Abstract
Objectives
This study aimed to characterize the clinical, radiographic and histopathologic features of early-onset gingivitis (EOG) and periodontitis in cats.
Methods
The medical records database was searched for cats diagnosed with histologically confirmed EOG or periodontitis from 1997 to 2022. Information such as medical history, lifestyle factors, clinical presentation, radiographic and histopathologic features were included for 27 client-owned cats. Response to treatment and long-term follow-up was also recorded.
Results
Moderate-to-severe periodontal disease was radiographically confirmed in 78% (21/27) of cats with moderate-to-severe EOG, compared with the evidence of periodontal disease noted in 30% (8/27) of cases during awake oral examination. Horizontal bone loss, along with missing teeth, were the predominant radiographic features noted in 89% (24/27) of cases. The predominant histopathologic feature was moderate-to-severe, erosive-to-ulcerative, neutrophilic and lymphoplasmacytic inflammation with varying degrees of epithelial and stromal hyperplasia. Two cats developed feline chronic gingivostomatitis (FCGS)-like lesions, and seven cats exhibited worsening of aggressive periodontitis (AP). Lack of improvement in the severity of gingivitis or clinical signs evident at the first follow-up appointment was significantly associated with progression of disease (P = 0.004).
Conclusions and relevance
The results of this study demonstrate the importance of oral evaluations in cats as early as 6 months of age. For cats exhibiting substantial gingivitis, an anesthetized evaluation, periodontal treatment and long-term monitoring are recommended. Given the high frequency of moderate-to-severe periodontitis encountered in these cats, clients should be informed about the potential need for tooth extractions. EOG may progress to AP. Finally, this study suggests that there could be a link between EOG and FCGS; however, further studies are needed to better characterize this condition and establish any potential link between the two entities.
Introduction
The most common disease of the tooth-supportive tissues in dogs and cats is periodontal disease. Periodontal disease has plaque-induced and non-plaque-induced causes.1,2 Initially, the inflammation is confined to the gingiva and is termed gingivitis and considered reversible. Once the disease progresses to periodontitis (inflammation and destruction of the attached gingiva, periodontal ligament and alveolar bone), the condition is no longer reversible. Early-onset gingivitis (EOG) and aggressive periodontitis (AP) are recognized clinical entities in young cats;1,3 however, despite the clinical presence and its negative health impact, this disease has not been characterized.
EOG describes the inflammation of the gingival tissues of the permanent dentition seen in young cats at around 6–8 months of age. 4 The etiology of EOG is unknown, and the incidence has not been reported. 4 Affected cats develop marked gingival inflammation, with erythema, edema and bleeding. In contrast, AP is characterized by extension of the inflammation to deeper tissues, leading to early attachment loss, periodontal pocket formation, gingival recession, furcation exposure and early tooth loss. 4 Anecdotally, EOG has been reported to progress to AP. 4 Phenotypically, the disease may be seen with or without concurrent gingival enlargement. 5 Alternatively, gingivitis may potentially progress to a more devastating and diffuse oral inflammatory disease known as feline chronic gingivostomatitis (FCGS). Occasionally, resolution is reported at the time of sexual maturity, and may suggest a hormonal influence.4,5
AP, or periodontitis stage III–IV and grade C, as per the 2017 ‘Classification of Periodontal and Peri-implant Diseases and Conditions’ in humans,6–8 has been estimated to occur in up to 13% of young adult cats. 1 This disease can present as a localized or aggressive form, and is characterized by a rapid evolution as compared with chronic periodontitis. 7 From a comparative pathology standpoint, the disease likely has environmental, microbial and genetic influences.9–16 Similarities in the histology of aggressive and chronic periodontitis in humans have been reported; however, the clinical manifestations are dissimilar, with AP having an earlier age of onset and a more rapid disease progression than chronic periodontitis, supporting the hypothesis of individual variations in susceptibility. 17 The histopathologic findings of EOG and periodontitis in cats have yet to be described.
The umbrella term historically used for EOG in cats is ‘feline juvenile gingivitis’ or ‘feline juvenile hyperplastic gingivitis’. 4 Given the prevalence and medical burden of the disease, there is a clinical need for a better understanding of EOG and periodontitis in the cat. In this context, characterization of the disease is prudent as it will serve as the foundation for further in-depth studies on early detection, preventative treatment, prognostic factors and effective therapeutics, to avoid early tooth loss in these patients.
Therefore, the present descriptive study aimed to characterize the clinical, radiographic and histopathologic features of EOG/periodontitis, as well as to evaluate potential lifestyle and environmental factors that may play a role in this disease.
Materials and methods
Case selection
The medical records of all cats examined by the Dentistry and Oral Surgery Service at the William R Pritchard Veterinary Medical Teaching Hospital, University of California-Davis, between January 1997 and March 2022, were reviewed. Cats that were 2 years old or younger at the initial presentation of disease were considered for inclusion. Diagnosis of EOG/periodontitis was made based on history and clinical findings of moderate-to-severe gingivitis that did not cross the mucogingival junction, dental radiographic signs and confirmed by histopathologic evaluation. For cats that presented more than once, the initial, as well as first follow-up anesthetized evaluation, including the set of full-mouth radiographs, dental charting and histopathologic findings when available, were assessed. For the two cats that had full-mouth extractions performed during the initial anesthetic episode, follow-up evaluation was performed via awake oral examination.
Data collection
The following data were obtained: signalment (breed, sex, neuter status, age); clinical signs at the time of presentation (ie, stomatorrhagia, inflammation, halitosis, weight loss, tooth loss and oral discomfort) and duration of these, and lifestyle factors (ie, indoor vs outdoor access, diet, presence of other cats in the household and oral care at home). In addition, pertinent medical history, including feline leukemia virus (FeLV)/feline immunodeficiency virus (FIV) status, as well as feline calicivirus (FCV) and feline herpesvirus (FHV) status – when available – and specific medications (non-steroidal anti-inflammatory drugs [NSAIDs], antibiotics, steroids and topical antimicrobial) received at the time of diagnosis, were obtained. Awake oral examination findings (ie, gingivitis, gingival enlargement, periodontal disease or tooth resorption, mucosal inflammation and mandibular lymphadenomegaly) were noted.
Hematologic findings such as anemia, hyperproteinemia, hyperglobulinemia and hyperglycemia were also obtained. Furthermore, anesthetized oral examination findings (ie, plaque and calculus burden, gingivitis index, stage and distribution of periodontal disease, and the presence of feline resorptive lesions and retained root tips) were included. Finally, histopathological diagnosis, and treatment and response to treatment were recorded. Cats with evidence of oral neoplasia were excluded from this study.
Radiological and histological evaluations
The full-mouth dental radiographs were reviewed by two board-certified dentists (MSR and BA). Histopathologic samples were re-examined by a board-certified pathologist (NV) and board-certified dentist (MSR).
Statistical analysis
Descriptive statistics were calculated. The type of distribution was determined with a Shapiro–Wilk test. Continuous data were not normally distributed; thus, they were expressed as median values, and ranges were reported along with 95% confidence intervals (CIs). Categorical data were expressed as frequencies. Fisher’s exact test was used to evaluate the association of periodontal disease stage and disease progression with anesthetized oral examination findings. Association between type of inflammatory infiltrate and radiographic features with anesthetized evaluation findings, periodontal disease stage and progression of disease was also evaluated. All tests were two-tailed and a P value <0.05 was considered to be statistically significant. Statistical analysis was conducted using GraphPad Prism version 9.4.0.
Results
Signalment and clinical presentation
The medical records search produced 53 cases, and 27 cats met the inclusion criteria. Purebred cats accounted for 41% (n = 11) of the population, with the following breeds represented in our study group: Maine Coon (n = 3), Abyssinian (n = 2), Tonkinese (n = 1), Havana Brown (n = 1), Devon Rex (n = 1), British Shorthair (n = 1), Himalayan (n = 1), Somali (n = 1). Female spayed cats represented 63% (n = 17) of cases, and castrated males followed closely, accounting for 33% (n = 9) of the population. Median age at presentation was 17 months (range 6–36, 95% CI 12–24), and the median duration of clinical signs prior to diagnosis was 9 months (range 0.5–24, 95% CI 2–17). Clinical signs at presentation in order of frequency included inflammation (n = 27), halitosis (n = 13), oral discomfort (n = 11), tooth loss (n = 6), stomatorrhagia (n = 3) and weight loss (n = 1). Fourteen cats had evidence of inflammation noted by the owners or primary care veterinarian but were asymptomatic.
Lifestyle and environmental factors
We noted that 74% (n = 20) of cats were kept indoors, and six (22%) had access to outdoor areas. Only one cat was kept exclusively outdoors. Forty-four percent (n = 12) of cats ate both dry and wet food, nine only ate dry food and one cat only ate canned food. In addition, 13 cats (48%) were members of multi-cat households, and only six had some form of oral care, including Veterinary Oral Health Council-approved dental food, treats and rinse, as well as a chlorhexidine-based product. Multi-cat household (P >0.99), outdoor access (P = 0.54) and pure breed (P = 0.63) were not significantly associated with periodontal disease stage. Multi-cat household (P = 0.12), outdoor access (P = 0.6199) and pure breed (P = 0.30) were not significantly associated with progression of disease.
Pertinent medical history
Six cats (22%) were reported to have a history of upper-airway disease. Of these six patients, one had viral pneumonia and another had concurrent conjunctivitis. Of the cats without reported upper airway signs, one had a cough of unknown origin and one had a healed tongue lesion. FeLV/FIV status (all negative) was known for 70% (n = 19). Retroviral status was acquired from the primary care veterinarians’ medical records for 13 patients, and antigen testing was performed at the initial visit for nine. Retroviral status was not re-evaluated for any of these patients. Eight cats were tested for FHV via PCR or virus isolation from samples obtained via oral swabbing; of these, only three tested positive. Seven cats were tested for FCV via PCR or virus isolation from samples obtained via oral swabbing; of these, four tested positive.
Hematologic and serologic analysis
Analyses consisted of a complete blood count and serum biochemistry panel or point-of-care diagnostics, including packed cell volume, total protein, blood glucose and blood urea nitrogen test strip. Four cats were anemic. Seven cats were hyperproteinemic, of which four had confirmed hyperglobulinemia. Five cats were hyperglycemic, with two patients having blood glucose values above 11 mmol/l (200 mg/dl).
Awake oral evaluation
Awake oral evaluation revealed severe and moderate gingivitis in 63% (n = 17) and 37% (n = 10) of cases, respectively (Figure 1). Gingival enlargement was noted in 48% (n = 13); one case was generalized. Gross evidence of periodontal disease was noted in 30% (n = 8) of cases, and feline resorptive lesions were suspected in 18% (n = 5) of cases. Inflammation of the caudal buccal mucosa was noted in 26% (n = 7) of cats. These lesions were described as contact ulceration, one was characterized as severe and the rest were mild. Of these seven cases, four progressed to AP and only two progressed to develop lesions involving of part of the caudal aspect of the oral mucosa. Malocclusions were noted in seven cats. Mandibular lymph node enlargement was reported in 33% (n = 9) of cats.

(a–c) Clinical appearance of early-onset gingivitis and periodontitis in an 8-month-old female spayed domestic mediumhair cat (case #22) with a 1-month history of moderate-to-severe gingivitis incidentally found during a wellness evaluation. (d–f) Intraoral radiographs of the same cat, demonstrating mild-to-moderate alveolar bone loss (†) and type I tooth resorption (*), as well as missing left mandibular first and second incisor teeth
Anesthetized evaluations
Dental charting and full-mouth intraoral dental radiography were performed in all cases. The plaque index was described as 1 in 26% (n = 7), 2 in 33% (n = 9) and 3 in 30% of cases (n = 8). The plaque index was not specified for three patients. The calculus index was described as 1 in 44% (n = 12), 2 in 30% (n = 8) and 3 in 19% (n = 5) of cases. Calculus was not noted in two patients. Gingivitis index was graded as 2 in 41% (n = 11) and as 3 in 67% (n = 18). Gingival enlargement was assessed as localized in six cats, semi-generalized in two and generalized in six. Plaque index (P >0.99), calculus index (P >0.99), severity of gingivitis (P = 0.07) and the presence of gingival enlargement (P = 0.21) were not statistically significantly associated with periodontal disease stage. Plaque index (P >0.99), calculus index (P >0.99), severity of gingivitis (P = 0.59) and the presence of gingival enlargement (P = 0.37) were not statistically significantly associated with disease progression.
Radiographic findings
The clinical and radiographic findings for the initial evaluation of all patients are summarized in Figure 2. In general, 89% (n = 24) of cats exhibited radiographic signs of periodontitis. These findings included stage 4 periodontal disease in 48% (n = 13) of cats. This was further classified as localized (n = 8), semi-generalized (n = 2) and generalized (n = 3). Stage 3 periodontal disease was described in 30% (n = 8) of cats. This was further classified as localized (n = 5) and generalized (n = 3). Three cats had stage 2 periodontal disease, which was localized in two and semi-generalized in one. Two cats had evidence of stage 1 periodontal disease. Missing teeth were noted in 67% (n = 18) of cases.

Summary of the clinical and radiographic findings of the initial anesthetized evaluation for all patients. (a) Overall radiographic features of the population. (b) Radiographic features of the population by age, with blue representing cats 12 months old or younger, and orange representing cats older than 12 months. Furcation involvement was more frequently evident in cats older than 12 months. Within the population of cats affected by moderate-to-severe periodontitis, those that were older than 12 months had generalized disease. GE = gingival enlargement; PD 1–4 = periodontal disease stage 1–4; HBL = horizontal bone loss; VBL = vertical bone loss; FI = furcation involvement; TR = tooth resorption
Horizontal bone loss was the most frequent radiographic pattern, with 59% (n = 16) of cats affected. Nine cats had both horizontal and vertical bone loss; however, in six of these cats, horizontal predominated over vertical bone loss. Furcation involvement was noted in 41% (n = 11) of cats, and buccal bone expansion was noted in six. Type 1 inflammatory resorption was noted in six patients, and type 2 were noted in two patients. Root remnants were noted in five cats due to incomplete extraction, fracture or resorption.
Evidence of missing teeth and horizontal bone loss (P = 0.60), vertical bone loss (P >0.99), type of bone loss combined (P = 0.24), periodontal disease stage (P >0.99), evidence of tooth resorption (P >0.99) or plaque (P >0.99) or calculus index (P >0.99) were not statistically significant. Evidence of missing teeth and horizontal bone loss were also not statistically significantly correlated with disease progression (P value = 0.60). Periodontal disease severity was also not statistically significantly associated with disease progression (P = 1.0)
Histopathologic findings
Fourteen patients underwent histopathologic evaluation of the gingival tissues. Different degrees of severity of histopathologic changes were noted (Figure 3). Inflammation varied but was mild in only two cases, moderate in four cases and severe in eight cases. Neutrophilic and lymphoplasmacytic infiltration was seen in eight cases. Neutrophilic infiltration alone was seen in one case, and only one case showed pyogranulomatous inflammation. The case with pyogranulomatous inflammation had a concurrent caudal, traumatic malocclusion. Ulceration or erosion was noted in 11 cases. Gingival enlargement was noted in 14 cases. In four cases the enlargement was due to epithelial hyperplasia, while in two cases it was due to subepithelial stromal expansion by inflammatory infiltrate. In three cases the enlargement was due to epithelial hyperplasia and a stromal inflammatory infiltration. The type of inflammation was not associated with evidence of buccal bone expansion (lymphoplasmacytic P >0.99; neutrophilic P >0.99), gingival enlargement (lymphoplasmacytic P >0.99; neutrophilic P = 0.33) or missing teeth (lymphoplasmacytic P = 0.51; neutrophilic P >0.99). Lymphoplasmacytic (P >0.99) and neutrophilic inflammation (P >0.99) were not correlated with progression of disease. Bacterial colonization of the tissues was noted in four cases; all had concurrent ulceration.

Hematoxylin and eosin-stained sections of the gingiva of affected patients. (a) Cat #15 (×10) shows surface ulceration with stromal expansion owing to neutrophilic inflammation and granulation tissue formation. (b) The same cat as shown in (a) (cat #15; ×20), showing neutrophilic infiltration of granulation tissue at higher magnification. (c) Cat #21 (×10) showing epithelial hyperplasia and ulceration. (d) The same cat as shown in (c) (cat #21; ×20) showing epithelial hyperplasia and ulceration with neutrophilic and lymphoplasmacytic infiltration
Treatment, response and follow-up
Treatment consisted of periodontal treatment (ie, sub- and supragingival scaling), with selective teeth extractions, in 22 patients. Two cats had full-mouth tooth extractions. Medical management consisted of a variety of medications as standalone agents or in combination with pain management. Four patients were prescribed systemic oral antibiotic therapy alone. An oral antimicrobial rinse alone was prescribed in five patients. An NSAID was prescribed in five patients. Combination therapy included a topical antimicrobial and NSAID in four patients; systemic oral antibiotic and topical antimicrobial and NSAID in one patient, systemic oral antibiotic and NSAID in two patients and systemic oral antibiotic and topical antimicrobial treatment in one patient. The course of treatment followed standard recommended doses for these medications, and extended between 3 days and 2 weeks after surgery in all treated patients. Though patients were reported to receive systemic steroid therapy prior to presentation, none were currently receiving these at diagnosis and these treatments were not continued in any of the patients after surgery.
The mean duration of follow-up was 10 months (range 0.5–132). Disease progression showed no statistically significant association with gingivitis index (P = 0.59), gingival enlargement (P = 0.37) or periodontal disease severity (P = 1.00), but it was statistically significantly associated with lack of response to treatment at the 2-week recheck (P = 0.004). For those that failed to show a response to treatment at the 2-week re-evaluation, anesthetized evaluation at no later than 6 months after the initial procedure was recommended. Notably, two cats (aged 13 and 12 months, respectively) did not respond to treatment and developed an FCGS-like lesion (Figure 4). Extension of inflammation of the caudal oral mucosa was noted 6 weeks after the initial procedure in one case (case #15). This cat underwent general anesthesia and histopathologic evaluation approximately 3 months later, the caudal oral mucosa was sampled and the histopathologic evaluation noted lymphoplasmacytic inflammation. The other patient (case #1) developed worsening inflammation of the caudal oral cavity, in addition to the persisting mild-to-moderate gingivitis, 4 weeks after the initial procedure. Clinically, this patient was doing well at home and the owner elected not to pursue an anesthetized procedure. Seven additional non-responsive patients underwent anesthetized re-evaluation as soon as 2 months after initial anesthetized treatment; of these, six cats were diagnosed with AP and one cat developed an oral mass that was diagnosed as caudal mucogingival lesions secondary to traumatic dental occlusion. Five cats showed improvement after treatment, including two cats that underwent full-mouth extractions, and three cats had resolution of clinical and gross evidence of disease.
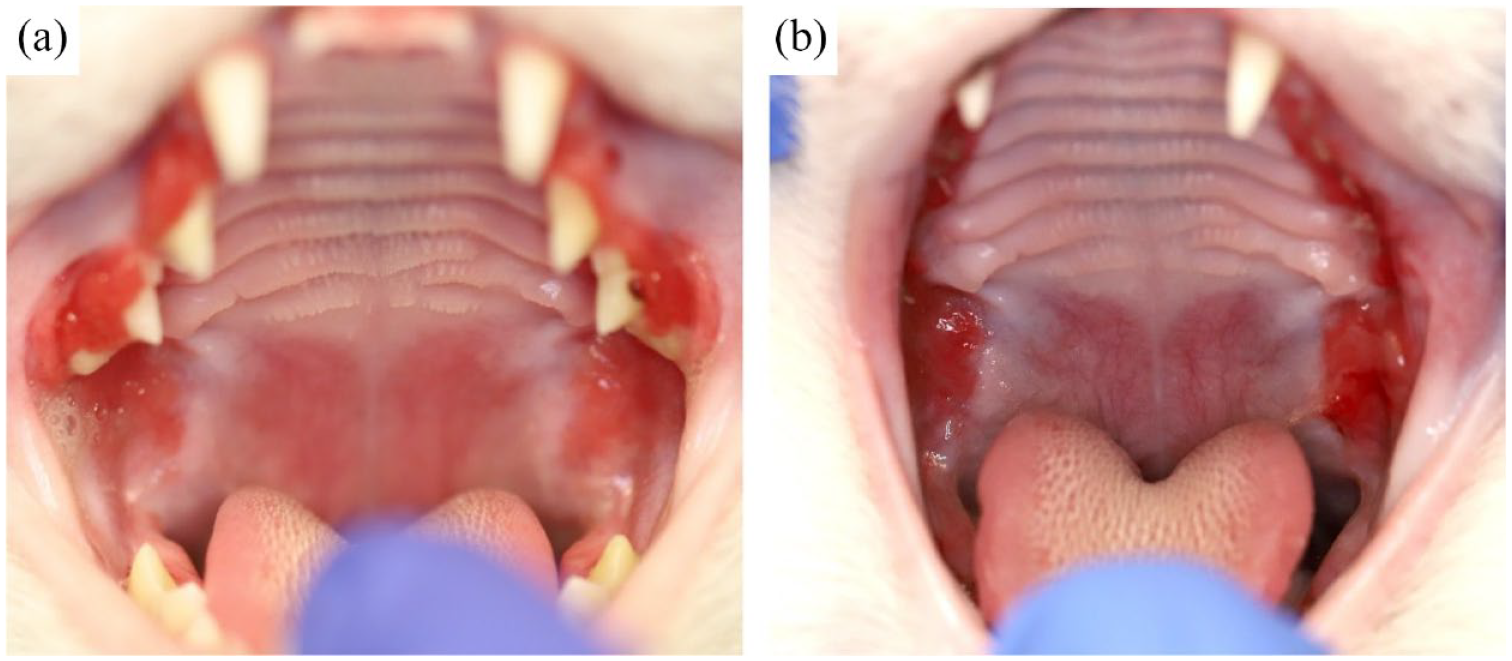
(a) Photograph of a 7-month-old female spayed domestic shorthair cat (case #15) exhibiting severe generalized gingivitis and semi-generalized gingival enlargement with mild inflammation of the caudal buccal mucosa, lateral to the palatoglossal folds, reminiscent of the inflammation seen in a patient with feline chronic gingivostomatitis. (b) The same cat (case #15) 6 months after the initial diagnosis of early-onset gingivitis and 1 month after extraction of all premolar-molar teeth. The photograph shows progression of the extent and severity of the inflammation to the caudal oral cavity, confluent with the gingivitis involving the maxillary teeth. Histopathologic evaluation revealed lymphoplasmacytic inflammation. The patient was only receiving pain medication (gabapentin 9 mg/kg PO q12h) at the time of biopsy
Discussion
This retrospective study characterized the clinical, radiographic and histologic features of EOG and periodontitis in cats. We describe a few important and clinically relevant features. First, anesthetized oral examination identified 82% of irreversible periodontal disease that was not always recognized on an awake oral examination. Second, moderate-to-severe periodontal disease and missing teeth are features noted in cats with EOG. Third, the most predominant histopathologic feature was moderate-to-severe, erosive-to-ulcerative, neutrophilic and lymphoplasmacytic inflammation. Finally, lack of improvement of clinical signs and evidence of gingivitis at the healing recheck was significantly associated with disease progression. Progression from EOG to gingivostomatitis was confirmed, as well as progression to AP.
Anesthetized evaluation of cats with EOG was essential to increase the diagnostic yield of periodontitis. Moderate-to-severe periodontitis and missing teeth were prominent features in our study population. Only one-third of patients in this study had evidence of periodontal disease on awake examination. This is striking, because nearly all patients had moderate-to-severe periodontitis diagnosed radiographically under anesthesia. This contrasting difference in diagnostic yield between awake and anesthetized evaluation, combined with the fact that approximately half of cases were described by owners as clinically asymptomatic, underscores the importance of performing an anesthetized evaluation, including full-mouth intraoral dental radiographs and dental charting on oral evaluations in young cats with significant gingivitis. Clients should also be informed of the possible need for extraction(s) of severely diseased teeth due to the high frequency of moderate-to-severe periodontitis encountered in these patients.
Biopsy of gingival tissues and submission for histopathologic evaluation is a good practice and should be prospectively performed to characterize this disease further. Though a larger sample size may be needed to discern and identify positive and negative predictive factors in the histopathology of these patients, the current data underscore the importance of microscopic characterization of these lesions. It is still essential in cases with gingivitis and gingival enlargement to differentiate hyperplasia from other pathologic processes that would require further treatment, such as odontogenic tumors, infectious diseases (ie, fungal) or malignancies.18–20 Histopathologic patterns of inflammatory cellular infiltration in some juvenile gingivitis cases in this cohort had a striking resemblance to the histomorphology of FCGS. Specifically, the most prominent was the extensive lymphoplasmacytic infiltration of the subepithelial stroma with or without a neutrophilic component. However, more detailed immunohistologic and molecular work-ups are needed to comment on the degree of resemblance or progression between FCGS and EOG and/or AP. An additional value for immunohistochemical work-up combined with systemic immunophenotyping and metagenomic analyses for identifying viral association is underscored in the recent discoveries made for FCGS, where the FCV component was confirmed.21,22
Concurrent ulceration or erosion and neutrophilic infiltration were noted in about half of cases in this cohort. These cases also had frequent evidence of bacterial biofilm associated with the oral biopsies. The significance of the association between oral ulceration and bacterial colonization remains to be determined. The bacterial colonization may be secondary to the disruption of the normal epithelial barrier defense and the use of necrotic debris by the bacteria as a nidus for infection. However, the possibility of bacteria and their products inducing ulceration cannot be ruled out. Since neutrophilic infiltration is expected in any case of epithelial ulceration/erosion, it is unknown at this time how bacterial types contribute to this process and if neutrophilic function is adequate. Interestingly, in humans, defective neutrophil function has been shown to be inherited along with the clinical occurrence of AP. 23 More studies targeting microbiome composition, identification of bacterial virulence factors and neutrophilic function are necessary to answer these questions.
In addition to radiographic evidence of moderate-to-severe periodontitis in a high percentage of patients, missing teeth was a feature commonly seen in this population. The prevalence of missing teeth without radiographic evidence of retained root tips has been reported to be approximately 37% in a study evaluating the periodontal health of a colony of cats. The same study reported a prevalence of AP of 13%. 1 This is lower than the frequency of missing teeth noted in this study, which more closely resembles the dental radiographic findings in cats with feline chronic gingivostomatitis. 24 In that study, semi-generalized or generalized and moderate or severe periodontitis was seen in 77% and 92% of affected cases, respectively. Additionally, cats with FCGS had more than twice the number of radiographically absent teeth than did cats without FCGS, consistent with the observed increased severity of periodontitis. 24 Evidence of missing teeth was not correlated with the presence of periodontal disease or tooth resorption in this study, indicating either the effect of a small sample size or the possibility of teeth that are congenitally missing.
Although severe gingivitis was most seen in the studied population, only about half of these cases had evidence of gingival enlargement, which varied from local to generalized. Consequently, and considering the radiographic findings of this study, the historical term ‘feline juvenile hyperplastic gingivitis’ does not appear to reflect the general manifestation of the disease, and the term ‘aggressive periodontitis’ in human periodontology maybe more accurate.1,3,25 AP in humans is defined as a destructive disease characterized by (1) early age of onset; (2) involvement of multiple teeth with a distinctive pattern of periodontal tissue loss; (3) a high rate of disease progression; and (4) the absence of systemic diseases. 25 Additionally, in humans with AP, periodontal tissue loss may commence before puberty or in the circumpubertal period. 25 In this study, the median age of cats at presentation was 17 months; however, the average duration of clinical signs prior to diagnosis was 9 months. Hence, it is important to emphasize that the term juvenile feline or adolescent in human literature may reflect the time of disease initiation, even though these individuals may be older during the progressed stage of the disease.
Patients in this study were followed-up for up to 10 months, on average, and – to our knowledge – this is the first time the progression of EOG to AP (22%) and a FCGS-like lesions (7%) has been reported, though concurrent disease cannot be completely ruled out. 24 The age of development of FCGS-like lesions for both of these patients was younger than the typical age of diagnosis of this disease, as reported in the literature. 26 However, in a study evaluating the radiographic findings of cats with gingivostomatitis, the included cats were a median of 6 years old (range 1–16), with 36% (n = 36/101) of affected cats aged 1–4 years. 24 Confirmation of FCGS would necessitate a more in-depth characterization of these cases consistent with the current knowledge of this disease (ie, FIV, FeLV and FCV status, and immunohistochemistry),22,27 as well as close follow-up to further characterize the distribution of the pattern of inflammation as the disease progresses. The latter highlights the relevance of early diagnosis, treatment and the need for close monitoring of feline patients affected by EOG. These findings also highlight the need for anesthetized radiographic and histopathologic re-evaluation of cases with a lack of response to initial periodontal and extraction treatment, to rule out other causes of stomatitis such as uremia, caudal chewing lesions (ie, pyogenic granulomas), contact stomatitis, immune-mediated disorders, eosinophilic infiltration and diffuse neoplastic diseases. When these conditions are not distinguished, there is a high risk of treatment failure.
The limitations of this study include its retrospective nature and rather small group size, prohibiting definitive conclusions being drawn on breed relevance and environmental factors, as well as characterization of positive or negative predictive factors in the anesthetized, hematologic, serologic and histopathologic findings. However, this study has underscored some unique features of juvenile gingivitis/periodontitis that justify further investigation. Larger studies, including populations from primary care practices and more crowded housing environments such as shelters, should be performed to make the data more representative. FeLV, FIV and FCV status were not known for all cases, and metagenomic analysis were not available in the present cases.
It can be clinically difficult to separate EOG from AP, and we suspect these present concomitantly in some cases. Considering the mean age of presentation in this study, it is likely that some patients presented with EOG and AP. This can certainly be true considering that owners will likely seek veterinary attention when obvious clinical signs are present, and this typically occurs with the development of periodontitis. In humans, differentiation between chronic and aggressive forms of periodontitis is based on rate of progression with slow-progressing forms of periodontal inflammation characterized as chronic periodontitis and the rarer form of inflammation characterized by a rapid evolution, mainly seen in young adults, considered as AP. More specifically, grade C periodontitis is diagnosed when ⩾2 mm of bone loss is noted over 5 years. Additionally, these patients are characterized by specific clinical patterns suggestive of periods of rapid progression and/or early onset of disease. 6 Proportionally, this would indicate an increase in bone loss of approximately 0.2 mm within 6 months in cats. Consequently, in this study, patients were diagnosed with AP when progressive bone loss of ⩾0.2 mm was noted at the anesthetized follow-up evaluation.
Hyperproteinemia, as seen in some of the cats presented here, has been reported to occur in 60% of cats with FCGS, and this is due to a hyperglobulinemia. 28 This has been proposed to occur as a result of an upregulation of interleukin 6, which has been reported to occur both systemically and locally in these patients.28,29 Additionally, some of the cats reported here were hyperglycemic. Hyperglycemia in these cats could be stress induced; however, these patients were not further evaluated to rule out the presence of diabetes. Larger, prospective studies of EOG in cats could aim to further characterize these serologic changes, as well as the local and systemic cytokine profiles of affected patients.
Though retroviral status was known for a number of the cases reported here, testing was not repeated and thus confirmation of positive status was not performed. Some of these patients also underwent FHV and FCV testing; however, valuable conclusions cannot be drawn with such a small number of the population tested. The possibility of associations between feline oral mucosal inflammation and multiple infectious agents, including FCV, FeLV, FIV, FHV type 1 and Bartonella species, has been investigated.22,30–32 Unbiased metagenomic next-generation sequencing and transcriptomic analysis identified a specific and strong positive association between FCV and FCGS in cats, and the findings provided additional evidence to support the hypothesis that FCV is involved in the development and/or maintenance of FCGS. 22 Similar studies evaluating the role of infectious diseases and genetic factors in AP in cats would pave the way to a better understanding of this disease’s etiology. Another important future step would be more in-depth immunological studies characterizing and comparing immune cell phenotypes between cases that resolve vs cases that progress. These studies could lead to discoveries of translational value for multiple species.
Conclusion
This retrospective study highlights the importance of performing oral evaluations in cats at 6 months of age or earlier. Obtaining dental radiographs and biopsies is essential. EOG is an indication for professional periodontal care and long-term follow-up owing to the potential of this disease to progress to more severe, deep and diffuse forms.
Footnotes
Acknowledgements
The authors thank Andrew Blandino from the UC Davis Statistics Department, and Hugues Beaufrere DVM, PhD, DACZM, DABVP, DECZM from the UC Davis Department of Veterinary Medicine and Epidemiology for their assistance in the statistical analysis for this work.
Conflict of interest
The authors declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding sources
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognized high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers) for all procedure(s) undertaken (prospective or retrospective studies). For any animals or people individually identifiable within this publication, informed consent (verbal or written) for their use in the publication was obtained from the people involved.
