Abstract
Practical relevance:
Cats are great pretenders; they often hide illness until they are critical. This makes patients of this species challenging to assess and manage in the emergency setting where quick and stress-free diagnosis and treatment are necessary. Veterinary point-of-care ultrasound (POCUS) is a rapid, evidence-based, non-invasive, repeatable, cage-side ultrasonographic examination designed to answer clinically driven questions without compromising feline wellbeing. Integrating feline friendly POCUS as an extension of the physical examination to streamline diagnostic and therapeutic interventions, thereby limiting stress and improving overall patient care, is advocated by the authors of this article.
Equipment:
Given the multitude of ultrasound machines and probes available that are portable, meaning they can be moved around the clinic and used patient-side, it should be possible for most practitioners to integrate POCUS into daily practice. The authors’ preferred equipment for feline POCUS is a microconvex probe and a portable machine with a fixed pre-set. This set-up allows the clinician to complete all POCUS (abdominal, lung and pleural space, and heart) without needing to move the patient, change probes or restrain the patient in a particular position, ultimately saving time, personnel and cost while maintaining patient comfort and safety.
Aim:
This review aims to serve as a valuable resource for veterinarians seeking to improve their feline patient care through the judicious utilisation of POCUS. In this article, the complex challenges posed by cats are addressed, and the different POCUS techniques, applications and clinical recommendations are discussed.
Evidence base:
This review draws on the published literature, as well as the authors’ own collective experience when providing recommendations.
POCUS for general practitioners
Veterinary point-of-care ultrasound (POCUS) is a rapid, evidence-based, non-invasive, repeatable, radiation-sparing, patient-side ultrasonographic examination of a patient. The ultrasound skillset required to perform POCUS can be easily mastered by general practitioners, which is especially valuable when there is a need to prioritise patient safety without delaying diagnostic and therapeutic interventions.
POCUS can be integrated into daily practice when performed patient-side as an extension of the physical examination. it can be performed with the cat in the position that they are most comfortable in; this is typically a sternal, standing or sitting position, although a lateral position may be preferred in cats with particular injuries (eg, pelvic/spinal fractures). dorsal recumbency, which is often stressful for cats, is avoided during POCUS.
POCUS should be used as a tool to ask key clinical questions; for example, for a cat with respiratory distress and known trauma, are pulmonary contusions present? A cat with a murmur and respiratory distress – is it congestive heart failure (CHF)? A cat with abdominal distension – is peritoneal fluid present? The questions asked are dynamic and may change as more information is gathered; in order to answer these quickly and efficiently, it is therefore important to assess POCUS findings alongside a patient’s history and physical examination findings.
General preparation
Ultrasound machines
There are numerous machines (console, portable, hand-held devices) available to the practitioner. Portability is key as POCUS should be performed patient-side, whether it is in a consultation room, oxygen cage or preoperative area. The authors also recommend an easily accessible durable machine that is, ideally, impact-resistant. New handheld devices are a cost-effective option if the practice budget is low (Figure 1).
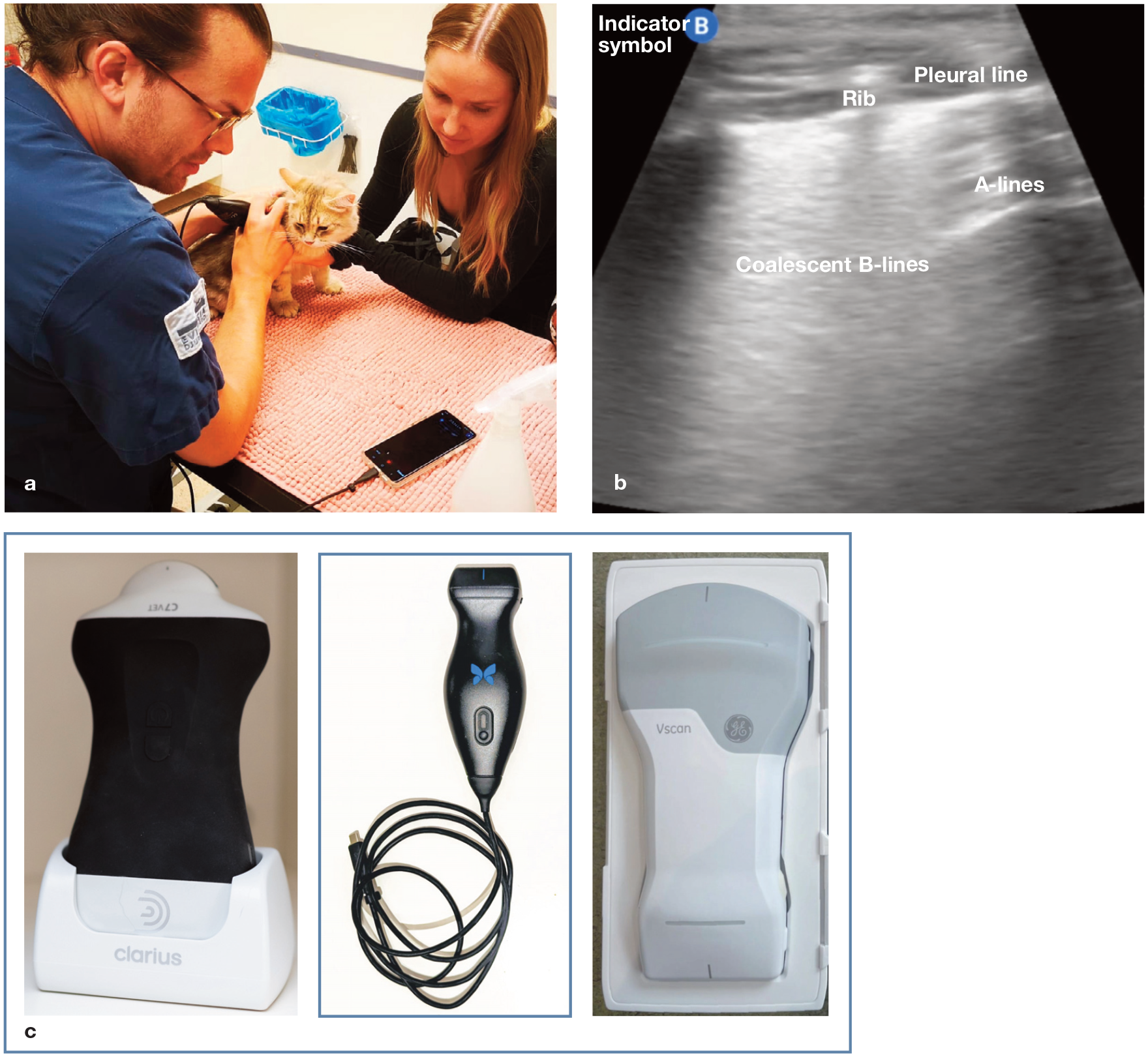
(a) Portable handheld ultrasound scan of a cat’s lung and (b) the resultant POCUS image showing the pleural line and coalescent B-lines. (c) Three different handheld ultrasound probes: C7 Vet probe (Clarius; left); iQ+ Vet ultrasound probe (Butterfly Network; middle); and Vscan Air CL (GE HealthCare; right). There are a number of different handheld ultrasound probes that can connect to a smartphone or tablet. Image (a) courtesy of Katharina Lunde and image (c) courtesy of Roderick Lee (left), Katharina Lunde (middle) and Seth Colby (right)
Machine settings
POCUS relies on three key machine settings: depth, gain and frequency. The depth and gain (overall brightness) should be adjusted to optimise image quality. in cats, due to their small size, the frequency, which balances penetration and resolution, is often set to the highest level. Perfect image quality is not critical to interpreting POCUS images and using a pre-set can streamline efficiency, allowing the clinician to begin scanning as soon as the machine powers up. Pre-sets are more helpful in cats compared with dogs as there is less size variation between breeds. A microconvex probe (Figure 1c) with depth at 4–6 cm and frequency at 5–8 MHz is an example of a commonly used pre-set used for lung and pleural space, and abdominal POCUS.
Probe selection
To save time, personnel and cost while minimising patient stress the authors use a micro-convex probe for all POCUS examinations (abdomen, lung and pleural space, and heart) as it allows complete POCUS assessment without having to change the probe. Linear probes may be helpful for feline abdominal POCUS but are less commonly used for lung and pleural space, and cardiac POCUS due to the probe’s large footprint and the small intercostal spaces of cats. 1
All probes contain an indicator or orientation marker (a notch, ridge, indentation, groove or nub) that corresponds to the indicator or orientation marker on the ultrasound screen (often depicted as a symbol). The probe marker indicates which part of the probe is left or right on the screen (see the indicator symbol in Figure 1b). The direction of the probe’s indicator or orientation marker is not critical to performing POCUS, but an awareness of the orientation, based on the location of the probe indicator marker, is important for image acquisition and interpretation (Figure 2).
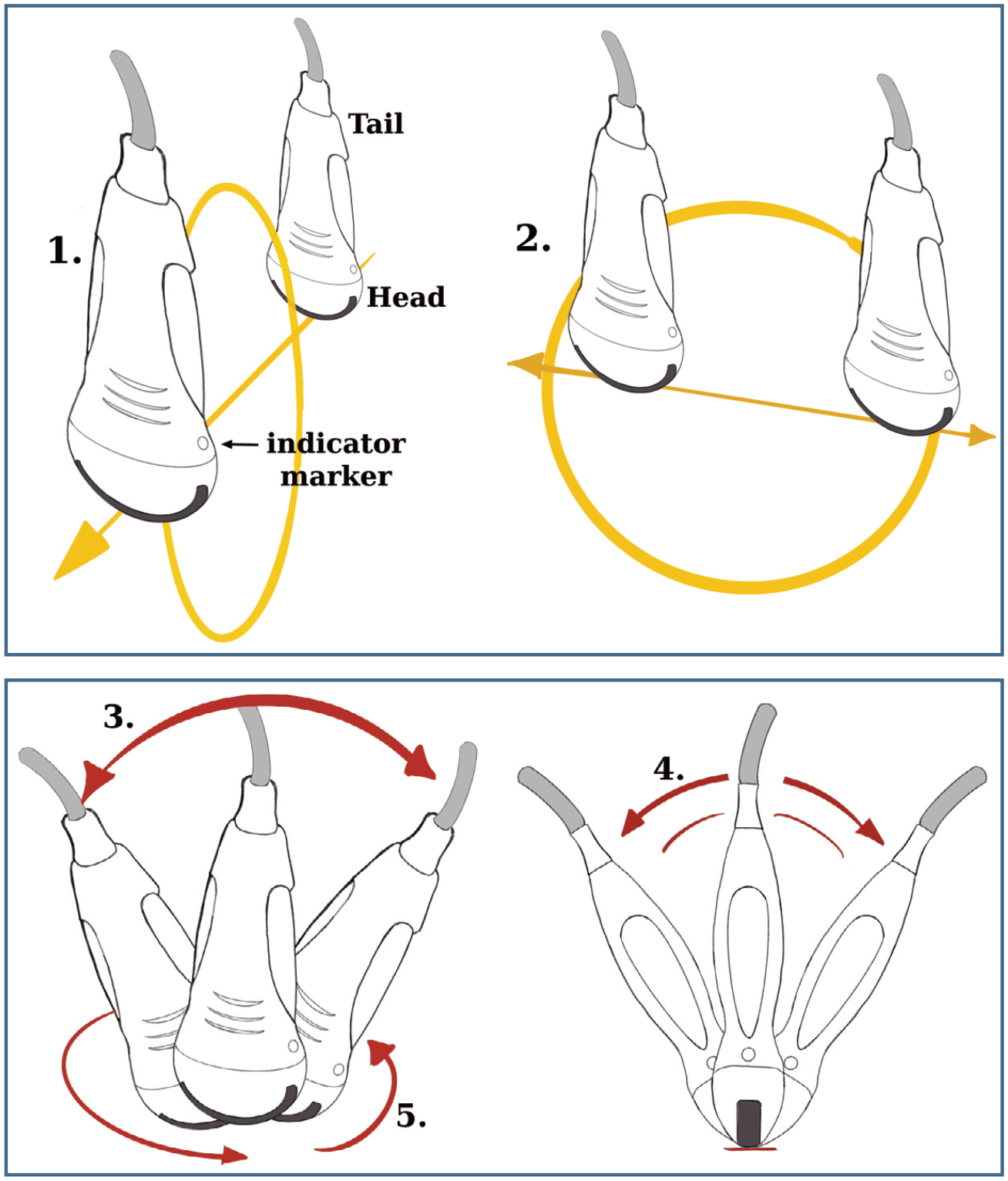
For the purpose of POCUS there are five general probe movements described to identify and assess structures. Sweeping and sliding are the only two movements where the entire probe moves along the skin from the starting point, without changing the angle of insonation of the probe (the angle of the probe relative to the skin surface or organ of interest): (1) sweeping refers to movement of the probe in the short-axis direction; and (2) sliding refers to moving the probe in the long-axis direction. The following three movements are performed with the probe fixed at the same probe footprint-to-skin interface: (3) rocking refers to moving the tail of the transducer from side to side relative to the shortest axis; (4) fanning refers to moving the tail of the transducer side- to-side relative to the widest axis; and (5) rotating refers to a degree of rotation along the axis of the probe in either a clockwise or anticlockwise direction. Due to cats’ small size, rocking, fanning and rotating are the most commonly used probe manipulations. It is also recommended that novice ultrasonographers only use one probe movement at a time to find and assess structures until they become more comfortable with the art of POCUS
Patient preparation
Minimal patient preparation is required to perform POCUS. The scanning position is determined by the cat, and the POCUS operator works around the patient rather than following a strict POCUS protocol, which may include scanning the cat in their carrier or kennel (Figure 3).

Lung and pleural space POCUS being performed inside a cat’s carrier with minimal restraint. The ultrasound image on the screen shows rib shadows and A-lines
Restraining cats in lateral or dorsal recumbency to perform POCUS is not recommended, unless the cat presents in lateral recumbency due to injury. instead, minor adjustments to the cat’s position can be made to optimise image acquisition. When imaging the heart or ventral abdominal regions with the cat in sternal recumbency, placing a rolled towel under the front limbs can elevate the cranial trunk, making it possible to scan ventral POCUS sites with minimal stress. if pain, discomfort, anxiety or stress is evident, administration of analgesia and/or anxio lytics (eg, 0.2 mg/kg butorpha nol) should be prioritised over POCUS.
Clipping fur is not required; instead, fur is parted to expose the skin, where alcohol is then added as the coupling agent (a 2/3:1/3 water:alcohol ratio is the authors’ preferred agent, delivered via a spray bottle), and the probe is applied directly to the skin. if clipping is required, it is kept to a minimum (just enough fur is clipped to allow the POCUS window of interest to be assessed) and quiet clippers are recommended. Gel is generally not used; if it is, however, it should not be applied directly to the patient as it can trap air between hair strands, reducing image quality and it should, instead, be applied directly to the probe’s scanning surface (footprint). When using alcohol, the probe should be wiped off immediately after the examination to minimise the risk of damage to the probe surface; alternatively, an ultrasound probe cover can be used. A sterile probe cover can also be used if maintaining a sterile field is required. Alcohol should be used sparingly as alcohol vapours are an irritant and can cause nausea, which is exacerbated when cats are placed in a confined space, such as an oxygen cage. it is also important to remove excess liquid (eg, with a towel) following POCUS, since both alcohol and excessive clipping have a cooling effect. Due to their small size and large body surface area compared with dogs, cats are at greater risk of hypothermia, which is often exacerbated by clinical interventions. Proactively placing cats on a bed or a cat-safe heating pad while performing POCUS can help to minimise additional heat loss.
Lung and pleural space POCUS
Indications
Stabilisation of cats in respiratory distress has not changed since the advent of POCUS. Priority should be given to oxygen supplementation, analgesia and/or anxiolytics. 2 However, POCUS is a safe cat friendly adjunct to the physical examination, as it allows supplemental oxygen and emergency interventions to be administered without having to restrain the cat in an unnatural position. Pathologies commonly assessed with lung and pleural space POCUS include pleural space and pulmonary parenchymal diseases (eg, air/fluid and B-lines/ consolidation, respectively).
Techniques
Lung and pleural space scanning often starts with the probe orientated perpendicular to the ribs, within the intercostal space, at the site of interest. The site of interest varies with the suspected pathology, as determined by history and other clinical findings, as well as patient positioning (air rises and fluid falls). The probe is then fanned to visualise the pleural line between ribs (Figure 1b).
Following a standardised scanning protocol is useful to ensure repeatability. A number of different lung ultrasound protocols have been published and include Vessozi, VetBLUE and Calgary PLUS, which assess between four and 19 thoracic windows (Figure 4).3–5 The optimal number of sites to scan is unknown but should be based on the clinical question being asked and the stability, size and tolerance of the cat. Having a clinically driven POCUS question to answer helps streamline the examination. For example, if pleural effusion is suspected clinically, the cat should be scanned ventrally where fluid will accumulate when it is a sternal or standing position. if the cat remains laterally recumbent due to injury, or it is too ill to stand or rest in a sternal position, however, the widest gravity-dependent site of the thorax should be scanned.
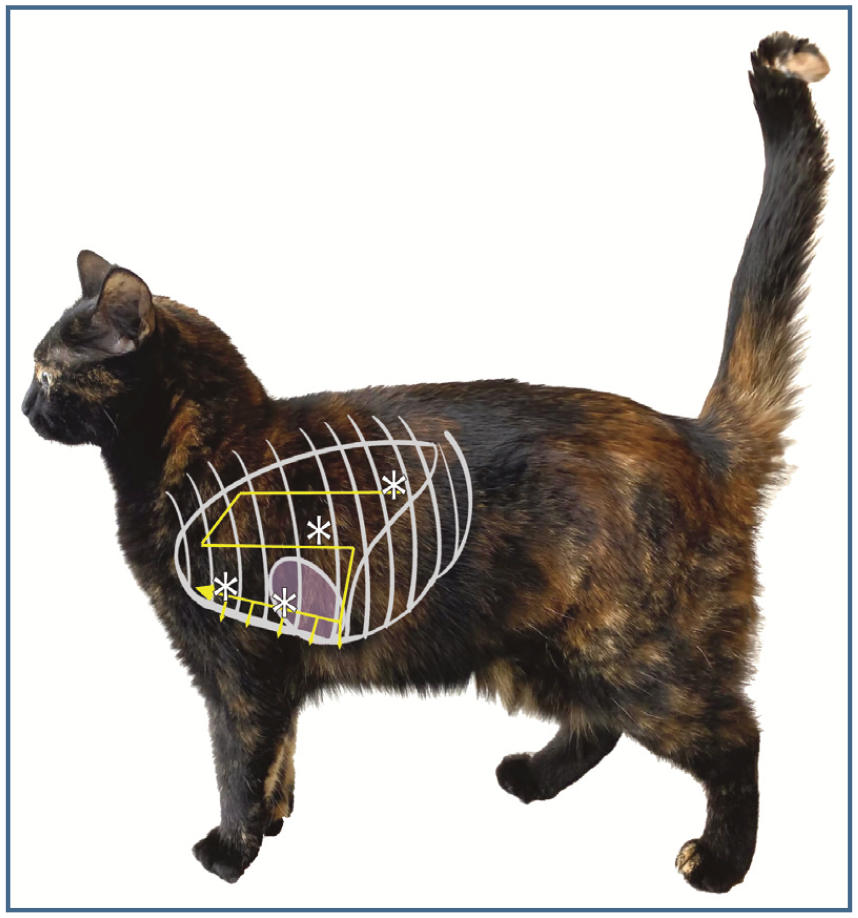
VetBLUE and Calgary PLUS protocols overlaid on a photograph of a cat. These two protocols, along with the Vessozi protocol (not shown for clarity), are performed on each hemithorax, with the subxiphoid view included in all three. The asterisks represent the four different areas of the thorax scanned with the VetBLUE protocol. The yellow line represents the S-pattern of the Calgary PLUS protocol, which includes turning the probe parallel to the ribs at the most ventral regions to search for smaller pockets of pleural effusion. The Vessozi protocol (not overlaid on the image) is performed by scanning dorsally to ventrally within each intercostal space from caudally to cranially
Normal findings
Arguably, the most important sonoanatomy of the pleural space and lungs includes identification of the pleural line, proximal rib surfaces with distal rib shadowing, A-lines, lung sliding and the abdominal curtain sign (see box and Figure 5). A-lines when the ultrasound beams are orientated perpendicular to the pleura, are the predominant artefact seen when lung surface aeration is above 85%.8–12 Pathology will arise from the pleural line in different ways and, therefore, the identification of the pleural line is key when performing lung and pleural space POCUS. When performing POCUS, it needs to be considered, however, that lung pathology that does not reach the lung surface will not be visible.
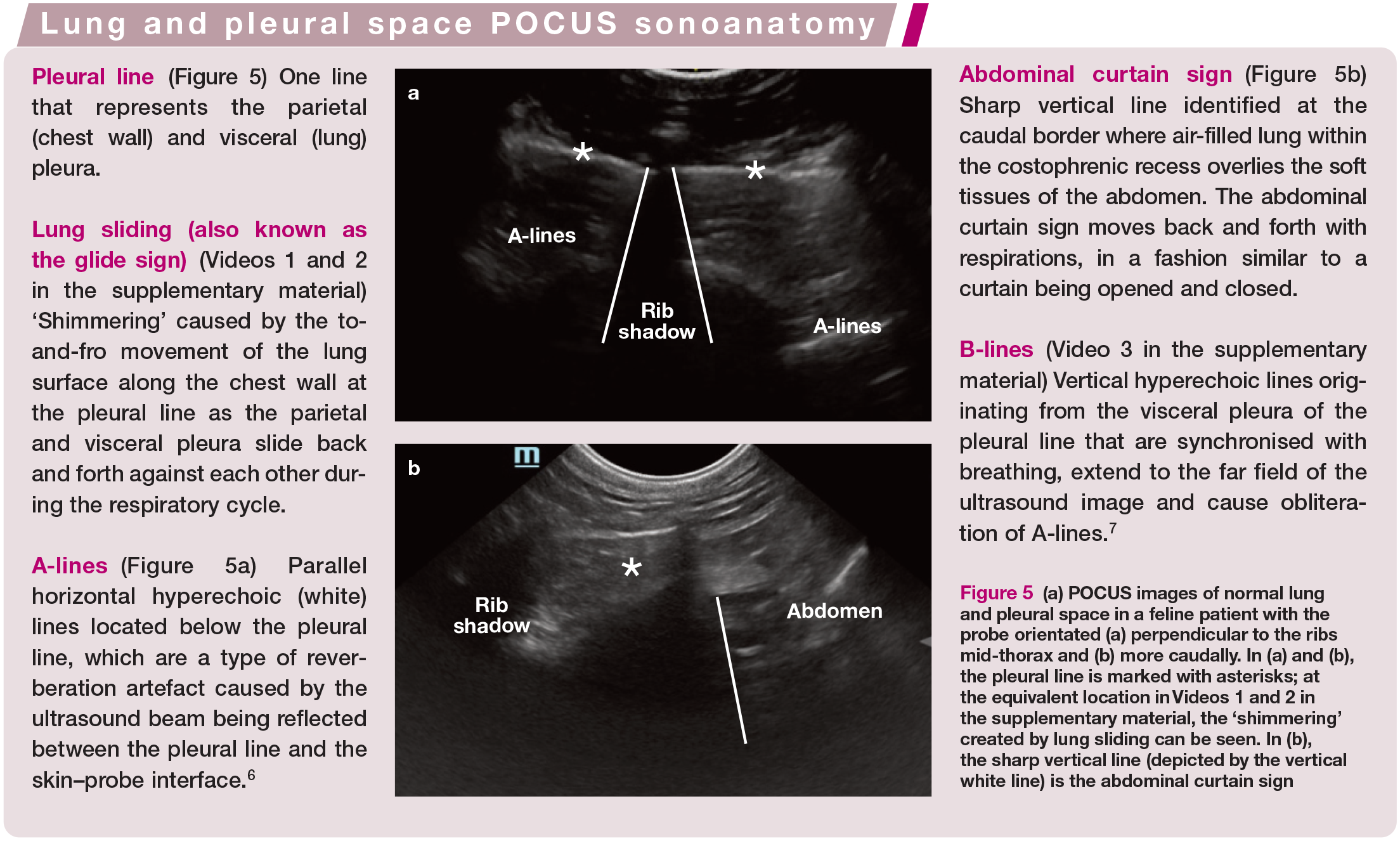
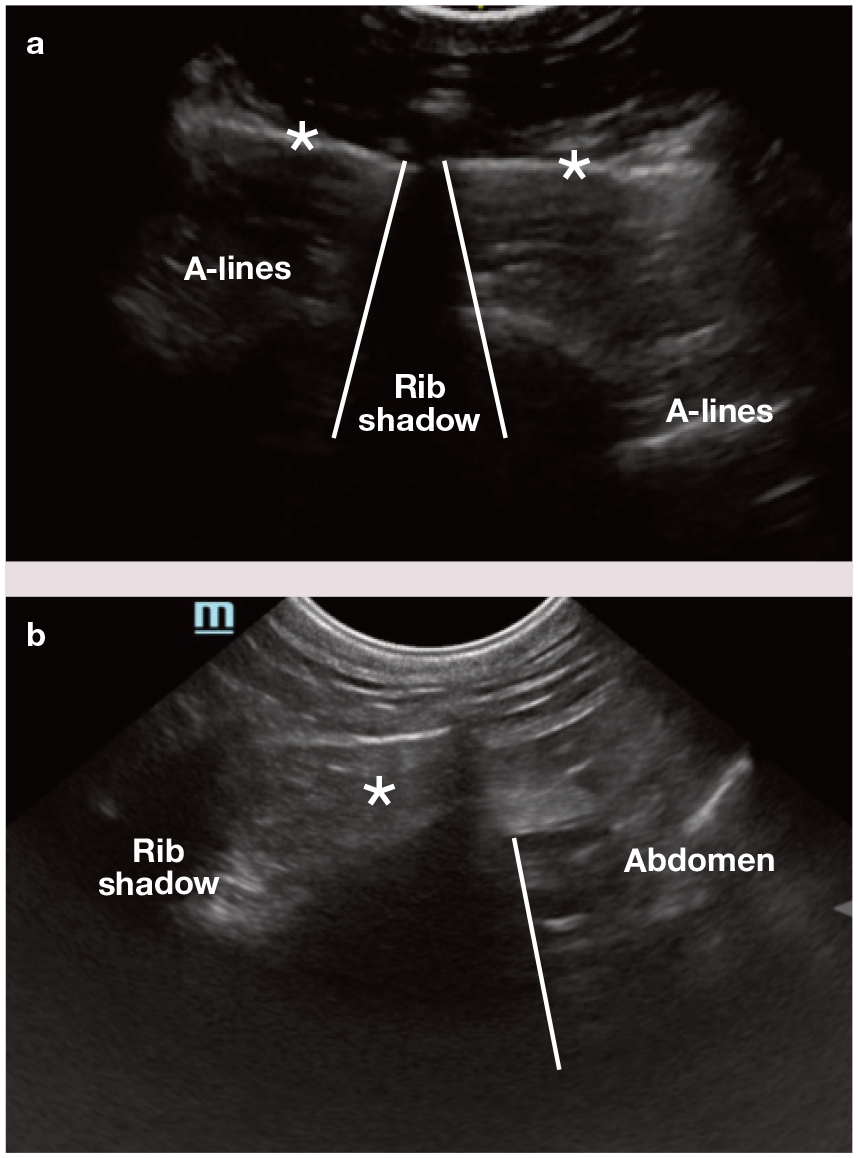
(a) POCUS images of normal lung and pleural space in a feline patient with the probe orientated (a) perpendicular to the ribs mid-thorax and (b) more caudally. In (a) and (b), the pleural line is marked with asterisks; at the equivalent location in Videos 1 and 2 in the supplementary material, the ‘shimmering’ created by lung sliding can be seen. In (b), the sharp vertical line (depicted by the vertical white line) is the abdominal curtain sign
Pulmonary pathology
B-lines (see the ‘Lung and pleural space POCUS terminology’ box) represent decreased lung aeration within 1–3 mm of the lung surface. The occurrence of B-lines in healthy cats ranges from 10% to 50% depending on the scanning protocol used, with more B-lines detected as more lung surface is evaluated. 13 B-lines can be classified as 1, 2, 3, >3 and co-alescent based on the number visualised at the site scanned (Figure 1b). in general, clinically healthy cats are considered normal if they have fewer than three B-lines at any single ultrasonographic site examined, or fewer than two positive sites on a hemi thorax regardless of the number of B-lines within those two sites. The number of B-lines increases when the ratio of aerated lung tissue at, or just below, the lung surface falls below 85%, with more than three B-lines in a single scanning site indicative of pulmonary parenchymal disease and/or atelectasis.8–12 However, thoracic CT abnormalities have been detected in cats with fewer than three B-lines at a single scanning site.4,13 Therefore, it is important for the clinician to combine history, physical examination findings and distribution of POCUS changes when appraising the clinical significance of B-lines.
Ultrasonographic lung consolidation, a more severe manifestation of interstitial– alveolar infiltrate, occurs when the percentage of aerated lung falls below 5–10% (Figure 6).8–12 Although identification of lung tissue is always abnormal and parenchymal pathology is the most common cause of lung consolidation, a similar ultrasonographic pattern can be seen with lung atelectasis. Like B-lines, this emphasises the importance of incorporating the history, distribution of lesions, concurrent POCUS findings and other clinical findings when deciding the cause of ultrasonographically identified lung consolidation. There are additional lung signs and lines that exceed the scope of this article.5,14–16
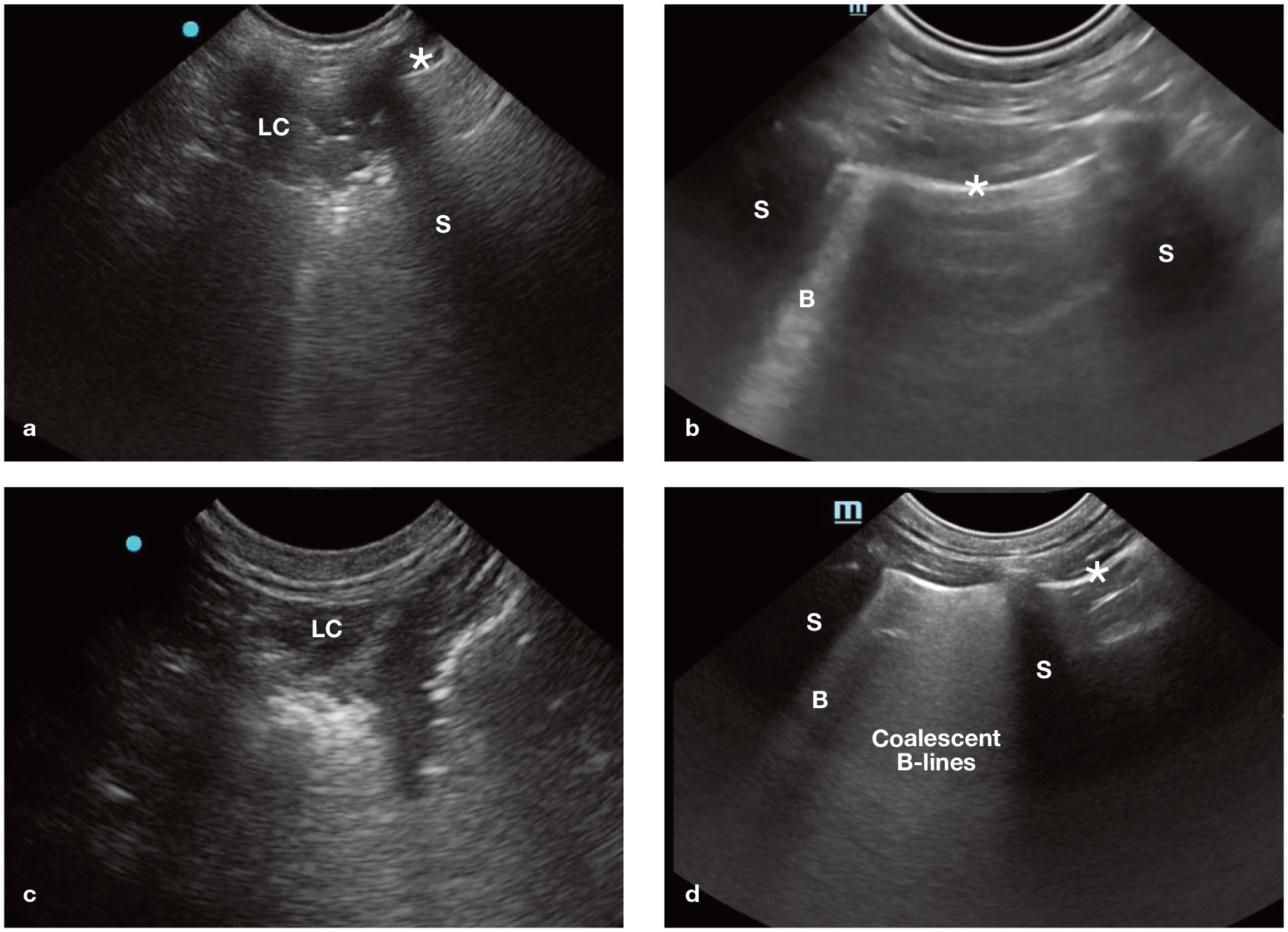
POCUS images of lung consolidation and increased numbers of B-lines in cats. (a) The hyperechoic dots within the hypoechoic consolidated lung area are air bronchograms and represent trapped air within the consolidated lung tissue. (b) A single B-line, originating from the pleural line, is visible. Differential diagnoses for B-lines include extravascular lung water (most common), tissue, increased cell numbers or decreased air (atelectasis). (c) The pleural line has become hypoechoic and difficult to visualise, with a loss of lung sliding, where lung consolidation contacts the chest wall. This is because it is the air-filled lung surface against the chest wall (ie, air-to-soft tissue interface) that makes the pleural line hyperechoic and the accompanying lung sliding visible, but the appearance of this interface is lost with the loss of air in the lung with lung consolidation. (d) Coalescent B-lines and a single B-line are visible; these both arise from the lung surface, appear hyperechoic, extend to the far field of the image and cause obliteration of A-lines. LC = lung consolidation; S = rib shadow; * = pleural line; B = B-line
Pleural space disease
Pleural effusion (Figures 7 and 8) often appears as anechoic (black) to hypoechoic (darker than surrounding tissue) at the most gravity-dependent sites of the thorax, most often the ventral thorax between the heart and liver, or within the costo phrenic recess when the patient is sternal or standing. Although thoracocentesis is required to determine the nature of the fluid, pleural fluid is most commonly associated with CHF and neoplasia in cats.17,18
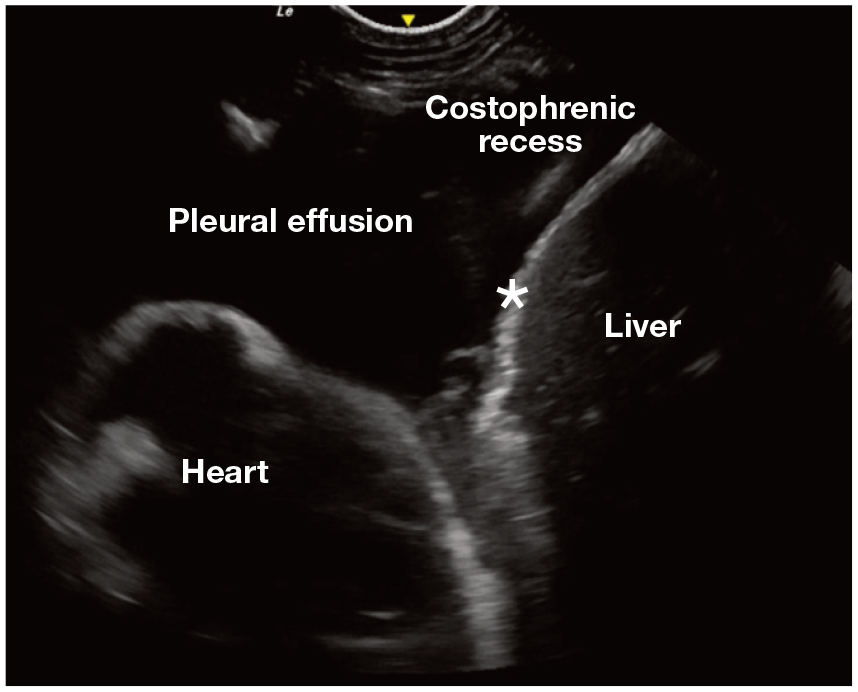
POCUS image with anechoic pleural effusion trapped in the pleural space between the diaphragm (asterisk) and heart and filling the costophrenic recess between the diaphragm and chest wall
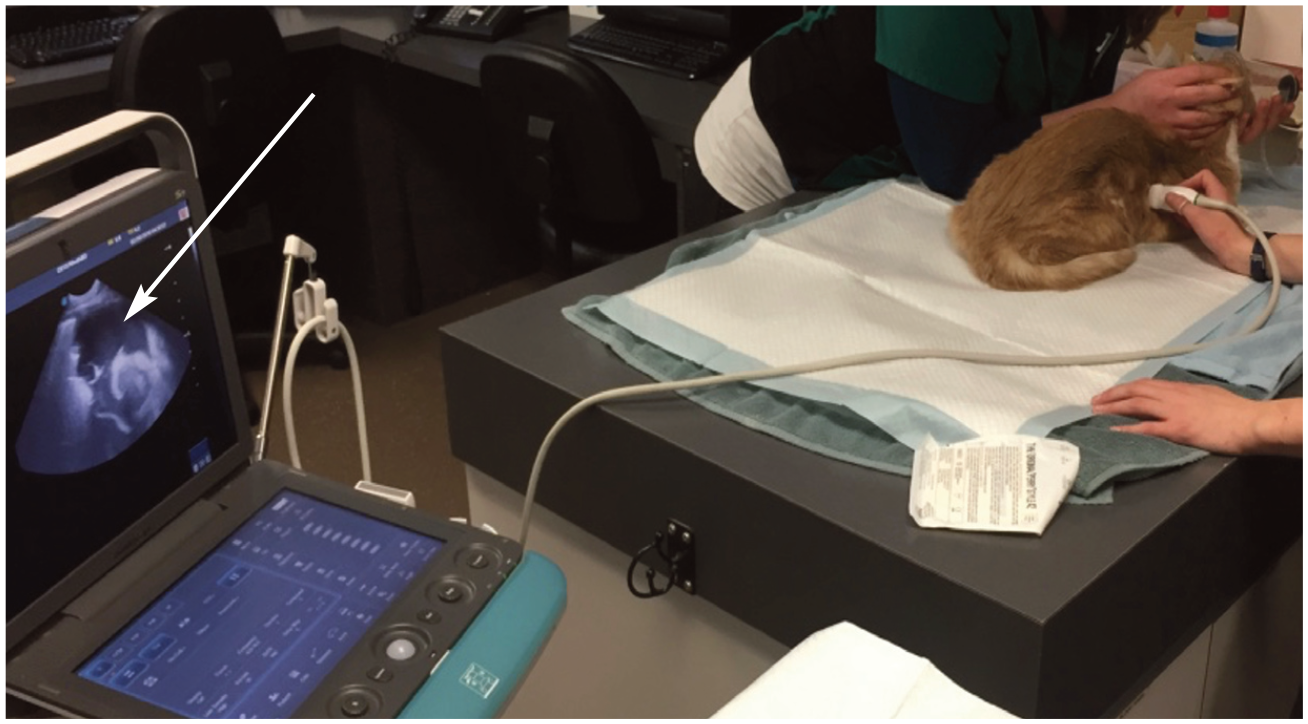
Lung and pleural space POCUS scan of a cat using minimal restraint, while providing supplemental oxygen. Large volume pleural effusion is visible on the ultrasound image to the left (arrow)
Pneumothorax
The pleural line should be evaluated when considering pneumothorax. A-lines are visible in healthy air-filled lung and patients with pneumothorax since both air-filled lung and free air within the pleural space create a bar rier to the ultrasound beam. This barrier, created when the parietal lining (inner chest wall) is in contact with either air-filled lung or free pleural air, reflects the beam back and forth between the parietal lining and probe surface, causing the pleural line to be ‘mirrored’ below its true location, which gives rise to A-lines.
To help visualise lung sliding (defined in the ‘Lung and pleural space POCUS sono-anatomy’ box and shown in Videos 1 and 2 in the supplementary material), the probe should be held as still as possible while fanning it such that the ultrasound beam will not strike the pleura at a 90° angle (the probe should be slowly fanned away from 90°). The depth (eg, decreased to 2 cm) and gain can also be decreased to better visualise the pleural line and enhance the presence of lung sliding. The gain and depth should be reset after looking for lung siding to ensure other lung pathology is not missed. Absence of lung sliding is suggestive of pneumothorax since the presence of free pleural space air will separate the parietal and visceral pleura, preventing them from sliding across each other and subsequently obliterating the shimmer seen with lung sliding.2,4,19,20 However, this finding lacks sensitivity. 21 When scanning for pneumo -thorax, it is important to scan the most gravity-independent sites, being mindful that this site will change based on patient positioning. A lung point, where the lung surface re-contacts the thoracic wall in the presence of free pleural air, confirms the presence of pneumothorax. in other words, where the visceral and parietal pleura re-connect is identified as the presence and absence of a lung sliding side by side within the same ultrasound image. This finding is more supportive of pneumothorax.
A novel ultrasonographic finding, an abnormal abdominal curtain sign (double or asynchronous; defined in the ‘Lung and pleural space POCUS sonoanatomy’ box), has been reported in dogs with pneumothorax and although the authors have identified abnormal curtain signs in cats with pneumothorax, there are no published feline data for curtain sign abnormalities.22,23
Pneumothorax can be more reliably ruled out than ruled in on lung and pleural space POCUS. B-lines, when not associated with the lung point, rule out pneumothorax at the probe’s location because B-lines originate at the lung surface, and their presence means there is no pleural air between the lung surface and the chest wall. if pneumothorax is clinically suspected in an unstable cat, pain relief/sedation combined with thoracocentesis should be prioritised over POCUS.
Cardiac POCUS
Indication
Cardiac POCUS or focused cardiac ultrasound, alongside history, physical examination, other POCUS and diagnostics such as N-terminal pro B-type natriuretic peptide (NT-proBNP), can support a diagnosis of left-sided CHF as a cause of dyspnoea in cats.24,25
Techniques and views
The cat should be scanned in the position it is most comfortable in (often sternal or standing) with a microconvex probe while receiving oxygen supplementation. Cardiac POCUS, similar to other POCUS examinations, requires no clipping and uses alcohol (or rarely gel) as the coupling agent. 26 The standard right parasternal short-axis views are obtained with the probe initially placed perpendicular to the ribs at the maximal palpable heart beat (fourth to fifth intercostal space). The probe is then slowly rotated clockwise to obtain a symmetrical short-axis view of the heart, followed by fanning the ultrasound beams dorsally through several short-axis planes to visualise the different cardiac chambers (Figure 9). Since cardiac POCUS is extremely sensitive to probe orientation and angle, the clinical POCUS questions asked should be limited: does the left atrium look enlarged and/or is there pericardial effusion?
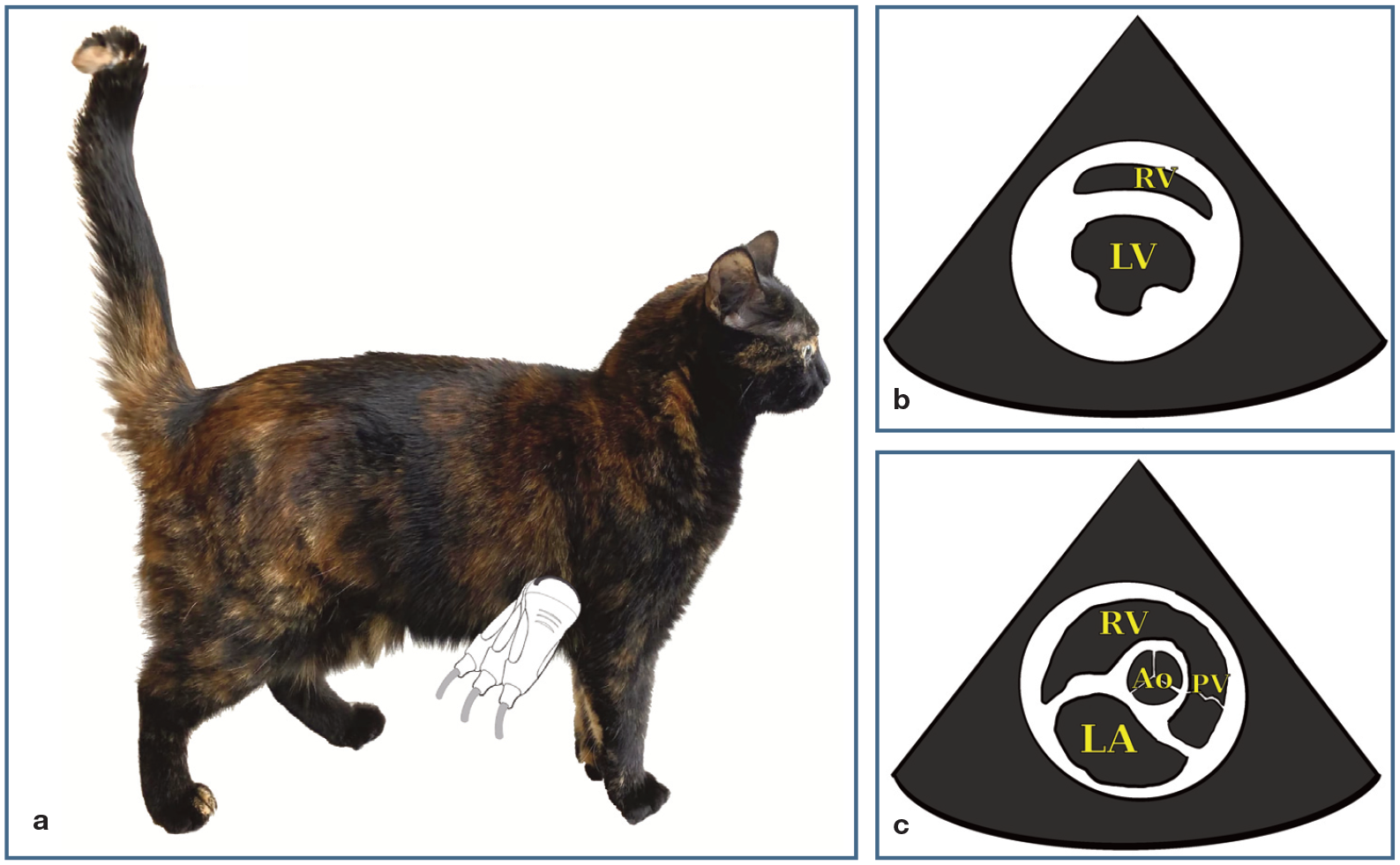
(a) Visual representation of probe fanning to reach the different right parasternal short-axis views (b,c) with the indicator symbol on the right of the ultrasound image and the probe indicator marker directed towards the elbow. (b) The right parasternal short-axis view at the level of the ventricles is used for assessment of contractility and intravascular volume estimation. Single probe manipulations (fanning and rotating) should be used to make the image as symmetrical as possible, which will create an image referred to as the mushroom view. From here, the probe should be fanned dorsally through the mitral valve towards the cervical spine. Sometimes a slight rocking of the probe is needed to centre the heart in the image screen. When the correct plane is achieved, the heart base should be visualised with the round aorta (Ao) in the middle (c). This is the left atrial aortic view, where the left atrial and aortic diameters are subjectively compared and, if desired, objectively measured. LA = left atrium; LV = left ventricle; PV = pulmonary valve; RV = right ventricle
Cardiac disease findings: CHF
Cardiac POCUS is a useful adjunct in cats with cardiorespiratory instability to support a diagnosis of left-sided CHF (LCHF) as a cause of dyspnoea. Cardiology guidelines classify the left atrium as enlarged when the left atrium to aortic root ratio (LA:Ao) is >1.55. 27 Cats that present with clinical signs of dyspnoea and have B-lines secondary to LCHF have LA:Ao >2:1. 28 Subjective assessment of left atrial size is often used during POCUS by visualising the number of circumferences of the aorta that can fit into the lumen of the left atrium. 24 if more than 2.5 aortic circumferences can fit in the left atrium, enlargement should be suspected; however, like for LA:Ao, if a cat is dyspneic secondarily to LCHF, it is the authors’ experience that most cats will subjectively have more than four aortic circumferences within the left atrium (Figure 10). 28
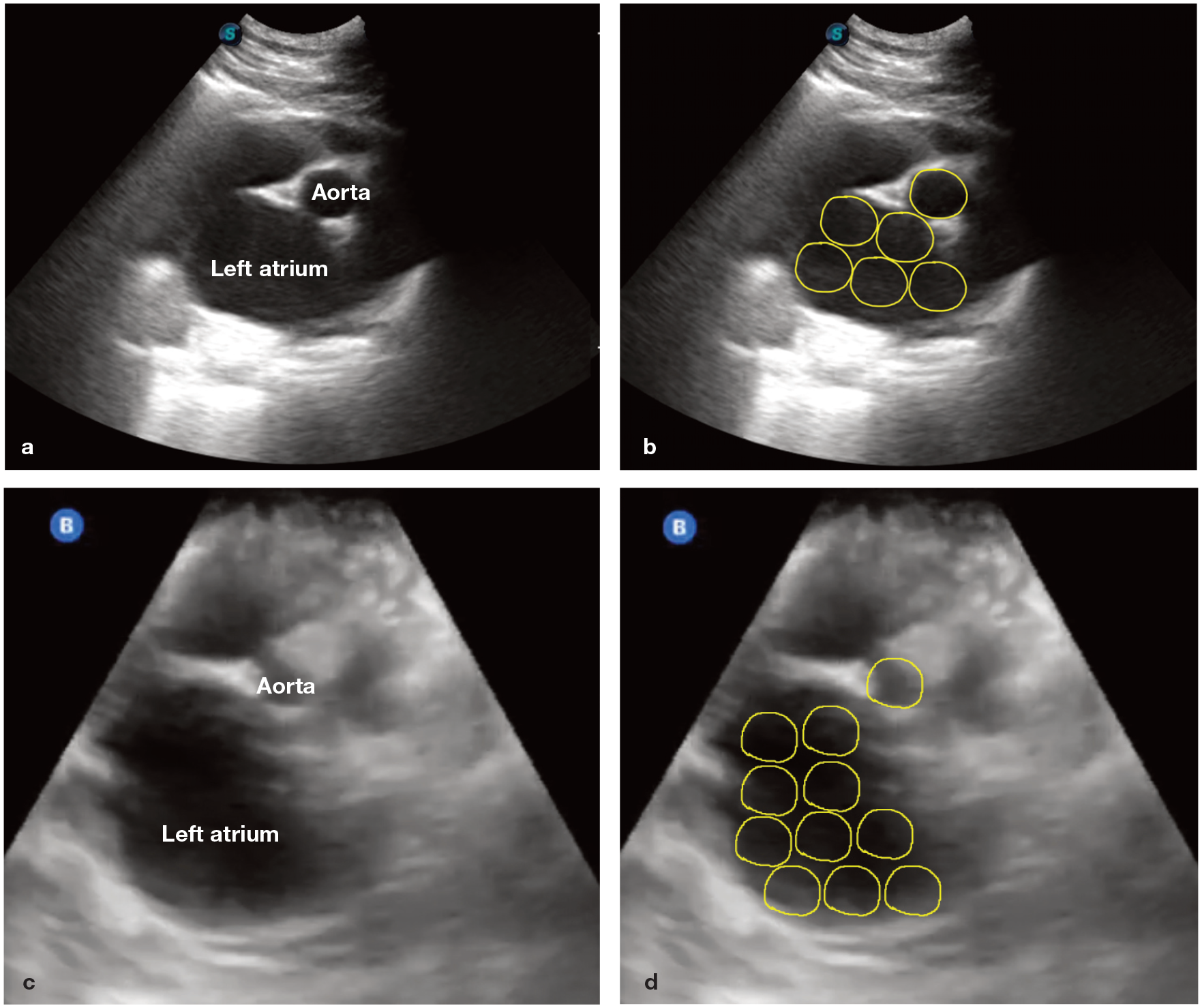
Right parasternal short-axis POCUS images of the left atrium to aortic root ratio (LA:Ao) labelled (a,c) and with a subjective estimation of the number of aortic circumferences (yellow circles) that can be drawn within the left atrial lumen (b,d). The images are from two different cats that presented in respiratory distress and scanned in a standing position. (a,b) The aortic circumferences can fit more than five times in the left atrium. (c,d) The aortic circumferences can fit >10 times in the left atrium. For the cat in (a,b), which was diagnosed with congestive heart failure, Video 3 in the supplementary material shows an enlarged LA:Ao, coalescent B-lines and pleural effusion
In addition to assessing LA:Ao, lung and pleural space POCUS findings (ie, more than three B-lines at multiple sites) can help to increase the index of suspicion of LCHF (see Video 3 in the supplementary material). Pericardial effusion in cats is also strongly associated with CHF.24,28 The authors therefore recommend evaluating LA:Ao subjectively in combination with clinical findings (gallop, murmur), cardiac POCUS findings (presence of pericardial effusion), lung and pleural space POCUS findings (B-lines and/or pleural effusion) ± other paraclinical information (NTproBNP), to support a diagnosis of CHF.17,18
Abdominal POCUS
Indications
Abdominal POCUS can expedite the diagnostic and therapeutic management of cats with acute abdomen including those with abdominal distension and azotaemia. Binary questions, including whether there is the presence of peritoneal effusion, visible urinary bladder and renal asymmetry and/or renal pelvic dilation may help streamline subsequent diagnostics and patient management.
Technique and normal views
Four or five focused sites, depending on the protocol used, are scanned during abdominal POCUS, obtaining the following views: (1) subxiphoid (level of the xiphisternum; Figure 11a; see also Figures 13 and 14a); (2) left para lumbar (Figure 11b); (3) right paralumbar (subcostally at the level of the last rib; Figure 11c); (4) urinary bladder (caudal midline; Figure 11d; see also Figure 14b); and (5), if assessed, umbilical. The probe is initially placed longitudinally at each site, then fanned and rocked to thoroughly assess for POCUS pathology. The authors recommend rotating the probe 90°, where possible, to also achieve the transverse views at each site.
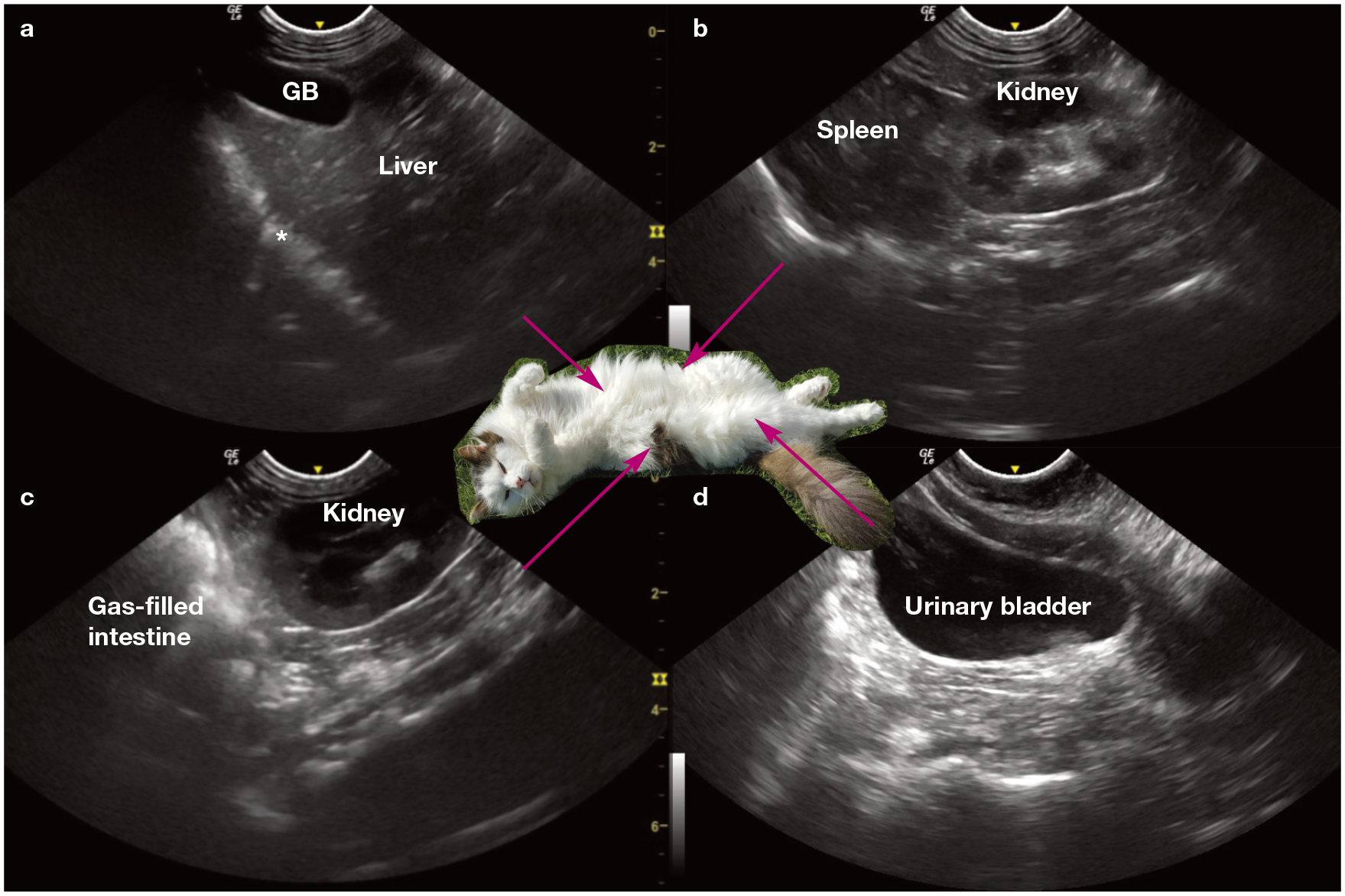
Four abdominal sites in a cat showing normal structures: (a) subxiphoid view with gallbladder (GB) and diaphragm (asterisk); (b) left paralumbar view; (c) right paralumbar view; and (d) urinary bladder view. The photo of the cat in dorsal recumbency in the centre is only for visualisation of the sites; this positioning is not recommended during POCUS scanning
Subxiphoid view
in the subxiphoid view, the liver, gallbladder, diaphragm, caudal vena cava (CVC) ± heart, pericardial space, caudal lung surface and pleural space can be assessed. A common problem encountered at the subxiphoid site, however, is incomplete visualisation of the cranial–ventral regions if the probe is not rocked cranially enough at the level of the xiphisternum. To achieve complete visualisation, pressure often needs to be applied to the probe while rocking it such that the probe is more parallel to the spine and the ultrasound beams are directed into the cranial and more ventral regions of the thorax. The authors recommend a fist-like grip on the probe to spread the pressure over a larger area to improve comfort when scanning; this grip should make the probe application feel like palpation rather than a stabbing sensation (Figure 12 and Video 4 in the supplementary material). Most cats prefer being scanned in sternal recumbency, and a rolled towel placed under the cat (see earlier) or gently lifting the cat into a more bipedal position (Figure 12) may be required to obtain the subxiphoid view. Fluid is usually localised between liver lobes or between the diaphragm and liver (Figure 13; see also Figure 14a); however, if the cat is lifted to view the subxiphoid site, patient positioning may shift fluid to the more caudal pelvic areas. Protocols should be modified to maximize the chances of detecting fluid, which will fall to the gravity-dependent regions, and vary based on patient position.

The owner of this cat is gently lifting the cat’s front end so it is in a bipedal position, allowing the veterinarian to perform POCUS and obtain the subxiphoid view. The probe is held in a fist-like grip to increase the surface area and decrease discomfort for the cat. Video 4 also shows this procedure, with the cat kneading its front paws, suggesting that it was happy to be assessed in this position. Courtesy of Katharina Lunde
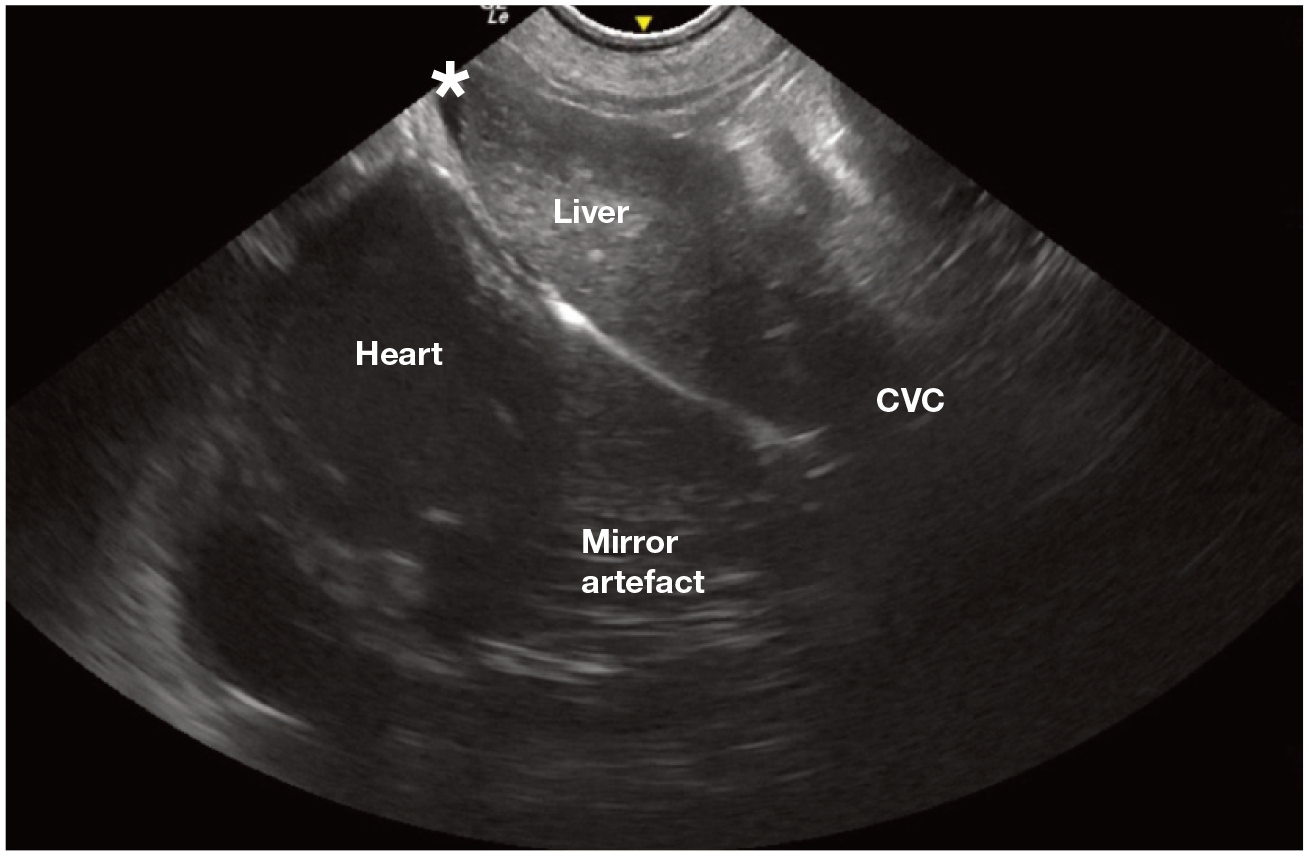
Subxiphoid view on POCUS in a Maine Coon cat with a small amount of free abdominal fluid marked with an asterisk. The CVC and the region beyond the diaphragm shows a mirror image artefact of the liver, reflected from above the diaphragm. CVC = caudal vena cava
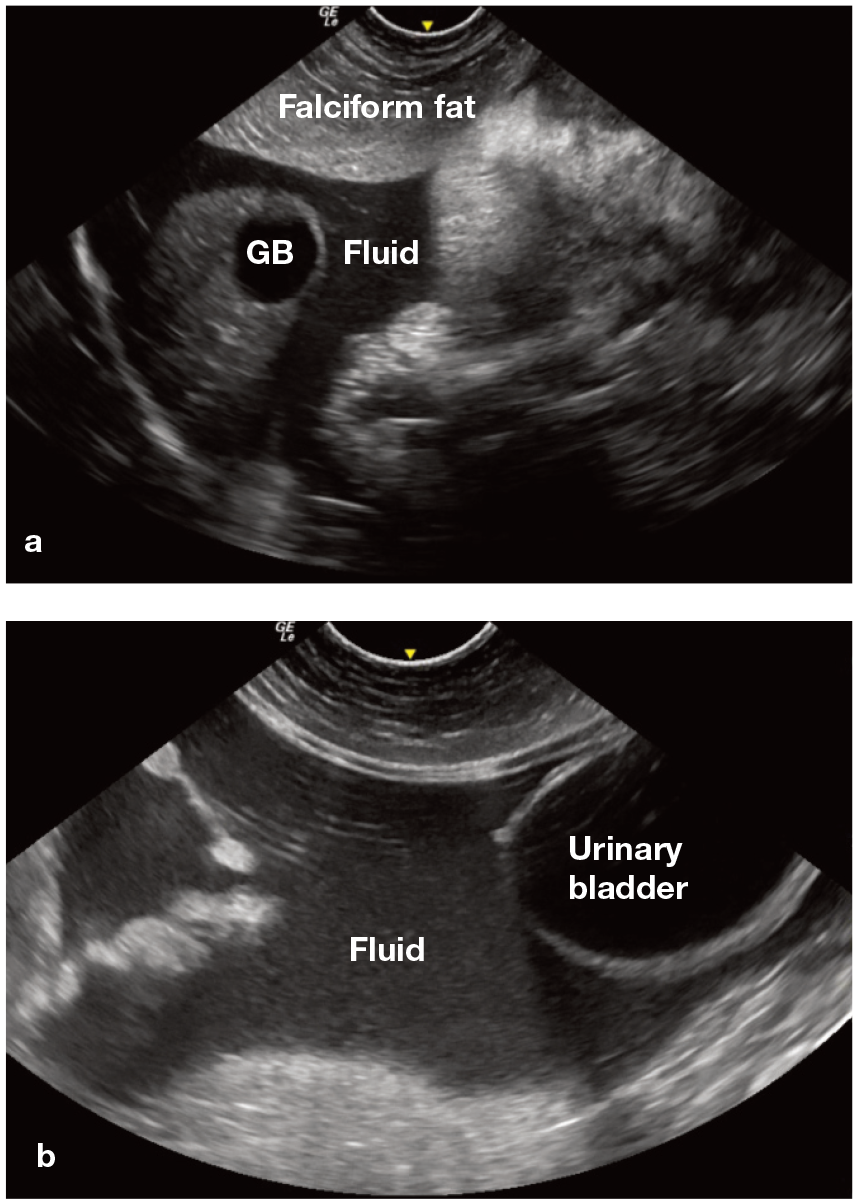
Two fluid-positive sites on POCUS in a cat. (a) Subxiphoid view where free abdominal fluid forms sharp angles/triangles between abdominal structures. The falciform fat is more prominent in cats than dogs. (b) Urinary bladder view where sharp angles/triangles can be seen surrounding the urinary bladder – a hallmark used to identify free abdominal fluid. GB = gallbladder
The CVC (Figure 13) can be visualised at the sub xiphoid site passing through the diaphragm between ventral midline and the gallbladder (located to the right of midline). The CVC collapsibility index (CVC-Ci), a non-invasive means of assessing volume status in cats, is normally >20%. it can be assessed in cats that are standing or in lateral recumbency and evaluates the change in CVC diameter during inspiration and expiration. However, it is operator dependent and inter-operator variability occurs. 29 Cate gorising the CVC into two sub jective groups: fat (hypervolaemic) and flat (hypovolaemic) is more clinically practical, particularly when combined with clinical findings (pulse quality, mucous membranes colour, refill, mentation and temperature). A flat CVC collapses during inspiration, while a fat CVC remains relatively static throughout respirations. Cats with cavitary effusions or CHF may also have a fat CVC, 30 and increased abdominal pressure, including that caused by scanning, can flatten CVC diameter. To avoid iatrogenic collapse, the pressure applied to the probe (compression) should be reduced after identifying the CVC and before subjectively assessing it. if the CVC supports euvolaemia or hypovolaemia, a fluid bolus can be administered provided clinical findings and history support the need for a bolus, and there are no contraindications to its administration (normal LA:Ao, no B-lines).
Right and left paralumbar views
Right and left paralumbar views are usually assessed with a microconvex probe, although a linear probe may provide better renal architecture visualisation. Although the probe is largely placed subcostally, occasionally, especially with smaller kidneys, intercostal placement of the probe may be required for renal assessment. The kidney and spleen, or kidney and liver, are visualised in the left and right paralumbar views, respectively. in both views, the aorta and CVC may be identified. Fluid is often trapped at the poles of the kidney, between the spleen and left kidney or liver and right kidney. Subjective assessment of the renal pelvis is often performed in the transverse plane; gross evidence of renal pelvic dilation in an azotemic cat should prompt further investigation to rule out a ureteral obstruction.31,32
Urinary bladder view
The urinary bladder view requires caudal abdominal placement of the probe on the mid-line. The bladder (Figure 11d), colon, great vessels and lymph nodes may be visualised. Free fluid often accumulates at the apex of the urinary bladder (see Figure 14b). Non-invasive measurement of urinary output can be achieved by serially scanning the bladder in longitudinal and transverse planes. Built-in ultrasound algorithms or the following equation can be used to estimate bladder volume at different time points: length × width × height (cm) × 0.2 × π = volume (ml). 33 The longitudinal and transverse measurement of height can be combined and divided by two for a more accurate measurement. Urinary bladder measurement is a non-invasive way to estimate if a patient is oliguric or anuric.
Peritoneal effusion
The main objective of abdominal POCUS is the detection of free abdominal fluid. This fluid often appears as dark (anechoic or hypo echoic) sharp angles (triangles) between organs and structures (Figure 14). However, highly cellular effusions, such as exudates or acute haemorrhage, can have a more heterogenic, isoechoic appearance relative to surrounding soft tissues. Probe manipulation (eg, rapid compression and release to cause cellular ‘swirling’ within fluid) helps differentiate highly cellular fluid from surrounding soft tissues. Abdomino-centesis and cytology are required for fluid classification. Fluid scoring the sites (0/4 or 0/5) is helpful in cases of traumatic peritoneal effusion. Each positive site is scored 1 point (Figure 14). 34 Serially calculating fluid scores monitors progression or resolution of fluid accumulation and may help with management decisions. 2 The frequency of scanning depends on the individual patient, reason for presentation, underlying pathology and stability of the cat. As a rule of thumb, cats should be reassessed as dictated by repeat physical examination and clinical findings, which in a haemodynamically unstable cat post-trauma may be within minutes if clinical status suddenly deteriorates, or a minimum of every 4 h if the cat is more stable and responding to therapy.

Key Points
✜ Veterinary POCUS is a rapid, evidence-based, non-invasive, repeatable, radiation-sparing, patient-side ultrasonographic examination of a patient.
✜ POCUS can be integrated into daily practice when performed patient-side as an extension of the physical examination.
✜ Pathologies commonly assessed with lung and pleural space POCUS include pleural space and pulmonary parenchymal disease (eg, air/fluid and B-lines/consolidation, respectively).
✜ Cardiac POCUS can help to support a diagnosis of LCHF as a cause of dyspnoea.
✜ Abdominal POCUS can expedite the diagnostic and therapeutic management of cats with acute abdomen, including those with abdominal distension and azotaemia.
Footnotes
Acknowledgements
The authors would like to thank Katharina Lunde and the VECCUS Board for their assistance.
Supplementary material
The following files are available as supplementary material:
Video 1: Lung sliding shown on POCUS in a cat.
Video 2: Lung sliding and abdominal curtain sign shown on POCUS in a cat.
Video 3: Enlarged LA:aorta and the presence of B-lines and pleural effusion shown on POCUS in a cat diagnosed with congestive heart failure.
Video 4: POCUS being performed in a cat in a bipedal position in order for the veterinarian to access the subxiphoid view. Courtesy of Katharina Lunde.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
This work did not involve the use of animals and therefore ethical approval was not specifically required for publication in JFMS.
Informed consent
This work did not involve the use of animals (including cadavers) and therefore informed consent was not required. For any animals or people individually identifiable within this publication, informed consent (verbal or written) for their use in the publication was obtained from the people involved.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
