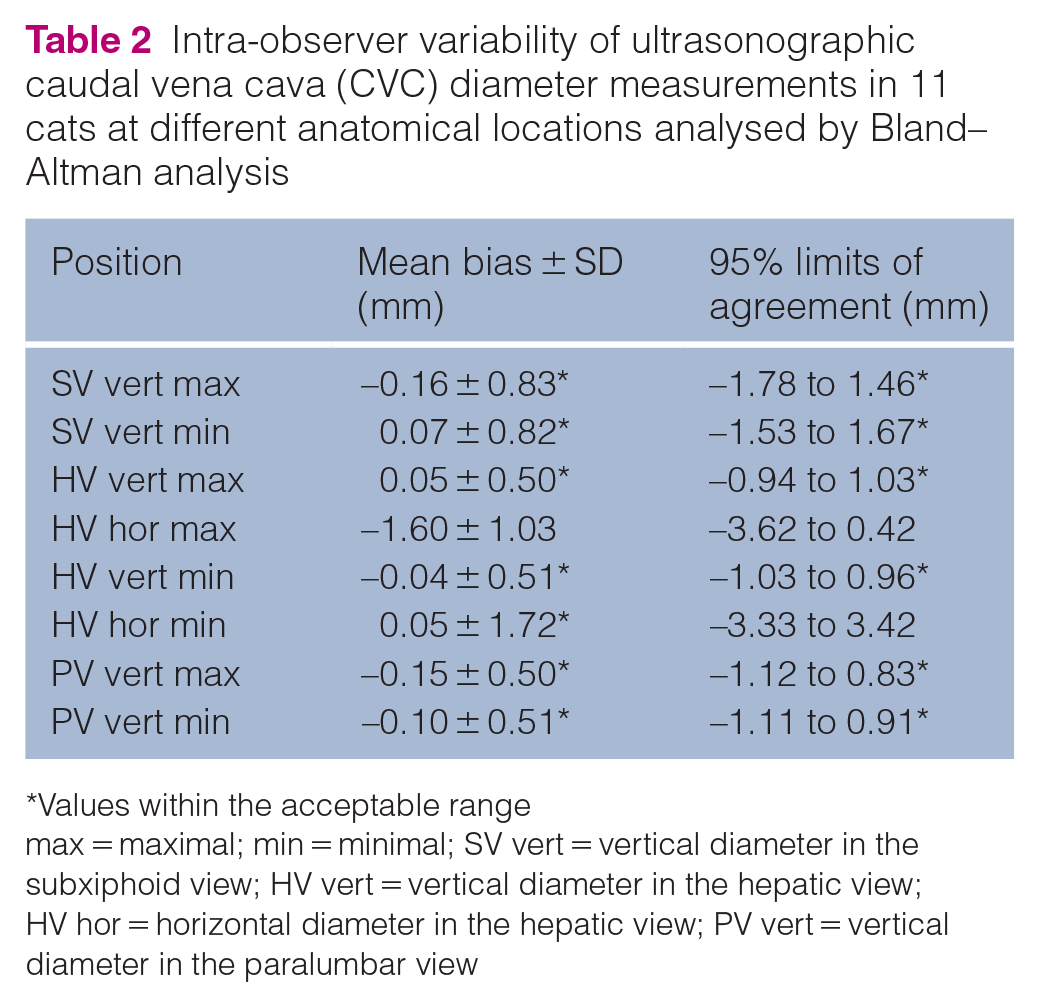
Values within the acceptable range
max = maximal; min = minimal; SV vert = vertical diameter in the subxiphoid view; HV vert = vertical diameter in the hepatic view; HV hor = horizontal diameter in the hepatic view; PV vert = vertical diameter in the paralumbar view
Inter-observer variability of ultrasonographic caudal vena cava (CVC) diameter measurements in 11 cats at different probe positions analysed by Bland–Altman analysis
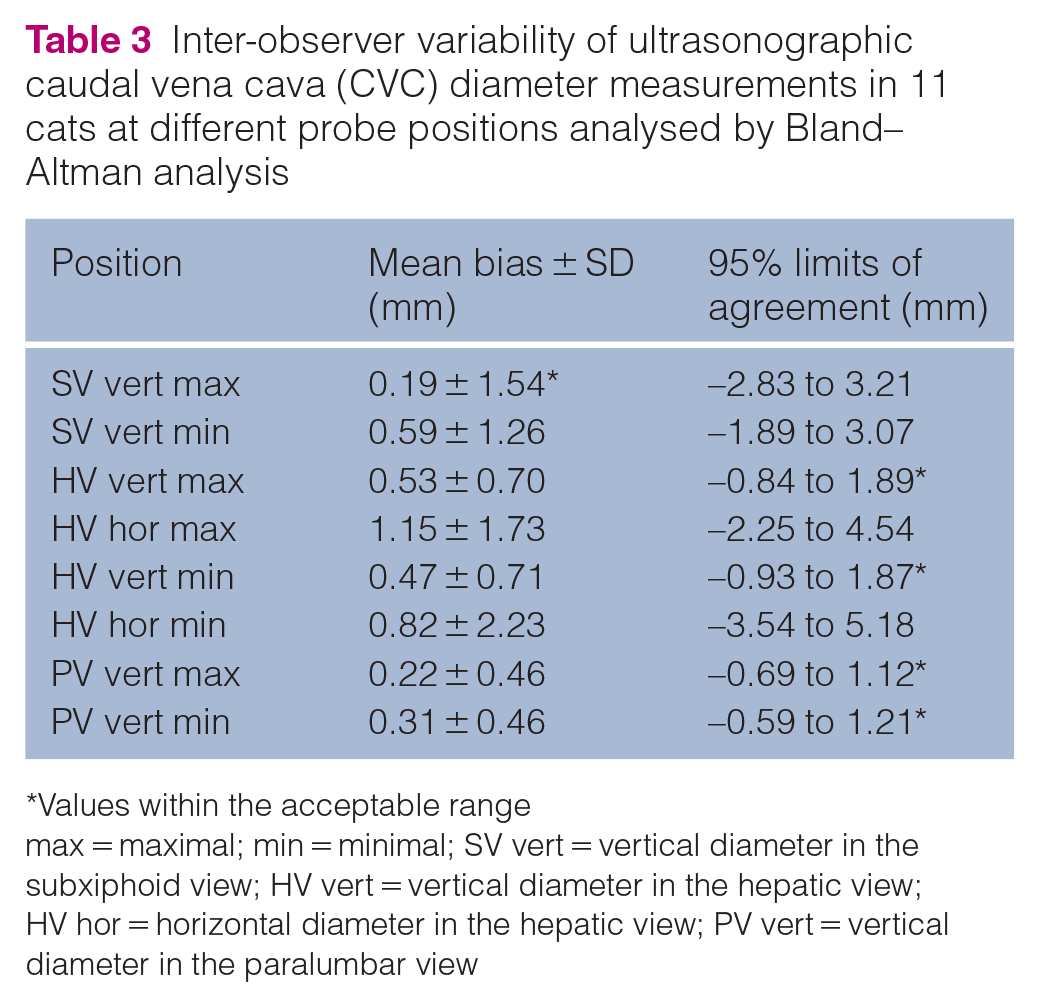
Values within the acceptable range
max = maximal; min = minimal; SV vert = vertical diameter in the subxiphoid view; HV vert = vertical diameter in the hepatic view; HV hor = horizontal diameter in the hepatic view; PV vert = vertical diameter in the paralumbar view
Blood donation and CVC diameter assessment
The study population consisted of 18 cats presenting for blood donation. In three cats, blood donations were performed twice, and ultrasonographic measurements during 21 blood donation procedures were evaluated. Two cats were intact males, eight were castrated males, two were intact females and six were spayed females. The breed distribution for these cats included 12 domestic shorthairs, two British Shorthairs, two Maine Coons, one Siberian Forest Cat and one mixed-breed cat. The mean age was 5.0 ± 2.8 years, and the median weight was 5.0 kg (range 3.6–8.0).
At the SV, the complete measurement of the CVC diameter was obtained during all 21 blood donations. At the SV, the minimal CVC diameter was significantly smaller at T1 than at T2 (P <0.001). The maximal CVC diameter was significantly smaller at T1 than at T0 and T2 (P <0.001; Table 4).
Median diameter (range) of the caudal vena cava (CVC) before (T0) and after (T1) blood donation and after volume resuscitation (T2) during 21 blood donations in 18 cats using three ultrasonographic views

Data are reported as median (range). Data were analysed with the Friedman test and the post-hoc Dunn’s multiple comparison test. Significant differences between timepoints are marked with * and †, and are printed in bold
Significantly different to T0
Significantly different to T1, both analysed by the post-hoc Dunn’s multiple comparison test
max = maximal; min = minimal; SV vert = vertical diameter in the subxiphoid view; HV vert = vertical diameter in the hepatic view; HV hor = horizontal diameter in the hepatic view; PV vert = vertical diameter in the paralumbar view
It was not possible to correctly identify the CVC and measure its diameter at the HV probe position at three timepoints during 2/21 blood donations. At the HV, the minimal vertical CVC diameter was significantly smaller at T1 than at T0 and T2 and at T0 than at T2 (P <0.001). The minimal horizontal CVC diameter was significantly larger at T2 than at T0 and T1 (P <0.001). The maximal vertical CVC diameter was significantly smaller at T1 than at T0 and T2 (P <0.001). The maximal horizontal diameter was significantly smaller at T1 than at T2 (P = 0.002).
It was not possible to correctly identify the CVC and measure its diameter at the PV probe position at seven timepoints during 5/21 blood donations. At the PV, the minimal and maximal CVC diameters were significantly smaller at T1 than at T2 (P <0.001).
At six of these 10 timepoints, the CVC could not be identified and measured, possibly because the cats were hypovolaemic (T1). The mean weight of the cats with unsuccessful CVC identifications was not different from that of the cats in which the CVC could be properly identified (P = 0.230).
The CVC diameter depended on the respiratory cycle. The minimal CVC diameter was measured at inspiration, and the maximal CVC diameter was measured at expiration.
There were no significant differences in the CVC index between timepoints for any anatomical location (Table 4). The changes in the CVC diameter over time are presented in Figures 3–5.

Vertical caudal vena cava diameter during blood donation in the subxiphoid view before (T0) and after (T1) a blood donation of 10 ml/kg and after fluid resuscitation with 30 ml/kg lactated Ringer’s solution (T2). max = maximal; min = minimal; SV = vertical diameter in the subxiphoid view; 0 = T0; 1 = T1; 2 = T2
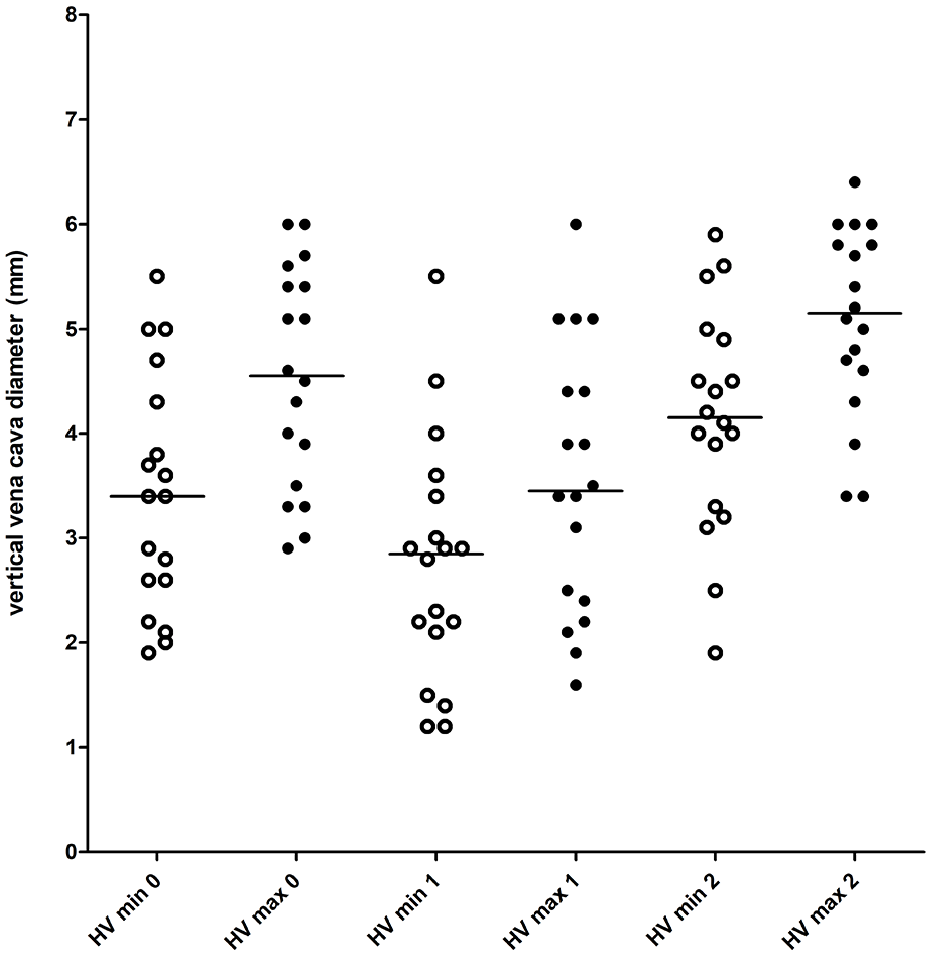
Vertical caudal vena cava diameter during blood donation in the hepatic view before (T0) and after (T1) a blood donation of 10 ml/kg and after fluid resuscitation with 30 ml/kg lactated Ringer’s solution (T2). max = maximal; min = minimal; HV vert = vertical diameter in the hepatic view; 0 = T0; 1 = T1; 2 = T2

Vertical caudal vena cava diameter during blood donation in the paralumbar view before (T0) and after (T1) a blood donation of 10 ml/kg and after fluid resuscitation with 30 ml/kg lactated Ringer’s solution (T2). max = maximal; min = minimal; PV = vertical diameter in paralumbar view; 0 = T0; 1 = T1; 2 = T2
Discussion
This study evaluated the ultrasonographic assessment of the CVC in cats, its inter- and intra-observer variability, and the changes in CVC diameter during blood donation and subsequent volume replacement.
In the present study, the intra-observer variability had a maximum bias of 0.16 ± 0.83 mm. The bias could be considered acceptable, despite a high SD. The inter-observer variability had a maximum bias of 0.59 ± 1.54 mm and was considered to be unacceptable. The mean maximal IVC diameter in euvolaemic human patients is 16.8 ± 1.6 mm in the longitudinal subxiphoid view. 16 In human patients, an intra-observer variability of <0.6 mm and an inter-observer variability of <0.8 mm are considered acceptable. 17 The acceptable inter-observer variability of <0.8 mm correlates to approximately 5% of the diameter. A maximal median CVC diameter of 5.2 mm (range 2.8–7.1) was determined at the SV in this study. After extrapolating from the acceptable variations in humans, an upper limit of 0.2 mm was defined as acceptable for inter- and intra-observer variability in the present study.
The inter- and intra-observer variability of the CVC diameter has already been described in dogs and humans, but the results in the literature are controversial.9,13,14,18,19 One study reported acceptable inter- and intra-observer variability in dogs and suggested that further study is needed. 9 A study comparing the differences in the CVC measurements between cardiologists and non-cardiologists had larger limits of agreement, and it was concluded that this technique has low accuracy and reproducibility. 14 In the present study, the probe placement and differences in applied pressure of the probe to the skin are possible reasons for the high inter- and intra-observer variability. The small size of feline patients vs canine patients might be another reason for the unacceptably high inter-observer variability in the present study, as it is technically more challenging to assess the vessel size of a few millimetres vs larger vessels of 1–2 cm.
CVC assessment during blood donation was possible at all three studied probe positions but not in all cases. Visualisation of the CVC from different views has already been studied in dogs and people.9–11 Probe positions and ultrasonographic views in the present study were chosen owing to similarities to those used in canine studies. 13 However, visualisation of the CVC was associated with technical and patient-related difficulties. Technical difficulties previously described in dogs include the lack of visualisation of the CVC due to intestinal gas located in the right upper abdominal quadrant and due to non-fasted or deep-chested dogs. 13 Problems that can cause a lack of visualisation of the CVC include air in the gastrointestinal tract, obesity and inadequate contact of the ultrasound probe to the skin. Better visualisation of the CVC and probably a lower inter- and intra-observer variability could have been measured if the fur had been clipped and ultrasound gel used, but this was not the case in this study as POCUS is usually performed without clipping the fur. Furthermore, the owners did not agree to fur clipping.
The best visualisation of the CVC was possible on the SV. The CVC diameter at the SV was measured in all cats at all timepoints, but the bias and the 95% LOA at that location were unacceptable for inter-observer variability. On the PV, the visualisation of the CVC was difficult in hypovolaemic patients (T1), but this location provided the best results for intra- and inter-observer variability measurements. Therefore, measurement of the CVC diameter is more reliable on the SV and the PV than on the HV.
In the present study, the CVC diameter changed significantly during blood donation. This was also observed in a canine study. 9 The best visualisation of the CVC was possible at the SV. The CVC diameter was obtained in all cats at that location, but the SD of the measured CVC diameter between the cats at that location was high. There was a significant overlap between the measured ranges at the different timepoints, so this location provided no relevant information for the estimation of changes in volume status. On the HV, the CVC diameter was measured in 19/21 blood donations. Measurements at that location were difficult because of the interference and a lack of visualisation from the respiratory cycle. The CVC could not be visualised during inspiration or expiration at the same probe positions. In a canine study establishing reference intervals, the CVC could not be identified on the HV in 43% of dogs. 13 A modified view caudal to the porta hepatis had to be utilised in these dogs. At the PV, the CVC diameter was measured in 16/21 of the blood donations in the present study. The SD between cats was smaller than at other positions. Therefore, that location provided the most reliable values for the changes in volume status if visualisation of the CVC was possible. In a previous canine study, only one measurement was obtained on the PV because the diameter did not change during the respiratory cycle at that location. 13 This was also observed in the cats in the present study. In another previous study in dogs, the CVC diameter was measured with this view only behind the kidney during blood donation, and the CVC diameter could be obtained on the PV in all dogs. The CVC diameter was significantly lower after blood donation. 9 A reason for the high SD in the present study might be that the measured CVC diameter could have been influenced by the probe placement. It is also possible that an oblique probe position could have increased the measured CVC diameter compared with a perfect perpendicular probe position. Pressure of the probe as applied to the abdominal wall may also lead to compression of the vessel and therefore smaller measured diameters. 14 Probe pressure variations between patients could not be objectively determined in the present study. Owing to the small size of feline patients vs canine patients, the pressure of the probe might have an important influence on the results. To further establish this technique for routine clinical use, the exact location of the probe position needs to be defined.
There were no significant differences in the CVC index between the timepoints at any location in the present study. The CVC index decreased after blood donation and increased again after volume resuscitation, but the changes were not significant. In humans, the CVC index seems to be a more accurate indicator than the IVC diameter in the assessment of volume status. 4 The CVC diameter was measured at its minimal and maximal extensions at each location in the present study. We also showed that variations in the CVC diameter correlated with the respiratory cycle. At T0, the difference between maximal and minimal extension was 1.2 mm for the SV, 1.1 mm for the HV and 0.2 mm for the PV. The influence of respiratory variations was lowest on the PV. The variations in the CVC diameter during the respiratory cycle have also been described in humans, dogs and healthy foals.13,20 During inspiration, the negative pressure in the thorax enhances blood flow from the abdominal CVC into the thoracic CVC, and the abdominal CVC diameter decreases. During expiration, the positive pressure in the thorax leads to the accumulation of blood in the abdominal CVC, and its diameter increases. 20 In addition, the CVC collapsibility depends on the respiratory effort. With increasing inspiratory effort, the CVC diameter decreases owing to the increased diaphragmatic motion. 19 Because of the small size of the vessel, the respiratory cycle has a lower influence on the CVC diameter in cats than in dogs or humans. Therefore, the CVC index depends greatly on the respiratory cycle. This could also explain why the CVC index does not change significantly during blood donation in cats. In addition, the cats in the present study were anaesthetised. The dogs, foals and humans in previous studies were not anaesthetised. Furthermore, anaesthesia might influence respiratory activity and therefore could also contribute to less severe changes in the CVC diameter during the respiratory cycle.
Measurement of the aortic diameter or CVC:aorta ratio were not performed in this study. Because these parameters are beneficial for volume estimation in dogs and humans, it might also be useful in cats. Further studies are needed to evaluate these parameters in cats.
The present study has some limitations. First, it is possible that in some cats, a blood loss of 10 ml/kg is not enough to cause severe hypovolaemia or significant changes in the CVC diameter. In human studies, a blood loss of 10 ml/kg was not associated with clinical signs of hypovolaemia. 21 However, a canine study previously showed a significant change in the CVC:aorta ratio after a blood donation of 6 ml/kg. 9 Removal of a larger volume during blood donation was considered unacceptable in this study, as this would have caused a high risk of haemodynamic instability in the blood donors. Secondly, in this study, cats were anaesthetised with butorphanol, alfaxalone and isoflurane. Anaesthetic drugs are known to have an influence on blood pressure and vessel tone, which can influence the CVC diameter. Most anaesthetic drugs, especially the inhalation anaesthetics, cause vasodilation and a decrease in arterial blood pressure.22,23 This leads to a relative hypovolaemia vs an absolute hypovolaemia because of blood loss. It cannot be objectively determined whether the induced hypovolaemia in this scenario is due to vasodilation because of anaesthesia or due to blood loss because of blood donation. As the depth of anaesthesia was similar at all timepoints, differences in the CVC diameter, noted here, seemed to result from volume depletion and volume replacement. Therefore, the results of this study cannot necessarily be translated to non-anaesthetised cats. The cats for blood donation were anesthetised, whereas cats for inter- and intra-observer variability testing were awake. As awake cats are more likely to move during examination, movement artefacts might lead to an increasing inter- and intra-observer variability in this study.
Conclusions
Ultrasonographic assessment of the CVC for volume estimation in cats under general anaesthesia and after 10 ml/kg blood volume loss is a challenging technique with major limitations. Even though routine clinical application is difficult, this method could be used to monitor fluid responsiveness in the same patient, by the same operator by assessing the CVC diameter at two or more consecutive timepoints. The PV probe position provides the most reliable information regarding the changes in volume status. In addition, the inter-observer variability of this technique is poor. Additional studies are needed to further evaluate this technique in non-anesthetised cats or in clinical settings of acute blood loss.
Footnotes
Author note
This paper was presented, in part, at the 18th European Veterinary Emergency and Critical Care online congress 2020.
Conflicts of interest
The authors declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
This work involved the use of non-experimental animals only (including owned or unowned animals and data from prospective or retrospective studies). Established internationally recognised high standards (‘best practice’) of individual veterinary clinical patient care were followed. Ethical approval from a committee, while not specifically required for publication in JFMS, was nonetheless obtained, as stated in the manuscript.
Informed consent
Informed consent (either verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (either experimental or non-experimental animals) for the procedure(s) undertaken (either prospective or retrospective studies). No animals or humans are identifiable within this publication, and therefore, additional informed consent for publication was not required.
