Abstract
Objectives
The aim of this study was to compare the quality of sedation with three different anaesthetic protocols (alfaxalone combined with butorphanol, methadone or pethidine) administered intramuscularly in cats, and to evaluate the influence of the injection site (between supraspinatus and quadriceps muscles) on the onset and quality of sedation.
Methods
A total of 151 cats were selected for this study. Cats were sedated with alfaxalone (3 mg/kg) combined with either butorphanol (0.3 mg/kg; n = 50), methadone (0.3 mg/kg; n = 53) or pethidine (5 mg/kg; n = 48). The combination was injected intramuscularly into the supraspinatus (n = 79) or quadriceps muscle (n = 72). The data included a scoring system for the quality of sedation and physiological parameters, such as heart rate (HR), respiratory rate, body temperature and occurrence of mydriasis, monitored during the first 30 mins of anaesthesia.
Results
The opioid associated with alfaxalone influenced the overall sedation score, the degree of myorelaxation, the occurrence of mydriasis and HR. The overall sedation score was poorer with butorphanol than with methadone (P = 0.008), and butorphanol induced a lower degree of myorelaxation than methadone (P = 0.013). The injection into the supraspinatus showed better qualitative results for sedation and a faster onset time (in about 3 mins) than that into the quadriceps (P <0.001). HR decreased from baseline (P <0.001) and over time (P <0.001), mainly in cats of the butorphanol–supraspinatus and pethidine–quadriceps groups (P = 0.004). The occurrence of mydriasis was lower after butorphanol than after methadone and pethidine (P = 0.025), while the incidence of side effects did not differ among groups.
Conclusions and relevance
All three protocols provided a good quality of sedation and allowed performing the scheduled procedure. Moreover, the injection into the supraspinatus muscle showed superior results in all the qualitative scores of sedation and quicker onset time than that into the quadriceps muscle.
Introduction
During daily clinical procedures, such as physical examination, venepuncture or intravenous (IV) catheter placement, some cats can be difficult to handle and require intramuscular (IM) sedation. Moreover, defining the American Society of Anaesthesiologists (ASA) physical status in unhandled patients, such as stray animals, might be complicated given their fractious behaviour, and this may lead to possible errors in the anaesthetic risk evaluation. Therefore, identifying a sedative protocol suitable for the most excitable cats could reduce the anaesthetic risk, minimise the stress related to prolonged physical restraint1,2 and contribute to early pain management. 3 However, sedation is not devoid of risks, and the choice of drugs and doses can be challenging; the ideal protocol should maintain adequate cardiorespiratory function while unpleasant or minimally invasive procedures are performed. 4
To date, multiple drug combinations have been used for IM sedation in cats; the most used protocols include alpha (α2)-adrenoreceptor agonists, opioids and ketamine.
5
α2-Adrenoreceptor agonists not only allow reliable sedation, muscle relaxation, analgesia and chemical restraint, but they also cause peripheral vasoconstriction, hypertension with reflex bradycardia and decreased cardiac output.
4
These drug-induced cardiovascular modifications limit the suitability of α2 agonists in animals that have or could have cardiovascular impairment. Ketamine, an N-methyl-
Variables associated with the absorption of drugs after IM injections are related to the drug composition, the correct execution of the injection and the vascularisation of the muscle where the injection takes place. Studies, both in humans and veterinary medicine, have shown differences in the efficacy of sedation protocols based on different muscular injection sites.24,25
The aims of this study were to compare the quality of sedation with three different anaesthetic protocols (alfaxalone combined with butorphanol, methadone or pethidine) administered intramuscularly for chemical restraint in cats, which were required for diagnostic/medical/presurgical purposes, and to evaluate if the site of the IM injection (supraspinatus or quadriceps muscle) influences the onset and quality of sedation. The hypotheses of the authors were that all protocols would be effective in restraining cats, with mild differences among the three associations, and that injections at the supraspinatus would induce a quicker onset of sedation.
Materials and methods
This was a multicentre, randomised, investigator-blinded, prospective clinical study conducted at the University of Veterinary Medicine of Perugia, the Centre Hospitalier Vétérinaire ADVETIA (Paris, France) and the Briar House Veterinary Practice (Kent, UK). The study was approved by the Ethical Committee of the University of Perugia (protocol n. 2019/02), and owner consent was obtained for each cat enrolled in the study.
Animals
All cats presented to the clinics from November 2018 to February 2020 requiring chemical restraint for medical, diagnostic or surgical purposes were included in the study. Exclusion criteria were eventual history related to adverse reactions to one or more of the molecules included in the protocol. Signalment included breed, sex, age (categorised as paediatric [0–3 months], young [4–12 months], adult [1–10 years] and geriatric [>10 years]) and body condition score (BCS; from 1 to 5; categorised into three groups [1–2, 3 and 4–5] for statistical purposes). A temperament score ranging from 1 (very friendly) to 5 (very aggressive) was assigned while approaching and visiting the animal; a pre-anaesthetic evaluation allowed for ASA physical status classification (from 1 to 5).
Study protocol
After enrolment in the study, cats received alfaxalone 3 mg/kg (Alfaxalone; Jurox) mixed with one of three opioids: methadone (M) 0.3 mg/kg (Semfortan; Eurovet), butorphanol (B) 0.3 mg/kg (Dolorex; MSD) or pethidine (P) 5 mg/kg (Petidina Cloridrato; Molteni). The injection site was further randomised between the supraspinatus muscle (S) and the quadriceps muscle (Q); this led to the identification of six main groups defined as MS (n = 28), MQ (n = 25), BS (n = 24), BQ (n = 26), PS (n = 27) and PQ (n = 21), according to the association of opioid and the site of injection, respectively. Injections took place in a quiet room with dimmed light, and the selected drug mixture was prepared and administered by an anaesthetist different from the evaluator. A 2.5 ml syringe with a 22 G needle was used to perform the IM injection, with the cat held in a standing position. The time of injection (T0) was recorded, and the animal was placed back in its transport cage. The reaction to the injection was scored from 0 (no reaction) to 4 (prolonged reaction; Table 1). The cat was left undisturbed and evaluated at prefixed time points at 3, 5, 8, 12, 15, 20, 25 and 30 mins from T0. The degree of chemical immobilisation was assessed according to a composite simple descriptive sedation score adapted from Deutsch et al (Table 1). 2 The assessment included four main evaluations: patient ‘position’, ‘resistance to lateral recumbency’, ‘response to noise’ (ie, producing a kiss-like sound, 1 m away from the cat) and ‘degree of myorelaxation’. These parameters were summed to obtain a total score, a maximum of 13 that indicated deep sedation. A total score of ⩾5 was required to start the procedure. If at T15 sedation was still inadequate (total score <5), a dose of dexmedetomidine 3 µg/kg (Dexdomitor; Orion) was administered IM.
Scoring system of the qualitative parameters used to describe the degree of sedation of cats

Parameter contributing to the total score, adapted from Deutsch et al. 2 A total score ⩾5 was considered adequate to start the procedure
The time to achieve lateral recumbency (TL) was recorded from T0 and was considered the time for safe animal handling. The cat was then removed from the cage, and the clinical procedure could begin (time to start the procedure). In most of the cases, this included an IV catheter placement in a cephalic vein or in other easily accessible veins, according to the case. Monitoring was performed for a maximum of 30 mins and included physiological parameters such as HR, RR, body temperature (T) and the presence or absence of mydriasis.
Further assessments included the ‘ease of catheter placement’, the eventual ‘orotracheal intubation’ along with the amount of propofol required for each cat, the time when eventual other anaesthetics (TOA) were necessary to complete the scheduled procedure and the ‘overall sedation’ score (from 1 [excellent] to 4 [inadequate]) (Table 1). Secondary effects related to the protocol were recorded and analysed, but their extensive discussion was beyond the aim of this study.
Sample size and randomisation
The minimum sample size calculation and post-hoc power analysis were performed using G*Power software (version 3.1.9.4), counting six experimental groups (ie, MS, MQ, BS, BQ, PS and PQ) and setting α = 0.05. Continuous and normally distributed variables were hypothesised. Before data collection, f-tests evaluating the effect of the opioid (three levels: methadone, butorphanol and pethidine), injection site (two levels: supraspinatus and quadriceps) and their interaction were planned. Setting a power (1 − β) = 0.95 and an effect size f = 40, a minimum of 17 cats per group was required. This number was approximated to 21 to take into consideration an estimated attrition rate of 20%. Thus, after inclusion in the trial, cats were randomly assigned to each group using the function ‘CASUAL’ in Microsoft Excel (2019) until at least 21 cats were included in each group. At the end of the experimental period, 28, 25, 24, 26, 27 and 21 cats were randomly included in the MS, MQ, BS, BQ, PS and PQ groups, respectively. The achieved power of the analyses, computed using post-hoc procedures and setting the same parameters, was 99%.
Statistical analysis
Descriptive statistics were used to present data as medians, ranges (minimum and maximum), quartiles, absolute and relative frequencies. The qualitative and quantitative (including physiological parameters) variables describing the quality of sedation were then analysed using generalised linear models. Diagnostic graphs were used to verify distribution, and the following probability distributions and link functions, respectively, were used: (1) multi-nomial and cumulative logit for categorical and ordinal variables (ie, recovery quality and response to injection); (ii) binomial and logit for binary variables (ie, mydriasis and side effects); and (3) normal and identity for continuous variables (ie, TL, HR, RR, T and propofol requirement). The models assessed the effects of the opioid (three levels: methadone, butorphanol and pethidine) and injection site (two levels: supraspinatus and quadriceps), including, when appropriate, time (for repeated measurements), age, BCS, temperament score, ASA status and/or surgery (yes/no) as a covariate. For the continuous variable (TL), the interaction between the opioid and injection site was also evaluated, while time (as a repeated factor) and the baseline value were included as covariates. The Sidak correction was used for pairwise comparisons. Results were expressed as estimated marginal means and standard error (SE), odds ratio with 95% confidence interval, and P value from Wald statistics. For the continuous variables, coefficient b, with its SE, was also reported. Data were analysed using IBM SPSS Statistics, version 25, while GraphPad Prism, version 7.0, was also used for visualisation. The level of statistical significance was set at P ⩽0.05.
Results
A total of 151 cats, including 84 males (30 castrated) and 67 females (28 spayed) weighing a mean ± SD of 3.7 ± 0.1 kg, were selected for the final analysis. Details of the demographic characteristics and temperament scores of the animals according to the group are shown in Table 2. Among all the cats in the study, 60% underwent surgery and 40% underwent diagnostic or medical treatment.
Demographic characteristics and temperament score of the population of 151 cats undergoing chemical restraint according to the group

Results are reported as n (%) for categorical variables and median (range) for continuous variables
*BCS in the MQ group was assessed only for 24 catsBCS = body condition score
Regardless of the opioid, the ‘response to injection’ was influenced by the muscular site, with higher scores, that is, worst reaction, for the quadriceps than for the supraspinatus muscle (P = 0.050), and by the BCS, with a higher score for cats with BCS 3 than for cats with BCS 1–2 (P = 0.046; Figure 1) (see also Tables 1 and 2 in the supplementary material).

Forest plot of odds ratio with 95% confidence interval showing factors associated with an increased risk of a high score (ie, worst reaction) for ‘response to IM injection’ (1 = no effect).
With regard to the ‘position’ (Figure 2; see also Tables 1 and 3 in the supplementary material), high scores, that is, best sedation, were observed in animals injected at the supraspinatus vs those injected at the quadriceps (P = 0.011) and in animals with low presedation temperament score (ie, more friendly; P = 0.008); moreover, the position score was higher in cats of BCS 3 (P = 0.048) and BCS 4–5 (P = 0.012) than in cats with BCS 1–2 and significantly increased with time (P <0.001), as well as increasing ASA status (P = 0.002).
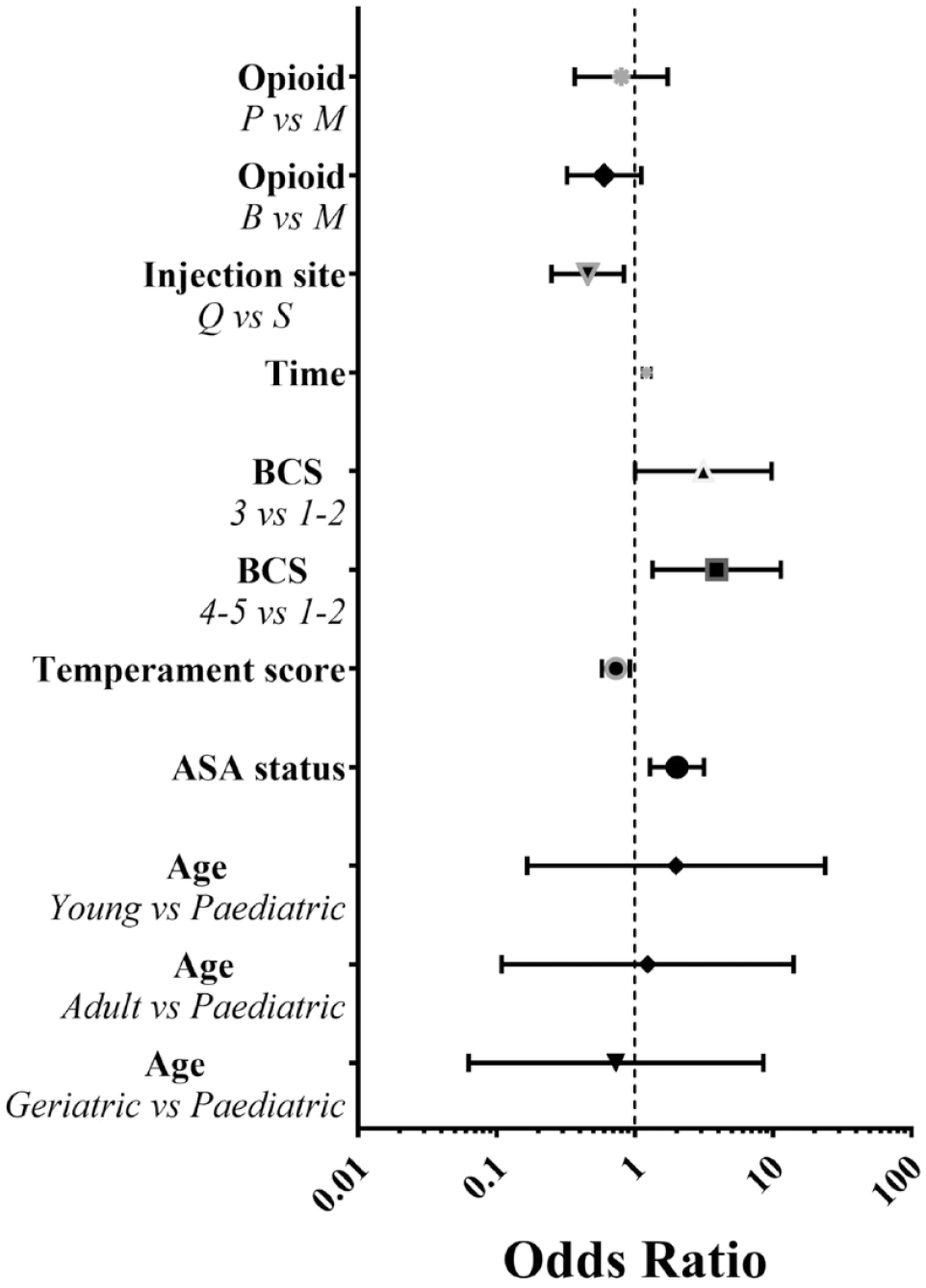
Forest plot of odds ratio with 95% confidence interval showing factors associated with an increased risk of a high score (ie, best sedation) for ‘position’ (1 = no effect).
The ‘resistance to lateral recumbency’ (Figure 3; see also Tables 1 and 4 in the supplementary material) was influenced by the site of injection regardless of the opioid, with lower scores, representing minor sedation, for the quadriceps than for the supraspinatus muscle (P <0.001); the score significantly increased with time (P <0.001) and was influenced by both the BCS, with lower scores for BCS 1–2 than for BCS 3 (P = 0.016) and BCS 4–5 (P = 0.02), and by increasing ASA status (P = 0.001). Moreover, this score was significantly influenced by the temperament score (P = 0.005), with higher scores in cats with lower temperament scores (ie, more friendly cats), and by age, with lower scores in paediatric cats than in young cats (P = 0.031).

Forest plot of odds ratio with 95% confidence interval showing factors associated with an increased risk of a high score (ie, best sedation) for ‘resistance to lateral recumbency’ (1 = no effect).
The ‘degree of myorelaxation’ (Figure 4; see also Tables 1 and 5 in the supplementary material) was affected by the opioid and body condition; regardless of the muscular site, better quality was observed in groups M than in groups B (P = 0.013) and in cats with BCS 3 than in cats with BCS 1–2 (P = 0.026). Furthermore, the scores for ‘degree of myorelaxation’ increased with increasing ASA status (P = 0.003) and time (P <0.001).

Forest plot of odds ratio with 95% confidence interval showing factors associated with an increased risk of a high score (ie, best sedation) for ‘degree of myorelaxation’ (1 = no effect).
The ‘response to noise’ (Figure 5; see also Tables 1 and 6 in the supplementary material) was significantly influenced by time, with greater scores (ie, best sedation) as time increased (P <0.001); by the BCS, with higher scores in cats with BCS 3 (P = 0.004) and BCS 4–5 (P = 0.017) than in cats with BCS 1–2; and by ASA status, with greater scores as the ASA status increased (P <0.001).

Forest plot of odds ratio with 95% confidence interval showing factors associated with increased risk of high score (ie, best sedation) for ‘response to noise’ (1 = no effect).
The ‘overall sedation’ (Figure 6; see also Tables 1 and 7 in the supplementary material) showed a lower level of sedation (ie, higher score) in groups B than in groups M (P = 0.008) and in cats injected in the quadriceps than in the supraspinatus (P = 0.008). A better level of sedation (ie, lower score) was recorded in cats with BCS 3 (P = 0.001) and BCS 4–5 (P <0.001) than in cats with BCS 1–2, and with increasing ASA status (P <0.001). Finally, the ‘overall sedation’ score improved with time in all the groups (P <0.001).

Forest plot of odds ratio with 95% confidence interval showing factors associated with increased risk of high score (ie, lower level of sedation) for ‘overall sedation’ (1 = no effect).
‘Time to achieve lateral recumbency’ (Figure 7) was significantly influenced by the site of injection, regardless of the opioid, resulting in a faster response if injected in the supraspinatus than in the quadriceps (2.9 ± 0.3 and 4.5 ± 0.4 mins in S and Q, respectively; P <0.001); more-over, increasing ASA status significantly reduced the onset (b = −45.7 ± 14.3; P = 0.001). The opioid, however, was not significant (P = 0.483) when evaluated as a main effect or as an interaction with the injection site (P = 0.209).
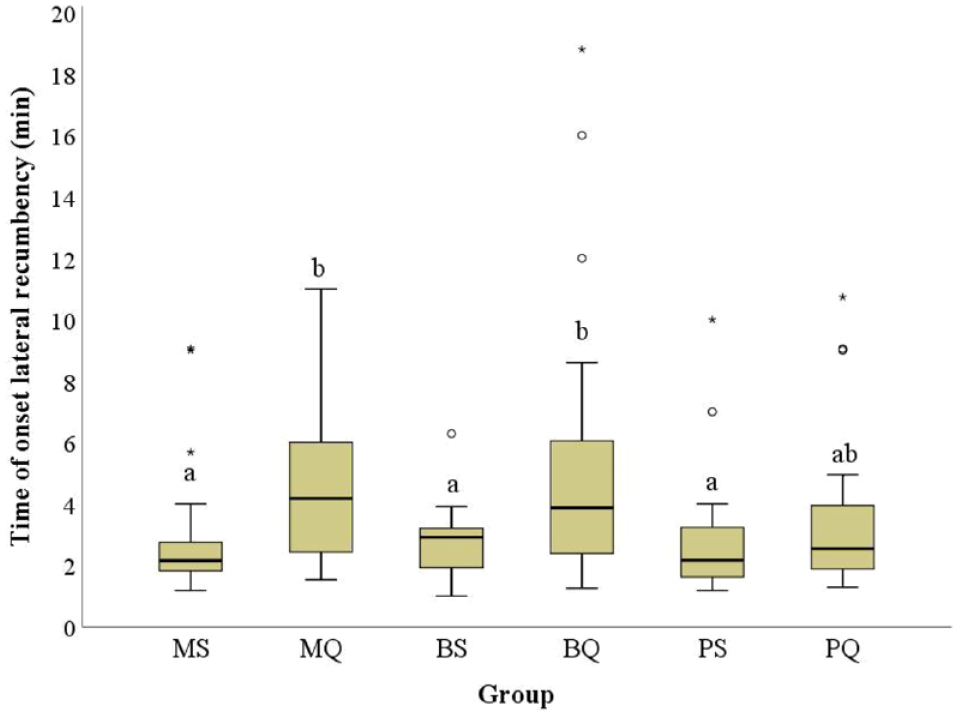
Marginal times (mean) taken to achieve lateral recumbency according to the opioid (M = methadone, B = butorphanol, P = pethidine) and injection site (S = supraspinatus, Q = quadriceps). Boxes with no common letters are significantly different (pairwise comparisons, P <0.05). Circles (O) indicate mild outliers (first quartiles −1.5 interquartile range [IQR] or third quartiles + 1.5 IQR), while asterisks (*) indicate extreme outliers (first quartiles −3.0 IQR or third quartiles + 3.0 IQR). The model evaluated the effect of opioid, injection site and their interaction, as well as body condition score, age, American Society of Anesthesiologists and temperament score
The mean ± SD time to starting the procedure did not differ between groups and was 6 ± 3 and 8 ± 4, 6 ± 2.5 and 7 ± 4 and 4 ± 2 and 6.5 ± 4 mins in the MS and MQ, BS and BQ and PS and PQ groups, respectively. This time mostly coincided with the placement of the IV catheter, which was easily performed in most animals (97%), while three cats (two in group BQ, one in group BS) required 3 µg/kg of dexmedetomidine IM as they still received a total score of <5 at T15.
To complete the procedure, propofol was required in 78% of cats. The mean ± SD dose did not differ among the groups and was 2.9 ± 1.8 mg/kg (P >0.1 for all effects). Similarly, the TOA did not differ and was 16 ± 7, 17 ± 5, 17 ± 6, 18 ± 6, 16 ± 7 and 19 ± 7 mins in the MS, MQ, BS, BQ, PS and PQ groups, respectively (P >0.1 for all effects).
The marginal means and results of statistical models of physiological parameters are detailed in Table 8 in the supplementary material. HR was significantly influenced by the interaction between the opioid and the site of injection (P = 0.004). Pairwise comparisons showed that the lowest marginal means of HR were recorded in the MQ and BS groups and the highest in the MS and PQ groups (P <0.05; Figure 8). Moreover, HR tended to decrease over time (P <0.001) and was influenced by baseline values (P <0.001).

Marginal means of heart rate (HR) according to the opioid (M = methadone, B = butorphanol, P = pethidine) and injection site (S = supraspinatus, Q = quadriceps). Boxes with no common letters are significantly different (pairwise comparisons, P <0.05). Circles (O) indicate mild outliers (first quartile ± 1.5 interquartile range). The model evaluated the effect of opioid, injection site and their interaction, and included time and baseline values as a covariate
The RR decreased over time (P = 0.002) and was influenced by injection site, with lower values when the supraspinatus was chosen over the quadriceps muscle (26 ± 1 and 30 ± 1 breaths/min for the supraspinatus and quadriceps, respectively; P = 0.010 [see Table 8 in the supplementary material]). The lowest RR was recorded in the BS group, and the highest was recorded in the MQ, BQ and PQ groups (P <0.05; Figure 9).

Marginal means of respiratory rate (RR) according to the opioid (M = methadone, B = butorphanol, P = pethidine) and injection site (S = supraspinatus; Q = quadriceps). Boxes with no common letters are significantly different (pairwise comparisons, P <0.05). Circles (O) indicate mild outliers (first quartiles −1.5 interquartile range [IQR] or third quartiles + 1.5 IQR), while asterisks (*) indicate extreme outliers (first quartiles −3.0 IQR or third quartiles + 3.0 IQR). The model evaluated the effect of opioid, injection site and their interaction, and included time and baseline values as a covariate
Body temperature tended to decrease over time in all cats (P <0.001) and was affected by baseline values (P <0.001) without differences among groups (see Table 8 in the supplementary material).
The presence of mydriasis was significantly influenced by opioid (P = 0.025; see Table 8 in the supplementary material), with lower occurrence in the BS group than in both the MS and PS groups (92% ± 4%, 89% ± 5%, 74% ± 9%, 82% ± 6%, 90% ± 4% and 90% ± 5% for MS, MQ, BS, BQ, PS and PQ groups, respectively; P <0.05).
The incidence of side effects is presented in Table 3. Logistic regressions showed no significant associations with either the opioid or the injection site (see Table 9 in the supplementary material).
Incidence of side effects

Data are number of cats showing the side effect and percentage within each group
Including ataxia, itching, tremors, twitching, nystagmus, vocalisation and nausea
Discussion
The aim of this study was to evaluate and compare the efficacy and quality of sedation of three anaesthetic combinations for chemical restraint in cats. The combinations included alfaxalone (3 mg/kg) with either methadone (0.3 mg/kg), butorphanol (0.3 mg/kg) or pethidine (5 mg/kg). The influence of injection sites, that is, the supraspinatus and quadriceps muscles, was also investigated. The three anaesthetic protocols enabled adequate chemical restraint for about 17 mins, and, to the best of our knowledge, this is the first time that such an assessment has included a large population of cats of different ASA status, temperament, age, sex and BCS. Moreover, the multivariable statistical approach allowed the evaluation of the independent effect of all these factors on the quality of sedation. None of the cats showed persistent signs of pain or motor impairment at the injected limb.
The mean onset time for safe handling of the animals was influenced by the injection site and not by the opioid, and it decreased as ASA status increased. Each protocol allowed safe manipulation in about 7 mins, with lateral recumbency gained earlier when the supraspinatus was injected vs the quadriceps (about 3 vs 5 mins). The reported mean time is faster than described by Deutsch et al, 2 where 0.2 mg/kg of butorphanol, combined with either 2 or 5 mg/kg alfaxalone, induced lateral recumbency in a median time of 6 mins after injection at the quadriceps. The recorded onset time was also faster than what reported by Granfone et al after 0.4 mg/kg of butorphanol and 2–3 mg/kg of alfaxalone administered IM in cats presented for blood donation. 12 Nevertheless, the onset time in our study was similar to that reported by Adami et al, 5 where different doses of dexmedetomidine, alfaxalone and butorphanol induced lateral recumbency in 5–7.5 mins.
The insertion of an IV catheter, considered to be the time the procedure was started, was easily achieved in all the cats, with no differences among the groups; nevertheless, three cats receiving butorphanol required dexmedetomidine as inadequate sedation persisted at T15. The addition of other molecules, such as propofol, to complete the procedures was required in 78% of all animals; however, it should be noted that 60% of the cats in the study underwent surgery. Similarly, Deutsch et al 2 reported that 58% of cats that were administered 0.2 mg/kg of butorphanol and 2 mg/kg of alfaxalone required a second injection to perform diagnostic procedures. Conversely, orotracheal intubation was not possible after IM injection of 5 mg/kg alfaxalone or two combinations of dexmedetomidine/alfaxalone in only 9.5% of cats. 14 Moreover, these cats did not undergo specific clinical procedures, but received repeated noxious stimuli, and in case of reaction to both orotracheal intubation or noxious stimuli, no additional drugs were administered. 14
With regard to the quality of sedation, the opioid significantly influenced the ‘degree of myorelaxation’ and ‘overall sedation’ scores, and methadone produced superior results than butorphanol. Moreover, both descriptors were scored higher in animals with a BCS of 3 than in thinner animals and likely better scored in animals with higher ASA status. Response to noise was significantly greater in thinner cats than in fatter animals, while it was significantly decreased over time and in cats with higher ASA status. It was not surprising that more friendly cats received greater scores for sedation quality, while changes according to age had poor clinical relevance. The authors did not find other studies that analysed the incidence of ASA status, age and BCS on the quality of sedation using alfaxalone associated or not with other molecules; this is often a consequence of the inclusion in clinical studies of animals with low ASA status.
Cardiorespiratory depression is one of the most described secondary effects of opioids during sedation and general anaesthesia in domestic animals.19,20 However, previous studies testing different opioids in cats showed a reduction of cardiorespiratory parameters within an acceptable range for anaesthetised cats.26,27 HR was influenced by the opioid–injection site interaction, resulting in a greater reduction (about 13% from baseline) in the BS than in the MS group (1.6% from baseline). HR decreased by 12% from baseline in the MQ group vs 1.5% and 0.2% in the BQ and PQ groups, respectively. Pethidine has a similar chemical structure to atropine, 28 and clinical effects typical of the antimuscarinic agent can be considered; however, the cardiovascular effects of pethidine could be mostly related to histamine release after IV administration, which corresponds to a marked increase in HR; IM injection does not provoke a significant increase in HR. 29
RR was influenced by the injection site and it was reduced from baseline, more often when the supraspinatus was injected. After injection in the supraspinatus, RR was lower in cats receiving butorphanol than in cats receiving methadone and pethidine (43%, 40% and 29% from baseline, respectively). The combination of 3 mg/kg pethidine with 0.1 mg/kg midazolam in cats induced minimal cardiorespiratory depression, nausea and vomiting as side effects, besides a reasonably short duration (~30 mins) of sedation suitable for venepuncture, minor invasive procedures (ie, thoracentesis, abdominocentesis and cystocentesis) and other non-invasive procedures (ie, proper radiographic position) in cats, without a hangover after the end of the procedure. 30 However, the lack of any blood gas evaluation impedes assessment for possible ventilatory depression.
Body temperature tended to decrease in all cats over time without differences among groups; this is in disagreement with the hyperthermia described in cats after opioid administration, 31 but it is probably due to the sedative effect of alfaxalone and the correspondent immobility of the cats rapidly after the injection.
The number of cats that presented mydriasis was lower after butorphanol than after methadone and pethidine. Mydriasis is a secondary effect of opioids described in cats as outlasting their analgesic duration, 32 but no studies have investigated eventual disparity among different molecules. In the cat, morphine-induced mydriasis results from catecholamine release, mainly from the adrenal glands, which act on the iris. 32 This antagonises the parasympathetic input expected to produce miosis, which is the main effect of µ-agonists in dogs. 33 Sharpe 34 reported that separate neural mechanisms control the pupillary components and that μ-opioid receptors are more involved in mediating opiate-induced mydriasis in the cat. This could justify the lower pupillary dilation induced by the κ-agonist butorphanol in our cats.
When assessing the injection site, the supraspinatus muscle induced better scores in all the parameters of the sedation scoring system. A faster time to onset has been reported in raccoons injected in a forelimb rather than a hindlimb for IM injection of pre-anaesthetic drugs. 35 This finding could be due to the different blood flow between the two muscles, as demonstrated in humans among deltoid and either the gluteus or the vastus lateralis. 36 It is likely that the bioavailability of drugs could be affected accordingly. 25 A previous study in cats showed that IM alfadalone/alfaxalone in the biceps femoris was less effective than in the quadriceps muscle, probably due to the administration in the fascial planes surrounding the muscle rather than in the muscle body. 24 Another study in dogs evidenced the faster bioavailability of a sedative mixture after administration in the postural muscles (quadriceps and triceps) than in the non-postural muscles (cervical and axial, gluteus medius) as they are characterised by a greater blood flow. 37 This blood flow could also be influenced by muscle movement, which can, in turn, affect drug absorption by increasing local perfusion. The cats in the present study did not move as they were kept in the carrier cage after the injection. However, the supraspinatus muscle, although not properly a postural muscle, induced qualitatively better and faster sedative effects, probably due to greater perfusion than the quadriceps muscle and likely to a faster return of blood to the closer right heart. Ideally, IM injection should not cause pain, but cats in our study showed a greater reaction when injected into the quadriceps rather than into the supraspinatus muscle. As the quadriceps is a postural muscle, it is likely that when the animal is standing, as at the time of injection in this study, the quadriceps is under tension and the muscular body is surrounded by inelastic fibrous connective tissue, impeding easy spread of the injected liquids. The intensity of the reaction to IM injection is similar to other studies investigating the IM administration of alfaxalone with various sedatives.14,38,39 As the pH of alfaxalone is 6.6–7.0, the reason for the reaction to IM injection should be attributed to the volume of the injectate itself. 14 The maximum volumes of substances to be injected in animals differ based on the species, the route and the characteristic of the mixture. 40 Diehl et al 40 considered 0.25–0.50 ml/kg to be good practice for IM injection in dogs, but no specific volumes have been defined for cats. The volume of injectates in our study was within the range reported for dogs, 40 and it is likely that, given the low concentration of alfaxalone (1%), a higher volume would be reached when associated with pethidine, but no influence was found with this association. The response to injection was lower in thinner cats, and this could most likely result from a true IM injection rather than an IM or subcutaneous administration, respectively.
The secondary effects of alfaxalone have been well described 14 and were almost equally recorded in this study with all the opioid combinations and included opisthotonos, hyperextension of neck and limbs, vocalisation, myoclonus and ataxia. Animals undergoing procedures requiring more manipulations had a greater incidence of secondary effects.
This study had some limitations. First, there was no control group that included only alfaxalone. However, previous studies evaluating the quality of sedation associated with IM alfaxalone are discordant. The level of sedation with 2 or 5 mg/kg of alfaxalone was inadequate to perform diagnostic procedures in cats. 2 However, 5 mg/kg of alfaxalone produced a moderate-to-deep degree of sedation. 14 While 1 mg/kg of alfaxalone induced lateral recumbency only in 50% of the cats the injections of 2.5, 5 and 10 mg/kg induced lateral recumbency in 100% of them. 38 Alfaxalone has no analgesic properties and therefore its use in anaesthetic protocols for minimally invasive procedures should be associated with analgesics. Second, the three anaesthetic mixtures provided good immobility for about 17 mins; however, if longer chemical immobilisation or relevant manipulations of the animal are required, the adjunct of other drugs is highly advocated to limit behavioural side effects. This was demonstrated by Wheeler et al,13 where the addition of midazolam to IM alfaxalone and hydromorphone produced greater sedation and myorelaxation in cats than alfaxalone–hydromorphone alone. Furthermore, the choice of the associated opioid should depend on the scheduled procedure in terms of duration and anticipated pain, but such an evaluation was beyond the scope of this study. Finally, because of the clinical nature of the study, standardisation of the medical/diagnostic/surgical procedure was not possible, and the main qualitative comparisons ended after 30 mins when the IV catheter would have been available for any further supplemental drugs, such as propofol. Small doses of propofol should be titrated to effect to obtain light sedation avoiding unexpected induction of general anaesthesia and possible relevant consequences if cats are not properly monitored, and oxygen supplementation via face mask should be provided accordingly. 4
Conclusions
The three anaesthetic protocols induced a good quality of sedation for medium–short procedures in cats of any age, ASA status and BCS. The IM association of alfaxalone with the three opioids was equally safe and effective, with acceptable secondary effects. The injections into the supraspinatus muscle resulted in better scores for all the evaluated parameters and a faster onset than those into the quadriceps muscle.
Supplemental Material
Table 1
Descriptive statistics for the evaluated parameters according with the opioid and site of injection.
Supplemental Material
Table 2
Factors affecting response to injection.
Supplemental Material
Table 3
Factors affecting position.
Supplemental Material
Table 4
Factors affecting resistance to lateral recumbency.
Supplemental Material
Table 5
Factors affecting degree of myorelaxation.
Supplemental Material
Table 6
Factors affecting response to noise.
Supplemental Material
Table 7
Factors affecting overall sedation.
Supplemental Material
Table 8
Factors affecting physiological parameters.
Supplemental Material
Table 9
Logistic regression (reference category: no side effects). Associations between opioid and site of injection.
Footnotes
Acknowledgements
The authors wish to thank Dr Ilaria Russo and Dr Enrico Cecchetti for their clinical contribution with the enrolled cats.
Supplementary material
The following files are available online:
Table 1: Descriptive statistics for the evaluated parameters according with the opioid and site of injection.
Table 2: Factors affecting response to injection.
Table 3: Factors affecting position.
Table 4: Factors affecting resistance to lateral recumbency.
Table 5: Factors affecting degree of myorelaxation.
Table 6: Factors affecting response to noise.
Table 7: Factors affecting overall sedation.
Table 8: Factors affecting physiological parameters.
Table 9: Logistic regression (reference category: no side effects). Associations between opioid and site of injection.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognised high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers) for all procedure(s) undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
