Abstract
Objectives
The sedation quality of intramuscular (IM) alfaxalone and butorphanol in combination with midazolam was investigated in hyperthyroid cats undergoing suitability assessment for radioiodine treatment.
Methods
A total of 60 hyperthyroid cats undergoing diagnostic investigations were randomly allocated to receive butorphanol (0.3 mg/kg IM) and midazolam (0.2 mg/kg IM) with either alfaxalone (2 mg/kg IM) (BMA2) or alfaxalone (3 mg/kg IM) (BMA3), or butorphanol (0.3 mg/kg IM) with alfaxalone (3 mg/kg IM) (BA3). If required, additional alfaxalone (0.2 mg/kg) was administered intravenously. Cat Stress Score, response to injection, time to lateral recumbency, sedation score at 10, 15 and 20 mins and subsequent 10-min intervals, additional alfaxalone requirements, and time to first administration, recovery quality (excellent, fair, poor) and adverse effects were assessed. Thyroxine concentrations, gabapentin treatment and assessors were recorded. Heart and respiratory rate and arterial haemoglobin saturation were monitored every 5 mins. Data were compared using χ2 and Kruskal–Wallis testing. The multidimensional sedation score and predictors of sedation score were analysed using a mixed effect and linear regression model, respectively (P <0.05).
Results
No significant predictors for sedation quality were identified. In all groups, the median sedation score was considered good and the median recovery score was fair. The sedation score over time across groups and cardiorespiratory variables were not significantly different. Additional alfaxalone was administered in 53 cats. In group BA3, additional alfaxalone was required significantly earlier (P = 0.043). Although sedated, muscle twitching was a commonly observed adverse effect in all groups, but head pawing was significantly increased in BA3 (P = 0.014).
Conclusions and relevance
Sedation and recovery quality were satisfactory with all protocols but the addition of midazolam prolonged sedation.
Introduction
Feline hyperthyroidism requires long-term treatment with thiamaz alfaxalone (2 oles or feeding an iodine-restricted diet. Radioiodine is a potentially curative treatment that can change the quality of life for both owners and cats. 1 To assess eligibility for radioiodine, diagnostic investigations are often performed. Hyperthyroid cats may be more fragile or reactive in the hospital environment and intolerant of handling, which may trigger a stress response exacerbating the effect of the thyrotoxic state. Hence, intramuscular (IM) sedation for diagnostic procedures or even initial clinical assessment and intravenous (IV) cannula placement is often desired. Concurrent disease, such as chronic kidney disease, hypertrophic cardiomyopathy and hypertension are frequently encountered.2 –4 Until diagnosis and severity of concurrent (cardiac) disease are established, a sedation protocol with minimal effects on the cardiovascular system is favoured.
Alfaxalone has limited cardiovascular effects when given alone or in combination with butorphanol.5 –8 For diagnostic procedures, alfaxalone in combination with butorphanol (0.2 mg/kg) provides reliable sedation in healthy cats and cats with mild systemic disease if given at a high dose of 5 mg/kg IM. 8 However, a large injection volume and long recovery period is disadvantageous. 8 A lower dose of 2 mg/kg IM combined with butorphanol at 0.2 mg/kg failed to provide sufficient sedation for most cats, despite achieving adequate sedation levels for echocardiography.6,8 In addition, adverse effects to noise and touch, such as hyperaesthesia, have been reported with alfaxalone-based sedation protocols regardless of dose.8,9
Adding a benzodiazepine to the sedation protocol may enhance muscle relaxation without major cardiorespiratory effects. 10 The addition of a benzodiazepine to the sedation protocol could allow reduction of the alfaxalone dose, thereby reducing the injection volume and recovery duration. 11 Furthermore, the requirement for incremental doses of alfaxalone to achieve appropriate sedation levels and adverse events may be reduced.
The aims of the present study were two-fold: to establish whether the addition of midazolam to alfaxalone and butorphanol IM (1) improves the quality of sedation and (2) reduces the amount of alfaxalone needed to achieve adequate sedation for clinical investigations in hyperthyroid cats.
The hypothesis was that adding midazolam to IM alfaxalone and butorphanol for sedation in hyperthyroid cats would improve the quality of sedation, as assessed by a multidimensional sedation score, and reduce the need for additional alfaxalone doses to complete procedures, thereby resulting in a better quality of recovery.
Materials and methods
This prospective blinded randomised clinical study was approved by the Animal Welfare and Ethics Review Body of the University of Bristol, UK (VIN/18/021) and the Veterinary Medicine Directorate, UK (ACT-S-167).
Hyperthyroid cats presented for assessment of suitability for radioiodine treatment at Langford Vets, University of Bristol Teaching Hospital, between May 2019 and December 2021 were recruited after informed, written owner consent was obtained by the internal medicine clinician. During the coronavirus disease 2019 (COVID-19) pandemic (March 2020 to July 2021), case collection was paused. Cats with a history of hyperthyroidism, treated or untreated, classified according to the American Association of Anesthesiologists physical status grading system as 2 or 3 and requiring an IM sedation for diagnostic investigations, were included in the study. Cats treated with phenobarbitone were excluded because of potential sedative effects. For all cats, diagnostic investigations included thoracic radiographs and abdominal ultrasound, and, if indicated, fine-needle aspiration of altered tissue, cystocentesis and/or completion of echocardiography and fundic examination. Cats were randomly (www.randomizer.org) assigned to 1/3 study groups using a sequentially numbered envelope system. Group BMA2 received butorphanol (0.3 mg/kg, Dolorex 10 mg/ml; MSD Animal Health UK), midazolam (0.2 mg/kg, Midazolam 5 mg/ml; Hameln Pharma) and alfaxalone (2 mg/kg, Alfaxan Multidose; Jurox); group BMA3 received butorphanol (0.3 mg/kg), midazolam (0.2 mg/kg) and alfaxalone (3 mg/kg); and group BA3 received butorphanol (0.3 mg/kg) and alfaxalone (3 mg/kg).
After a settlement period following admission (at least 14 h), each cat was assigned a Cat Stress Score (CSS) by an experienced feline nurse (Table 1). 12 Before sedative drug administration, cats underwent a full clinical examination, including non-invasive Doppler systolic blood pressure (BP) measurement (Doppler Flow Detector Model 811-B; Parks Medical Electronics) using a cuff size approximately 40% of the limb circumference. BP was measured when the cat was undisturbed and relaxed in its kennel. Gabapentin (approximately 10 mg/kg PO) was administered at the clinician’s discretion in the morning before the procedure and was not standardised. Thyroxine concentrations were recorded, and levels below 12.9 nmol/l were nominally assigned 12.9 nmol/l because of the limit of the assay. All scores were assessed and recorded by qualified veterinary nurses trained and familiar with the scoring system.
Cat Stress Score (CSS), simple description (Kessler and Turner 12 ), response to injection and recovery quality score

The drug combination was drawn up by a nurse unrelated to the study. The clinician administered the IM sedatives into the lumbar muscles using a 2.5 ml syringe and a 25 G needle in a quiet room with no other cats or members of staff present other than a qualified veterinary nurse who provided minimal restraint. Any personnel handling and assessing the cat were unaware of treatment allocation.
The response to injection (Table 1) and the time to lateral recumbency were recorded in minutes. A multidimensional scoring system (adapted from Young et al 13 ) was used to score the quality of sedation at 10, 15 and 20 mins and subsequently in 10-min intervals until transfer of the cat into recovery. Posture, resistance to maintenance of lateral recumbency, degree of muscular relaxation and response to noise stimulus (firm handclap 1 m distanced from the ears) were evaluated (Table 2) with a maximum score of 13 possible. If after 10 mins the score was less than 5, or a 22 G IV cannula could not be placed because of inadequate sedation, additional alfaxalone (2 mg/kg IM) was administered. After placement of the IV cannula, incremental alfaxalone (0.2 mg/kg IV) doses could be administered to allow the procedure to be performed (eg, to ensure sufficient immobilisation for radiography positioning and ultrasound guided cystocentesis or organ sampling) at the discretion of the attending clinician. Heart rate (HR), respiratory rate (fR) and saturation of haemoglobin with oxygen (SpO2) (Nonin 2500A Pulse Oximeter; Nonin Medical) were recorded at 5-min intervals after injection, starting once adequate sedation levels were reached. Supplemental oxygen was provided using a face mask if SpO2 was below 92% or measurement was not possible. Active warming was provided using a heat mat covered by a blanket to avoid direct contact with the patient. Procedure time and rectal temperature at recovery were recorded. Recovery quality (Table 1) was scored. Recovery time, which was defined as the time from injection until standing and walking without ataxia, was noted. In case of prolonged dysphoria (>10 mins) or excitement, flumazenil (0.01 mg/kg; Flumazenil 0.1 mg/ml Injection; Hameln Pharma) was administered. The occurrence of adverse effects (head bobbing, opisthotonus, muscle twitching, exaggerated response to noise or touch, salivation, rubbing the face with paws) was recorded. All cats were fully recovered from the sedation, and the injection site was monitored for swelling or discharge, before discharge from the hospital.
Multidimensional sedation score adapted from Young et al 13

Statistical analysis
According to a sample size calculated by Deutsch et al, 8 at least 18 cats per group were required to show a clinically relevant difference of 4 using the multidimensional sedation score.
Statistical analysis was performed using a statistical software package (GraphPad Prism Version 9). Data were assessed for normality using the Shapiro–Wilk test and visual inspection of graphical plots. Continuous variables were compared across the three sedation protocol groups using the Kruskal–Wallis test and data, presented as median (range). Post-hoc testing was performed using Dunn’s multiple comparisons test. Categorical variables were compared using χ2 testing. Sedation score over time was analysed by fitting a mixed-effects model using a compound symmetry covariance matrix and is fit using Restricted Maximum Likelihood. The Geisser–Greenhouse correction was applied if the assumption of sphericity was not met. Dependent variables considered in the linear regression analysis included assessor, CSS, breed, sex, age, body weight, body condition score, gabapentin administration, thyroxine concentrations and hyperthyroidism control status (uncontrolled hyperthyroidism defined as thyroxine >30 nmol/l). 14 Variables with P <0.2 on univariable analysis were entered into a multivariable linear regression analysis. Significance was set at P <0.05.
Results
A total of 60 cats were included in the study and evenly distributed between groups (group size, n = 20). Baseline HR, fR and BP before sedation were not significantly different between groups. Domestic shorthair and longhair cats were analysed together and compared with pedigree cats, which included a Ragdoll and Egyptian Mau in BMA2, two Bengals, a Russian Blue, a Persian, a Burmese crossbreed and a British Shorthair crossbreed in BMA3, and none in BA3. Age, sex, body weight, body condition score and CSS were not significantly different between groups (Table 3). Thyroxine concentrations were significantly higher in BA3 compared with BMA2 (P = 0.022) (Table 3, Figure 1a); however, there was no significant difference in proportion of uncontrolled hyperthyroid cats between groups.
Group and sedation characteristics for 60 hyperthyroid cats*

Data are reported as n or median (range)
Cats were sedated with butorphanol (0.3 mg/kg IM), midazolam (0.2 mg/kg IM) and alfaxalone (2 mg/kg IM) (BMA2; n = 20), or butorphanol (0.3 mg/kg IM), midazolam (0.2 mg/kg IM) and alfaxalone (3 mg/kg IM) (BMA3; n = 20), or butorphanol (0.3 mg/kg IM) and alfaxalone (3 mg/kg IM) (BA3; n = 20). Age (years), sex, breed, body weight (kg), body condition score, thyroxine concentration (nmol/l), hyperthyroidism control status, gabapentin treatment, Cat Stress Score, response to injection, onset of lateral recumbency, duration of procedure, time to first additional alfaxalone requirement (AAR) (0.2 mg/kg IV), number of AARs, recovery quality and time until full recovery (standing, walking without ataxia) are shown
DLH = domestic longhair; DSH = domestic shorthair; FN = female neutered; MN = male neutered

(a) Thyroxine levels (TT4), (b) mean multidimensional sedation score, (c) sedation score at first incremental dose of alfaxalone and (d) time to first additional alfaxalone requirement, (e) duration of procedure and (f) time to full recovery (standing, walking without ataxia) in 60 hyperthyroid cats after administration of butorphanol (0.3 mg/kg IM), midazolam (0.2 mg/kg IM) and alfaxalone (2 mg/kg IM) (BMA2; n = 20), or butorphanol (0.3 mg/kg IM), midazolam (0.2 mg/kg IM) and alfaxalone (3 mg/kg IM) (BMA3; n = 20), or butorphanol (0.3 mg/kg IM) and alfaxalone (3 mg/kg IM) (BA3; n = 20). Data are median (range)
Reaction to injection and time to lateral recumbency were not significantly different between groups (Table 3). Cannula placement was successful in all cats after the initial injection.
The multidimensional sedation score over time across groups was not significantly different (Figures 1b and 2a). Out of 60 cats, 53 had additional alfaxalone requirements (AARs). The total number of AARs did not differ significantly between groups (Table 3). The time to the first AAR was significantly longer in BMA3 (P = 0.043) (Table 3, Figures 1c,d).

(a) Multidimensional sedation score, (b) heart rate (HR) and (c) respiratory rate (RR) over time are displayed for 60 hyperthyroid cats sedated with butorphanol (0.3 mg/kg IM), midazolam (0.2 mg/kg IM) and alfaxalone (2 mg/kg IM) (BMA2; group 1 red; n = 20), or butorphanol (0.3 mg/kg IM), midazolam (0.2 mg/kg IM) and alfaxalone (3 mg/kg IM) (BMA3; group 2 green; n = 20), or butorphanol (0.3 mg/kg IM) and alfaxalone (3 mg/kg IM) (BA3; group 3 blue; n = 20). Data are median (range)
The duration of the procedure and time to full recovery was shorter in BA3 compared with BMA3 and compared with BMA2 and BMA 3, respectively (P = 0.003) (Table 3; Figure 1e,f). The median recovery score was fair for all treatment groups (Table 3).
Adverse effects are listed in Table 4. Head pawing occurred significantly more frequently in group BA3 (P = 0.014) (Table 4). No injection site reactions were observed.
Adverse effects in hyperthyroid cats after administration of sedative
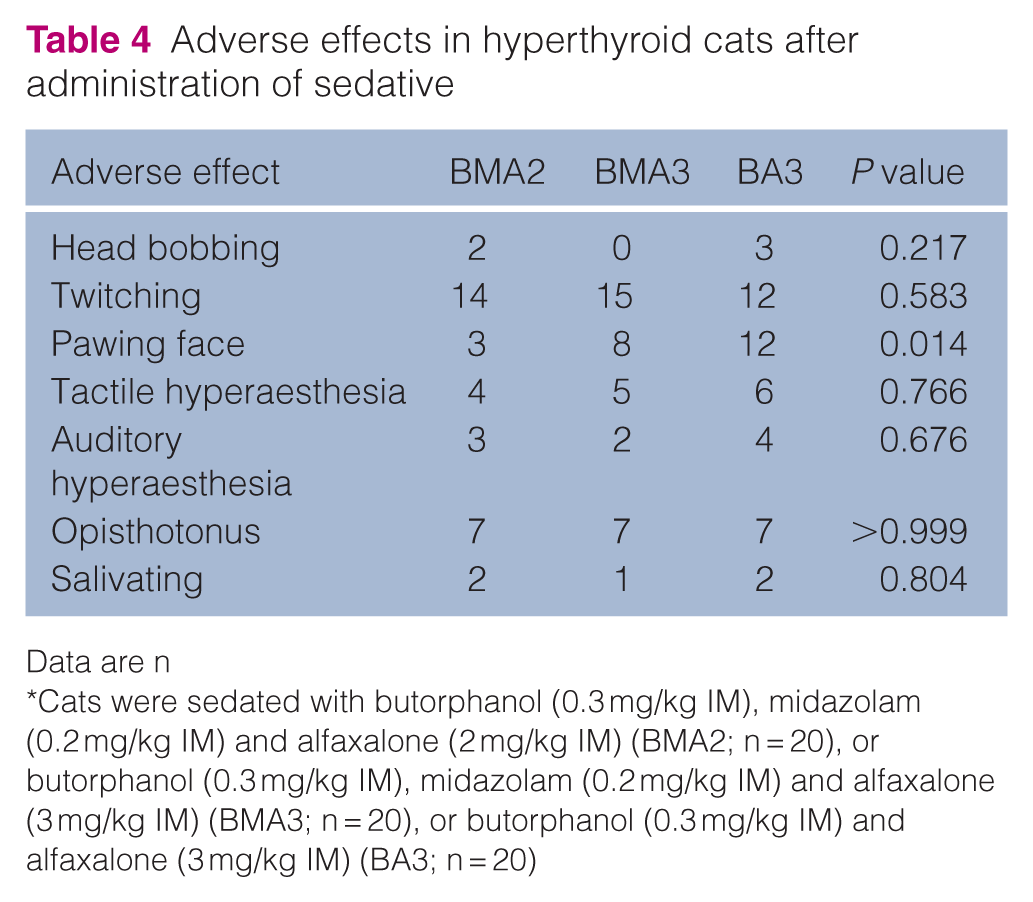
Data are n
Cats were sedated with butorphanol (0.3 mg/kg IM), midazolam (0.2 mg/kg IM) and alfaxalone (2 mg/kg IM) (BMA2; n = 20), or butorphanol (0.3 mg/kg IM), midazolam (0.2 mg/kg IM) and alfaxalone (3 mg/kg IM) (BMA3; n = 20), or butorphanol (0.3 mg/kg IM) and alfaxalone (3 mg/kg IM) (BA3; n = 20)
No significant predictors for sedation quality were identified in the univariable or multivariable linear regression model.
Cardiorespiratory variables were not significantly different over time between groups (Figure 2b,c). As a result of missing values, SpO2 could not be analysed. Flumazenil was not required for any cat based on the study criteria for its use.
Discussion
The addition of midazolam to an IM sedation protocol using butorphanol and alfaxalone in hyperthyroid cats failed to significantly improve the quality of sedation but prolonged its duration. Both AAR and recovery quality were not significantly affected; however, the duration of the procedure varied between groups and needs to be considered in the interpretation of the results. Therefore, the hypothesis was rejected. In hyperthyroid cats, two different doses of alfaxalone and butorphanol with or without midazolam provided adequate IM sedation for diagnostic procedures performed in a teaching hospital. Increasing the dose of alfaxalone from 2 mg/kg to 3 mg/kg or adding midazolam prolonged the median duration of sedation by approximately 21 mins (group BMA2) and 17 mins (group BA3), respectively. Similarly, adding midazolam (0.2 mg/kg IM) to hydromorphone and alfaxalone produced a better quality of sedation for a ‘longer’ time period. Notably, though, cats were only monitored for 20 mins after administration. 11
Almost all cats in this study required incremental doses of alfaxalone to achieve adequate levels of sedation and muscle relaxation for the diagnostic procedures. The procedure length of 1–2 h or longer may have contributed to this observation as it exceeds the duration of action of alfaxalone. 15 This may be attributed to two facts: the study was performed in a teaching hospital and sedated cats had to be transported to a different building and between rooms for the diagnostic investigations, which may have resulted in increased levels of stimulation during the sedation. Administration of a higher dose of alfaxalone (5 mg/kg IM) resulted in a profound sedation for 45 mins only. 15 In this study, the combination of alfaxalone (3 mg/kg IM) with butorphanol and midazolam provided a good quality of sedation for a median duration of 45 mins. However, the median of five additional alfaxalone doses in group BA3 was comparable to that in other groups. The first additional dose was required as soon as 15 mins after injection in 10% of cats in this study. This is similar to a recent study in which adequate sedation was reported to last only approximately 17 mins after alfaxalone (3 mg/kg IM) and butorphanol (0.3 mg/kg IM) were administered. 16 In contrast, in cats sedated for blood donation, lateral recumbency was maintained for 32 and 54 mins with alfaxalone (2 mg/kg IM) and butorphanol (0.2 mg/kg IM) and alfaxalone (2–3 mg/kg IM) and butorphanol (0.4 mg/kg IM), respectively.17,18 Apart from procedure length, the clinical study design, the desired profound sedation at our institution and cat population may have influenced the need for additional alfaxalone.
Although the onset of lateral recumbency varied widely, the mean onset time of 5–7 mins in all groups allowed IV access and is similar to other studies evaluating alfaxalone and butorphanol sedation.4,6,8,17
Recovery was of acceptable quality despite the repeated IV alfaxalone administration and comparable to other alfaxalone based protocols.8,14,17 Previously, recovery was assessed as mainly excellent and was slightly shorter at 81 mins (from injection), but the length of the procedure and increased requirement of additional alfaxalone may have contributed.8,18 The mean recovery duration after the end of the procedure was approximately 30 mins in the groups using midazolam but was considerably shorter for the group not using midazolam. Conversely, the shorter procedure time may have been the main influencing factor in the group without midazolam. Nevertheless, a similar number of additional alfaxalone doses was required in these cats, potentially indicating a lighter plane of sedation.
The combination of the three drugs and its sedative and cardiorespiratory effects have not been investigated previously. Despite that, the protocol is often used clinically to sedate cats that have escalated to exhibit protective behaviour and in elderly cats with multiple coexisting diseases. Mild cardiorespiratory side effects are reported using alfaxalone (1–5 mg/kg IM).5,7 Increasing doses up to 10 mg/kg IV, higher than those typically used in clinical settings, did not result in major cardiovascular side effects. 5 Mild respiratory depression was observed at doses of 5 mg/kg IV and 10 mg/kg IM.5,7 Alfaxalone (2 mg/kg IM) and butorphanol (0.2 mg/kg IM) resulted in only mild cardiorespiratory changes, with echocardiographic findings considered clinically insignificant.6,8,18 –20 However, hypoxaemia was observed in more cats when alfaxalone was increased to 5 mg/kg. 8 At the dose used in this study, midazolam has no notable effect on the cardiovascular system in dogs, but studies in cats focusing solely on midazolam are lacking. 21 However, as for any sedation, regular monitoring is required as sedation may deepen with increasing drug doses and addition of drugs to the protocol, resulting in enhanced cardiovascular and potentially respiratory depression.7,8,22
Muscle twitching and hyperaesthesia to touch and noise were commonly observed in all groups, which conforms with other reports of sedation with alfaxalone in cats.7,8,16 Midazolam is not licensed in dogs and cats but is widely used for sedation and general anaesthesia owing to its favourable cardiovascular properties and muscle relaxation. The additional muscle relaxant effect of midazolam failed to abolish these adverse effects. However, the incremental alfaxalone doses could have masked the potential beneficial effect of midazolam. Midazolam provided good sedation in combination with alfaxalone and opioids,11,23 and it has been successfully combined with ketamine, butorphanol and ketamine or butorphanol and dexmedetomidine.24,25
The 10 mg/ml concentration of the alfaxalone formulation results in a large volume of injection. Hence, the lumbar muscle was chosen as the injection site because of its lack of surrounding fascia. Despite anticipated difficulties in administering the large injection volume in cats with a low body condition score, no difference between groups was detected. Not unexpectedly, though, all cats reacted at least mildly to injection. Often, reactions are attributed to the injection volume; however, temperament or speed of injection could offer alternative explanations.8,9,16,26 Although the pH of the solution may be associated with a reaction to injection, the pH of multidose alfaxalone is 7.0 and unlikely to contribute. 27 When the administration of alfaxalone was compared with that of dexmedetomidine, no difference in reaction to injection was detected. 17 Comparably, when the preservative alfaxalone formulation was used in rabbits, reactions were observed in the placebo (saline) and alfaxalone group. 28
In this study, alfaxalone with preservative was used, which contains ethanol (150 mg/ml), chlorocresol (1 mg/ml) and benzethonium chloride (0.2 mg/ml), which can be found in meloxicam, buprenorphine, and ketamine and butorphanol, respectively. 29 In rabbits, 6–10 mg/kg IM produced acceptable sedation without local tissue reaction, comparable to this study. 28 Administered IV in cats, similar pharmacokinetic and pharmacodynamic characteristics compared with the preservative-free alfaxalone formulation have been reported, supporting interchangeable use.30 –32 Although pharmacokinetic data were not evaluated, this study highlights that the alfaxalone formulation with preservative can be used for IM sedation in cats.
Thyroxine concentrations varied greatly in this cat population; some cats were not receiving any medical or dietary therapy for their hyperthyroidism, and of those being treated, levels of control varied. Indeed, the variability in the thyroxine concentrations may explain the unexpectedly higher thyroxine concentrations in group BA3; however, there was no statistically significant difference in the proportion of uncontrolled hyperthyroid cats between the groups. Thyroxine increases sympathomimetic drive and may increase sedation requirements, as well as drug metabolism, resulting in a reduced sedation duration. However, in the linear regression analysis, thyroxine concentrations were not identified as a significant predictor of sedation score.
In addition to the thyroxine concentrations, pretreatment with gabapentin could have influenced the quality of sedation positively and prolonged the length of recovery from sedation. Gabapentin, a calcium channel antagonist, has been shown to reduce stress levels in hospitalised cats.33 –35 Pretreatment with gabapentin was not found to be a significant predictor of sedation score in the linear regression analysis, although further research studies are required to establish the influence of gabapentin on the quality and duration of sedation and recovery. Ideally, all cats would have been receiving the same treatment; however, at the time of the study, gabapentin treatment was based on the cat’s individual behaviour at the clinician’s discretion.
The limitations of this study include the use of several different assessors, which may have led to subjective variation in scoring. This was inevitable because of the long duration of case collection and the busy schedule of a referral hospital. All assessors were qualified veterinary nurses and specifically trained by the lead author to use the various scoring systems. Furthermore, the assessor, as a predictor of sedation, was evaluated in the linear regression analysis and not found to be significant. Second, no validated sedation scoring system is available; however, a familiar scoring system was chosen.8,13 The 10-min scoring intervals combined with the subjective evaluation of AAR may have masked a decrease in sedation score. Third, the basic investigations (thoracic radiographs, abdominal ultrasound and fundic examination) were routine, but additional diagnostics (eg, echocardiography or organ sampling) were non-standardised according to the individual requirements of each patient. Fourth, BP was not measured. Although this could have offered valuable information, given the nature of the procedures, this was not deemed possible at regular intervals. Although alfaxalone (5 mg/kg) in combination with butorphanol caused a decrease in BP, values remained above the accepted ranges of anaesthetised animals.8,18 Lastly, power was not determined for variables other than the sedation score and cautious interpretation of the results is warranted.
Conclusions
The addition of midazolam (0.2 mg/kg IM) to alfaxalone (3 mg/kg IM) and butorphanol (0.3 mg/kg IM) in hyperthyroid cats prolongs the duration of sedation compared with the protocol without midazolam, or the same protocol using a lower dose of alfaxalone (2 mg/kg), without significant effect on sedation depth and recovery quality. All three protocols could be used as tailored sedation plans for the individual hyperthyroid cat regardless of level of control of thyroxine concentrations.
Footnotes
Acknowledgements
The authors would like to thank Jurox Ltd for providing Alfaxan. A special thanks goes to the outstanding nurses working in the Feline Centre (Langford Vets, University of Bristol; Charlotte Roberts, Jade Sutherland, Suzanne Rudd, Rebecca Finney, Kimberly Whalen, Hannah Symes and Heather Hockey) for their support and contribution to data collection. We are grateful for the support of the clinicians with case recruitment and the clients who allowed their pets to be enrolled in this study.
Author note
This work was presented as an oral abstract presentation titled ‘Intramuscular alfaxalone-butorphanol sedation with or without midazolam in hyperthyroid cats’ at the World Congress of Veterinary Anaesthesia and Analgesia in Sydney in March 2023.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognised high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers, tissues and samples) for all procedure(s) undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
