Abstract
Objectives
The study primarily evaluated the long-term safety of intravenous, allogeneic uterine-derived mesenchymal stromal cells (UMSCs) for client-owned cats affected with refractory feline chronic gingivostomatitis (FCGS). Effectiveness during this long-term extension, when other medications were allowed, was also evaluated.
Methods
This prospective, multisite, open-label extension study evaluated cats with refractory FCGS after partial- or full-mouth extractions and persistent clinical signs despite medical management. During the initial trial, cats received two intravenous doses of 20 million UMSCs 14 days apart. No additional UMSC dosing occurred during the present follow-up period. A total of 35 cats were enrolled through day 365. Safety assessments included physical examination, owner-reported observations and clinicopathology. Clinical outcomes were evaluated descriptively using owner-reported assessment of overall response (ORA) and veterinarian-assessed global oral lesion score (GOLS). Concomitant medications were permitted.
Results
No serious adverse events (AEs) were associated with UMSC administration, and no clinically relevant abnormalities were observed via hematology or urinalysis. The most common AEs temporally associated with dosing were transient nausea, vomiting, tachypnea, diarrhea, hypersalivation and pyrexia. At 1 year, 69% of cats gained weight (mean increase of 16.2%). Improvement in ORA at days 180 and 365 was 75.8% and 65.6%, respectively. Improvement in GOLS at days 180 and 365 was 51.5% and 46.9%, respectively. Mean reduction in lesion score at 1 year was 33%, with 22% of cats achieving clinical cure. Of the cats, 66% did not restart medical therapy after UMSC treatment, with only 8.6% continuing an immunosuppressant drug.
Conclusions and relevance
Repeated intravenous UMSC therapy in cats with refractory FCGS appears safe in the long term, with no reported immunogenic or tumorigenic safety concerns. Clinically meaningful improvements in quality of life were maintained out to 1 year, with concurrent medication use as needed, and most cats were able to discontinue immunosuppressant medications. These results support the safe use of UMSCs for refractory FCGS.
Plain language summary
Feline chronic gingivostomatitis (FCGS) is a painful, long-lasting inflammatory disease of the mouth that can make it difficult for cats to eat, groom or interact normally. Even after full or partial tooth extractions, many cats continue to suffer from ongoing inflammation and pain, and often require lifelong medications that can have significant side effects. This study followed 35 client-owned cats with severe, treatment-resistant FCGS for 1 year after they received stem cell therapy made from uterine tissue donated during routine spay surgeries. The cats received two intravenous stem cell treatments 2 weeks apart during an earlier study and no additional stem cells were given during the 1-year follow-up period. During this time, cats were allowed to receive other medications as needed, reflecting real-world clinical care. Over the course of the year, the treatment was found to be safe. No serious side effects were linked to the stem cell therapy, and routine blood and urine tests remained normal. Mild, short-lived reactions such as vomiting or fever occurred in some cats shortly after treatment but resolved without lasting problems. Many cats showed meaningful improvement. Approximately two-thirds of owners reported better quality of life for their cats at 1 year, including improved appetite, activity and comfort. Veterinarians also observed gradual healing of oral lesions, with nearly half of the cats showing clear improvement and about 1/5 achieving complete resolution of visible mouth inflammation. Most cats were able to avoid restarting long-term immune-suppressing medications. Overall, this study suggests that intravenous stem cell therapy is a safe long-term treatment option for cats with severe, ongoing FCGS and may improve quality of life while reducing the need for medications that carry higher risks when used long term.
Keywords
Introduction
Feline chronic gingivostomatitis (FCGS) is an immune-mediated condition characterized by persistent, severe, ulcerative-proliferative oral inflammation. 1 Beyond localized oral pain that causes anorexia and weight loss, FCGS is associated with systemic immune dysregulation and extra-oral manifestations that complicate long-term management and severely diminish quality of life.2 –6
Current therapeutic protocols for FCGS involve surgical dental extractions, yet approximately two-thirds of affected cats remain refractory to this intervention. These patients typically require lifelong medical management, which is often limited by medication-associated toxicities and frequent clinical relapse upon drug withdrawal.2,4,7,8 Refractory disease results in euthanasia in approximately 10% of the FCGS population. 1
Mesenchymal stem cell (MSC) therapy is a novel treatment for cats with refractory FCGS.1,6,9 –11 MSCs possess potent immunomodulatory properties that address the complex immune aberrancies driving this and other inflammatory diseases.12,13 Uterine-derived MSCs (UMSCs), which can be harvested from young, healthy cats during routine ovariohysterectomy, offer a high level of potency, sterility, stability and purity. 14
We previously reported the short-term safety of UMSCs in refractory FCGS up to 90 days after treatment. 15 Although favorable long-term safety data have been described for adipose-derived MSCs in cats with refractory FCGS, the present study is the first to evaluate the long-term safety of UMSCs in this population treated concurrently with medications commonly used to manage refractory FCGS. 11
In addition to safety, our prior publication reported short-term effectiveness of UMSCs in refractory FCGS. By day 90, more than 70% of cats demonstrated clinically meaningful improvement in owner-assessed quality of life, and approximately 50% exhibited at least a 25% reduction in veterinary-assessed oral lesion scores. 15 In our experience, and in other MSC studies for refractory FCGS, improvement in quality of life typically precedes measurable oral lesion healing, potentially due to rapid effects on systemic inflammation and immune dysregulation.16 –18 Effects on mucosal healing appear to be slower, typically requiring more than 6 months to become appreciable and up to 18 months for full resolution, necessitating long-term follow-up for more accurate assessment. 10
The primary objective of this study was to evaluate the long-term safety of repeated, intravenously administered allogeneic UMSCs for the management of refractory FCGS. A secondary objective was to document effectiveness in an open-label clinical setting with concurrent medical management out to 1 year.
Materials and methods
This was an open-label extension of the previously reported 90-day safety and efficacy study. 15 After the short-term study, 35/46 cats continued to be evaluated for long-term safety and effectiveness under real-world conditions up to 365 days after treatment.
Investigators and animals
Investigators were board-certified veterinary dentists experienced in FCGS management. Client-owned cats with refractory FCGS that had undergone either full-mouth extraction (FME) or partial-mouth extraction (PME) were enrolled after client-signed informed consent was obtained. PME involved removal of all diseased teeth and associated roots. Patients were classified as refractory if clinical lesions and associated pain persisted for a minimum of 60 days after dental extractions, provided they were also receiving active medical therapy, aligning with previously published studies in this population.3,6,9,10
Investigational drug
Cats enrolled in this extension phase had previously received a two-dose regimen of allogeneic UMSCs during the initial 90-day trial. No additional cell therapy was administered during the present monitoring period. The manufacturing protocols, donor qualification and Current Good Manufacturing Practice expansion used for those initial treatments are described in detail in our prior report. 15
Study design
This multisite, single-arm, baseline-controlled clinical field trial was conducted at nine veterinary dental specialty hospitals across the USA. A historical control method (each cat served as its own control) was used and justified given the refractory nature of disease and poor response to conventional therapies, which is associated with an approximate 10% euthanasia rate.1,16 –19 The trial was conducted in compliance with Good Clinical Practice (VICH GL9) and approved by an independent ethical review committee. Owner informed consent was obtained for all cats.
In the first 90 days after treatment, immunosuppressant medications were withdrawn and certain medications were allowed if the cat had been receiving a consistent dose and schedule, as described previously. 15 In this open-label extension there were no restrictions on medication, allowing assessment of safety with concurrent medications as well as effectiveness under real-world conditions.
Study visits were conducted at days 180, 270 and 365.
Safety outcomes
Safety was assessed at each study visit through physical examinations, hematology, serum biochemistry, urinalysis, owner-reported observations and documentation of adverse events (AEs).
Effectiveness outcomes
Effectiveness was assessed during this open-label period, which followed the previously reported 90-day controlled efficacy phase. 15 Two outcomes were assessed at each study visit:
Owner overall assessment (ORA): each owner rated the cat’s overall response to treatment as greatly improved, mildly improved, unchanged or worsened. Cats rated as greatly or mildly improved were considered treatment successes.
Investigator-assessed global oral lesion score (GOLS): oral lesions were evaluated at eight predefined sites using the Modified Stomatitis Disease Activity Index described in our prior report, resulting in a total possible score of 24.6,15 Treatment effectiveness was defined by at least 25% reduction in total score relative to baseline.
Each visit also screened for changes in medical management, including discontinuation of pre-existing medications or addition of new therapies for refractory FCGS.
To minimize potential observer bias in this open-label study, both owners and investigating veterinarians were blinded to all prior assessment scores.
Statistical analysis methods
To maintain analytical continuity with the primary study, outcomes were modeled as binary events using a generalized linear mixed-effects model (SAS GLIMMIX) with a logit-link. This approach accounted for repeated measures through day 365 while incorporating clinic site, extraction history and extraction type as covariates to ensure comparability across the full 1-year data set. All statistical tests were conducted at the 0.05 level of significance. All analyses were performed using SAS v9.4.
Results
Animals
A total of 35 cats from the 90-day study were enrolled across nine clinical sites. Table 1 shows the total population completing the 365-day study along with the evaluable population for effectiveness and reasons for non-evaluability. All 35 enrolled cats were included in the safety analysis. For effectiveness, all cats completing study visits were considered evaluable. Cats receiving a prohibited medication that could impact assessment were automatically classified as treatment failures but remained in the effectiveness population to ensure a conservative success rate. Outcomes (success or failure) at the time new medication was administered were also considered.
Consort of cases in the safety and effectiveness population through day 365
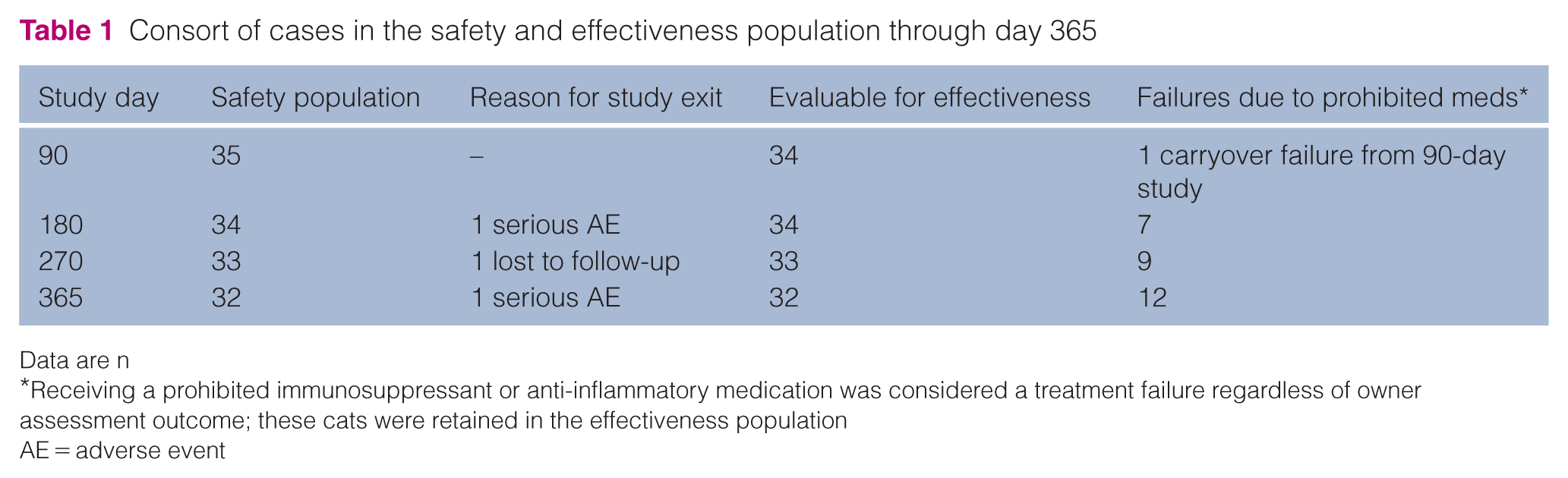
Data are n
Receiving a prohibited immunosuppressant or anti-inflammatory medication was considered a treatment failure regardless of owner assessment outcome; these cats were retained in the effectiveness population
AE = adverse event
In the population of 35 cats enrolled, 26 were castrated males and nine were spayed females. The most common breeds were domestic shorthair (n = 10), American Shorthair (n = 7), domestic mediumhair (n = 4) and domestic longhair (n = 4). The mean age of the cats was 6.9 years (range 1–14). The mean weight of the cats was 4.9 kg (range 2.9–7.6). Mean time from extraction was 271 days (range 60–1017). In total, 30 cats had undergone FME and five cats had undergone PME.
Before day 365, one cat was lost to follow-up and two were removed from the study due to an unrelated serious AE.
Several cats had pre-existing conditions, including feline calicivirus, feline infectious peritonitis, feline immunodeficiency virus (FIV) and diabetes mellitus. In addition, 12 cats exhibited mandibular lymphadenopathy and two had heart murmurs. Various other dermatologic, musculoskeletal and ophthalmic abnormalities were noted at screening but were not considered exclusionary or likely to affect study results.
Safety analysis
No serious AEs occurred related to treatment. Non-serious AEs that occurred within 24 h of treatment are summarized in Table 2. The following non-serious AEs were reported but not associated with treatment: weight loss, anemia, lameness, elevated alanine aminotransferase, hyperglycemia, bilirubinuria, azotemia, hair pulling, alopecia, thrombocytopenia, asthma, elevated white blood cell count, hypotrichosis, hypoalbuminemia, blepharospasm, respiratory congestion and hemoptysis; none of these were associated with UMSC administration. The causality of non-serious AEs, as determined by the investigators, were as follows: 3.0% (1/33) possible, 0.0% (0/33) probable, 6.1% (2/33) unclassifiable, 9.1% (3/33) inconclusive and 81.8% (27/33) unlikely.
Non-serious adverse events reported within 24 h of treatment

Efficacy data as previously published 15
One cat had tachypnea on both administration days
One cat had pyrexia associated with injection on both administration days; a second cat had pyrexia on one of the two administration days
Routine clinical pathology assessments, including hematology, serum biochemistry and urinalysis, showed no clinically relevant trends in values during the 365 days after the start of UMSC treatment. No AEs were considered related to immunogenicity by the investigators.
Three serious AEs occurred in three cats, all unrelated to UMSC administration, with one cat recovering and two cats euthanized. The surviving cat was reported to have an undiagnosed abdominal mass that resolved and a mass on the forelimb that resolved with time and corticosteroid treatment. No biopsies were performed. Neither of the masses were considered related to UMSC administration. The forearm mass may have been related to catheter placement. Of the two cats that were ultimately euthanized, the first presented with proliferative FCGS and was diagnosed with widespread squamous cell carcinoma (SCC) of the oral cavity and brain during the prior 90-day study. Euthanasia was elected shortly thereafter, on day 115. This case of SCC was considered likely to be pre-existing and undiagnosed at the time of enrollment (no biopsy at the time of enrollment). No evidence of treatment-related local or systemic tumor formation was observed during the study period. The final cat experiencing a serious AE was euthanized on day 315 after the owner called to report poor quality of life with ptyalism, anorexia and likely respiratory disease, all of which were deemed unrelated to UMSC therapy.
The incidence of non-serious and serious AEs was 91.4% and 8.6% per cat, respectively.
Medical management
Of the 35 cats, 28 (80%) received an analgesic medication, most commonly gabapentin or buprenorphine; 14 (40%) cats received either an immunosuppressant or an anti-inflammatory medication; and 10 (28.6%) cats received medications in response to an AE, most frequently cyclosporine, cefovecin, maropitant or fluids. Of the cats, 71% did not require treatment for AEs.
Effectiveness analysis
At days 180, 270 and 365, effectiveness analyses included 34, 33 and 32 evaluable cats, respectively. Effectiveness outcomes over time are provided in Table 3.
Effectiveness outcomes over time

GOLS = global oral lesion score; ORA = owner overall assessment
At day 365, 11 cats in the effectiveness population were automatically classified as treatment failures because they received new concomitant immunosuppressant or anti-inflammatory medications. Despite this designation, 90.9% of owners rated these cats as mildly or greatly improved. Of the 35 cats, 12 (34%) received at least one dose of cyclosporine or corticosteroids during the study. Of the 12 cats, only three (8.5%) received ongoing immunosuppressant medication. The other nine cats received immunosuppressant medication only once or for a short period before discontinuation. Of the enrolled cats, 23 did not have an immunosuppressant medication added in the open-label period.
Veterinary-assessed GOLS improved steadily over the study period, with mean scores decreasing from 10.9 at baseline (n = 35) to 8.5 at day 90 (n = 33) and 6.8 at day 365 (n = 32). The mean percentage reduction in total lesion score was −19.8% by day 90, −30.3% by day 180, −41.4% by day 270 and −33.5% by day 365. Two cats in the failure group had a clinically relevant increase in GOLS between days 270 and 365, decreasing the mean reduction/improvement in GOLS.
Success rates for ORA and GOLS at day 365 were recorded for both the PME and FME groups; however, because the number of PME cases was small (n = 5), these outcomes are presented for descriptive purposes only. In the PME group, 100.0% of cats were considered successes for ORA and 80.0% for GOLS. In the FME group, 96.3% of cats were considered successes for ORA and 55.0% for GOLS.
Of the cats, 22% achieved a clinical cure (GOLS of zero at day 365), all of which were in the FME group. Of these, only one cat has received an add-on immunosuppressive medication.
Discussion
The primary findings of this study support the long-term field safety of intravenous UMSC therapy. There were no serious AEs associated with UMSC administration, and all reported non-serious AEs related to treatment occurred during the first 24 h of treatment.
The UMSCs utilized in this trial were well tolerated through day 365, with no clinically relevant safety concerns identified regarding systemic tolerance or concurrent medication use. These longitudinal observations are consistent with a recent review of feline literature covering 215 cats, which found that the majority of MSC-treated patients experienced no serious AEs during follow-up periods of up to 9 years, including no evidence of treatment-related tumor formation.11,20
The present safety outcomes are supported by descriptive clinical effectiveness data. More than 65% of cats exhibited sustained meaningful clinical improvements out to 1 year, as indicated by quality-of-life owner-based assessments and veterinarian-assessed lesion assessments, with oral lesion scores improving in approximately 50% of cats. Of note, 22% of cats achieved clinical cure, based on a GOLS of zero.
In the prior 90-day study, owners frequently reported increases in appetite, activity and social interactions within hours of treatment, suggesting a rapid onset of benefit beyond what would be expected from administration of 20 ml fluids alone (Gallant, unpublished data).15,21 This rapid response contrasts with prior reports describing delayed clinical effects after MSC therapy.21 –23
FCGS is recognized as an immune-mediated condition with systemic involvement beyond the oral cavity, including behavioral disturbances.2 –4 Oral therapies alone, such as tooth extractions, may inadequately address underlying immune dysregulation. In contrast, intravenously administered UMSCs exert systemic immunomodulatory effects. Previous studies have shown that MSCs modulate T-cell activity, dampening lymphocyte proliferation and supporting CD8+ function.24 –28 These systemic effects may contribute to early improvements in quality of life that precede improvement in oral lesions, which may take up to 18 months to resolve.10,29
It should be noted that differences in administration route, manufacturing processes, cell quality and potency, and clinical outcome measures may influence study results and complicate comparisons across MSC sources. The biological characteristics of the UMSCs evaluated for refractory FCGS in this study are specific to this product and have been established using validated, proprietary assays.
A reduction of 25% in GOLS was selected to represent a clinically relevant improvement in oral lesion scores. In this study, 100% of cats with at least 25% improvement in GOLS were also considered successes in terms of ORA, supporting the correlation and clinical relevance of the selected threshold. This is consistent with our previously reported short-term study. 15 Two cats experienced an increase in GOLS and were therefore classified as lack-of-efficacy cases and managed as treatment failures for the purposes of study analysis. Of note, this classification did not preclude the possibility of perceived clinical benefit in individual cases. Some cats demonstrated improved quality of life based on owner assessment without corresponding improvement in GOLS, whereas all cats achieving at least a 25% improvement in GOLS demonstrated concordant improvement in owner-assessed quality of life.
It is clinically relevant to note that the majority of cats (91.5%) did not require the addition of an ongoing immunosuppressant or anti-inflammatory drug to manage clinical signs during the long-term extension, and 22% of cats achieved a GOLS of zero. Of the cats with healed oral lesions, 100% were considered successes for owner assessment of quality of life.
The assessment of effectiveness in this study was potentially impacted by the use of immunosuppressive and anti-inflammatory medications allowed during the open-label extension period. However, cats that were given a new medication that may have impacted effectiveness were considered treatment failures and not included in the success group. These cats had such medications withdrawn before UMSC treatment and during the first 90 days, as previously reported. 15
The case of oral SCC observed in this cohort was deemed likely pre-existing and unrelated to UMSC therapy. The rapid detection of extensive brain metastasis within 1 month of enrollment suggests the malignancy was present but undiagnosed at baseline, a risk further compounded by the cat’s FIV-positive status.30,31 Given the recently reported association between chronic oral inflammation and SCC in cats, the co-occurrence of SCC in this population reinforces the necessity of performing pre-treatment biopsies on proliferative lesions to exclude SCC before initiating UMSC therapy.30,32
The non-tumorigenic nature of UMSCs is supported by their multipotency, which restricts their differentiation potential compared with pluripotent cells.33,34 The UMSCs remained phenotypically and genotypically stable through at least two passages beyond the final clinical formulation. Furthermore, biodistribution studies indicate that MSCs function primarily through transient paracrine activity (often called a ‘hit-and-run’ mechanism) rather than long-term tissue engraftment.35,36
Although the lack of a contemporaneous control group is a limitation, placebo use in cats with painful, refractory disease presents ethical challenges, prompting the use of historical controls and real-world evidence to assess safety, aligning with recent guidance from the Food and Drug Administration.16 –19,37 An additional limitation is the low number of cats with PME, such that no definitive conclusions can be made about this subgroup of refractory FCGS cats.
Additional studies are warranted to further evaluate bolus dosing, mechanisms by which MSCs modulate systemic immunity, the impact of extraction type on long-term treatment response and tissue healing, and the role of viral dynamics in refractory FCGS.
Conclusions
This prospective, baseline-controlled study suggests that repeated intravenous administration of allogeneic UMSCs is safe in cats with refractory FCGS undergoing concurrent medical management, and approximately two-thirds of cats demonstrated sustained clinical improvement. UMSCs represent a cell therapy that supports tissue healing by targeting the underlying immune dysregulation of FCGS and improving quality of life. Use of UMSCs may also reduce the need for drugs with unfavorable safety profiles, particularly with chronic usage.
Footnotes
Acknowledgements
This clinical study was conducted with contributions from several board-certified dental specialists serving as clinical investigators. Manuscript drafting support provided by Will Pass, DVM, of Pass Medical Writing, with compensation provided by Gallant.
Accepted: 16 February 2026
Author note
The data supporting this study are on file with the study sponsor and are not publicly available. Access may be granted upon reasonable request to the corresponding author or study sponsor.
Conflict of interest
The following authors are employed by Gallant Therapeutics: VSW, JLS, CW, NT, LB; and the following authors received consulting fees from Gallant Therapeutics; RP, CT, MSR, BA.
Funding
This study was funded by Gallant Therapeutics, San Diego, CA, USA.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals and procedures that differed from established internationally recognized high standards (‘best practice’) of veterinary clinical care for the individual patient. The study therefore had prior ethical approval from an established (or ad hoc) committee as stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers, tissues and samples) for all procedure(s) undertaken (prospective or retrospective studies). For any animals or people individually identifiable within this publication, informed consent (verbal or written) for their use in the publication was obtained from the people involved.
