Abstract
Rhesus macaques (Macaca mulatta) are used extensively in biomedical research, often with a focus on the gastrointestinal tract, and yet a full characterization of their normal resident intestinal cell populations has not been published. In addition, chronic enterocolitis (CE), also known as idiopathic chronic diarrhea, affects up to 25% of colony-housed rhesus macaques, often requiring euthanasia for welfare concerns and severely limiting their value as a breeding animal or research subject. We aimed to characterize subjective and objective variables in sections of the ileum, cecum, colon, and rectum in 16 healthy rhesus macaques and compare these results with a cohort of 37 animals euthanized for CE to produce relevant diagnostic thresholds and to improve case definitions for future studies. We found neutrophils to be an infrequent but expected component of the large intestinal leukocyte population. Animals with CE had significantly increased total leukocyte populations between crypts in the cecum, colon, and rectum; variable increases in specific cell populations across all levels of the distal intestinal tract; and significantly increased intraepithelial CD3+ T cells in the colon and rectum. Concentrations of enteroendocrine cells, enterochromaffin cells, and intestinal mast cells were not significantly different between healthy and affected individuals. This study characterizes individual leukocyte populations in the rhesus macaque lower intestinal tract, is the first to evaluate rhesus macaque intestinal mast cells, and provides key diagnostic thresholds for evaluating animals with potential CE.
Rhesus macaques (Macaca mulatta) are a critical resource in biomedical research across numerous fields, and more recently, cynomolgus macaques (Macaca fascicularis) and rhesus macaques have experienced increased demand due to COVID-19 research.8,29 However, up to 25% of colony-housed rhesus macaques develop a syndrome of chronic intestinal inflammation resulting in chronic diarrhea, weight loss or poor weight gain, and unthriftiness, which severely limits an individual’s value as a research subject or member of a breeding colony.15,23,24 This syndrome has been termed idiopathic chronic diarrhea or idiopathic colitis in recent literature.2,11,15 The disease has been investigated for over 3 decades, but the cause remains unknown, although various infectious organisms and noninfectious catalysts have been implicated.2,4,7,11,15 We propose the term chronic enterocolitis (CE), as it is more descriptive of the histologic abnormalities in both the large intestine and distal small intestine, and does not include the descriptor “idiopathic,” which is likely to become a misnomer as the disease continues to be characterized. Infectious causes of chronic colitis with potentially similar histologic findings should be ruled out prior to diagnosis via culture or other ancillary diagnostics.2,15 As yet, there are suspicions of, but no definitive correlation to, a human disease counterpart.15,26 Histologically, CE in rhesus macaques has been compared with microscopic colitis, diarrhea predominant irritable bowel syndrome (IBS), and postinfectious IBS.2,15,27,28 The syndrome is most common in male macaques less than 5 years of age, but females and older individuals are also affected. 15 CE is often a subjective clinical and/or histological diagnosis with variable case definitions, heavily relying on the clinician’s and pathologist’s prior experience with the syndrome to describe clinical signs, severity, and possible confounding factors. Grossly, the affected animals typically have a distended colon containing watery, nonhemorrhagic material and the mucosa is thickened, with histology classically demonstrating a lymphoplasmacytic colitis.13,15 However, in the authors’ experience, clinically healthy captive macaques can have subjectively higher numbers of resident leukocytes in their intestinal tract than would be considered normal for many domestic species, complicating the interpretation of the significance of intestinal inflammation. There is a need for a standardized histopathological approach to evaluating the normal inflammatory cell population of the healthy captive rhesus macaque intestinal tract to provide a baseline for future studies on CE and other intestinal diseases of macaques.10,30,31 Previous semiquantitative studies of the intestinal tract have been primarily focused on the lymphocyte composition and cytokine profiles.15,21,32 However, by developing a normal baseline for distal intestinal leukocyte populations as well as other qualitative and semiquantitative measures and documenting the expected findings with respect to CE, thresholds can be developed. These, in conjunction with clinical findings and absence of a detectable infectious etiology, will allow for the creation of a specific case definition for use in future studies as well as in diagnostic cases, particularly for those with less nonhuman primate experience.
General histological characteristics of the macaque gastrointestinal tract have been previously described.3,13,16 Briefly, the ileum has shorter villi than the remainder of the small intestine and can appear blunted and fused near the ileocecal junction, and lacteals may be prominent throughout the small intestine. Occasional foci of pigmented macrophages are present throughout the lamina propria of the entire intestinal tract. The large intestinal mucosal glands should be of uniform height and contain a relatively high number of goblet cells.3,16 However, to our knowledge, no uniform, quantitative or semiquantitative evaluation of the rhesus macaque inflammatory cell population in the intestinal tract is available. A standardized grading scheme exists for dog and cat gastrointestinal endoscopic biopsies, produced by the World Small Animal Veterinary Association (WSAVA) Gastrointestinal Standardization Group, and we applied similar methods here, in conjunction with methods previously proposed for semiquantitative analysis of intestinal sections of rhesus macaques.5,15 In this study, we describe subjective findings of the ileum, cecum, colon, and rectum, and assess crypt hyperplasia, and individual leukocyte populations. In addition, using immunohistochemical (IHC) markers, we assessed normal populations of mast cells (MCs), enterochromaffin cells (ECs), enteroendocrine cells (EECs), and intraepithelial CD3+ T lymphocytes (IEL) across all age groups, with the exception of neonates, and both sexes of rhesus macaques. These cell populations have been variably shown to have significant differences in cases of CE and are often the target of inflammatory bowel disease (IBD), IBS, and postinfectious IBS (PI-IBS) human studies.6,9,14,15,27,28 We then compared the cell compositions of clinically healthy animals with a cohort of animals clinically diagnosed with CE. These findings should allow for future correlations of macaque surgical or endoscopic biopsies with human studies in hopes of providing an accurate animal model to continue research of IBD, IBS, and PI-IBS, and allow for more precise comparisons across study populations in future research.
Materials and Methods
Animals
The Keeling Center for Comparative Medicine and Research (KCCMR) at The University of Texas MD Anderson Cancer Center is an The Association for Assessment and Accreditation of Laboratory Animal Care accredited facility where animals are cared for in accordance with the USDA Animal Welfare Act and regulations and the Guide for the Care and Use of Laboratory Animals, and established Institutional Animal Care and Use Committee policies were followed. 25 Animals were part of an approximately 1000 Indian-origin rhesus macaque breeding colony specific pathogen free for Simian immunodeficiency virus (SIV), Simian retrovirus (SRV) type I, Simian T-lymphotrophic virus (STLV), and herpes B virus since 1991. Animals were housed on the KCCMR campus in outdoor gang-cages or “corn crib” structures each housing a single group of monkeys with approximately 25 square feet available per animal and daily enrichment activities. Cases were selected by reviewing necropsy records between 2014 and 2021 for animals submitted with a clinical diagnosis or suspicion of CE. During this time, the rhesus macaque colony at KCCMR was under the supervision of the same laboratory animal veterinarian, providing continuity of diagnostic methods, treatments, and case endpoints. Animals at KCCMR are diagnosed with presumed CE if they have had repeated episodes of diarrhea, particularly those that require hospitalization, with diminishing response to supportive care, concurrent weight loss, historic or recent negative stool cultures, and lack of evidence for other intestinal diseases such as gastric dilation, palpable intestinal masses, or positive fecal occult blood tests. Archived slides were reviewed from each case. Cases were included if the generally accepted features of CE were present (ie, lymphoplasmacytic infiltrates, crypt abscesses, epithelial damage) and tissues were in fair to good preservation condition. Cases where autolysis affected the luminal epithelium were kept for mucosal leukocyte analysis but excluded from immunohistochemistry analysis. Cases with severe autolysis were excluded. This method yielded 37 cases for inclusion in the study. In addition, all clinical records were reviewed and variables, including number of episodes of diarrhea, episode duration, previous fecal culture results, antimicrobial use, and immunosuppressive drug use were recorded. Healthy control animals were selected for analysis by reviewing 120 necropsy records between 2014 and 2021 for animals that were submitted for examination under a “colony health” designation. From this set, controls were chosen for inclusion, with an attempt at age and sex matching, if they had no history of hospitalizations for enteric-related disease and distal small intestine, large intestine, and rectum histology were recorded as normal to subjectively minimally increased lymphoplasmacytic infiltrates. Animals with no archived histology available, neonates, those with intestinal neoplasia, or those with significant autolysis were excluded, yielding 17 animals for use as controls. However, on review of archived slides for the control animals, 1 animal was found to have significant erosion and inflammation of the ileum and cecum; therefore, this animal was removed from the study, leaving 16 animals as healthy controls.
Semiquantitative and Qualitative Histological Assessment
Each individual had been previously subjected to a complete necropsy and microscopic evaluation. Tissues fixed in 10% neutral-buffered formalin were routinely processed for histopathology. Representative sections were embedded in paraffin, sectioned at 5 µm, stained with hematoxylin and eosin, and examined by a senior pathology resident (RLB) who earned board-certification over the course of the study. Consultation was conducted on approximately 10% of the slides to ensure agreement with a board-certified pathologist (CLH) who was blinded to the disease status of the examined sections. Examined sections of the intestinal tract included terminal ileum proximal to the ileocecal junction, cecum, colon, and rectum proximal to the recto-anal junction. Several cases had multiple sections of colon available for examination, from which the best-preserved section was selected for analysis. While the primary examiner was not blinded to the disease status of sections, the examiner was blinded to all other status markers of each animal during slide evaluation, including age, sex, and duration of clinical signs. Given this, every effort was made to randomly select fields, and specific areas within fields, for microscopic evaluation. To avoid differences due to oblique sectioning, examined regions of the intestine were included if the entire crypt or crypt pair was visible from basal lamina propria to lumen and a majority of each crypt lumen was similarly visible. Occasionally, due to sectioning and necessity of available fields, crypts that fell partially out of section were included, but quantitative assessments of these areas were only made in the original plane of section, not including additional areas that filled the previous crypt space. No more than 2 crypt pairs were included in assessments in any given field. Inflammatory infiltrates often did not diffusely affect sections, and as such, efforts were made to include both heavily affected and unaffected regions, including closely apposed crypt pairs, to avoid visual bias toward heavily infiltrated segments. Regarding assessment of ileal villi, similar measures were applied and included villi were always of similar height across an individual section, with the central lacteal typically visible.
Methods for histological assessment were adapted based on a combination of previous reports and the WSAVA guidelines for dogs and cats.5,15 All quantitative assessments were performed using standard 10×, 20×, or 40× objectives on microscopes with a field number (FN) 22 mm wide field of view. Any time a reference is made to a 40× field, this is equivalent to 0.237 mm2 and quantitation made over ten 40× objective fields are equivalent to 2.37 mm2. For each section of ileum, villous length was graded from none to severe blunting on a scale of 0 to 6, and lacteal dilation and villous fusion were graded none to severe on a scale of 0 to 3. For all sections of large intestine, surface epithelium tufting was graded none to severe on a scale of 0 to 6. For all tissues, surface epithelium damage and submucosal lymphocytes were graded none to severe on a scale of 0 to 6 and crypt abscesses were graded none to severe on a scale of 0 to 3. In addition, the absence, multifocal presence, or diffuse presence of a “blue brush border” was recorded, and the presence or absence of crypt herniation, amyloid, or fibrosis was recorded. The blue brush border is composed of basophilic bacteria, Helicobacter macacae, which adhere to the superficial epithelium of the intestine, and loss of this is a recently reported feature in macaques with CE, indicative of dysbiosis.13,15 In all tissues, the presence of luminal bacteria, intraepithelial bacteria, luminal Balantidium coli (large ciliated protozoa), and intraepithelial B. coli were graded from none to abundant on a scale of 0 to 4. The complete grading scheme for these qualitative variables is available in Supplemental Table S1.
For quantitative assessments of leukocytes in the ileum lamina propria, the number of villous lymphocytes, plasma cells, eosinophils, neutrophils, and macrophages distending villi were counted and averaged over 5 villi, again, with efforts made to evaluate only villi of uniform height in any given section. Similarly, the number of each cell population expanding the entirety of the lamina propria separating crypts from the crypt base to the mucosal surface was averaged across 10 crypt pairs, and the number of each population in the basal lamina propria, the region adluminal to the crypts, was averaged over 10 crypts. Totals of each population were summed to provide a total number of leukocytes in each section. If mucosa-associated lymphoid tissue (MALT) was present in a section, these areas were strictly avoided in leukocyte assessments with at least 2 crypt spaces between the MALT and the next examined region included for assessment. Sections of cecum, colon, and rectum were identically evaluated, with the exception of villous leukocyte measurements. Goblet cells were also quantitatively evaluated using a mean number of goblet cells per 100 enterocytes over a total of 500 enterocytes. Goblet cell counts were performed on randomly selected fields with contiguous luminal and crypt epithelium in good orientation. Counts made per 100 enterocytes were made only in areas where delineation between individual enterocytes with a single layer of visible nuclei were clearly apparent. Areas where there was any concern for overlap of layers of enterocytes were not included in these evaluations. Mucosal thickness was recorded relative to the height of a single 40× field (ie, 0.25–1.5 or more), and crypt mitoses were averaged per 40× field over a total of 10 fields. For all evaluations of the rectal mucosa, evaluations began in the first field of acceptable orientation closest to the squamocolumnar junction when the junction was available for assessment. Otherwise, randomly selected fields were evaluated in the section as with the ileum, cecum, and colon.
Immunohistochemistry
IHC evaluations were performed on ileum, colon, and rectum samples from 6 healthy animals and 15 case animals. The cecum was excluded from IHC analysis to minimize cost as a partial goal of this study was to provide comparison for future biopsy studies, as well as capability for human histologic correlations, and the cecum is an unlikely biopsy site. Recent adjustments to the WSAVA guidelines for dogs with IBD recommend inclusion of ileal biopsies, and the terminal ileum in particular is a common biopsy site during human colonoscopy procedures.1,17 Slides were processed routinely and labeled as follows: EECs were identified using anti-chromogranin A mouse monoclonal antibody clone LK2H10+PHE5+CGA/414 (Novus Biologicals, Centennial, CO), ECs were identified using anti-serotonin mouse monoclonal antibody clone 5HT-H209 (Novus Biologicals, Centennial, CO), MCs were identified using anti-tryptase mouse monoclonal antibody clone AA1 (Abcam, Waltham, MA), and IEL were counted using anti-CD3 rabbit monoclonal antibody, clone EP41 (Biocare Medical, Pacheco, CA). Briefly, 4 µm tissue sections were adhered to positively charged slides and deparaffinized, cleared, and brought to water. Slides were immersed in 95°C Target Retrieval Solution (Dako, Glostrup, Denmark, Europe) for 20 minutes followed by a gradual cooling step. Slides were then immersed and rinsed in wash buffer. The next steps were followed sequentially by a rinse with wash buffer and performed on the Fisher Autostainer: endogenous peroxidase block using dual enzyme block (Dako), Background Sniper (Biocare), Fc Receptor Block (Innovex), primary antibody for 30 minutes, secondary polymer (Envison+, DAKO), 3,3′-diaminobenzidine substrate for 10 minutes (DAKO), and counter stained for 5 minutes with automated hematoxylin (DAKO). Slides were then dehydrated through alcohol gradients and xylene and then permanently coverslipped. Positive control tissues used to optimize the antibodies and included as a slide in each run on the autostainer included pancreas for chromogranin A, uterus for tryptase, and colon for serotonin. Peyer’s patches were used as internal positive controls for CD3 immunolabeling and appropriate internal negative controls were used for all antibodies.
MCs in the villi for the ileum and between the crypts and in the basal lamina propria in the ileum, colon, and rectum, were counted as described for the leukocyte populations above, but not included in the overall leukocyte totals as intestinal MCs cannot be readily identified using standard staining practices. Any cell that had the morphology of an MC, or was not distinctly one of the above populations, in hematoxylin and eosin-stained sections was not included in the other leukocyte population evaluations. CD3+ IELs were counted based on the mean number of labeled cells per 100 enterocytes over a total of 500 enterocytes. The average number of EECs per crypt over 10 crypts was counted. ECs were counted per 40× field and averaged over 10 fields.
Statistical Analysis
All statistical analyses were performed using R (version 4.2.2) in R studio. 22 Variables were subjected to the Shapiro-Wilk test for normality and the vast majority failed, resulting in utilization of the Wilcoxon signed-rank test for significance. Differences were considered statistically significant if P values were less than .05. Reference range calculations were performed with 95% certainty and 90% confidence intervals. Threshold calculations were performed using the area under the receiver-operating characteristic (ROC) curve in the 2-state setting with 95% certainty.18,19,20 The full data set for individual animals is available in Supplemental Table S2.
Results
Sixteen individuals were identified, which fit the control animal selection criteria: 6 males and 10 females ranging in age from 9 months to 24 years of age. Further characterization of the control animals, including reason for submission and final diagnosis, is available in Supplemental Table S3. Thirty-seven individuals were identified with a final diagnosis of CE with adequate histology for evaluation. The age at onset of disease ranged from 4 months to 19 years and the age at euthanasia ranged from 1 year to 21 years. There were 11 males and 26 females in the case population. The cases were recorded as having at least 2 episodes of diarrhea, which required hospitalization, although the vast majority (35/37) had 3 or more episodes, with an average of 7.7. All cases were euthanized due to poor prognosis and being refractory to treatment, including various antimicrobials and immunosuppressives. While 19/37 cases were intermittently positive on fecal culture for known enteric pathogens, all cases’ most recent cultures were negative and there was no evidence of a specific pathogenic agent referable to the clinical signs at time of the necropsy. No cases with previously undetected infectious agents were identified.
Means and standard deviations of the cell counts and crypt mitotic activity of the controls across the ileum, cecum, colon, and rectum are summarized in Table 1. Subjective findings are summarized below. Representative images of the normal animals’ large intestines are depicted in Fig. 1, and representative images of selected cell populations from the colon and rectum are shown in Fig. 2. In the ileum, 2/16 (12) controls had mild to moderate villous blunting, 3/16 (19%) had scattered villous fusion, 1/16 (6%) had minimal epithelial erosion, 4/16 (20%) had scattered submucosal leukocyte infiltration, including some MCs, and 4/16 (20%) had scattered to moderate lacteal dilation. Crypts occupied up to 0.5 40× fields and the mucosa was uniform. No controls had crypt abscesses, crypt herniation, amyloid, fibrosis, intraepithelial bacteria, luminal B. coli, or intraepithelial B. coli in the ileum. Ten of 16 (62%) controls had small to moderate amounts of mixed luminal bacteria, which is expected in the intestinal tract. With respect to leukocyte populations in the ileum, the quantity of villous leukocytes was variable, with less variability between crypts and in the basal lamina propria. A single control animal had a single neutrophil in the villous region, but otherwise neutrophils were not appreciated in the ileum of control animals. All controls had a small number of leukocytes in the basal lamina propria. As noted above, ECs tended to be dispersed randomly throughout the crypts and luminal epithelium while EECs were primarily concentrated in the crypts. CD3+ IELs were variable and occasionally numerous, with up to 42 labeled cells per 100 enterocytes compared with the average of 16.6, although they were evenly dispersed. Overall, lymphocytes represent the primary leukocyte in the ileum, composing 50% to 66% of the leukocytes in any given section.
Means and standard deviations of cell counts and crypt mitotic activity of controls and animals with chronic enterocolitis across the ileum, cecum, colon, and rectum. Results for variables with a significant difference (P < .05) from controls are in bold.
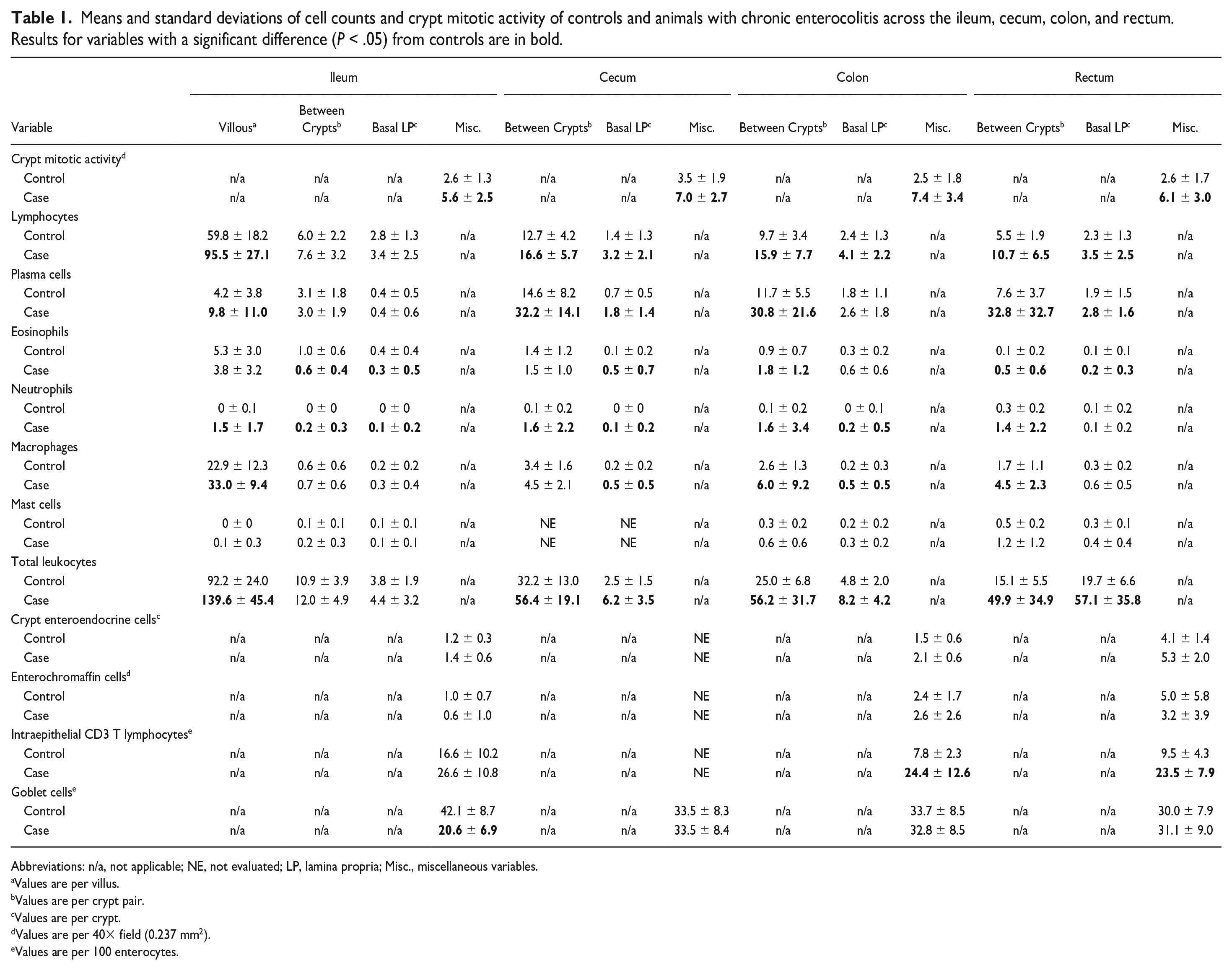
Abbreviations: n/a, not applicable; NE, not evaluated; LP, lamina propria; Misc., miscellaneous variables.
Values are per villus.
Values are per crypt pair.
Values are per crypt.
Values are per 40× field (0.237 mm2).
Values are per 100 enterocytes.
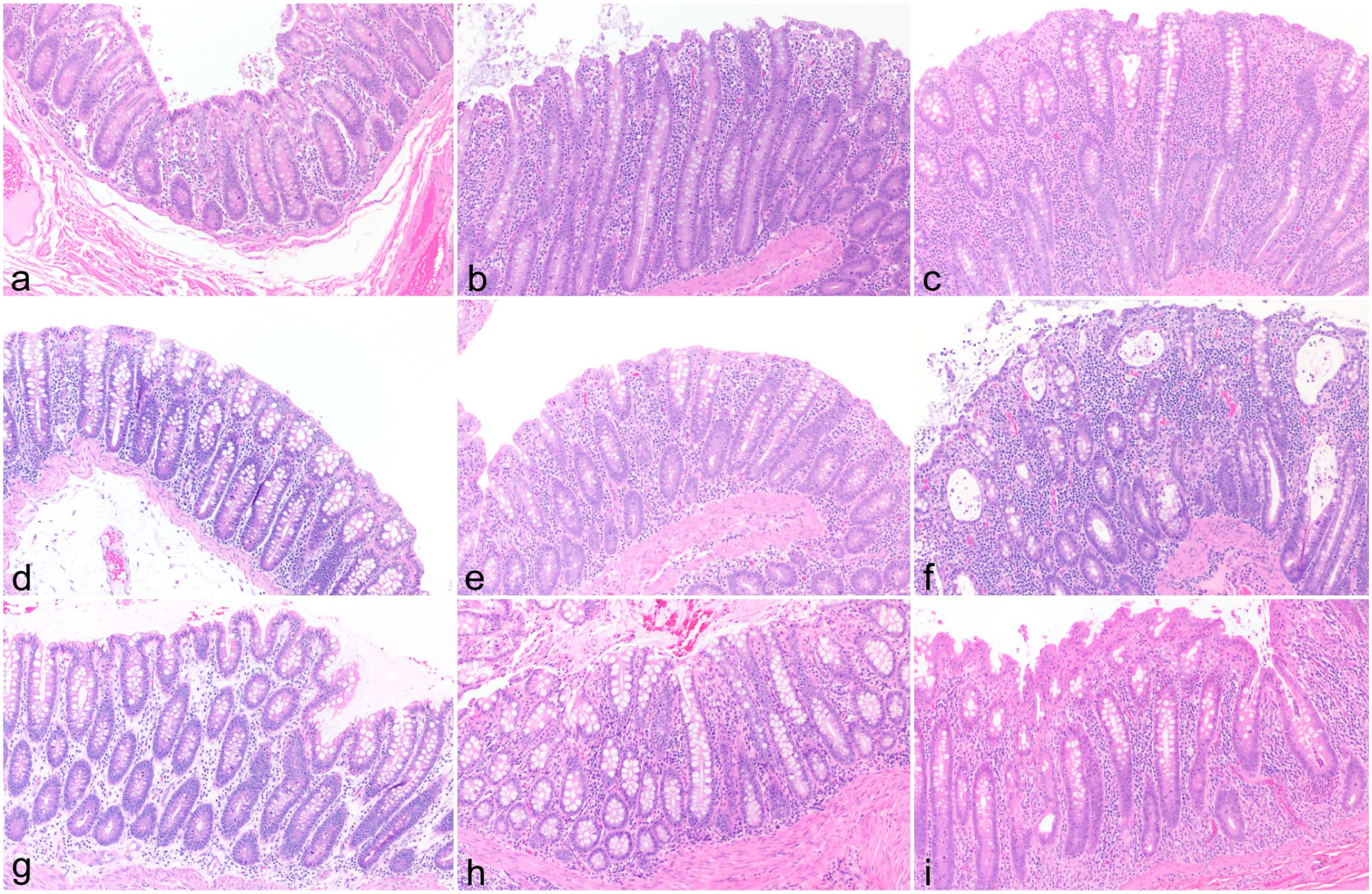
Large intestinal sections from rhesus macaques. Hematoxylin and eosin (HE). (a) Cecum from a healthy individual. (b) Cecum from an animal with total leukocytes between crypts at the threshold value (ie, borderline). The epithelium is mildly irregular and the mucosa is increased in thickness. (c) Cecum from an animal with total leukocytes above the threshold value. Crypts are separated by abundant leukocytes, with significant increases in lymphocytes, plasma cells, and neutrophils between crypts and all cell populations in the basal lamina propria. The epithelium is irregular and the mucosa is markedly thickened. (d) Colon from a healthy individual. (e) Colon from an animal with total leukocytes between crypts at the threshold value. (f) Colon from an animal with total leukocytes above the threshold value. In addition to significantly increased leukocytes, including lymphocytes, plasma cells, eosinophils, neutrophils, and macrophages between crypts and lymphocytes, neutrophils, and macrophages in the basal lamina propria, this section also demonstrates severe changes of chronic enterocolitis, including mucosal hyperplasia, crypt abscesses, and epithelial tufting and erosion. (g) Rectum from a healthy individual. (h) Rectum from an animal with total leukocytes between crypts at threshold value. (i) Rectum from an animal with total leukocytes above threshold value with increases in each cell population between crypts and lymphocytes, plasma cells, and eosinophils in the basal lamina propria.
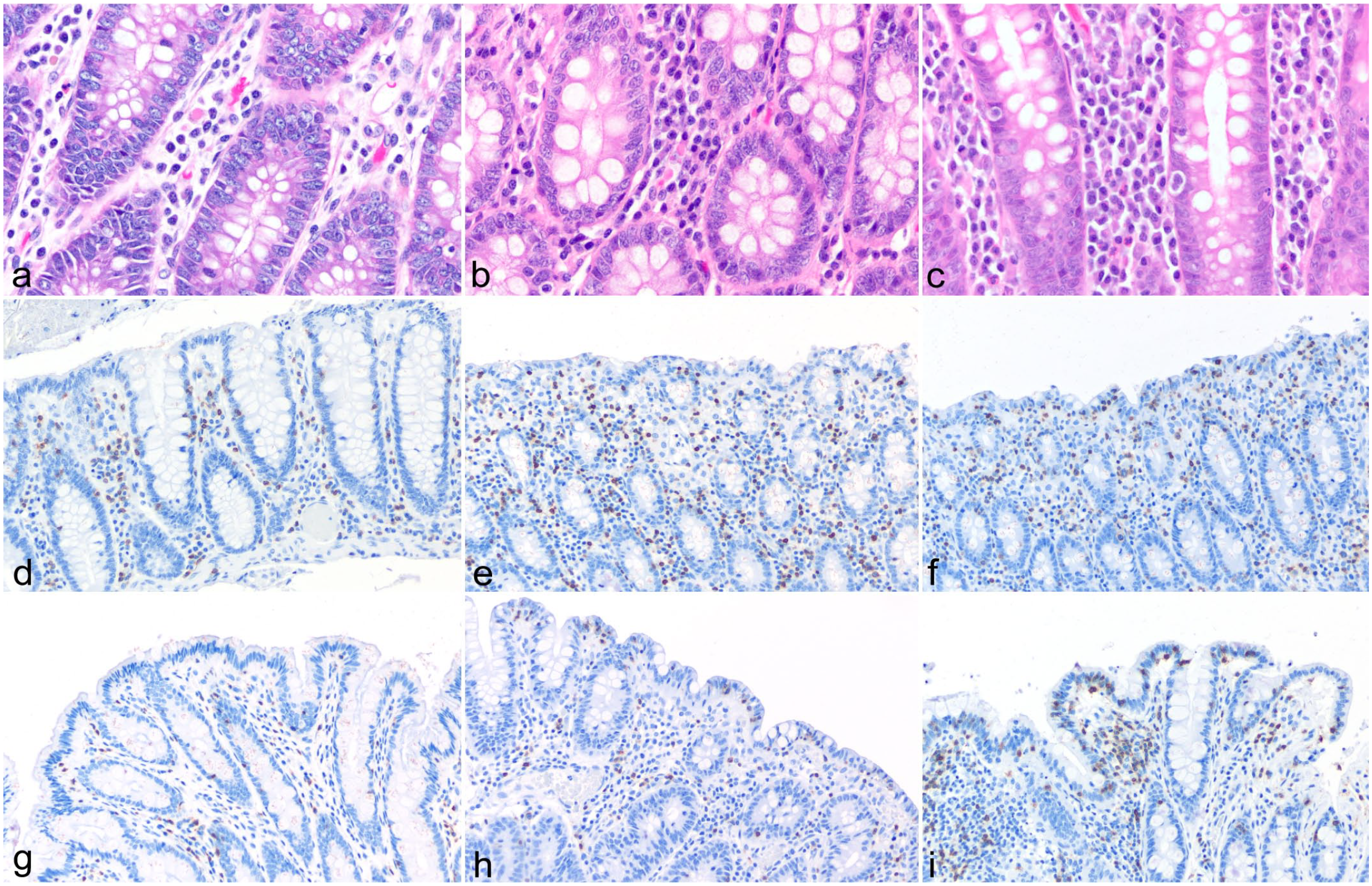
Large intestinal sections from rhesus macaques. (a–c) Hematoxylin and eosin. (d–i) CD3 immunohistochemistry. (a) Rectum from a healthy individual with prominent plasma cells between crypts. (b) Rectum from an individual with borderline increased plasma cells between crypts. (c) Rectum from an individual with plasma cells between crypts above threshold value. Significant increases in eosinophils, neutrophils, macrophages, and lymphocytes are also appreciated. (d) Colonic CD3+ intraepithelial lymphocytes (IELs) from a healthy individual. (e) Colonic CD3+ IEL from an animal with borderline elevated numbers. (f) Colonic CD3+ IEL from an animal with significant increases. (g) Rectal CD3+ IELs from a healthy individual. (h) Rectal CD3+ IEL from an animal with borderline elevated numbers. (i) Rectal CD3+ IEL from an animal with significant increases.
In the cecum, no controls demonstrated surface epithelial damage, although 1/16 (6%) demonstrated minimal epithelial tufting. Again, mild submucosal leukocyte infiltrates were appreciated. One animal had multifocal eosinophilic granulomas in the submucosa surrounding scattered hair shafts, but these were considered incidental. In contrast to the ileum, the cecal mucosa occupied up to an entire 40× field, but remained uniform. Scattered crypt abscesses, no more than 1 per entire examined section, were appreciated in 2/16 (12%) controls, as was some mild crypt herniation. Again, no evidence of amyloid or fibrosis was appreciated in controls. Seventy-five percent of control animals had an intact blue brush border. Intraluminal bacteria were variable, though the majority (69%) only had mild to moderate numbers. Five of 16 control animals (31%) had luminal B. coli, mostly scattered but with 1 animal having abundant quantities. One control animal had intraepithelial B. coli and bacteria in the cecum; however, this was considered secondary to autolysis and sectioning artifact. Regarding leukocyte populations in the cecum, in comparison with the ileum, lymphocytes remained the primary population, but plasma cells comprised a larger proportion and there were more leukocytes between crypts.
The colon of control animals was similar to the cecum in many respects, with no epithelial damage or tufting appreciated, mild submucosal leukocyte infiltration, similar mucosal depth, scattered crypt abscesses, an intact blue brush border, and absence of amyloid and fibrosis. Luminal bacteria were more prominent than in the cecum, with 80% having up to moderate luminal bacteria and 20% having large mats of luminal bacteria throughout the entire section. Interestingly, in comparison with the cecum, fewer animals had colonic luminal B. coli (12%), with one having rare B. coli and one having large numbers. CD3+ IELs were less variable than in the ileum and averaged approximately 8 per 100 enterocytes. The overall variability of leukocyte populations was less in the colon than cecum but the average numbers and proportions of leukocyte populations were similar between the 2 tissues. Neutrophils were observed in the colon, but did not occur in clusters. ECs were mildly increased compared with the small intestine, although not significantly, and EECs were minimally but significantly (P < .01) increased compared with the ileum.
Sections from the rectum tended to be more variable in orientation and thickness than the other sections of the large intestine, but none were thinner than 0.5 40× fields, and 2/16 (12.5%) showed minimal surface epithelium damage. Again, scattered crypt abscesses and crypt herniation were appreciated in 3/16 (18.8%) controls. The presence of a blue brush border was less consistent in the rectum of control animals than in the other examined tissues, but was present in 50% of individuals. The presence of luminal bacteria was more variable compared with other tissues, likely due to collection artifact, and no luminal B. coli were visualized. CD3+ IELs were slightly more numerous than in the colon but were consistent across individuals. The distribution of leukocytes in the rectum tended to shift more to the basal lamina propria region. There were fewer leukocytes between the crypts than other portions of the large intestine, and neutrophils and MCs were present. The EC density increased mildly with progression aborally through the intestinal tract, but not significantly. EECs increased significantly with aboral progression (P < .01).
Regarding the contribution of age and sex to cell populations within the control animals, total leukocyte numbers did not differ significantly between groups. While the numbers of infiltrating leukocytes mildly increased with age throughout the large intestine, this change was not significant. Similarly, leukocytes in the large intestine tended to be more numerous in females, but again, this was not significant.
CE Comparison
Means and standard deviations of cell counts and crypt mitotic activity in animals with CE are in Table 1 with cell populations demonstrating significant differences from the control animals (P < .05) in bold text. While the ileum has not historically been considered a primary site of this disease process, some changes are of note. In the case cohort, both blunting (P < .05) and fusion (P < .01) were significantly increased compared with healthy controls, with over 1/3 of cases having mild to moderate blunting, 4/35 (11%) had severe blunting, and over 50% having some degree of fusion, with 8/35 (23%) having moderate to severe villous fusion. Two cases were eliminated from this portion of analysis due to the degree of villous autolysis. The ileum did not show any differences in surface damage or submucosal leukocytes compared with controls, but scattered crypt abscesses were appreciated, where controls had none (P < .05). Individual leukocyte populations were significantly increased, although the populations affected varied depending on the specific mucosal location being observed. The amount of luminal bacteria in CE cases was significantly (P < .05) increased compared with controls, but there was no difference in intraepithelial bacteria or luminal and intraepithelial B. coli.
Qualitative differences were consistent across all examined sections of the large intestine. The degree of epithelial erosion and ulceration, epithelial tufting, submucosal leukocyte infiltration, and crypt abscessation were all significantly increased (P < .01) in CE cases compared with controls, and the presence of a blue brush border was significantly decreased (P < .01). In the colon and rectum, the presence of crypt herniation was significantly increased compared with controls (P < .05). While the presence of mucosal amyloid and/or fibrosis might be expected with chronic ulceration and inflammation, neither was significantly different between cases and controls. There were no significant differences in luminal or intraepithelial numbers of bacteria or B. coli.
Regarding more quantitative measurements, across all examined sections of the ileum and large intestine, crypt mitotic activity was significantly increased (P < .05) in CE cases compared with controls. Similarly, mucosal thicknesses in all sections of the large intestine were increased, with cecal and colonic mucosa in controls occupying an average of 0.5 40× fields compared with ≥ 1 field in cases, and rectal mucosa of controls occupying an average of 0.7 40× fields compared with 1 field in cases. While the mitotic count of the ileum differed, the thicknesses were not different between the 2 groups, with the average of both groups being near 0.5 40× fields.
Leukocyte counts were significantly increased in all large intestine sections and in the ileal villi of cases as compared with controls, although there was variability in the degree of inflammatory infiltrates among cases (Fig. 3). Lymphocytes and plasma cells in the large intestine, both between crypts and in the basal lamina propria, varied significantly between cases and controls (P < .05), with the exception of plasma cells in the basal lamina propria in the colon. Neutrophils were significantly increased (P < .05) in all examined sections and regions except for the basal lamina propria of the rectum where they were present in similar numbers to controls. The significance of the remaining cell populations was variable. There was no significant difference in the number of CD3+ IEL between cases and controls in the ileum, but CD3+ IEL were significantly increased in the colon and rectum of cases compared with controls. Goblet cells only showed significant decreases in the ileum and no section subjected to IHC showed differences in EC or EEC densities. In addition, we found intestinal MCs to be low in number throughout all sections examined with IHC and no difference was found between cases and controls.
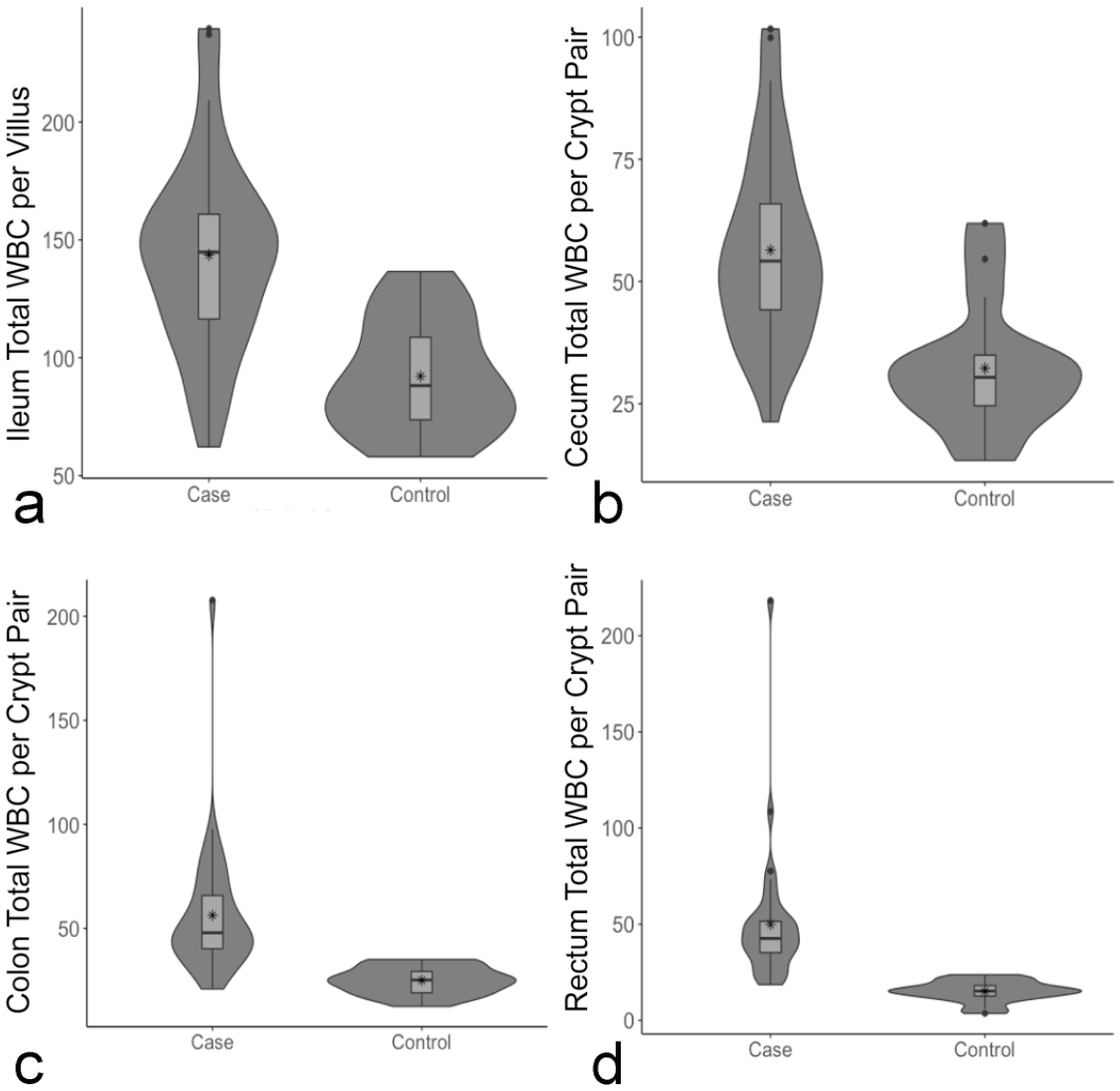
Violin and box plots demonstrating differences in total leukocytes between crypts in each examined section of the large intestine. Total leukocytes (a) per ileal villus, (b) per cecal crypt pair, (c) per colonic crypt pair, and (d) per rectal crypt pair. The difference between the cases and controls in each plot is significant (P < .05, Wilcoxon signed-rank test). Mean values denoted by *. WBC, white blood cell (ie, leukocytes).
Utilizing all of this information, a primary goal of this study was to identify thresholds of cell populations that might provide clear distinctions in case definitions and diagnostic criteria for CE. Table 2 summarizes the calculated thresholds for the primary cells of interest in CE between crypts in the large intestine, as this is an area easily evaluated using necropsy, surgical biopsy, or endoscopic biopsy specimens. Thresholds for total leukocytes between crypts in the colon and rectum, plasma cells between crypts in the rectum, and CD3+ IELs in the colon and rectum are in bold as they were significantly different between case and control animals. The thresholds for these measurements fell outside of the calculated reference range for our healthy population, making these selected populations an easily accessible, rapidly calculable snapshot of the section of interest. Qualitative variables that were significantly different (P < .01) between cases and controls across all sections of the large intestine are also included. A complete list of calculated reference ranges and thresholds for each cell population with significant differences between cases and controls can be found in Supplemental Table S4. In addition, representative photomicrographs of borderline and above threshold cases for total leukocytes between crypts can be found in Fig. 1, and similar depictions of rectal plasma cells between crypts and CD3+ IELs in the colon and rectum are found in Fig. 2. Each image has been selected for illustrating cell populations close to the average of each normal, abnormal, or threshold population. In addition, the abnormal images of Fig. 1 display other common qualitative changes associated with CE. These images are meant to provide helpful visual guidelines, but we refer readers specifically to Table 2 for diagnostic decision-making purposes.
Summary of rapidly identifiable diagnostic thresholds and qualitative findings strongly correlated with a diagnosis of chronic enterocolitis.
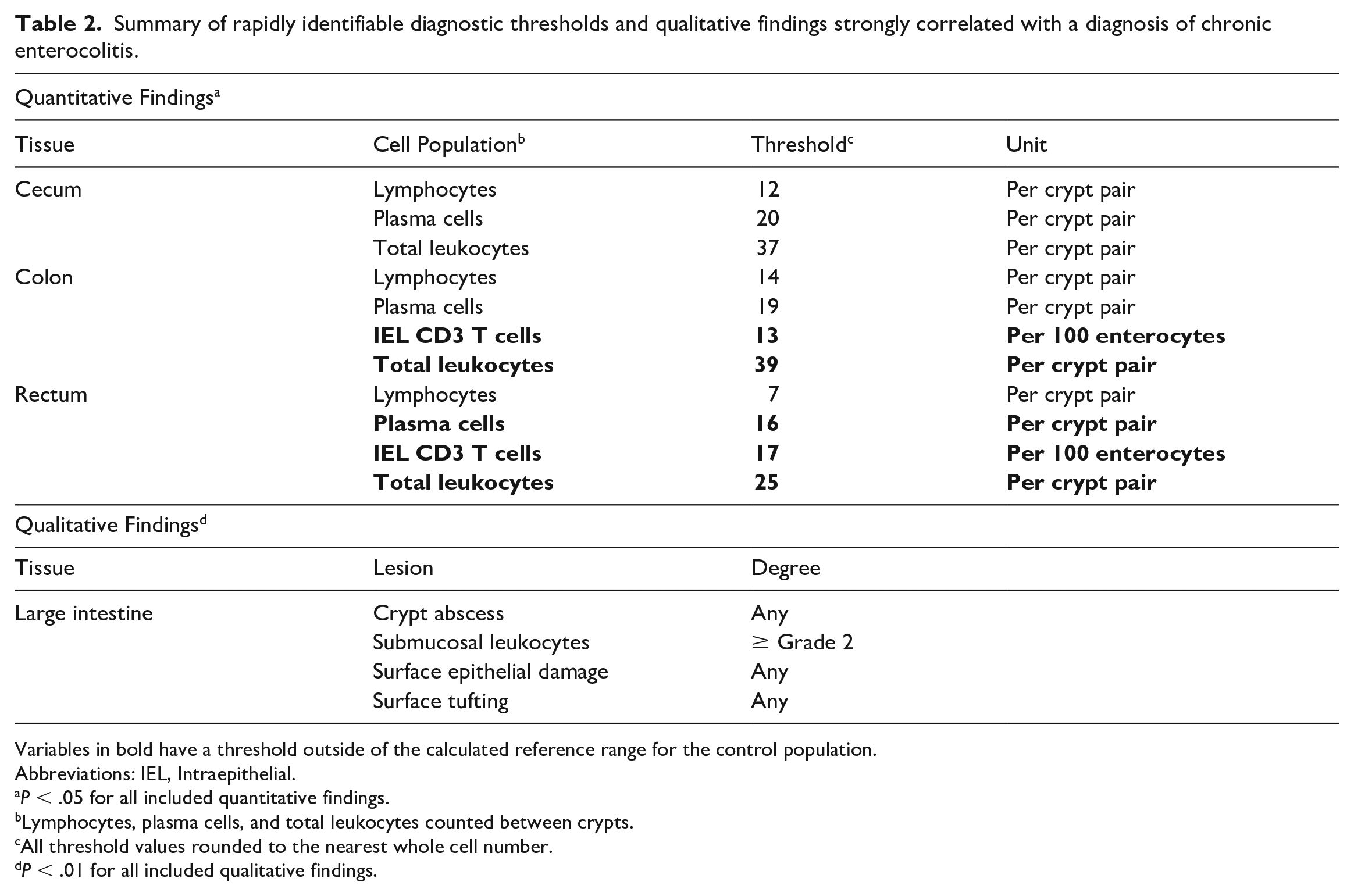
Variables in bold have a threshold outside of the calculated reference range for the control population.
Abbreviations: IEL, Intraepithelial.
P < .05 for all included quantitative findings.
Lymphocytes, plasma cells, and total leukocytes counted between crypts.
All threshold values rounded to the nearest whole cell number.
P < .01 for all included qualitative findings.
Discussion
In this study, we aimed to characterize the cell populations of the normal rhesus macaque lower intestinal tract to facilitate standardization of future histopathologic studies. We also compared these results with a cohort of animals euthanized for clinically diagnosed CE, also known as idiopathic chronic diarrhea, to create guidelines for diagnosis and ease of comparison across studies. We evaluated sections of the ileum, cecum, colon, and rectum using hematoxylin and eosin-stained slides, and further characterized several additional cell populations in the ileum, colon, and rectum utilizing immunohistochemistry.
Previous descriptions of the rhesus macaque intestinal tract noted scattered villous blunting and fusion, lacteal dilation, minimal epithelial erosion, and scattered submucosal leukocyte infiltration as potential normal findings in the ileum, particularly near the ileocecal junction.3,13,16 Our results corroborated these findings. Other evaluated qualitative measures included the presence or absence of a blue brush border and evaluation of relative bacterial and B. coli protozoa loads for each examined section. The presence of a blue brush border is one indication of a healthy intestinal tract, with its absence suggestive of dysbiosis.13,15 A majority of the control animals had a blue brush border in the examined sections, although approximately 25% of controls showed an absent blue brush border. The presence of a blue brush border was significantly decreased in CE cases, consistent with previous reports, but it should be evaluated in context of collection and processing methods as well as the presence of other findings indicative of intestinal damage.13,15
In addition to these findings, when comparing our results with those reported in the WSAVA guidelines from which some of our methods were adapted, we identified differences between rhesus macaques and domestic dogs and cats, species with which many veterinary pathologists tend to be more familiar. 5 Overall, colony-housed rhesus macaques appear to have a larger number of resident leukocytes than these domestic species in each examined portion of the intestinal tract, and while the presence of neutrophils should be considered abnormal in the ileum, as in domestic species, scattered individualized neutrophils were present throughout all sections of the large intestine and based on our results should not be considered abnormal without other evidence of an inflammatory or infectious process. However, neutrophils were significantly increased in cases, frequently occurring in clusters. Our study also evaluated the basal lamina propria, the region of the lamina propria adluminal to the crypts, which is not typically evaluated in endoscopic biopsies and is not included in the WSAVA guidelines as endoscopic biopsies rarely reach the submucosa. 5 Based on our findings regarding this area, a small number of leukocytes in the basal lamina propria should be considered normal and should not be interpreted as a disease state without other findings to support intestinal inflammation. In addition, we found the basal lamina propria region in the rectum to hold a larger portion of the resident leukocytes than between the crypts, compared with the cecum and colon.
Some trends in age/sex distributions for cell populations were observed, with females and older animals tending to have higher numbers of large intestinal leukocytes than males and older animals, respectively, but these changes were not statistically significant. A larger group of healthy animals potentially could demonstrate a significant difference, but these trends can be useful in considering the importance of mild differences. Possible confounding factors in the evaluations of these control animals, as they are group housed and part of a breeding colony, include normal variation in hypothalamic-pituitary-adrenocortical reactivity to psychosocial stress of group housing, and previous subclinical, undiagnosed, intestinal infections. However, all control animals were managed identically with annual physical evaluations, prophylactic anthelminthic medication, vaccinations for rabies and measles, and intradermal tuberculosis testing, and none of the control animals had records of hospitalizations or treatments outside of these standard practices. It is important to note that our study population as a breeding colony for sale purposes may have a different demographic composition than populations in other studies. Males over 4 years of age are typically sold to external institutions, and at any given time, males only make up approximately 1/3 of the animals in this colony. Thirty percent (11/37) of the case animals in this study were male, which approximately mirrors the overall demographics of the colony, although more detailed epidemiologic work is warranted. The age distribution as a breeding colony is likely also mildly skewed toward older animals, as proven successful breeding animals are typically kept for their entire lifespan.
To our knowledge, this is the first study to attempt to quantify resident intestinal MCs in the rhesus macaque, despite abundant research in human IBS and IBD being focused on the contribution of intestinal MCs to these disease processes. 27 Results in human studies vary widely across studies, potentially indicating the heterogeneity of etiologies that result in similar clinical syndromes. In one meta-analysis, the majority of studies did find an increase in MC density in rectal biopsies but a significant portion found no significant differences or a negative correlation. 27 While no differences in MC numbers were found between cases and controls in our study, this provides a baseline for future studies and additional avenues of investigation. It is possible this represents a true difference from human counterparts, or that this disease may only mirror a small subset of the human disease, or potentially, that our methods were not sensitive to detect all macaque intestinal MCs and other detection methods may yield a different result.
We also quantified CD3+ IELs, ECs, and EECs in a systematic way. Our methods were designed to allow ease of communication and consistency across pathologists and institutions and were found statistically comparable with previously described methods that utilized an average number of reactive cells per 600× field, a degree of magnification not available at every facility. 15 Consistent with previous reports, EECs and ECs in controls increased from ileum to colon to rectum. 15 CD3+ IELs were numerous in the ileum, similar to felines, with fewer in the colon and rectum.
Using the baseline data from control animals, we looked for easily measurable differences between control animals and those with CE to clarify the case definition for CE to increase uniformity across studies and help future laboratory animal diagnosticians. Importantly, prior to utilization of our proposed criteria, infectious causes of enterocolitis in rhesus macaques should be ruled out with culture or molecular methods as some agents, such as Campylobacter sp, can produce similar changes. 13 Previous reports have characterized CE as centered on the proximal colon; however, our study, similarly to another recent study, demonstrated significant changes throughout the ileum, cecum, colon, and rectum. 15 This could be at least somewhat explained by our use of terminal cases that have an expected high degree of disease severity.2,13,15 Significant changes in the ileum of cases include villous blunting and fusion, crypt abscesses, and increases in specific leukocyte populations, though which population depended on which section of the mucosa was being examined. These findings appear consistent with previous reports of “reflux inflammation” where the ileum receives some bystander inflammation due to the severe inflammation present in the large intestine. 2
Expected normal and abnormal numbers of each cell population and relevant thresholds for significant variables have been provided. As a limitation, we recognize our provided reference ranges were calculated from a relatively low sample size of controls. As such, we recommend utilization of these values simply as guidelines as more animals would be required to calculate true reference ranges. These data do help to provide information on which variables are most important (Table 2) when considering if an animal should be diagnosed histologically with CE. As previously described, CE is primarily a chronic lymphoplasmacytic inflammatory disorder with lymphocytes and plasma cells in the large intestine between the crypts and in the basal lamina propria varying significantly between cases and controls, with the exception of plasma cells in the basal lamina propria in the colon. Crypt mitotic activity was also significantly increased across all examined sections, consistent with previous descriptions of mucosal hyperplasia as a reaction to chronic inflammation in this disease process.2,13,15 While neutrophils are not classically considered part of the histology of CE, their presence was significantly increased in all examined sections and regions except for the basal lamina propria of the rectum where they were present in similar numbers as controls. The infiltration of neutrophils is likely secondary to the ongoing erosion, ulceration, and active inflammatory process, but given the lack of difference in bacterial and B. coli protozoal populations between cases and controls, should not necessarily be linked to an active underlying infectious process. The significance of the remaining cell populations was variable and more investigation into underlying etiologies is necessary to determine why this may be true. Based on these findings, a pathologist may choose to look first at quantities of total leukocytes between crypts in the colon and rectum, plasma cells between crypts in the rectum, the listed qualitative findings, and CD3+ IELs in the colon and rectum. For animals that fall at or below the threshold values, all variables should be looked at and taken as a whole, in conjunction with the animal’s history and clinical presentation.
Regarding enteric microbial populations, a frequent focus of this disease process is the potential component of dysbiosis leading to or resulting from chronic inflammation. Specifically, the importance of luminal B. coli is often argued and their role in the pathogenesis of diarrheal diseases is being investigated.13,24 In this study, only luminal bacteria in the ileum were significantly increased (P < .05) in cases compared with controls. There were no differences in luminal B. coli in the ileum, or luminal B. coli and bacteria in any section of the large intestine. Given the lack of significant differences in luminal or intraepithelial numbers of bacteria and B. coli in this study, the presence or absence of these organisms in a standardly collected and processed sample should not be considered a mark of a disease process. If evaluation of these populations is specifically warranted in a case, special collection considerations should be made.
Interestingly, CD3+ IELs were strongly correlated with CE. We confirmed significant differences in this population across the colon and rectum, consistent with previous reports, and with human literature.15,27 While there was moderate variability in the cases, this cell population was reasonably consistent in controls and this may be a useful marker in diagnostic decisions in cases with questionable histologic findings in other portions of the mucosa, and perhaps should be included in all future case definitions when using histologic findings as qualifying criteria. CD3+ IELs did not differ significantly in the ileum, which is consistent with the ileum being a secondary, or bystander, site of disease. This disease has been suggested as similar to PI-IBS in humans and a previous human study interpreted CD3+ IEL as evidence of a previous infectious insult, warranting further investigation into potential infectious agents as inciting causes of this debilitating disease.15,27
A previous report noted differences in goblet cell, EC, and EEC densities in CE in rhesus macaques. 15 However, in our cohort, goblet cells were only significantly different in density in the ileum and no section subjected to immunohistochemistry showed differences in EC or EEC densities. The previous report chose to stratify their data and found differences between the lowest and highest severity cases; however, given all of the animals in our cohort were euthanized due to their clinical severity of disease, we elected to not stratify them. In some species, histologic findings do not always correlate well with clinical severity of intestinal disease, making stratification of necropsy samples potentially somewhat arbitrary. 1 Populations of EC and EEC can be negatively affected by acute inflammation, and again, given the presence of active ulceration and neutrophils in many of these cases, this could be a possible explanation for the lack of increase. 28 In addition, the study that reported increased numbers only saw increased numbers in a specific section of the colon, and our study, while relying on general uniformity of sampling at necropsy and tissue grossing, did not have a record of where in the colon the histologic sections were from, which may explain these differences. 15 Further investigation, ideally with endoscopic biopsies across multiple time points, should be pursued to determine whether there are true differences in EECs and ECs in CE.
Potential limitations of our study include the retrospective nature and limitation to one breeding colony of rhesus macaques. However, changes to laboratory animal veterinarians and veterinary pathologists overseeing these cases during the study duration were limited, somewhat limiting variability in management and sample collection. Due to the method used for selecting affected animals, the nonblinded nature of the primary examiner could be a concern; however, measures were taken to ensure appropriate and varied field selection for evaluation, methods that should similarly reduce interobserver variation. In addition, breeding colonies are managed similarly across the country and we anticipate these results to be applicable to other outdoor, colony-housed, rhesus macaques. Future work should include collaboration across multiple nonhuman primate centers and pathologists to further validate the grading scheme and provide a more thorough picture of this disease syndrome. Inclusion of animals with active infectious diarrhea would also strengthen further study and determine the ability of these criteria to differentiate CE from an active infectious cause of enterocolitis. In the absence of an infectious cause, we believe the proposed criteria, in conjunction with compatible clinical history and signs, are diagnostic of CE. As such, the standardized grading scheme we describe here should facilitate comparison across study populations and allow for more complete comparisons with human data. Future prospective studies to validate these findings with endoscopic biopsies would be desirable and allow assessment of the disease at earlier and multiple time points without the need to sacrifice study subjects. A larger scale study with multiple time points and clinical severities to correlate histologic disease severity, using our grading scheme, with prognosis will be crucial for future investigation of this disease. A recent study in dogs showed the current WSAVA guidelines correlated minimally with ultimate prognosis and a simplified grading scale was superior for that purpose, but the same information is yet to be investigated in nonhuman primates.1,5,12 Finally, the description of the control rhesus macaque lower intestinal tract can be used when investigating any number of intestinal diseases and for evaluating potential background changes in experimental studies.
Supplemental Material
sj-pdf-2-vet-10.1177_03009858231203315 – Supplemental material for Analysis of cell populations in the normal rhesus macaque (Macaca mulatta) lower intestinal tract and diagnostic thresholds for chronic enterocolitis
Supplemental material, sj-pdf-2-vet-10.1177_03009858231203315 for Analysis of cell populations in the normal rhesus macaque (Macaca mulatta) lower intestinal tract and diagnostic thresholds for chronic enterocolitis by Rebecca L. Bacon, Loni Taylor, Stanton B. Gray and Carolyn L. Hodo in Veterinary Pathology
Supplemental Material
sj-xlsx-1-vet-10.1177_03009858231203315 – Supplemental material for Analysis of cell populations in the normal rhesus macaque (Macaca mulatta) lower intestinal tract and diagnostic thresholds for chronic enterocolitis
Supplemental material, sj-xlsx-1-vet-10.1177_03009858231203315 for Analysis of cell populations in the normal rhesus macaque (Macaca mulatta) lower intestinal tract and diagnostic thresholds for chronic enterocolitis by Rebecca L. Bacon, Loni Taylor, Stanton B. Gray and Carolyn L. Hodo in Veterinary Pathology
Footnotes
Acknowledgements
We thank Sarah Dysart and the MD Anderson histology service for their help in procuring archived slides and immunohistochemistry processing.
Author Contributions
RLB, CLH, and SBG contributed to the experimental design; RLB and CLH performed the entirety of the study design, including obtaining of samples, histologic evaluation, and statistical analysis. LT contributed to clinical data acquisition of study animals and provided secondary review of statistical analysis procedures. The manuscript, figures, and tables were written and compiled by RLB with review and contribution from all other authors.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Research support for Dr. Rebecca Bacon is provided by the NIH T32 Ruth L Kirschstein National Research Service Award (NRSA) Texas A&M AgriLife Research Institutional Training Grant (T32 OD011083). Grant funding from Cattlemen for Cancer Research (CCR) provided for the IHC (SBG).
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
