Abstract
Objectives
The aim of this study was to determine the feasibility of ultrasonographically measuring the caudal vena cava (CVC) at the subxiphoid view of healthy, lightly sedated cats in a standing position and lateral recumbency.
Methods
This was a prospective, observational, experimental single-centre study. Twenty healthy research-purposed cats were enrolled. Two trained operators scanned each cat in two positions – standing and lateral recumbency – in a randomised order. CVC diameter was measured at the narrowest diameter during inspiration and at the widest diameter during expiration, at two anatomical locations along the CVC – where the CVC crosses the diaphragm (base) and 2 mm caudal to the diaphragm. The CVC collapsibility index (CVC-CI) was calculated for each site. Normalcy was assessed with a Shapiro–Wilk test. A one-way ANOVA with post-hoc Tukey’s test was used to compare inspiratory with expiratory values within and between groups. A paired t-test compared the CVC-CI between groups (P ⩽0.05 indicated statistical significance). Spearman’s correlation and Bland–Altman analysis assessed inter-operator variability.
Results
All ultrasonographic data passed normalcy and were reported as mean ± SD. When compared with each other, inspiratory and expiratory values were statistically different for position, location and operator (all P <0.0001). There was no statistically significant difference between lateral recumbency or standing position for inspiratory, expiratory and CVC-CI values. Inter-operator variability was substantial, with operator 2 consistently obtaining smaller measurements than operator 1. The mean CVC-CI in lateral recumbency at the base was 24% for operator 1 and 37% for operator 2. For the same site in standing position, CVC-CI was 27% and 41% for operators 1 and 2, respectively.
Conclusions and relevance
This pilot study demonstrates that it is possible to ultrasonographically measure the CVC diameter in both lateral recumbency and a standing position in healthy, lightly sedated cats. However, measurements obtained are operator dependent with variability between individuals. Further studies are needed to determine if ultrasonographic CVC assessment will prove helpful in estimating intravascular volume status in cats.
Introduction
Assessment of intravascular volume status is vital in the management of critically ill patients. Multiple studies in both human and veterinary medicine have demonstrated the negative effects of both hypovolaemia (intravascular volume depletion) and hypervolaemia (intravascular volume overload) in critically ill patients admitted to the intensive care unit.1–8 Assessment of intravascular volume status is often based on patient history and physical examination findings, which have low sensitivity and specificity at predicting states of volaemia and fluid responsiveness. 9
Developing appropriate tools to diagnose intravascular volume derangements, monitor intravascular volume status and predict fluid responsiveness is crucial to the management of emergency and critical care (ECC) patients. Cats are particularly challenging given their unique response to shock states (bradycardia, hypothermia and hypotension) and their small size. Furthermore, they are prone to volume overload, likely due to their smaller blood volume, decreased rate of fluid elimination and potential to have occult cardiomyopathies.10–14 Owing to its elastic nature, ultrasonographic evaluation of the inferior vena cava (IVC) is an effective, non-invasive monitoring tool to determine intravascular volume status and fluid responsiveness in people.15,16 Moreover, feasibility and reference intervals (RIs) for caudal vena cava (CVC) diameter and CVC collapsibility index (CVC-CI) have been established in dogs and foals.17,18 It has also been demonstrated that the CVC-CI can accurately predict fluid responsiveness in critically ill dogs with perfusion abnormalities. 19 There is limited research regarding the CVC as a volume assessment tool in cats. A recent study described CVC measurements at three sites – subxiphoid (SV), hepatic (HV) and paralumbar (PV) views – in anaesthetised cats (n = 21) undergoing blood donation. The study found a difference in the CVC diameter when comparing a 10 ml/kg deficit of whole blood and a subsequent 30 ml/kg bolus of crystalloid fluids. 20 Another study examining the CVC-CI in a group of 15 healthy awake cats via the right HV showed a CI of 28% ± 3% at this location. 21
Historically, most small animal point-of-care ultrasound (POCUS) examinations, including CVC evaluation in dogs, have been performed in lateral recumbency.18,22 In contrast, a standing position has been used in foals. 17 Given a greater degree of restraint is often required to maintain patients in lateral recumbency, which is no longer recommended in cats, exploring alternative patient positions, such as standing, is warranted when performing feline POCUS examinations. 23
To our knowledge, measurement of the CVC at the SV in awake cats and the effect of patient positioning on ultrasonographic CVC measurements has not been investigated. The aim of this pilot study was to determine if it is possible to ultrasonographically visualise and measure the inspiratory and expiratory CVC diameter, and calculate the CVC-CI at two CVC locations obtained from the SV of healthy awake cats in standing and lateral recumbency. The results will be used to determine the optimal position for SV ultrasonographic assessment of the CVC, which will subsequently allow establishment of RIs from a much larger study population of cats. We hypothesised that image acquisition would be possible, that inter-operator variability would be acceptable and there would be no statistically significant differences in the CVC measurements obtained in standing and lateral recumbency.
Materials and methods
Animals
This was a prospective, observational, experimental single-centre study. The study protocol was approved by the Massey University Animal Ethics Committee (Protocol 19/136). Twenty healthy research-purposed cats from the Centre for Feline Nutrition at Massey University, New Zealand, were enrolled. Cats were deemed healthy based on physical examination, complete blood count and biochemistry analysis. Additionally, all cats were screened for occult cardiomyopathy using the two-minute screening echocardiogram for cats as described by Loughran et al. 11
Devices and operators
The study was performed using the same machine (Xario 200, Model: TUS-X200; Toshiba Care) with a medium frequency (7 MHz) convex transducer (PVU-712BT) with depth set at 5–7 cm. Without clipping, the fur was parted and a combination of alcohol and ultrasound gel was used as the coupling agent. The two operators (operator 1 [an ECC resident] and operator 2 [a board-certified ECC specialist]) were extensively trained in abdominal, pleural space, lung and cardiovascular POCUS. Operator 1 used their right hand, while operator 2 used their left hand to perform the POCUS examinations.
Study protocol
Approximately 10–30 mins before examination each cat was administered butorphanol (Butorgesic 10 mg/ml; Troy Laboratories) at a dose of 0.3 mg/kg subcutaneously. Low stress handling, provided by a certified research assistant, was applied to the cats for each POCUS evaluation. Cats were placed on a table and scanned in right lateral recumbency and a standing position by both operators.
Operators were blinded to each other’s ultrasonographic images and measurements. The two operators scanned each cat in two positions, standing and right lateral recumbency, in a randomised order, for both operator and position. Scans were completed in succession, with each operator starting their scan immediately following completion of the preceding examination by the other operator. The probe was placed just caudal to the xiphoid cartilage in longitudinal axis, at roughly a 45° angle to the spine and with the marker pointed cranially. The liver and diaphragm were identified, and the probe was then gently fanned to the right of midline until a longitudinal view of the CVC was obtained, visualised as two horizontal parallel lines (walls of the CVC) crossing the diaphragm (Figure 1). The ultrasound probe was then slowly fanned through all longitudinal planes across the CVC, to identify its luminal centre, subjectively identified as the largest distance between the two walls of the CVC. Two to five 10 s cine loops were recorded over several respiratory cycles for post-hoc measurement and analysis of the CVC.

Still ultrasound image from the subxiphoid view of a cat in lateral recumbency showing the caudal vena cava (CVC), recognised as two parallel lines crossing the diaphragm. Two CVC measurements were made: one at the level the CVC crosses the diaphragm (CVCB) and the second 2 mm caudal to the level the CVC crosses the diaphragm (CVC2mm)
The narrowest CVC diameter during inspiration (insp) and widest CVC diameter during expiration (exp) (Figure 2) were measured post-hoc by each operator at two anatomical locations along the CVC: at the entry of the CVC into the abdominal cavity (B = base) and 2 mm caudal to site B (2 mm). Images from one respiratory cycle were used by each operator to obtain all CVC measurements. The choice of which cine loop and respiratory cycle within a cine loop was left to the discretion of each operator. The CVC-CI was calculated at each CVC location (B and 2 mm) for all cats in both lateral and standing positions (Lat and Stan) using the following formula: (CVCexp – CVCinsp)/CVCexp × 100.

Schematic images of the caudal vena cava (CVC) during (a) inspiration and (b) expiration in a euvolaemic cat. The CVC is identified as the two parallel lines crossing the diaphragm. Two CVC measurements were made: one at the level the CVC crosses the diaphragm (CVCB) and the second 2 mm caudal to the level the CVC crosses the diaphragm (CVC2mm). GB = gallbladder
Statistical analysis
Statistical analysis was performed using the commercially available software program Prism (Version 8.4.2 for macOS; GraphPad Software). A Shapiro–Wilk test was used to assess ultrasonographic results for normalcy. A one-way ANOVA with post-hoc Tukey’s test was used to compare inspiratory with expiratory values within and between groups. A paired t-test compared the CVC-CI between groups. Spearman’s correlation and a Bland–Altman analysis were used to assess inter-operator variability. Intra-class correlation coefficient, which ranges from 0 (no association between the two observers) to 1 (perfect agreement between the two observers), was used to assess the level of agreement of CVC measurement between the two operators. A P value ⩽0.05 was considered significant for all statistical calculations.
Results
There were 14 intact females, five intact males and one neutered male. Median age was 1.4 years (range 0.9–4). Median weight was 3 kg (range 2.2–4.4). Median numbers of cine loops recorded per cat was 2.8 in the lateral position and 3.4 in the standing position for operator 1, and 2 and 2.5 recordings, respectively, for operator 2. Operator 2 failed to scan two cats in standing position owing to movement of the cats. Therefore, the success rate for identifying the CVC in both positions was 100% for operator 1 and 100% in lateral and 90% in standing for operator 2. Descriptive statistics are presented in Table 1.
Caudal vena cava (CVC) diameter measurements and collapsibility index (CI) calculations for each operator
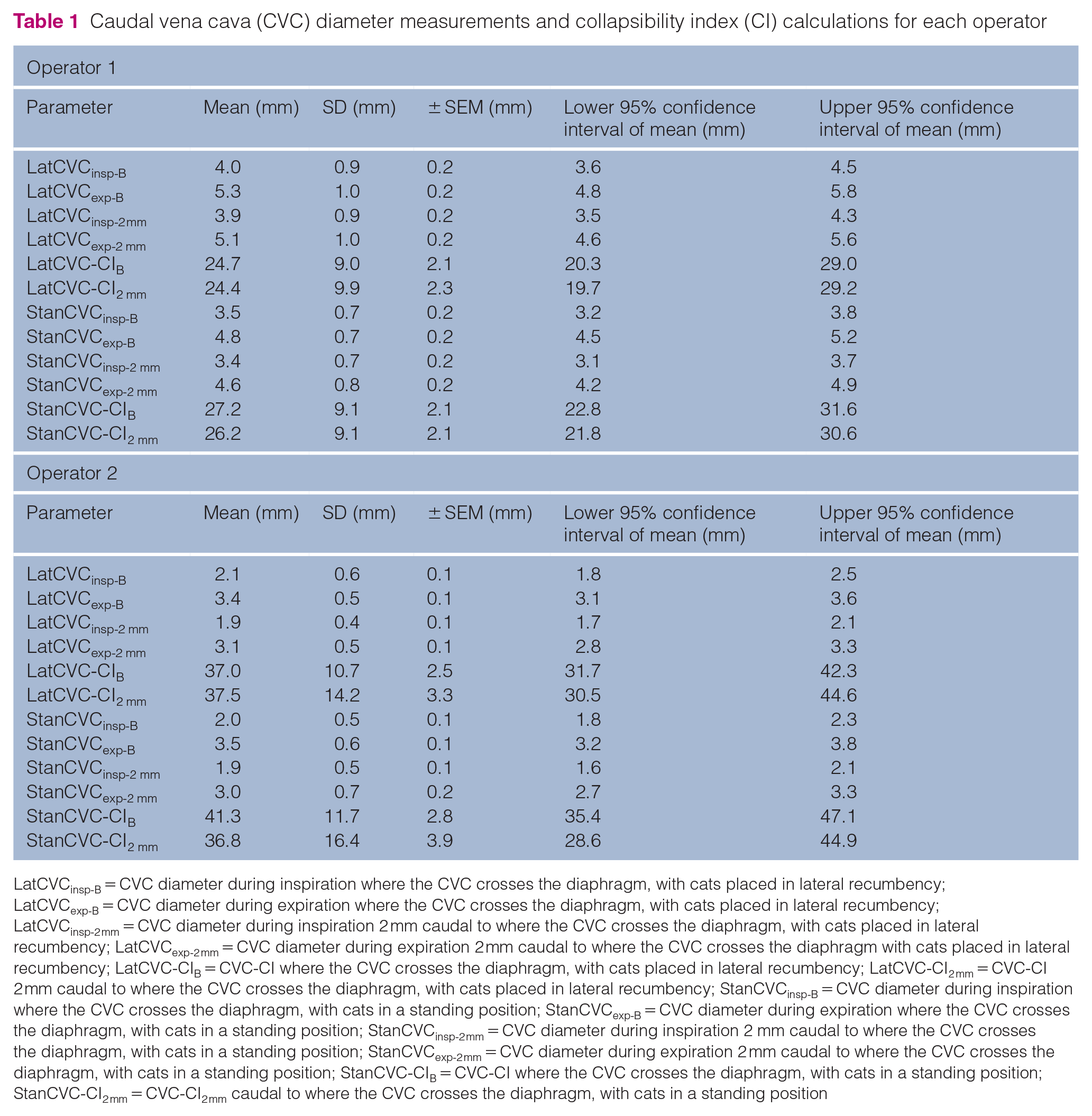
LatCVCinsp-B = CVC diameter during inspiration where the CVC crosses the diaphragm, with cats placed in lateral recumbency; LatCVCexp-B = CVC diameter during expiration where the CVC crosses the diaphragm, with cats placed in lateral recumbency; LatCVCinsp-2 mm = CVC diameter during inspiration 2 mm caudal to where the CVC crosses the diaphragm, with cats placed in lateral recumbency; LatCVCexp-2 mm = CVC diameter during expiration 2 mm caudal to where the CVC crosses the diaphragm with cats placed in lateral recumbency; LatCVC-CIB = CVC-CI where the CVC crosses the diaphragm, with cats placed in lateral recumbency; LatCVC-CI2 mm = CVC-CI 2 mm caudal to where the CVC crosses the diaphragm, with cats placed in lateral recumbency; StanCVCinsp-B = CVC diameter during inspiration where the CVC crosses the diaphragm, with cats in a standing position; StanCVCexp-B = CVC diameter during expiration where the CVC crosses the diaphragm, with cats in a standing position; StanCVCinsp-2 mm = CVC diameter during inspiration 2 mm caudal to where the CVC crosses the diaphragm, with cats in a standing position; StanCVCexp-2 mm = CVC diameter during expiration 2 mm caudal to where the CVC crosses the diaphragm, with cats in a standing position; StanCVC-CIB = CVC-CI where the CVC crosses the diaphragm, with cats in a standing position; StanCVC-CI2 mm = CVC-CI2 mm caudal to where the CVC crosses the diaphragm, with cats in a standing position
All ultrasonographic data passed normalcy. Regardless of patient position (lateral or standing), site of CVC diameter measurement (B or 2 mm) and operator (operator 1 or operator 2) there was a statistically significant difference between the inspiratory and expiratory CVC diameter (P <0.0001). For each operator, there was no statistically significant difference between lateral recumbency or standing position for inspiratory or expiratory CVC values. Measurement of inspiratory and expiratory CVC values for each operator with cats in lateral recumbency and standing position at CVC location B are shown in Figure 3. Values for each CVC parameter for operators 1 and 2 are presented in Table 1. There was a statistically significant difference between operator 1 and operator 2 for all CVC measurements (P <0.0001). Operator 2 obtained smaller CVC measurements than operator 1, although the CVC-CI was larger for operator 2 than operator 1. Correlation between operators was considered poor for all CVC parameters in both positions and locations (Table 2). Inter-operator variability was substantial for both positions and locations (Table 3).

Box and whisker plots of inspiratory and expiratory caudal vena cava (CVC) measurements at the site the CVC crosses the diaphragm. (a) Operator 1, measurements in lateral recumbency; (b) Operator 1, measurements in a standing position; (c) Operator 2, measurements in lateral recumbency; (d) Operator 2, measurements in a standing position
Caudal vena cava (CVC) correlation coefficients between operators for cats in lateral recumbency and a standing position
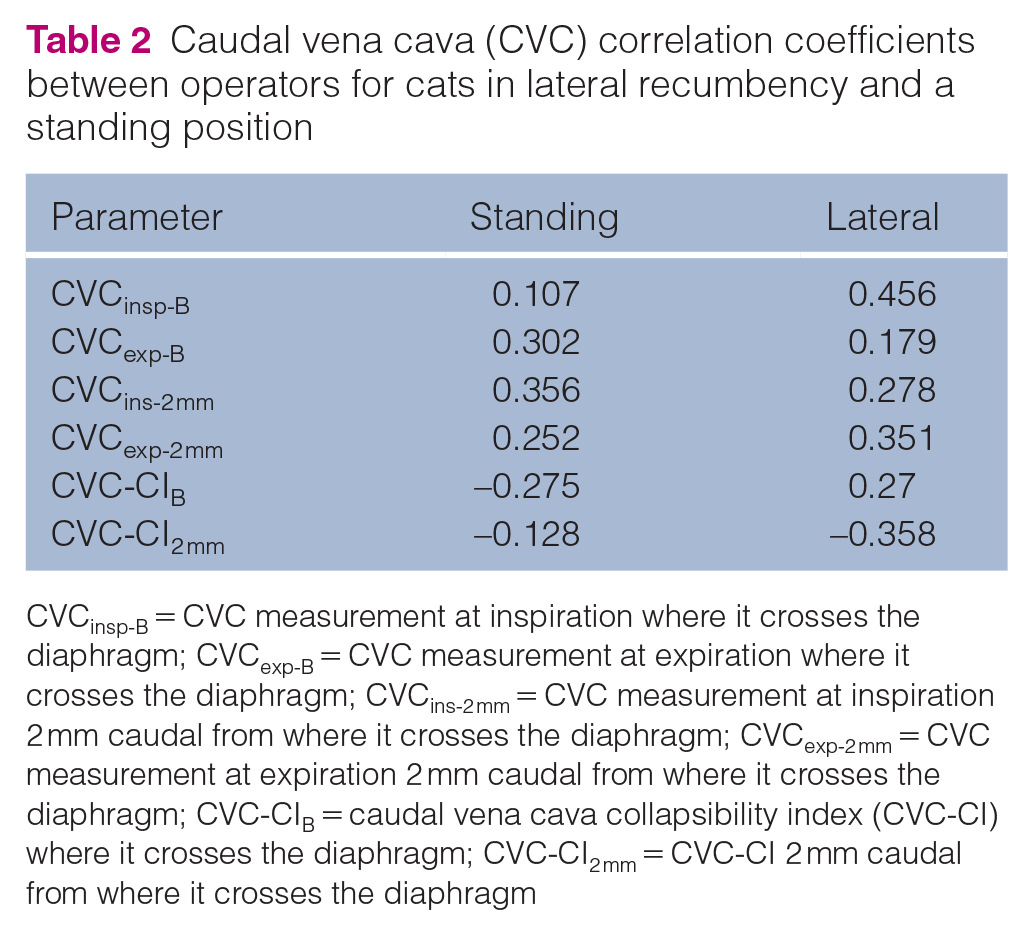
CVCinsp-B = CVC measurement at inspiration where it crosses the diaphragm; CVCexp-B = CVC measurement at expiration where it crosses the diaphragm; CVCins-2 mm = CVC measurement at inspiration 2 mm caudal from where it crosses the diaphragm; CVCexp-2 mm = CVC measurement at expiration 2 mm caudal from where it crosses the diaphragm; CVC-CIB = caudal vena cava collapsibility index (CVC-CI) where it crosses the diaphragm; CVC-CI2 mm = CVC-CI 2 mm caudal from where it crosses the diaphragm
Bland–Altman analysis for inter-operator variability for caudal vena cava (CVC) parameters

CVC-CI = caudal vena cava collapsibility index; LatCVCB = CVC diameter where it crosses the diaphragm, with cats placed in lateral recumbency; LatCVC2 mm = CVC diameter 2 mm caudal from where it crosses the diaphragm, with cats placed in lateral recumbency; StanCVCB = CVC diameter where it crosses the diaphragm, with cats in a standing position; StanCVC2 mm = CVC diameter 2 mm caudal from where it crosses the diaphragm, with cats in a standing position
Discussion
This pilot study in 20 cats demonstrated a statistically significant difference between inspiratory and expiratory CVC diameter. This suggests that it is possible to visualise and measure the CVC at the SV of healthy, lightly sedated cats, which agrees with previous studies. In foals, the CVC has been ultrasonographically identified and measured in a standing position, 17 while SV CVC measurements have only been documented in dogs and cats placed in lateral recumbency.18,20 One of the main findings of this pilot study is that patient position (standing or lateral) has little impact on CVC measurement in cats. This may have clinical relevance considering cats tend to be more reluctant to be restrained than other species, and current trends are to perform veterinary POCUS examinations in the position the patient is most comfortable, which may be lateral or standing. Interestingly, studies in people report conflicting results when comparing IVC diameters measured in supine, lateral, upright and prone positions.24–27 Owing to the anatomical differences, however, a direct inter-species comparison cannot be made. To our knowledge, no similar studies on patient position and CVC diameter have been performed on dogs.
A limited number of studies in cats suggests the CVC can be measured at the same three locations as reported in dogs. A study in 15 healthy cats reports CVC diameter measurements obtained from the HV. 21 A study in 21 healthy cats anaesthetised for blood donation reported CVC measurements at the SV, HV and PV. 20 The authors reported 100% success rates at the SV but an inability to obtain images at three and seven attempts for the HV and PV, respectively. They concluded that the SV is best suited for visualisation of the CVC, with the PV also being reliable. Obtaining images from three views, in two recumbencies by two operators, exceeded the handling capabilities of lightly sedated cats. For these reasons, in this pilot study focusing on positioning, only the SV was evaluated. Further studies in awake healthy and critically ill cats with variable volume states are required to determine if any single CVC view is superior to another for intravascular volume status and fluid responsiveness evaluations. Of note, the SV is incorporated in all abdominal POCUS protocols, while the HV is not, and the PV focuses on other structures than the CVC. Therefore, it is likely that practitioners are most comfortable with assessing the CVC at the SV.
The variation in CVC diameter during the respiratory cycle makes it possible to calculate the CVC-CI. Previous studies in people and dogs indicate that the CVC-CI can be used to assess intravascular volume status.28–30 This is in contrast to a 2021 study in healthy feline blood donors, which suggests the CVC-CI is not useful in cats and has considerable variability in measurements between operators. 20 A study in healthy dogs also showed significant inter-operator variability for CVC-CI at the SV. 18 Similarly, the present study demonstrated a significant inter-operator variability for both inspiratory and expiratory CVC diameter and for CI, regardless of patient position or where CVC values were measured. The CVC-CI had the highest inter-operator variability, which is likely a result of the CVC-CI being dependent on a calculation, which will compound any differences between operators. Inter-operator variability in the present study may have been influenced by the relatively small size of the feline CVC vs other species, as suggested in the above-mentioned study in cats undergoing blood donation. 20 Furthermore to fully assess inter-operator variability in obtaining the window and measurements, the choice of cine loop and respiratory cycle within a cine loop was left to the discretion of the operator. This approach likely increased the variability noted between operators as there may have been changes in the CVC diameter during different respiratory cycles and with regard to when cine loops were captured. Future research measuring the inter-operator variability on the same cine loop and respiratory cycle, and averaging the measurements from several respiratory cycles, should help determine the source of variability between operators.These limitations should not preclude the use of the CVC-CI measured at the SV in cats. The greatest variation of CVC-CI is expected to occur in healthy populations, with less variation occurring in patients with values outside RIs. Therefore, although variation is substantial in healthy people, foals and dogs, calculation of the CVC-CI at the SV may still have clinical utility and prognostic relevance when values are at the extremes of hypovolaemia and hypervolaemia. 28 This study had several additional limitations. The CVC shape may change depending on the manual pressure applied to the ultrasound probe. However, as operators applied minimal manual pressure to acquire images, this is not likely a significant factor. Each operator scanned cats using their dominant hand to manipulate the probe, which was the opposite for each operator. Research on the effects of handedness on ultrasonographic image quality and measurements is lacking. However, scanning with the dominant hand is the accepted level of standard. The small sample size may also have affected results. The two sites were originally chosen to account for any interference from cardiac oscillations that may impact CVC respiratory variation and inter-rater variability. However, descriptive statistical analysis showed no major differences between sites and no further analysis was made.
This pilot study, like prior feline CVC studies, was performed in small cohorts of healthy cats. Measurements of CVC parameters in larger populations of sick cats with varying intravascular volume status are needed to accurately evaluate the usefulness of the CVC to guide fluid therapy in cats. Obtaining accurate CVC measurements might be hindered by image resolution because of the small size of the vessel. However, future research focusing on the subjective classification of the CVC as small, normal or enlarged may prove more transferable to everyday practice. Indeed, a recent study demonstrated that general practitioners can be successfully trained to screen asymptomatic cats for occult heart disease using a ‘two-minute screening echocardiogram’ and subjectively determine whether the heart is normal, equivocal or diseased when compared with an examination from a board-certified cardiologist. 11
Conclusions
This pilot study indicates that measurements of CVC diameter are feasible in healthy, lightly sedated cats in both lateral recumbency and a standing position, and that the effects of position and location were minimal. Significant inter-operator variability was noted.
Footnotes
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This project received funding from the Massey University Wenzel Research Fund (RM22764).
Ethical approval
This work involved the use of experimental animals and the study therefore had ethical approval from an established committee as stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers) for all procedure(s) undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
