Abstract
Objectives
Feline arterial thromboembolism (ATE), an often devastating outcome, was recently shown to affect 11.3% of cats with hypertrophic cardiomyopathy over 10 years. Current American College of Veterinary Internal Medicine guidelines recommend the use of clopidogrel in cats at risk for ATE, with addition of a factor Xa inhibitor in very high risk or post-ATE cases. To date, no studies have examined the safety or efficacy of this combined antithrombotic therapy. This retrospective case series aimed to assess the frequency and type of adverse events that occurred in cats prescribed dual clopidogrel and rivaroxaban therapy. Secondary aims were to evaluate indications for dual therapy and clinical outcome.
Methods
The study included 32 cats prescribed clopidogrel (18.75 mg PO q24h) and rivaroxaban (2.5 mg PO q24h) on an outpatient basis over a 5-year period.
Results
Cats were prescribed dual therapy for at least one of the following: ATE event (n = 18), presence of an intracardiac thrombi (n = 17) or presence of spontaneous echocardiographic contrast (SEC) (n = 16). Five cats experienced adverse effects that could be attributed to medications, a median of 13 days from initiation (epistaxis, hematemesis, hematochezia or hematuria). No cat required hospitalization as a result of these events. Median survival time from onset of therapy was 257 days (interquartile range [IQR] = 38–497) for all cats, 502 days (IQR = 171–663) for ATE cats, 725 days (IQR = 133–856) for cats with an ATE to two or more limbs and 301 days (IQR = 221–431) for cats with only one limb affected. Recurrence rate of ATE while on dual therapy was 16.7%; no cat newly developed an ATE while on dual therapy.
Conclusions and relevance
Dual antithrombotic therapy with clopidogrel and rivaroxaban resulted in a low reported incidence of adverse events. Cats placed on dual therapy for an ATE event experienced a low rate of recurrence and effective thromboprophylaxis was achieved in cats with intracardiac thrombi or SEC.
Introduction
Feline arterial thromboembolism (ATE) occurs when a thrombus, often formed in the left auricle, embolizes to a peripheral artery leading to ischemia of the affected vascular bed. 1 Cats most commonly develop ATE secondary to cardiomyopathies, with less common underlying conditions being congenital cardiac disease, hyperthyroidism or pulmonary neoplasia.2,3 Feline cardiomyopathies are a significant cause of morbidity and mortality with subsequent ATE leading to a severe, life-limiting outcome.4–9 Hypertrophic cardiomyopathy (HCM) is the most commonly diagnosed cardiac disease in cats, with a reported prevalence of approximately 15% in the general adult feline population.6–8 Outcomes of cardiomyopathic disease in cats include a persistent subclinical state, congestive heart failure (CHF), sudden cardiac death and ATE, alone or combination with CHF. While ATE is less frequent than CHF, it has been reported to affect up to 11.3% of cats within 10 years following diagnosis of HCM, and is associated with a poorer prognosis.9–11
Intracardiac thrombosis in cats is hypothesized to occur due to increased platelet activation, blood stasis and endothelial dysfunction, exacerbating each key factor of the Virchow’s triad to create a prothrombotic state.1,12 Clinical predisposing factors that have been associated with an increased risk for development of ATE include spontaneous echocardiographic contrast (SEC), left atrial enlargement, reduced left atrial fractional shortening, decreased left auricular function and a previous ATE event.11,13,14 SEC is thought to be directly correlated with decreased left auricular flow velocities and is, therefore, considered an indicator of blood stasis. 14
Given the high risk of intracardiac thrombosis in cats with HCM, optimizing medical management to prevent thrombosis and subsequent ATE in at-risk cases or recurrence in post-ATE cases is crucial. Current American College of Veterinary Internal Medicine consensus guidelines on feline cardiomyopathies recommend initiation of the anti-platelet drug clopidogrel in cases where echocardiographic risk factors are present. 5 This recommendation is based on the only randomized, controlled clinical study to date assessing thromboprophylaxis in feline ATE; 15 namely, the FAT CAT study, which showed that clopidogrel was superior to aspirin in preventing recurrence in 75 cats that had survived an ATE event. 15 In cases believed to be at increased risk or for those cats that have already experienced an ATE event, escalation of therapy to include both clopidogrel and a factor Xa inhibitor, such as rivaroxaban, is recommended. 5 However, this recommendation is based on human clinical studies, which demonstrate that the addition of rivaroxaban to antiplatelet therapy reduces the incidences of cardiac-related death, myocardial infarction and thrombosis in people with coronary arterial disease. 16 No studies to date have evaluated the efficacy of this combined therapy in cats.
The degree to which clopidogrel suppresses platelet function has been shown to have high inter-individual variability in both cats and humans.17–19 This variability in the inhibitory effects of clopidogrel in cats, in addition to other key factors such as degree of cardiac remodeling and left atrial size, may explain the high recurrence of ATE reported in the FAT CAT study. Therefore, single agent therapy with clopidogrel may be insufficient to prevent thrombosis in all cats, especially in those at very high risk. Dual antithrombotic therapy with the addition of a factor Xa inhibitor in these instances is currently based on recommendations and anecdotal evidence given that no studies have evaluated their combined efficacy or risk for adverse effects such as spontaneous hemorrhage. Two studies evaluating the pharmacokinetics and pharmacodynamics of rivaroxaban and apixaban in healthy cats provide evidence that these two factor Xa inhibitors are well tolerated when given alone.20,21
The primary aim of this retrospective case series was to evaluate the frequency and type of substantial adverse events associated with dual therapy with clopidogrel and rivaroxaban in cats with or at risk for thromboembolic disease. Secondary aims were to characterize the clinical indications for dual therapy at the studied institution and to assess the outcomes in these cases.
Materials and methods
The medical records of client-owned cats between June 2015 and August 2020 at the University of California, Davis, Veterinary Medical Teaching Hospital were retrospectively evaluated for those receiving concomitant clopidogrel and rivaroxaban therapy. Cats were included if they survived to discharge and dual clopidogrel (18.75 mg PO q24h) and rivaroxaban (2.5 mg PO q24h) therapy for at-home care was prescribed and started between June 2015 and June 2020. Cats which were initially started on either clopidogrel or rivaroxaban and then later had the other medication added within the study time period were also included. Cats that had previously received other anticoagulants but were then switched to clopidogrel and rivaroxaban during the study period were also included. Cats receiving additional anticoagulants concurrently, such as enoxaparin or aspirin, and cats that received dual therapy but did not survive to discharge were excluded.
Medical records were reviewed for evidence and incidence of adverse events that could be attributed to dual clopidogrel and rivaroxaban therapy, as well as clinical outcome (alive, dead, euthanized). Criteria for adverse events attributed to antithrombotic therapy included evidence of spontaneous bleeding diatheses such as epistaxis, hematemesis, hematochezia or hematuria. Only adverse events substantial enough to have been reported by the owner or to have resulted in a veterinary visit were evaluated. Medical records were also evaluated for signalment, cardiovascular disease diagnosis, medication regimen, indications for and dates of starting dual therapy.Clinical indication for starting dual therapy was categorized as: (1) ATE based on physical examination findings or (2) intracardiac thrombus and/or SEC based on echocardiographic examination by the attending cardiologists at the time of diagnosis. Owners and referring veterinarians of cats whose outcome could not be determined from the medical record database were contacted by telephone and/or email to determine current survival status, medication regimen, any adverse events or date and suspected cause of death. Deaths were considered cardiovascular if known to be associated with decompensated CHF, ATE, reported as sudden, or euthanasia due to owner-perceived progression of cardiac disease. Deaths from any other causes were designated as non-cardiovascular. In addition, any ATE events that occurred after starting dual therapy, discontinuation of dual therapy, or dose change of clopidogrel or rivaroxaban were recorded. Cats with unknown clinical outcome and whose owners could not be contacted were considered lost to follow-up. Survival was calculated as the days between initiation of dual therapy and clinical outcome. Kaplan–Meier curves were used to estimate survival function and time to event, with cats still alive at the end of the study right censored. As this study included a relatively small number of cats and was retrospectively conducted, results are descriptive in nature with no formal statistical analyses performed.
Results
Study population characteristics
Thirty-two cats received dual therapy with clopidogrel and rivaroxaban between June 2015 and August 2020 and met the inclusion criteria for this study. The baseline characteristics of the study population, including sex, weight, age, clinical presentation and initial drug dosages, are presented in Table 1. At the time of initiation of dual therapy, 26 cats were diagnosed with CHF and 30 were diagnosed with HCM or remodeled HCM. One cat was diagnosed with restrictive cardiomyopathy and the other was diagnosed with severe left ventricular hypertrophy suspected to be due to HCM or subaortic stenosis, with a concurrent congenital defect consistent with either cor triatriatum sinister or supravalvular mitral stenosis. Eleven cats were initially prescribed clopidogrel alone and later had rivaroxaban added to their antithrombotic therapy, as clopidogrel is standard of practice for moderate to severe left atrial enlargement at the studied institution. None of the cats received rivaroxaban first as a single agent. Indications for starting dual therapy are listed in Table 2. Cats had at least one of the following reasons for initiation of dual therapy: ATE (n = 18), presence of an intracardiac thrombus (n = 17) or the presence of SEC (n = 16). Thirteen of the 18 cats (72.2%) had concurrent CHF at the time of ATE. Of the 18 cats with ATE, nine cats (50%) had both hind limbs affected, while six cats (33%) had either the right or left hind limb affected. Two cats (11%) had the right thoracic limb affected by ATE. One cat (6%) had both hind limbs and the right thoracic limb affected. Each cat was prescribed rivaroxaban (2.5 mg PO q24h) and clopidogrel (18.75 mg PO q24h). Five cats required an adjustment in their medication during the study period. Changes included increase in rivaroxaban dosage (n = 1), increase in clopidogrel dosage (n = 1), discontinuation of medications due to difficulty of administration (n = 1), disease improvement (n = 1) or adverse effects (n = 1). Five cats received other anticoagulant therapy at home prior to switching to dual therapy, with one cat having received aspirin and four cats having received enoxaparin. Additional cardiac medications given concurrently during the study period were pimobendan (n = 25), furosemide (n = 25), benazepril (n = 14), enalapril (n = 7), spironolactone (n = 6), diltiazem (n = 4) and atenolol (n = 2).
Baseline demographic data for 32 cats.

Data are displayed as median (range), proportion (%) or mean ± standard deviation
ATE = arterial thromboembolism; CHF = congestive heart failure; HCM = hypertrophic cardiomyopathy
Indication for initiation of dual clopidogrel and rivaroxaban therapy

ATE = arterial thromboembolism; SEC = spontaneous echo contrast
Adverse events
During the course of the study, five cats (15.6%) experienced bleeding diatheses potentially related to dual antithrombotic therapy. Table 3 shows these events, the clinical outcome for these cases and days from the onset of event to outcome. One of the two cats that experienced hematuria continued demonstrating lower urinary tract signs up to 8 months after discontinuing rivaroxaban. The median number of days between starting dual therapy and the onset of the adverse event was 13 days (range = 1–489 days). Rivaroxaban was discontinued in one of the five cats, with continuation of clopidogrel, after presenting with hematuria and a moderate anemia (packed cell volume 26%, total protein 7.8 g/dl) following 13 days of treatment. The remaining four cats continued dual therapy as prescribed due to the clinician-perceived insignificance of clinical signs and the confirmation of a normal packed cell volume at recheck examination following the onset of the event. Of the four cats that continued dual therapy, one died at home the day after event onset and recheck examination. In the cats that survived > 1 day post adverse event, the clinical signs were all reported to have resolved without any alteration of dose, discontinuation of therapy or other medical intervention. No cats experienced a recurrence of any adverse events and one of the five cats was still alive at the study end.
Adverse events of five cats receiving combined clopidogrel and rivaroxaban therapy. Clinical outcome for each case is listed along with days from onset of event to outcome

Patient received both rivaroxaban and clopidogrel q12h instead of q24h owing to owner error
CHF = congestive heart failure
Clinical outcome
Of the cats that were initially prescribed combined clopidogrel and rivaroxaban due to an ATE event, 3/18 (16.7%) had recurrent ATE events while on dual therapy. Examination of medical records revealed that one of these cats was occasionally not administered medication doses if ‘hiding in inaccessible places’. Time to recurrence for the two cats that were properly medicated was 46 and 616 days, and 173 days for the cat that was not receiving medications consistently. None of the cats that were prescribed clopidogrel and rivaroxaban for SEC or intracardiac thrombi (n = 14) experienced ATE. Four cats were newly diagnosed with CHF during the study period after dual therapy was started. The median survival time for all cats after starting dual therapy was 257 days (IQR = 38–497), while the median survival time for cats with an initial ATE event was 502 days (IQR = 171–663) (Figure 1). Nine of the 18 (50%) ATE-affected cats survived longer than 1 year after their first ATE event and the initiation of dual therapy. The median survival time following initiation of dual therapy for the 10 cats with ATE affecting two or more limbs was 725 days (IQR = 133–856) compared to 301 days (IQR = 221–431) for the eight cats with only one limb affected (Figure 2). At the end of the study period, eight cats were still alive (three cats with ATE and SEC; three cats with ATE; two cats with intracardiac thrombi), 14 cats were euthanized and nine cats had died at home. One cat was lost to follow-up. All deaths and euthanasias were reported to be due to cardiovascular causes as defined in the methods.
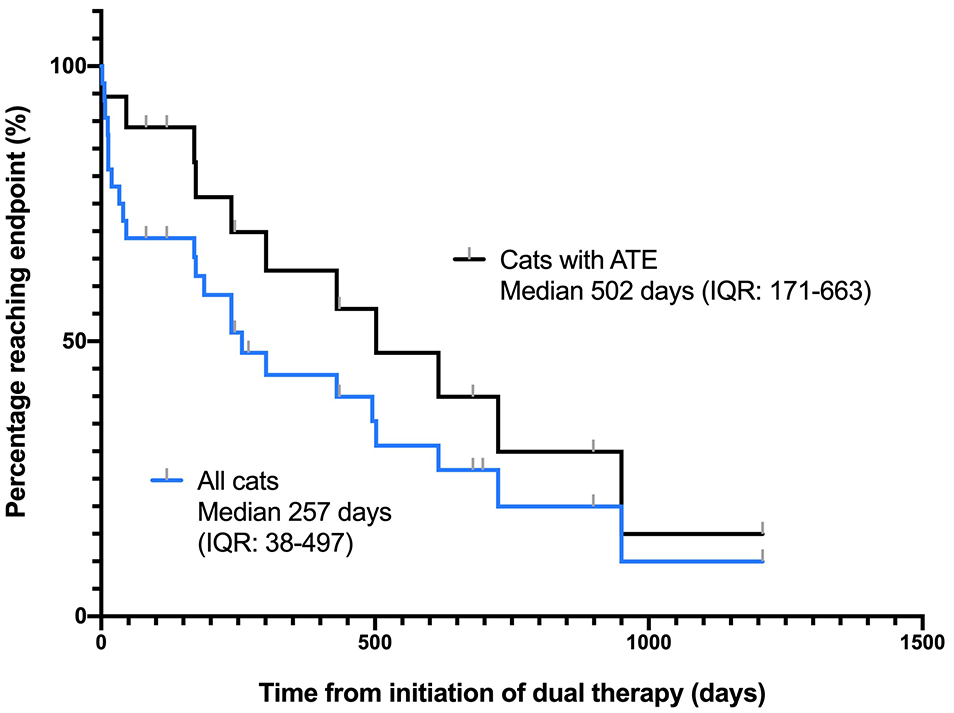
Kaplan–Meier curves for all cats in the study (32) and for cats with an arterial thromboembolism (18) event as their indication for initiation of dual therapy with clopidogrel and rivaroxaban. Median survival times (interquartile ranges) are shown

Kaplan–Meier curves for cats that suffered an arterial thromboembolism affecting two or more limbs (10) and for cats that suffered an ATE affecting only one limb (8). Median survival times (interquartile ranges) are shown
Discussion
Herein, we report that dual antithrombotic therapy consisting of clopidogrel and rivaroxaban resulted in few clinically significant adverse events in the evaluated population of cats at high risk of cardiogenic ATE. Of the 32 cats that were prescribed dual antithrombotic therapy with clopidogrel and rivaroxaban at a single academic veterinary teaching hospital over a 5-year period, five cats (15%) were reported to experience potentially related adverse events.Clinical signs consisted of minor bleeding diatheses such as epistaxis, hematemesis, hematochezia or hematuria. One cat had persistent lower urinary tract signs despite discontinuation of rivaroxaban, while another cat that experienced hematochezia was inadvertently receiving both clopidogrel and rivaroxaban q12h rather than q24h, twice the intended dose of both medications. Therefore, it is uncertain if antithrombotic therapy played a role in the adverse event experienced by the former cat and if dual therapy at the intended doses would have been tolerated by the latter. One cat died at home 1 day following the onset of hematemesis and epistaxis, and 2 days after presenting for an ATE. At a recheck examination prompted by the hematemesis, a packed cell volume and total protein were unremarkable (40%/8.7). The patient was sent home on both medications with continued monitoring. Since this cat was presented multiple days after the onset of ATE clinical signs, it is uncertain if the death was due to rapid deterioration of the patient’s condition or related to adverse antithrombotic therapy effects. None of the cats required a blood transfusion or hospitalization. These findings suggest that standard dose dual clopidogrel and rivaroxaban therapy was tolerated in the majority of cats at high risk of thromboembolism and for medical management post-ATE event, with a small portion (n = 5) detected to have clinical signs consistent with spontaneous bleeding diatheses.
Cats with HCM are at high risk of thromboembolic disease, with a recent international multicenter retrospective study finding an 11.3% risk of developing an ATE over 10 years. 9 This devastating outcome confers the need to optimize thromboprophylaxis treatment. In post-ATE cats, prevention of recurrence is a major goal of patient management since up to 47% of cats experience subsequent thromboembolic events. 22 Previous recommendations for long-term antithrombotic treatment in cats have included antiplatelet therapy such as aspirin and clopidogrel, 5 heparin (unfractionated and low-molecular weight) and vitamin K antagonists such as warfarin.23,24 While recent studies have shown that clopidogrel is superior to aspirin at reducing the likelihood of recurrent ATE, and that warfarin use is associated with an elevated risk of adverse effects, the optimal drug or drug combination to prevent thrombus formation in cats has not been determined. 25
Although direct and statistical comparisons between studies cannot be made, post-ATE cats on clopidogrel and rivaroxaban in the present study lived longer and had fewer ATE recurrences than previously reported in cats receiving other anticoagulant therapies.2,11,22,26 Additionally, no cat developed a first-time ATE event after starting dual clopidogrel and rivaroxaban therapy. This is an important finding as this combination was started in cats considered very high risk, given the presence of intra-cardiac thrombi or SEC. However, it should be noted that given the small number of cats this could be an underrepresentation, with the potential for an extended study period to have revealed some newly developed ATEs. The median survival time on dual therapy for post-ATE cats in this study was 502 days, which was longer than the median survival time on dual therapy (257 days) for all cats in the study. Previously published studies revealed a median survival in post-ATE cats of 184 days on aspirin, warfarin, low-molecular weight heparin or a combination of these mediations (n = 43 cats), 11 149 days on high-dose aspirin (n = 18 cats), 2 105 days on low-dose aspirin (n = 24 cats), 2 and 94 days on aspirin, clopidogrel, heparin, a combination of these or no anticoagulant therapy (n = 30 cats). 22 The only prospective clinical trial evaluating clopidogrel in feline ATE found a median time of 346 days to a composite endpoint of recurrent ATE or cardiac death. 15 The 1-year survival rate of cats in this study after initial presentation of ATE was 50%. Other studies have reported 1-year survival rates post-ATE to be 20% and 0%.24,26 However, it is important to note that the present study only included cats that survived to hospital discharge, whereas previous studies included cats euthanized at presentation, significantly worsening survival rate.
Comparison of multiple retrospective studies is troublesome due to differences in study design, population and aims; however, the recurrence rate of ATE in our study was 16.7%, which is lower than reported rates from previous studies evaluating aspirin (25–75%), clopidogrel (49%), and either aspirin, clopidogrel, heparin, a combination or no medical treatment in cats (47%).2,15,22 Our retrospective case series cannot determine if dual therapy truly is more effective, but we hope this finding propels future prospective studies to evaluate if combined clopidogrel and rivaroxaban is more effective at preventing ATE recurrence and reducing the risk of ATE development in high-risk cats. Furthermore, the median survival time of cats that had more than one limb affected by ATE was longer than those with just one affected limb (725 vs 301 days) in this study. This is in contrast to a previous study that found the number of limbs affected to be a negative prognostic indicator. 2 It cannot be ascertained from the current data whether or not the number of affected limbs is a risk factor for mortality in cats receiving dual therapy and further investigation into survival based on affected limbs in ATE is warranted.
This study has several limitations, in large part due to the retrospective design. These include the small study population size, the reliance on owner observations and clinician reporting in medical records and the fact that it was performed at a single tertiary institution. Ensuring treatment compliance was not possible and there was no established protocol to evaluate for adverse events at follow-up visits. Specifically, only adverse events that were perceived as substantial enough to have resulted in reporting by the owner to the clinician or in a veterinary visit were evaluated, and therefore less serious adverse events may have been missed. Low frequency catastrophic events, such as idiosyncratic reactions, may also have been missed and, given the small number of cats studied, there is an expectedly large confidence interval surrounding the number of reported adverse events (7–32%). Treatments and medication dosages were not standardized across all cats and therefore therapies administered or adjusted were based solely on the decision making of the attending clinician. It is possible that existing comorbidities or concomitant medications, which differed between cats, influenced clinical outcome. A portion of cats (n = 11) were placed on clopidogrel before starting rivaroxaban later, which could have influenced outcome. Echocardiograms were not re-evaluated as part of this study, and clinical diagnoses were made by the attending cardiology clinician at the time of each veterinary visit.
Despite these limitations, the results of this study suggest that dual antithrombotic therapy with clopidogrel and rivaroxaban is generally well-tolerated in cats, with few reported adverse effects. Furthermore, the median survival time of 502 days post-ATE and no reported first-time ATE events suggest that dual therapy may be effective for preventing thrombosis in a high-risk population of cats with cardiac disease. These results should serve to support a prospective study assessing the safety and efficacy of dual antithrombotic therapy with clopidogrel and rivaroxaban in cats at risk of developing new or recurring cardiogenic ATE. As this study was relatively small, further research is needed to validate these findings and lead to creation of more concrete treatment guidelines in these cases.
Conclusions
ATE is a life-threatening outcome of HCM, the most common feline cardiac disease. Owing to the generally acknowledged poor prognosis of ATE in cats, euthanasia at presentation is common and a minority of cats survive to discharge. We found that dual antithrombotic therapy with clopidogrel and rivaroxaban was tolerated in the studied population of cats with minimal adverse effects and a longer median survival time, with 50% of cats alive 1 year after an ATE event. Cats with ATE that survive to discharge may therefore have better prognoses than previously reported owing to new available therapies. It is important to elucidate this so that veterinarians can confer a realistic prognosis to owners and provide the optimal treatment recommendation. Further prospective research on the safety and efficacy of combined clopidogrel and rivaroxaban therapy is needed to evaluate the possible superiority of this dual therapy protocol to other antithrombotic treatment regimens.
Footnotes
Acknowledgements
The authors wish to acknowledge the Students Training in Advanced Research Program at the UC Davis School of Veterinary Medicine for allowing this opportunity, and the Cardiology and Emergency and Critical Care residents, faculty and technicians at the UC Davis Veterinary Medical Teaching Hospital for their involvement in each of these clinical cases.
Conflicts of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: this work was partially supported by the National Institutes of Health T35 grant number OD010956.
Ethical approval
This work involved the use of non-experimental animals only (including owned or unowned animals and data from prospective or retrospective studies). Established internationally recognised high standards (‘best practice’) of individual veterinary clinical patient care were followed. Ethical approval was therefore not specifically required for publication in JFMS.
Informed consent
Informed consent (either verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (either experimental or non-experimental animals) for the procedure(s) undertaken (either prospective or retrospective studies). No animals or humans are identifiable within this publication, and therefore additional informed consent for publication was not required.
