Abstract
Objectives
The objective of this study was to compare the efficacy of feline mesenchymal stem cells (fMSC) with prednisolone as a treatment for inflammatory bowel disease (IBD) in cats.
Methods
Cats with chronic enteropathy that failed a 2-week diet trial and were not found to have significant concurrent disease were eligible for the study. If endoscopic biopsies confirmed a histopathologic diagnosis of IBD, the cat was randomly assigned to either the fMSC or prednisolone groups. Owners were blinded to the grouping. Stem cell treatment consisted of two intravenous injections of 2 × 106 cells/kg of freshly cultured allogeneic stem cells separated by 2 weeks. Prednisolone treatment was 1–2 mg/kg PO q24h, tapered according to clinical response. Owners were asked to make no changes (eg, diet and other medications) for the first 2 months, at which time they either continued to the 6-month recheck with no changes, or ‘failed’ treatment and owners were unblinded and changes made as necessary.
Results
Six prednisolone and six fMSC treatment cats completed the study. All six prednisolone group cats were spayed females with a mean age of 8.3 years (range 2–14), a mean body weight of 3.6 kg (range 2.5–4.8) and a mean pretreatment Feline Chronic Enteropathy Activity Index (FCEAI) score of 3.6 (range 2–6). The six stem cell cats included three spayed females and three castrated males, and had a mean age of 8.0 years (range 4.5–13), a mean body weight of 4.9 kg (range 4.0–5.9) and a mean pretreatment FCEAI score of 3.7 (range 2–5). One cat in each group failed at the 2-month recheck. At the 6-month recheck, the mean FCEAI score for the prednisolone group was 3.7 (range 0.5–9) and 0.75 (range 0–1.5) for the fMSC group.
Conclusions and relevance
These results suggest that this specific fMSC protocol appears to be as effective in the treatment of feline IBD as a standard course of prednisolone therapy.
Introduction
The treatment of feline inflammatory bowel disease (IBD) requires owners to commit to daily medication for prolonged periods of time, entails the potential side effects of immunosuppressive drugs and may still result in an inadequate control of clinical signs.1,2
Previously, a proof-of-concept study confirmed the safety and potential benefit of treating feline chronic enteropathy with two doses of freshly cultured, allogeneic adipose-derived mesenchymal stem cells (MSCs). 3 Although the inclusion criteria for the pilot study included the absence of significant secondary gastrointestinal disease such as chronic kidney disease, hepatopathy and hyperthyroidism, there was no attempt to obtain a definitive diagnosis of the primary gastrointestinal disease (ie, to distinguish between IBD or alimentary lymphoma). Despite the overall positive results of that study, one of the cats receiving stem cell therapy did not improve, with one possible explanation being that not all of the cats had the same underlying etiology. The current study was designed to determine the effect of fMSC treatment for a specific feline chronic enteropathy (IBD) compared with the standard treatment using prednisolone.
MSCs have been investigated as a potential treatment for IBD in several human clinical trials and are often harvested from bone marrow or umbilical cord tissue. Two to three intravenous (IV) infusions of 1 × 106 cells/kg is the common dose used in human patients with IBD, although stem cells can be injected locally into fistulas in patients with Crohn disease (CD). In fact, much of the current literature appears to show that MSC use in human IBD may be most useful for perianal fistula repair.4,5 Interestingly, and relevant to the intent of this study, there is a report of allogeneic MSC use in human patients with refractory IBD – including both CD and ulcerative colitis. Six of the seven patients were being treated with corticosteroids at the time of study entry, and four of those patients were able to taper or discontinue steroid treatment following stem cell infusion. All seven patients experienced a significant reduction in clinical activity index scores; five patients went into remission 3 months after MSC treatment. 6 IV MSC infusion in published, Food and Drug Administration-registered human clinical trials appears to be safe, with few serious adverse events. 7
Adipose-derived MSC have been shown to be a safe and effective treatment for canine IBD, and, as noted, we previously demonstrated a positive clinical response in a blinded, placebo-controlled trial using allogeneic, freshly cultured adipose-derived feline MSC (fMSC) to treat cats with chronic enteropathy of undetermined etiology.3,8,9 These results and the promising findings in humans with CD suggest that fMSC are potentially a powerful therapeutic option in cases of feline IBD.10,11 Accordingly, the aim of this study was to compare fMSC therapy with standard prednisolone treatment in cats with IBD.
Materials and methods
Cats were deemed eligible for study entry if they had a history of vomiting, diarrhea or both for >1 month, had no other significant concurrent condition (as determined by physical examination), history, minimum database, feline leukemia virus/feline immunodeficiency virus SNAP, total thyroxine and a Texas A&M gastrointestinal (GI) panel (Figure 1). Cats also had to have had a recent fecal examination that was negative for GI parasites or been previously treated with a de-wormer, and were required to have failed to gain significant improvement in clinical signs following a 2-week diet trial with a hypoallergenic or hydrolyzed diet. Cats could not have been treated with a glucocorticoid in the 2 months prior to study entry. Failure to meet any of these requirements resulted in exclusion from study participation. Cats were not required to have had an abdominal ultrasound. Owners signed a study consent form and completed a questionnaire as part of the Feline Chronic Enteropathy Activity Index (FCEAI) score, as well as grading their pet’s diarrhea on a 7-point scale. All aspects of the study were approved by the Colorado State University Clinical Review Board (VCS#2017-108), and all owners signed an informed consent prior to study entry.

Diagram of study timeline. *Owner blinded to treatment (Rx) group; both groups received placebo Rx, mimicking the other group. fMSC = IV injection of feline mesenchymal stem cells; Histopath = histopathology; PARR = PCR for antigen receptor rearrangement; Hx = history; PE = physical examination; MDB = minimum database (complete blood count, chemistry, urinalysis); TX A&M = feline gastrointestinal panel; TT4 = total thyroxine; Failed Diet Trial = 2-week hypoallergenic/hydrolyzed diet; FCEAI = Feline Chronic Enteropathy Activity Index
Owners were made aware of the study prior to endoscopy, which was performed as with any clinical case. Endoscopic biopsies were obtained from the stomach, duodenum, ileum and colon for histopathology and PCR for antigen receptor rearrangement (PARR) analysis. Histopathology was performed by the pathologist on duty, and if histopathology was reported as clearly consistent with IBD, the cat was accepted into the study. Any stated concern for occult or emerging lymphoma on the histopathology report in any section of the intestinal tract, or any evidence of clonality at any site on PARR analysis, disqualified the patient from the study, as demonstrated for the cat in Figure 2.

Histopathology (hematoxylin and eosin). (a) Magnified (× 20) image of duodenum showing increased numbers of lymphocytes and plasma cells in the laminal propria. Mildly to moderately increased proprial connective tissue increases the spacing between glands. Confirmed polyclonal by PCR for antigen receptor rearrangement (PARR). Diagnosis was inflammatory bowel disease. (b) Magnified (× 20) image of ileum showing neoplastic small lymphocytes multifocally expanding the propria. Cells slightly displace glands and diffusely infiltrate into the villous epithelium (epitheliotropism). Confirmed clonal by PARR. Diagnosis was low grade alimentary lymphoma. Images courtesy of Dr Paula Schaffer
Eligible cats were randomly assigned (random number generator: odd numbers to one group, even numbers to the other group) to receive either prednisolone or fMSC therapy. All cats were either already on a hypoallergenic/hydrolyzed diet or had been transitioned to one as part of the entry criteria for the study – the transition having failed to resolve clinical signs. Owners were blinded to the group that their cat was assigned to, and all cats received both the treatment for their group designation and a placebo representing the treatment their cats were not receiving (ie, liquid placebo vehicle on a standard prednisolone schedule or catheterization and sterile saline injection following the fMSC schedule). Owners were asked not to change any aspect of their cat’s treatment or diet during the first 2 months of the study.
The prednisolone group was started on 1–2 mg/kg PO q24h (unflavored compounded liquid [CSU Veterinary Teaching Hospital Pharmacy]; rounded to 5 mg, 7.5 mg or 10 mg q24h such that the dose was >1 mg/kg and <2 mg/kg). All of the prednisolone-treated cats were maintained on their original dose for the entirety of the first 2 months, after which the dose could be tapered as determined by the attending clinician, based on the owner reporting a positive response. No cat in the prednisolone group received <5 mg every other day for the remainder of the study. Members of this group also went through the fMSC treatment schedule with the injection of sterile saline. The fMSC group was started on the same volume of unflavored liquid vehicle without prednisolone, with dosing q12h for the first 2 weeks and subsequently tapered to one dose every other day for the duration of the study. This group was given two IV injections of 2 × 106 cells/kg of freshly cultured, allogeneic, adipose-derived stem cells separated by 2 weeks. fMSCs were prepared as previously described, and all fMSC treatments in the study were derived from the same specific pathogen-free donor. 5 Stem cell injections were given over 20 mins, and all cats remained in hospital for observation for a minimum of 1 h following injection of either stem cells or placebo. Owners were asked to report any clinical signs or behaviors that they believed might, in any way, be adverse events associated with the treatment.
All study cats were followed over the course of the next 2 months through direct communication and 1–2 recheck appointments (body weight, history, physical examination). All participants had a 2-month study recheck appointment.
If the clinician and client decided that the cat’s treatment response was not acceptable (eg, no change or increase in clinical signs, weight loss and hyporexia), the owners were unblinded and given the option of changes or additions to the treatment regimen. These cats were considered treatment ‘failures’. In cases where it was decided that the cat’s response to therapy was acceptable, owners were asked to maintain the study treatment schedules, still blinded, until the 6-month recheck. At the 6-month recheck, clients again completed the FCEAI questionnaire, cobalamin and folate concentrations were determined, and owners were unblinded as to their cat’s group participation.
Results
Six cats in the prednisolone treatment group and six cats in the fMSC treatment group completed the first 2 months of the study; five cats from each group completed the full 6-month course of the study. One cat from the prednisolone group presented to the 2-month recheck appointment with a significant increase in the frequency of vomiting and continued weight loss and the owner elected to add chlorambucil to the cat’s treatment at that time. One cat from the fMSC group presented to the 2-month recheck with an increase in diarrhea, a decrease in appetite and weight loss, and the owner elected euthanasia. These two cats were not included in the comparison group data or determination of pre- and 6-month recheck data. Other than these two treatment ‘failures’, no immediate or long-term adverse events were noted in any of the study cats.
As summarized in Table 1, the duration of clinical signs prior to study entry ranged from >6 months to >2 years. Examination of previous medical records revealed that four of the cats in the prednisolone group and three of the cats in the fMSC group had lost weight prior to entry into the study (Table 1). Group characteristics, sex, age, weight and FCEAI score are summarized in Table 2. The mean pretreatment FCEAI score for the prednisolone group was 3.6 (range 1–6), while the mean pretreatment FCEAI score for the fMSC group was 3.7 (range 2–5; Table 2). The mean body weight of the cats in the fMSC group at study entry was significantly greater than the mean body weight of the cats in the prednisolone group both before and after the 6-month study period (P = 0.019 and P = 0.016, respectively; Student’s unpaired t-test), although there was no difference in BCS between groups or within groups pre- to post-study (Table 2). One cat from each group had a cobalamin concentration below the reference interval (both were <150 ng/l) at study entry. Both cats were started on subcutaneous cobalamin supplementation, as recommended by the Texas A&M GI laboratory, and both were found to have a cobalamin >1000 g/l at the 6-month recheck.
Summary of the clinical condition of cats in the prednisolone and feline mesenchymal stem cell (fMSC) groups
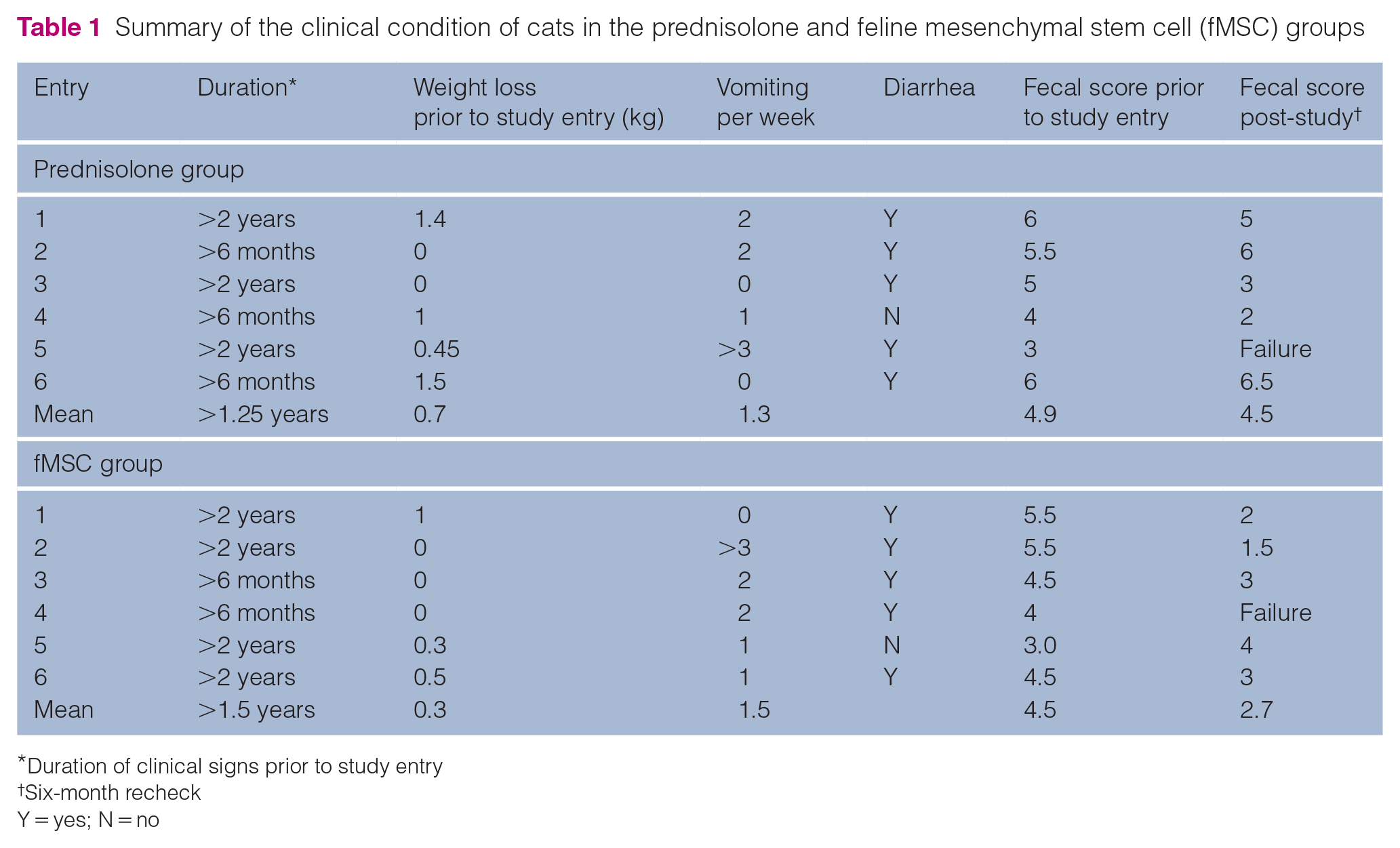
Duration of clinical signs prior to study entry
Six-month recheck
Y = yes; N = no
Group summaries for prednisolone and feline mesenchymal stem cell (fMSC) groups, comparing parameters prior to study entry with those at the 6-month conclusion of the study
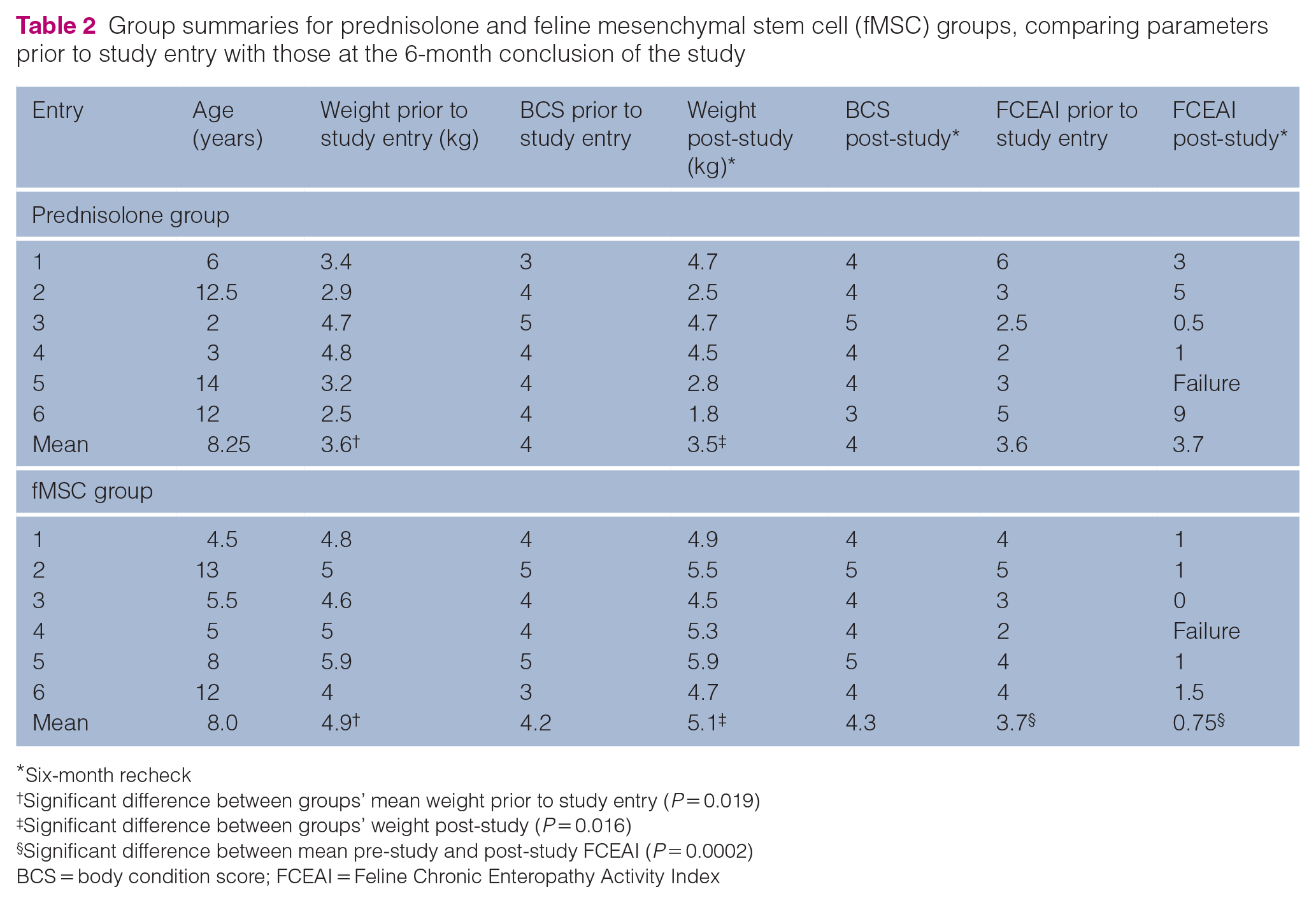
Six-month recheck
Significant difference between groups’ mean weight prior to study entry (P = 0.019)
Significant difference between groups’ weight post-study (P = 0.016)
Significant difference between mean pre-study and post-study FCEAI (P = 0.0002)
BCS = body condition score; FCEAI = Feline Chronic Enteropathy Activity Index
The mean fecal score (7-point Purina fecal scoring chart) for the cats in the prednisolone group went from 4.9 to 4.5, and for the cats in the fMSC group, it went from 4.5 to 2.7 over the 6-month study period (P = 0.098; Table 1). For the five cats in each group that completed the study, the 6-month recheck mean FCEAI score for the cats in the prednisolone group was 3.7 (range 0.5–9), and for the cats in the fMSC group it was 0.75 (range 0–1.5; Table 2). The mean FCEAI score for the five cats in the prednisolone group did not change after 6 months of treatment, while the mean FCEAI score for the five cats in the fMSC group decreased significantly (P = 0.0002; Student’s paired t-test).
Discussion
The results of this study suggest that this fMSC protocol is at least as effective in the treatment of feline IBD as the standard prednisolone therapy. A larger study is needed to determine whether the fMSC protocol is more clinically beneficial than prednisolone therapy for cats with IBD, considering that fMSC treatment decreased the need for daily medication with an immunosuppressive drug, significantly improved FCEAI scores, and may improve fecal scores, all without noted side effects in cats in this study.
The use of MSCs as a therapy for companion animals is under investigation for a number of immune-mediated diseases.12,13 MSCs are seemingly able to migrate to sites of injury or inflammation, where they produce a number of immunomodulatory molecules and bind ligands that direct T cell, B cell and other immune and inflammatory cell differentiation and behavior. 11 It is these migratory and immunosuppressive abilities that likely explain the impact of MSCs in cases of IBD. Promising results have already been established in cases of feline chronic gingivostomatitis. Arzi et al 14 administered two IV infusions of 2 × 107 allogeneic adipose-derived stem cells and identified a subset of cats that achieved complete remission. Feline MSC work has continued to expand in scope and has been evaluated in other conditions such as feline asthma. 15 Because of our previous success in a pilot study using MSCs in cats with unspecified chronic enteropathy, and the results of stem cell treatment in canines and humans with IBD, the current study focused on cats with histologically confirmed IBD.
The IV administration of allogeneic adipose-tissue derived MSCs has been shown to be a safe and effective treatment for IBD in canines.3,4 In an unblinded clinical trial with no control group, 11 dogs were treated with a single IV dose of 2 × 106 cells/kg. There were no acute adverse events reported. Those dogs demonstrated a significant clinical improvement by day 42 as measured using the Clinical Inflammatory Bowel Disease Activity Index, the Canine Chronic Enteropathy Clinical Activity Index, and albumin, cobalamin and folate concentrations. 4 Endo-scopic and histopathologic examination of the small intestines was performed on these dogs both before MSC infusion and 3–4 months after treatment. Significant improvement was found for both the Canine IBD Endoscopic Index and the Histologic Score. 3 Cristóbal et al 16 administered a single IV dose of adipose-derived allogeneic MSC (4 × 106 cells/kg) to dogs that either had no previous treatment or were currently being treated with prednisolone but with an unsatisfactory clinical effect. Using clinical indices and albumin and cobalamin concentrations as indicators of response to therapy, a significant improvement was seen in both groups of dogs over the 12 months of the study, and all of the dogs that had been receiving prednisone were successfully weaned off that medication. 16
Consistent with previous reports, the current study confirmed that IV infusions of allogeneic, freshly cultured adipose-derived fMSC were easily administered and safe in cats with IBD. One of the desirable traits of MSCs is their low expression of major histocompatibility complex class II and ability to avoid clinically relevant immune surveillance. 17 No adverse events were associated with the first or the second injection of fMSCs from the same donor, administered 2 weeks apart. In this study the response to therapy was similar between the group that received standard prednisolone therapy and the group that received MSC infusions; one cat from each group was considered a treatment ‘failure’ at 2 months, while the other five cats reached the 6-month mark with a clinical response that was comparable. Although the mean FCEAI and fecal scores were not significantly different between the two groups at study completion, the FCEAI score improved significantly over the 6-month study period for the MSC group only and the fecal score decreased, although this was not statistically significant (P = 0.098).
This study was not designed to provide controlled, long-term follow-up data, but, as of January 2022, the owners of 4/5 cats from the fMSC group that completed the 6-month study were successfully contacted; the owner of cat 5 was lost to follow-up. As of 3–4 years after study completion, all four cats were alive; all were reported to have a normal appetite, activity level and normal stool (fecal scores of 3 or 4 on a 7-point scale). For the two cats in which vomiting was the presenting complaint, the vomiting had resolved or was reported as happening ‘two times a month’. One of these four cats was started and maintained on oral prednisolone therapy after the 6-month study period when the owner reported finding blood in the cat’s stool; another of the study cats was started on inhaled steroids for asthma. This anecdotal information is encouraging and highlights the need for a larger sample size and longer-term, controlled studies.
Other limitations of this study include a reliance on relatively subjective measures that include owner input. The FCEAI has been established and used in a number of studies, but ongoing efforts to identify meaningful and easily accessible biomarkers will benefit future studies in trying to quantify and compare a response to therapy. Study participants were not required to return for a repeat of endoscopy for histopathology, as changes in the clinical condition of patients appears to be independent of changes seen on repeat endoscopy or histopathology, at least in dogs.18,19 Although several studies using MSC for IBD show promising results, owing to the nature of stem cells, studies using different MSC protocols may not have the same outcomes.
One of the only significant differences between the two groups at the beginning of the study, besides one group being of a single sex, was that the fMSC group was significantly heavier than the prednisolone group, although BCS scores between the groups were similar. This may have been due to the random assignment of male cats to this group or an indication that the fMSC group started the trial in a healthier state, despite the similarity in their clinical indices and assessments. The effects of both donor and recipient sex on disease and regenerative medicine treatment outcomes remains an important area of investigation.
The diagnosis of IBD vs alimentary lymphoma is a current area of intense research, and it is seemingly possible that, despite best practices, some of these cats were misdiagnosed. Assessment of biopsies from the stomach, duodenum, ileum and colon for histopathology and PARR analysis in each patient, as well as the random assignment of cats to the two groups, were considered important elements of the study design and would suggest that any misdiagnosis would also be randomly distributed between the two groups; however, with the small sample size, it is clearly possible that the distribution of any trait (eg, sex and body weight) would not be similar.
The results from this study are promising, but many questions remain regarding the use of MSC therapy in cats with IBD. Beyond the few anecdotal reports from this study, nothing is known about the long-term success, efficacy or side effects of this stem cell treatment in cats. With the prevalence of feline alimentary lymphoma, and the possibility that IBD progresses to this condition in cats, the potential impact of MSCs in cats with small cell lymphoma is an important area of research. Although the protocol for the preparation of MSCs for this study was relatively simple and appeared to be safe and effective, there are many ways that cellular therapy can be influenced prior to infusion into a patient, and a number of adjustments to MSC treatment protocols (eg, donor selection, culture medium, dose, frequency and number of infusions and route of administration) that may enhance accessibility, efficacy and duration of effect, and require further investigation.20,21
Conclusions
Although adipose-derived MSCs must be handled and processed under stringent laboratory conditions and practices, when processed correctly and administered IV as allogeneic, freshly cultured fMSC, they appear to have few if any immediate or longer-term adverse side effects in cats. The current study adds to the growing literature that supports the beneficial impact of MSC therapy for IBD in dogs and cats.
Footnotes
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was funded in part by grants from the Miller Trust, WINN Feline Foundation; Frankie’s Fund, and the CSU Feline Cancer Core.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognized high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers) for all procedure(s) undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
