Abstract
Practical relevance:
Feline chronic gingivostomatitis (FCGS) is a debilitating disease for cats and a challenge for veterinarians and cat caregivers alike. Recent literature indicates that the disease is immune-mediated in nature and likely associated with a chronic viral infection in patients with higher alpha diversity of their subgingival microbiome. The immune-mediated nature of FCGS includes both local as well as systemic effects, and the transcriptomic analysis of affected patients supports these findings.
Treatment options:
Localized therapy in the form of surgical extraction of all, or nearly all, teeth continues to be the mainstay of treatment. For cats that do not respond to surgical management, medical management, in the form of immunosuppressive or immunomodulatory therapy, remains an option. Analgesia is of fundamental importance. Immunomodulation utilizing mesenchymal stromal cell therapy provides an alternative treatment avenue for refractory patients and likely targets the chronic viral infection present in this disease. The potential for treatment stratification and use of novel systemic treatment options may be revealed as the molecular pathways involved in this disease are better described.
Aims:
This review outlines current and emerging concepts linking available science pertaining to FCGS and clinical management of the disease.
Evidence base:
The article draws on the best evidence base at this juncture and is also driven by the authors’ collective experience of working on the disease for over a decade.
Introduction
Feline gingivostomatitis is a chronic oral mucosal disease that affects up to 26% of domestic cats.1-4 Affected cats typically develop extensive inflammatory lesions throughout the oral cavity, including in the area lateral to the palatoglossal folds. 5 There appear to be two clinical phenotypes of the disease - ulcerative and proliferative - though some patients will display both, as shown in Figure 1. Lesions are typically associated with moderate to severe oral pain, decreased or absent appetite, poor grooming and reduced socialization.5-7 Cats affected with feline chronic gingivostomatitis (FCGS) may be euthanized due to an insufficient response to therapy or a lack of resources for treatment.
The current body of literature supports that FCGS is immune-mediated in nature, 8 as outlined in this review which includes a comparison of the local host response in FCGS and in the healthy oral mucosa (see box ‘The oral mucosa in health’). Furthermore, it is becoming unquestionable that the immune response is somehow related to feline calicivirus (FCV) infection. Targeted studies on the oral mucosa have evaluated the qualitative histomorphological structure, as well as characterizing and quantifying the immune cell types and host response in health and FCGS. In addition, untargeted, multiomic approaches have taken charge over recent years in describing the microbial landscape of FCGS,9,10 and the genetic 11 and metabolic 12 pathways involved in this disease.
In compiling this review, the authors’ aim has been to provide a cohesive summary of currently available scientific evidence that supports the immune-mediated nature of this disease. This is discussed in relation to etiologies and clinical management. The authors also highlight limitations within the current evidence base that inform recommendations for further research. Furthermore, they illuminate various aspects of surgical and clinical management, and make the case that a ‘personalized medicine’ approach (ie, tailored to the individual patient using different types of biomarkers) is appropriate for managing cats that are non-responsive to dental extraction therapy.


The clinical presentation of feline chronic gingivostomatitis (FCGS) can vary slightly, though the hallmark is inflammation that not only affects the gingiva but also extends to the alveolar and buccal mucosa. (a,b) A patient with refractory ulcerative FCGS also affecting the sublingual tissues. (c) A case showing both ulceration and proliferation. Note the proliferation on the right side affecting the buccal and sublingual mucosa. (d) A case of FCGS where spontaneous bleeding from the areas of ulceration was noted. (e) Concurrent inflammation of the gingiva and alveolar mucosa is typical of patients with FCGS and in this case surrounds the left maxillary fourth premolar tooth. (f) In this cat with FCGS there is mild inflammation on the left side of the buccal mucosa, while the right side has moderate inflammation
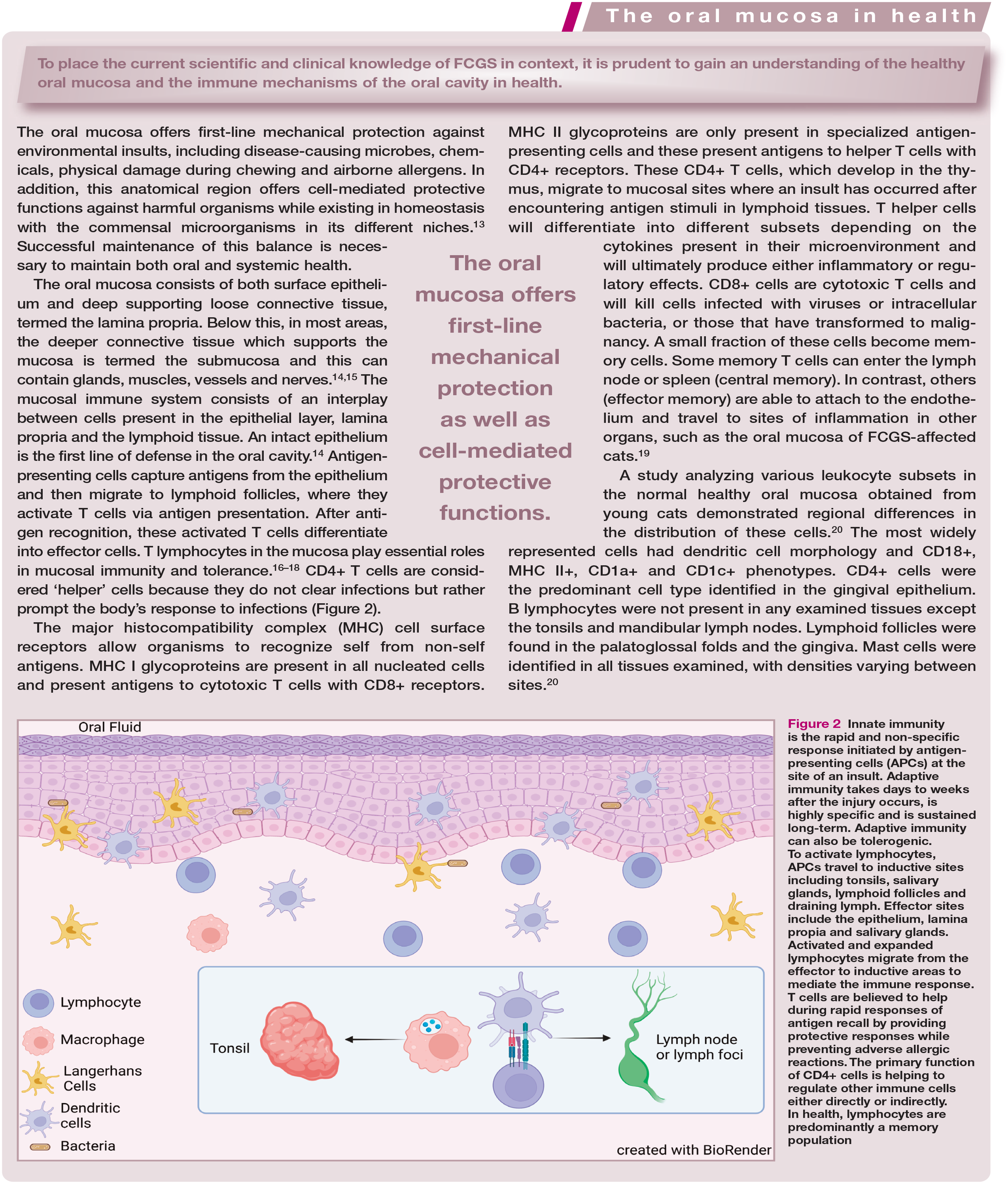

Innate immunity is the rapid and non-specific response initiated by antigen-presenting cells (APCs) at the site of an insult. Adaptive immunity takes days to weeks after the injury occurs, is highly specific and is sustained long-term. Adaptive immunity can also be tolerogenic. To activate lymphocytes, APCs travel to inductive sites including tonsils, salivary glands, lymphoid follicles and draining lymph. Effector sites include the epithelium, lamina propia and salivary glands. Activated and expanded lymphocytes migrate from the effector to inductive areas to mediate the immune response. T cells are believed to help during rapid responses of antigen recall by providing protective responses while preventing adverse allergic reactions. The primary function of CD4+ cells is helping to regulate other immune cells either directly or indirectly. In health, lymphocytes are predominantly a memory population
Local host response in FCGS
The typical appearance of the mucosal epithelium of FCGS-affected cats is hyperplastic, with prominent rete pegs that extend deep into the submucosa (Figure 3). The submucosal capillaries are typically congested and lined with plump endothelial cells. The mucosa can also show ulceration; in those cases, neutrophils are primarily observed dispersed or aggregated within the lamina propria/ submucosa underlying areas of epithelial degeneration.8,21 Increased numbers of mast cells have been documented in gingival biopsies from cats with FCGS. 22 Though their role in this disease is not entirely characterized, mast cells release a range of pro-inflammatory and immunomodulatory mediators, and it is possible that, even in small numbers, these cells could be involved in the initiation, propagation and perpetuation of local inflammatory and immunologic responses in FCGS. 21
The non-inflamed feline oral mucosa is an active immunologic site, with a cell population that is predominantly biased toward a type 1 cytokine expression profile. Conversely, the mucosa of patients with FCGS shows a mixed type 1 and type 2 profile, indicating the development of a solid cellular and humoral response. 23 The mucosa of cats affected by FCGS is characterized by a heavy infiltration of lymphocytes and plasma cells, with occasional to abundant Mott cells. T cells are present in the superficial mucosa and submucosa (CD3), whereas B cells and Mott cells appear to be restricted to the submucosa (CD20). 8 Most of the plasma cells are of the IgG isotype, with fewer IgA+ and IgM+ plasma cells described. 21
Immunolabeling of CD4+ and CD8+ T cells in affected mucosa has revealed an approximately equal ratio of these cells in the lamina propria in edentulous patients, 20 though cats with teeth remaining show a predominance of CD8+ cells. 21 These inflammatory cells are frequently distributed in a band-like pattern that obscures the border between epithelium and submucosa. 8 The number of cells labeled for CD3+, CD4+, CD8+, CD79a+, IgG+, IgM+, IgA+ or L1+ (neutrophils) within the lamina propria/submucosa is significantly increased in FCGS as compared with the healthy oral mucosa; so too is the number of mast cells. Furthermore, correlations between the severity of inflammation and the number of plasma cells (CD79a+ cells), neutrophils (L1+ cells), helper T cells (CD3+) and MHC II glycoprotein levels have been noted. 21 Mast cell densities were found to be significantly increased in gingival tissues adjacent to teeth affected by FCGS, although not significantly different from those observed in cats with periodontal disease and feline resorptive lesions (Figure 4). 22

Histologic section from the buccal mucosa of a patient with FCGS. Note the hyperplastic epithelium and prominent rete pegs (finger-like structures) that extend deep into the submucosa. 20× magnification; bar = 20 urn
Cytokine expression has been correlated with disease severity in FCGS-affected cats. This mirrors what is observed in human periodontal disease, which is not surprising considering that the majority of cats affected by FCGS have concurrent periodontitis.23,24 Although the immunologic host response at the level of the gingiva of patients with FCGS has not been fully characterized, dental radiography demonstrates that FCGS is associated with widespread and severe periodontitis, with a high prevalence of external inflammatory root resorption and retained roots, suggesting a highly inflammatory, destructive process (Figure 5). 24 This response could also reflect the collection of cytokines that are expressed during chronic inflammation of the oral mucosa arising from various etiologies, as these seem to have little to no variation. 25


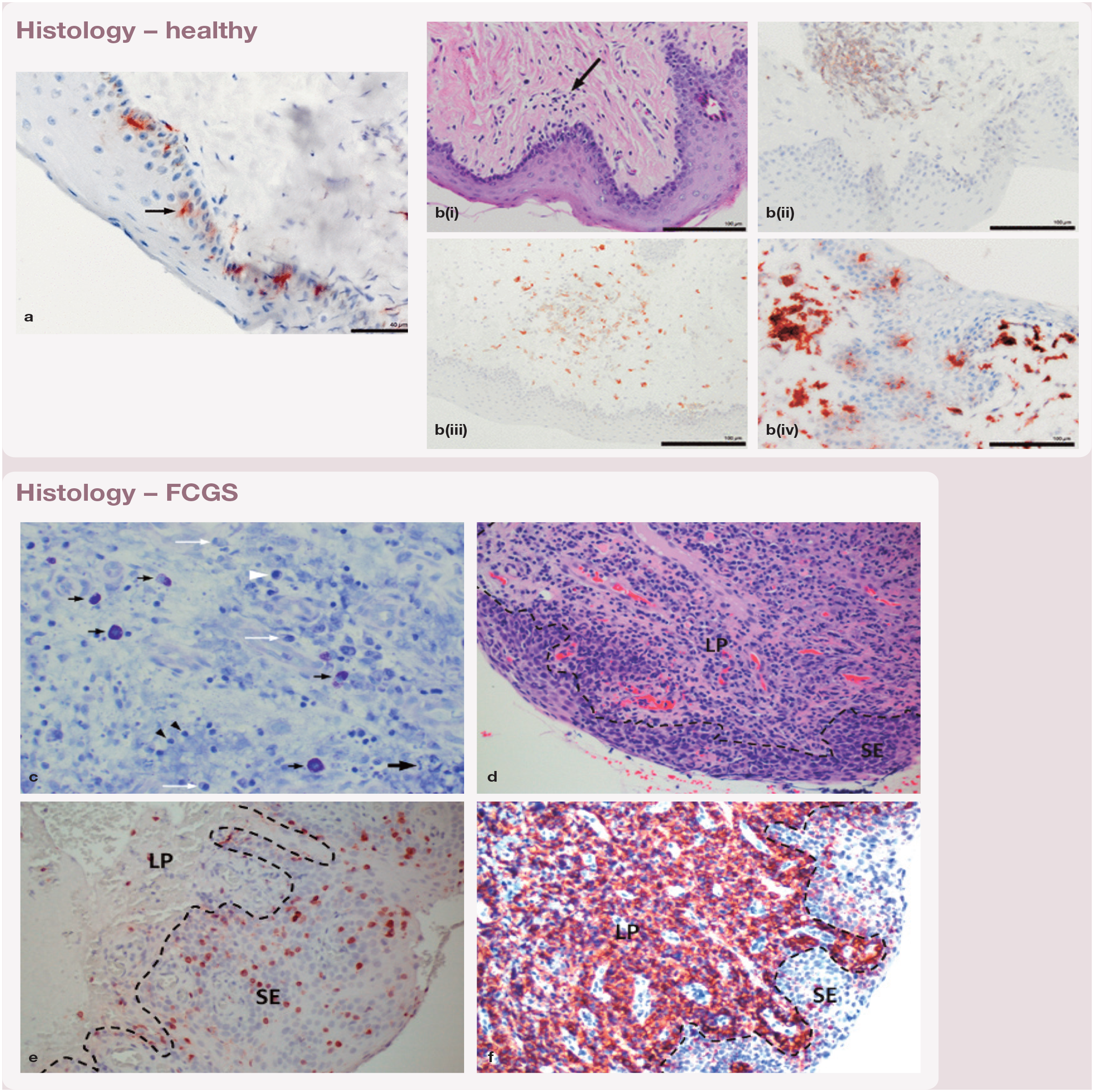
Histologic sections from the buccal mucosa and palatoglossal folds of a healthy cat (a,bi-iv), and from the gingiva and affected buccal mucosa of a patient with FCGS (c-f). LP = lamina propia; SE = subepithelium.


Intraoral radiographs of a 5-year-old domestic shorthair cat with clinically and histopathologically confirmed FCGS.
Systemic manifestations
As we deepen our understanding, it has become apparent that FCGS is not only a local disease but one with systemic ramifications.8,26-29 Affected cats have high circulating CD8+ effector memory cell levels with a concurrent decrease in central memory cells and evidence of circulating activated CD8+ T cells (CD25+, CD62L-). 8 Memory CD8+ T cells are the principal component of immunity against intracellular pathogens such as viruses. This means there is an unresolved inflammation in which CD8+ T cells are activated more than once and remain activated. The predominance of CD8+ T cells in lesions and increased circulatory effector memory cells in FCGS confirm that an intracellular organism - most likely one involved in a viral infection - causes inflammation.
Most affected cats also exhibit elevated serum and salivary immunoglobulin levels. 28 Salivary immunoglobulins are likely produced by the mucosal plasma cells and reach the oral cavity via the gingival crevicular fluid or by leaking across the mucosa. The hypergammaglobulinemia, a polyclonal gammopathy seen in these cases, could be secondary to upregulation of IL-6 in FCGS. 23 Additionally, it is suspected that cats with FCGS are more likely to have a systemic inflammatory response. 26 Other evidence of systemic inflammation in cats with FCGS includes increased expression of proinflammatory serum cytokines, such as interferon-gamma (IFN-γ), tumor necrosis factor-alpha (TNF-α), interleukin-1 beta (IL-1β) and IL-6, as well as blood neutrophil counts (Figure 6). 29
Though originally thought to be a local disease impacting only the oral cavity, in a controlled study evaluating the prevalence of esophagitis in cats with FCGS, evidence of esophagitis was found via esophagoscopy in 98% of affected cats, compared with 0% of control cats. 30 Affected patients showed microscopic evidence of inflammation and metaplasia in otherwise grossly normal-appearing tissues and in the absence of clinical signs consistent with gastrointestinal disease. Endoscopic re-examination of cats treated for FCGS that no longer showed clinical signs of that disease also demonstrated macroscopic healing of esophagitis. The investigators of the study proposed that esophagitis in these cases is due to a concurrent dysbiosis in the esophagus promoting the production of certain proinflammatory cytokines. 30


Evaluation of the systemic immune response of FCGS patients revealed hyperglobulinemia in 60% of patients, elevated circulating levels of IFN-γ,TNF-α and IL-lβ, neutrophilia in 30-40% of patients, as well as CD8+ effector memory cells. 29 These findings mirror those of studies evaluating the local immune response
Current knowledge of the etiology of FCGS
Several conditions and infectious agents have been implicated as the inciting factor of the immune response seen in FCGS,31–33 without proof of causation. An association between the clinical severity of FCGS and the presence of FCV (toll-like receptor [TLR] 7) and Tannerella forsythia (TLR2) has also been noted, sparking an interest in these as possible etiologic agents.34,35 Unbiased metagenomic and tran-scriptomic analyses of FCGS patients revealed that the only microbe strongly associated with FCGS was FCV, which was detected in 21/23 FCGS-affected cats and not in any of the control cats. 10 This study also showed that coinfection of FCGS-affected cats with FCV and puma feline foamy virus (PFFV) might adversely affect the response to treatment. 10 This finding was supported by an approximately 40-fold higher expression of guanylate binding protein-1 (GBP-1) in FCGS patients compared with controls, a gene known to have antiviral effects. 10
Yet, FCV oral load in cats with FCGS, as evaluated by real-time reverse transcription polymerase chain reaction (RT-PCR) testing on oropharyngeal swabs, was not correlated with the severity of oral lesions or treatment outcome. 7 Furthermore, studies attempting to prove Koch’s postulate by inoculating healthy cats with FCV to recapitulate FCGS have failed to prove causation.35,36 Although acute stomatitis occurred, the chronic carrier state did not. 36 It is important to note that one of these studies 35 involved 16-week-old specific pathogen-free cats. The oral cavity of kittens may not mimic the clinical scenario necessary for the disease to occur, in terms of genetic basis, oral microbiome and microenvironment. Additionally, three studies have documented decreased viral load or resolution of FCV along with the resolution of clinical signs and gross lesions in FCGS.10,37,38 In short, despite inconsistencies, the literature largely supports a role for FCV, at least in modulating the severity of disease.
Historically, feline leukemia virus (FeLV) and feline immunodeficiency virus (FIV) have been inconclusively linked to FCGS. 39 More compelling evidence has been provided in a study comparing the clinical outcome of cats with FCGS, with and without FIV and FeLV infection, which showed different phenotypic manifestations as well as an effect on response to full mouth extractions (FMEs) or partial mouth extractions (PMEs) and adjuvant medical support. 40 Phenotypically, FeLV-positive cats had significantly less proliferative stomatitis and fewer resorptive lesions, though they tended to have more lingual ulcers. This study concluded that FeLV-positive cats had a 7.5-times greater chance of having no improvement after dental extractions than cats that did not carry retroviral disease. 40
The subgingival microbiome has also been characterized in cats with FCGS. 9 A higher alpha diversity was found in these patients, together with a higher relative abundance of Peptostreptococcus species as compared with healthy cats (P = 0.0052) and cats with periodontitis (P = 0.0127). 9 The implications of these findings are unclear at this time.
Finally, a study evaluating whether the number of cohabiting cats represents a risk factor for FCGS showed that cats in shared households had significantly increased odds of FCGS (seven-fold) compared with those from single-cat households. 41 Each additional cat in the household increased the odds of FCGS by more than 70%. 41

Clinical management of FCGS
The clinical management goals for FCGS are to decrease or eliminate antigenic stimulation and modulate the abnormal immune response. Given that nearly all affected cats will exhibit moderate to severe periodontitis and tooth resorption, surgical management in the form of dental extractions is the starting point. Periodontitis is a substantial inflammatory burden on the oral mucosa and the immune system. Therefore, extraction of teeth will effectively reduce a portion of the chronic inflammatory burden, allowing for a subpopulation of patients to achieve a significant improvement or even a cure.
However, it is unclear at this juncture how removal of teeth is associated with reduction or elimination of FCV from the oral mucosa. 10 It is plausible that tooth extraction removes the characteristic subgingival microbiome that may play a role in this disease, thereby reducing the inflammatory burden and freeing the immune system to focus on concurrent chronic viral infections. Alternatively, by reducing inflammation, the environment may be less favorable for these viruses to thrive.
Preoperative evaluation
FCGS-affected cats should be evaluated for risks associated with a relatively long anesthetic procedure when either full-mouth or partial tooth extractions are being planned (complete blood count, serum biochemistry panel, etc). In addition, viral disease testing to assess FeLV, FIV, FCV and PFFV status should be performed due to the potential prognostic significance.10,40 Repeat testing 30 and 60 days after possible FeLV and FIV exposure, respectively, should be considered to confirm a negative status. 42 Combined virus isolation and RT-PCR techniques using samples obtained from the conjunctiva or oropharynx is recommended to increase detection rates for FCV. 43 To the authors’ knowledge, commercial PFFV testing is not available at this time.
A standardized activity index has been developed to monitor clinical manifestations associated with inflammation in the oral cavity of cats with FCGS. The Stomatitis Disease Activity Index (SDAI), initially devised by Dr Jamie Anderson and used in several previous studies,29,44-46 has been a valuable tool to assist the diagnostic process and status monitoring. The index considers both the client’s assessment of quality of life of the affected patient at home and the gross evaluation of the oral cavity by the veterinary practitioner. However, at times, disparities arise between the owner and clinical assessment of patient disease status. Consequently, the authors of this review have created a modified score that considers objective criteria (see Appendix 1 in the supplementary material).
The SDAI should be completed by the veterinarian at the patient’s first visit and used at each consecutive evaluation to monitor progress. The questions in the owner assessment portion can be incorporated into the veterinarian’s usual history-taking survey. The veterinarian’s assessment is performed during the awake oral examination, although light sedation may be necessary in uncooperative patients, or the clinician may opt to wait until the patient undergoes general anesthesia to evaluate the oral cavity. The objective assessment is completed once all the diagnostic work-up for that patient has been returned. Although it has been histopathologically validated,4,7,47 the SDAI has not undergone intra-and inter-reader variability studies and so, whenever possible, it should be the same veterinarian who completes this form for an individual patient. Moreover, future studies should aim to assess the contribution of the different subsections of the SDAI (ie, veterinarian assessment, objective assessment and owner assessment) in obtaining a score of prognostic significance for the patient.
A summary of the preoperative diagnostic approach for these patients is available in Figure 7. Confirmation of the diagnosis is achieved via incisional biopsy performed under anesthesia. The information provided by histopathology may also have prognostic value or identify disease subtypes, as is the case for infiltrative gastrointestinal disease in dogs and cats.48-52 The authors are currently studying the effects of eosinophils in the outcome of FCGS patients.
Surgical management
The success rate for dental extractions in cats affected by FCGS was first reported in 1997. 53 Historically, this treatment was instituted based on the assumption that FCGS patients have a dysfunction of a protective mechanism or a detrimental immunologic reaction induced by bacterial periodontopathogens or viruses, and tooth extractions were thought to aid in reducing the chronic antigenic stimulation these patients were facing. 53 While this may partly be the case given the characteristic subgingival microbiome of these patients, 9 it has also been recognized that extractions are an effective therapy for treating moderate to severe periodontitis, which is present in up to 93% of affected cats, and tooth resorption and retained roots, which are seen in up to 66% of affected cats.6,24,53


Schematic diagram summarizing the diagnostic and treatment recommendations for FCGS patients in the preoperative, intraoperative and postoperative periods. As discussed in the text, immunosuppressive therapy refers to the use of glucocorticoids to affect the immune response more globally (compared with immunomodulation, which refers to more targeted treatment of specific portions of the immune system via different types of medications). SDAI = Stomatitis Disease Activity Index; CBC = complete blood count; FeLV = feline leukemia virus; FIV = feline immunodeficiency virus; FCV = feline calicivirus; PFFV = puma feline foamy virus; PME = partial mouth extraction; FME = full mouth extraction; IFN = interferon
Early studies from 1997 53 and 2008 6 reported similar response rates to extraction therapy, with the first of these published works demonstrating that 80% of cats (24/30) were significantly improved or clinically cured at follow-up 11-24 months after treatment. 53 In both of these studies, most patients (cumulatively 96.8% [60/62]) were treated with PMEs. Re-evaluating the success rate of extraction therapy after radiographic findings came to light, and with a greater number of patients, a later study, published in 2015, revealed a response rate of 39% (37/95) for substantial clinical improvement and 28% (27/95) for complete resolution of stomatitis. 5
In line with these findings, approximately 33% of cats (31/95) did not respond to extraction therapy in this later study. 5 The study also evaluated the role of extended medical management in surgically treated cases and revealed that most (68.8%) of the patients that achieved substantial improvement or complete resolution required medical management with antimicrobial, anti-inflammatory or analgesic medications for a finite amount of time after the 2-week immediate postoperative period. 5 In contrast to previous studies, this study was able to evaluate for the first time the effect of the extent of dental extractions on the outcome, concluding that there was no significant difference between cats treated with PMEs vs FMEs in terms of the overall response to treatment. 5 This finding supports the notion that dental plaque is a less crucial etiologic factor than previously thought. The possibility that there can be a variation in disease phenotype or that patients treated with PMEs are showing an entirely different disease altogether, such as aggressive periodontitis or contact stomatitis, should also not be discounted. 5 Though early intervention could plausibly lead to better outcomes in this inflammatory disease, no studies have evaluated the effect that the duration of clinical signs has on the outcome of surgical treatment. However, in one study, duration of clinical signs was not correlated with severity of the lesions (clinical scores) at the time of treatment. 7
The impact of concurrent viral infection on the success of extraction therapy has been evaluated in a recent study which showed that FeLV-positive cats had a 7.5-times greater likelihood of no improvement after dental extractions. 40 Coinfection with FCV and PFFV has also been found to be significantly more likely in cats with refractory FCGS than in cats with responsive FCGS. 10
At this juncture, PMEs (extraction of all premolar and molar teeth) and FMEs have continued to provide the best long-term results.5,7,53 In the light of the existing body of knowledge, the authors have a number of suggestions regarding best practice, as outlined in the box ‘Recommendations for tooth extraction therapy’.

Medical management
Medical management of FCGS may be challenging to maintain owing to the significant oral pain caused by this disease, but is necessary for both acute and chronic settings. Medical management before tooth extraction has failed to affect the overall response to surgical treatment, and is likely only to temporarily alleviate some of the discomfort associated with stomatitis in cats. 5 In short, medical management has no role as a sole treatment entity in place of surgery at this time. However, it is a fundamental consideration, given the often severe pain exhibited by FCGS patients and the concurrent infections that may occur. 24 Moreover, approximately a third of cats receiving tooth extraction therapy do not respond to surgical treatment, and appropriate support via medical management becomes essential postoperatively in these refractory cases (see box ‘What denotes “refractory”?’).
The authors institute a personalized medicine (tailored) approach for cats that do not respond to dental extractions. In the absence of systemic disease, refractory cases have been managed with a combination of analgesia and immunosuppressive or immunomodulatory therapy. Current work from the authors’ institution supports the use of biomarkers in stratifying treatment. Biomarker discovery in patients with FCGS is ongoing work inspired by recent findings that approximately two-thirds of cases that are not responsive to extraction therapy are coinfected with FCV and PFFV, 10 and also that there is an effect of FeLV status on the prognosis of these cats. 40 Such research suggests that patients that are FeLV, FIV, FCV and/or PFFV positive should undergo medical management in conjunction with surgical management.
The following discussion focuses on analgesic, antimicrobial, immunosuppressive and immunomodulatory therapy, with Figure 7 presenting a summary of key treatment recommendations.
Analgesia
Pain management is essential at all stages of FCGS management: in the acute setting, in the post-surgical setting and in those cases refractory to extraction therapy. In a study evaluating the analgesic effects and absorption of buprenorphine after buccal administration in feline oral disease, cats with stomatitis exhibited lower bioavailability and a shorter absorption half-life. 55 Despite this, buprenorphine produced an analgesic effect and low inter-individual variability in plasma concentration compared with healthy cats and saline control. 55 Other pain management agents that may be beneficial, but where scientific data are still lacking to support their use in FCGS, include N-methyl-D-aspartate (NMDA) receptor antagonists (amantadine), gabapentin, opioids other than buprenorphine, and non-steroidal anti-inflammatory drugs (NSAIDs).

NSAIDs have been evaluated as an adjunct treatment modality for cats with FCGS. A combination of bovine lactoferrin oral spray (6 mg/cat, q12h) and piroxicam (0.3 mg/kg PO on alternate days) was investigated in a randomized, double-blind clinical trial, and clinical signs were significantly improved in 77% of the cats. 70 Oral piroxicam alone decreased clinical signs during the first 2 weeks; however, the combination of bovine lactoferrin oral spray and piroxicam produced an enhanced effect that reduced the severity of the oral lesions and improved clinical signs, quality of life and weight gain over the duration of the 12-week study. 70 It is worth mentioning that the authors did not state the success rate for the control group in this study and that control cases were converted to treatment cases after the fourth week. Oral administration of lactoferrin alone had previously been investigated for its effects in inhibiting bacterial growth in cats with stomatitis with and without FIV, and resulted in an improvement in clinical signs as well as a concurrent increase in neutrophil phagocytic activity. 71

Antimicrobials
Scientific data supporting the use of antibiotics in FCGS are limited. One study reporting the effect of different antibiotics documented improvement in 38% of cats treated with amoxicillin and 37% of cats treated with metronidazole. 27 Considering these effects are only transient, and that response rates are lower for antibiotics than immuno-suppressive therapy, antibiotic treatment is only recommended in the acute setting and/or if secondary infections are noted. 27
Research has been performed to investigate the antimicrobial susceptibility of the sub-gingival microbiome of cats. 72 However, the patients included in this study had only gingivitis, whereas patients with FCGS show evidence of a more severe form of periodontitis. 24 Though studies have described the subgingival microbiome in FCGS patients, 9 the antimicrobial resistance patterns of these organisms have yet to be evaluated. An analogous human disease featuring a compromised oral epithelim that would be an indication for complete (or near-complete) tooth extractions is lacking. However, a Cochrane review has investigated the use of antibiotics to prevent complications following tooth extractions in healthy humans. 73 Taken together, the studies included in the review found low-certainty evidence that antibiotics may reduce the risk of infection and dry socket compared with placebo in people undergoing extractions of impacted third molars. Furthermore, there was very low-certainty evidence of no increase in the risk of adverse effects in these patients. This may be different in immuno-compromised or immunosuppressed patients, which were not included in this study. 73
The authors’ approach is to utilize a short course (5 days) of antimicrobials postoperatively for FCGS cases (amoxicillin clavulanate, 13.75 mg/kg PO q12h; clindamycin 5–11 mg/kg PO q12h) owing to the aggressive nature of surgery and poor condition of the mucogingival tissues. The authors are, however, actively researching if there is a benefit of >24 h antibiotic therapy.
Immunosuppressive therapy
Glucocorticoids remain the most frequently prescribed medication for the management of stomatitis pre- and postoperatively. In postoperative patients, glucocorticoids have been utilized immediately after surgery and also in refractory cases. This approach is in contrast to the use of some novel therapeutics (discussed in the ‘Immunomodulation’ section) that enhance patients’ immune response, rather than immunosuppressing them further.
Treatment with steroids has been shown to produce complete cure or a marked improvement in about 23% of patients; of those, only 7% achieved clinical remission. 47 Considering these facts, along with the potentially harmful side effects of long-term administration, such as diabetes mellitus, the authors recommend that corticosteroids be reserved for patients not responding to pain management protocols, for symptomatic treatment, on a tapering course, or for use as a salvage option. If corticosteroids are used, it is recommended that periodic blood work is obtained to assess for potential side effects.
Ciclosporin is also an immunosuppressant that is used in cases of FCGS. Ciclosporin decreases T cell proliferation by reducing IL-2 expression; this, in turn, leads indirectly to decreased B cell function (ie, responsiveness and antibody production) as well as directly decreasing B cell migration. 74 A randomized, controlled, double-blind, prospective clinical trial where oral ciclosporin was administered to nine cats that had previously been treated with tooth extractions reported a significant difference in the number of cats experiencing clinical improvement over the 6-week study period between the treatment group (7/9 cats, 77.8%) and the placebo group (1/7 cats, 14.3%). 44 Long-term observation of 11 cats, as part of the same study, showed 45.5% (five cats) were clinically cured after receiving ciclosporin for 3 or more months. Furthermore, whole-blood ciclosporin levels >300 ng/ml were associated with significant improvement in oral inflammation in cats with refractory chronic stomatitis. 44 Treatment with ciclosporin before dental extractions has also been evaluated in a small number of patients (n = 8), with 50% of the cats reported to achieve clinical remission. 75
The authors of this review advise against immunosuppression without surgical intervention.
Immunomodulation
Immunomodulation is often reserved for refractory cases (ie, those that have not responded to surgery). Note that, as referred to earlier, the time at which a patient is defined as refractory varies. Moreover, the use of biomarkers has significant potential in this context to reduce waiting times for medical therapy and improve patient quality of life.
In refractory cases, treatment with recombinant feline interferon-omega (rFeIFN-ω) and mesenchymal stromal cell (MSC) therapy has shown promise. A controlled, randomized, double-blind study found that oromucosal rFeIFN-ω resulted in moderate improvement to clinical cure in 55% of treated cats and marked improvement to clinical cure in 45%. 47 Results of a study investigating subcutaneous administration of rFeIFN-ω in FCV-positive cats with FCGS found that stomatitis was improved through inhibition of FCV proliferation. 38 Subcutaneous rFeIFN-ω has also shown clinical efficacy in cats that are naturally infected with FeLV or coinfected with FeLV and FIV. 76 Additionally, oral rFeIFN-ω has been shown to be an effective alternative therapy for FIV-infected cats. 77
In a study evaluating the efficacy of rFeIFN-ω for clinical improvement and reduction of concurrent viral excretion in retrovirus-infected cats from a rescue shelter, caudal stomatitis was a common finding (6/16 cats), particularly among FIV-positive cats. 78 In this study, FIV-infected and FIV/FeLV coinfected cats improved during therapy. Interestingly, all of the FIV cats that tested positive for FCV (4/7) had gingivo-stomatitis; and, of the 11 FCV-positive cats, nine had reduced viral loads post-rFeIFN-ω treatment. 78 Taken together, these studies confirm the role of rFeIFN-ω therapy in patients with FCGS and confirmed retroviral (FIV/FeLV) or FCV infection. rFeIFN-ω is approved for use in Europe, but is currently not approved in the USA.

The efficacy of both autologous and allogeneic, fresh, adipose-derived MSCs administered intravenously has been studied in cats with refractory FCGS,29,45,46 and is reported in a recently published paper from the authors’ group at the University of California, Davis (see ‘Companion paper on FCGS’ highlight circle). 79 The immunomodulatory action of MSCs has shown promise for treating cats with refractory FCGS, with up to 57% and 71% of FCGS patients exhibiting clinical improvement or remission after allogeneic and autologous treatments, respectively.29,46 No response was seen when treating with MSCs before extraction therapy in a pilot study. 80

Key Points
✜ Current evidence regarding the etiology of FCGS points toward an immune-mediated disease process with both local and systemic ramifications in response to a chronic antigenic stimulant.
✜ There is increasing scientific support for a role for FCV as a part of the etiology of FCGS - at least as a disease-modulating agent.
✜ All cats with FCGS are affected by periodontitis to varying extents.
✜ Surgical treatment continues to be the gold standard therapy, with extraction of all teeth for patients where the inflammation extends to the rostral oral cavity. Partial (premolar and molar) dental extractions may be considered for patients where inflammation is confined to the regions of the premolar and molar teeth.
✜ Given the complexity of this disease, and until a tooth-sparing treatment option is found, a personalized medicine approach is recommended. The aim should be to optimize patient care, allowing for more timely medical treatment for those patients that are predicted to have a poor response to surgical treatment.
✜ The value of medical management in the acute setting before extractions are performed, in the immediate postoperative period and for treatment of refractory cases, is recognized.
Supplemental Material
Appendix 1
Feline Stomatitis disease Activity index.
Footnotes
Supplementary material
The following file is available as supplementary material alongside this article:
✜ Appendix 1: Feline Stomatitis Disease Activity Index.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
This work did not involve the use of animals and therefore ethical approval was not specifically required for publication in JFMS.
Informed consent
This work did not involve the use of animals (including cadavers) and therefore informed consent was not required. No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
