Abstract
Practical relevance:
Diabetes mellitus (DM) is a common feline endocrinopathy, and is often driven by underlying insulin resistance with associated pancreatic beta (β)-cell dysfunction. Although spontaneous hyperadrenocorticism (HAC) with hypercortisolemia (hypercortisolism) is relatively uncommon in cats, it is a well-established cause of insulin resistance and is routinely associated with DM in this species.
Clinical challenges:
Many of the clinical signs associated with feline HAC are subtle and may be attributed to concurrent DM or the aging process. Failure to recognize HAC in the diabetic cat can impact patient wellbeing and predispose the patient to progressive compromise. Unfortunately, it can be difficult to establish a diagnosis of HAC, as test results may be influenced by poor diabetic regulation, and protocols are different to those used in canine patients. Treatment options depend on the underlying cause, and often require careful, ongoing assessment and modulation of both adrenal function and insulin requirements. However, various approaches have been shown to either improve glycemic control in cats with sustained insulin dependence, or facilitate diabetic remission.
Evidence base:
This review summarizes the current literature on feline HAC, with a particular focus on cats with concurrent DM. The clinical findings that suggest HAC are discussed, along with an outline of diagnostic options and their limitations. Published outcomes for various medical options, surgical procedures and radiation therapy are provided. The authors also share their thoughts on the safe and effective management of cats with HAC and DM, with an emphasis on the anticipation and recognition of changing insulin requirements.
Keywords

Introduction
Prevalence
The estimated prevalence of diabetes mellitus (DM) in cats is 0.2–1%, making it one of the most common endocrinopathies in this species. 1 The pathogenesis of the most common presentation of this disorder has many similarities to human type 2 DM, as sustained insulin resistance often plays a role; unlike their human counterparts, most affected cats are managed with exogenous insulin. 2 Prompt recognition of underlying conditions that blunt insulin sensitivity is important, as restoration of adequate pancreatic β-cell function permits diabetic ‘remission’ and the cessation of insulin therapy.
Numerous conditions are known to reduce insulin responsiveness, including obesity, infection and inflammation. 3 Concurrent endocrinopathies such as hypersomatotropism, hyperthyroidism and HAC are also associated with insulin resistance and the genesis of DM in cats. The prevalence of concurrent HAC in diabetic cats is not well established, but it appears to be much less common than acromegaly and hyperthyroidism.4,5 In all, fewer than 100 cats with both DM and HAC have been reported in the peer-reviewed literature, and only two of almost 10,000 cats presented to a veterinary teaching hospital over an 11-year period were diagnosed with concurrent HAC and DM. 6
Impact of glucocorticoids
Glucocorticoids such as cortisol have wide-ranging impacts on energy metabolism and glucose homeostasis, including the upregulation of hepatic gluconeogenesis and hepatic glucose output. 7 Additionally, they reduce the sensitivity of peripheral tissues such as skeletal muscle to insulin, resulting in decreased glucose uptake and decreased glycogen synthesis. Hypercortisolemia also drives the deposition of fat both within and around the abdominal cavity. This ‘central adiposity’ is triggered by various adipokines; in humans, this feature is strongly associated with insulin resistance, and has significantly more impact on glucose tolerance than overall obesity. 8 A recent study on feline obesity suggests that leptin likely plays a key role in fat deposition in this species. 9 Exposure to supraphysiologic cortisol concentrations is therefore expected to initially trigger a hyperinsulinemic state; if sustained, this might eventually be associated with β-cell compromise and inadequate insulin secretion in susceptible individuals. Overt DM will be apparent when the blood glucose (BG) concentration exceeds the renal threshold, and is reported in the majority of cats with HAC. It is interesting to note that although overt iatrogenic Cushing’s syndrome is fairly rare in cats, studies looking at risk factors for DM report recent exogenous glucocorticoid administration in 6–17% of patients.10,11
Temporal relationship between concurrent DM and HAC
Our current understanding of the relationship between HAC and DM would suggest that adrenal dysfunction precedes the onset of hyperglycemia. However, most cats with spontaneous HAC are first diagnosed with DM; concurrent HAC is often identified many months later.12–15 Although obvious insulin resistance may trigger consideration of HAC, practitioners should not exclude this possibility simply because a cat achieves glycemic control on an unremarkable insulin dose.
Pathogenesis of spontaneous feline HAC
The majority of cats with HAC (85–90%) have an underlying pituitary tumor, most often an adenoma, causing inappropriate secretion of adrenocorticotropic hormone (ACTH).12–15 Less commonly, HAC may be adrenal dependent (ADH); affected cats have a functional cortical adenoma or carcinoma.12,16 Adrenal tumors account for only 0.2% of all feline neoplasms, and more often result in hyperaldosteronism, rather than HAC.17,18 Irrespective of secretory activity, the likelihood of malignancy for feline adrenocortical tumors is estimated at 50%. Several cats with multifunctional adrenal tumors have been described, so clinical and biochemical evidence of excessive aldosterone or sex hormone production does not rule out the possibility of concurrent HAC.19,20
Clinical presentation
Most cats with HAC are over 10 years of age, although one case series included a 4 year old.12,13 There does not appear to be a breed or sex predisposition.
Polyuria, polydipsia and polyphagia are routinely reported in cats with HAC and DM. However, these changes are often a reflection of poorly controlled DM, rather than HAC per se. Non-specific changes in mentation and behavior, such as lethargy or depression, may be noted; mild changes may be attributed to sustained hyperglycemia, whereas more overt abnormalities, such as visual deficits, circling or ataxia, suggest complications related to an expansile pituitary mass.21–24 However, the absence of behavioral changes or neurologic signs does not discount the possibility of a pituitary tumor. 14
Physical examination findings in cats with HAC are variable. Central adiposity is commonly noted and, along with hepatomegaly and poor muscle tone, contributes to a ‘pot-bellied’ appearance (Figure 1).12,15 A plantigrade stance may be evident; this may indicate a peripheral neuropathy related to sustained hyperglycemia or changes in ligament strength related to HAC. Limbs may appear thinner than expected, with a loss of both muscle and fat. A large adrenal tumor may be noted on abdominal palpation.

Castrated male Rex cat with hyperadrenocorticism (HAC). Note the abdominal distension, haircoat changes and wound on the dorsum, indicating skin fragility. Courtesy of Dr John August
Dermatologic changes are routinely observed in cats with HAC (Figures 2 and 3). Non-pruritic alopecia affecting the ventrum and flanks and/or thinning of skin were documented in almost 100% of cases in some reports.12,13,15,25–27 Bruising may be noted, particularly following venipuncture. In some instances, cats develop extreme skin fragility, and are prone to tearing during normal handling and self-grooming activities (Figure 4). This is likely due to steroid-related suppression of fibroblast activity, and is essentially pathognomonic for HAC in this species.12,21,22 Compromise to both skin integrity and the immune system may result in recurrent cutaneous abscess formation.12,25,28

Castrated male domestic longhair cat before (a) and after (b) the onset of HAC. Note the extensive truncal alopecia and small skin tear (arrow) on the left hindlimb. Images courtesy of Dr Brigitte McAtee

Castrated male domestic shorthair cat (a) on the left, with its sibling, 12 months prior to the onset of DM; (b) on the left, with its sibling, at the onset of DM; and (c,d) 6 months later. Note the progressive change in hair color, ventral alopecia, lack of hair regrowth following clipping, and curling of the whiskers. Images courtesy of Dr Linda Fleeman
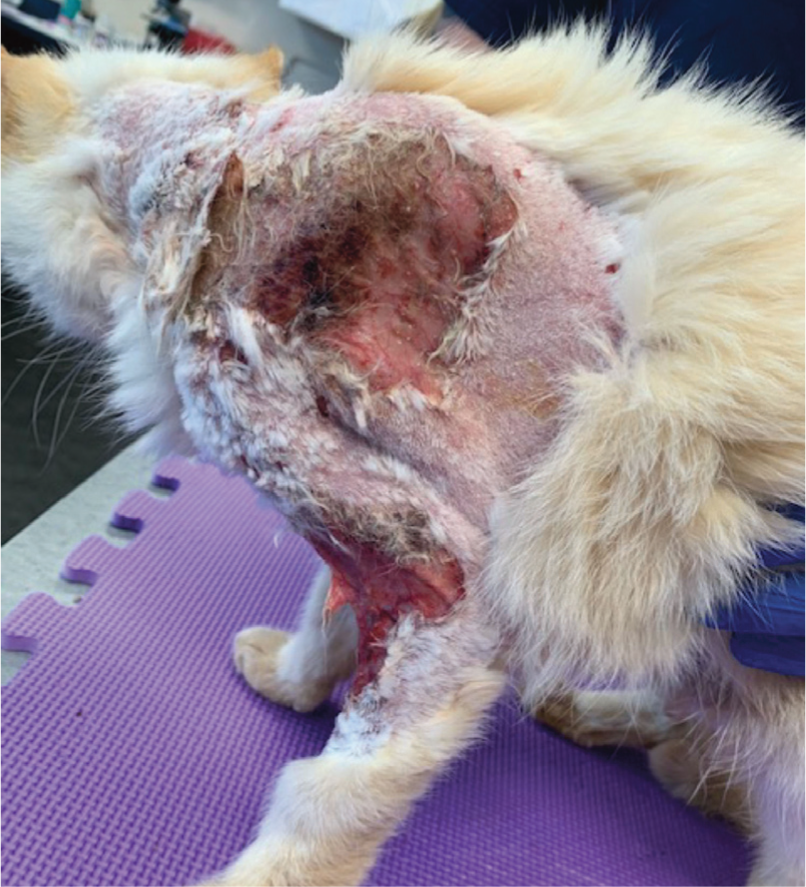
Twelve-year-old castrated male domestic shorthair cat with a history of unregulated DM. Note the extensive wounds and thinning of the skin. Courtesy of Dr Brigitte McAtee
Hyperpigmentation of the skin was reported in a small group of cats with iatrogenic HAC and secondary hypothyroidism, but is not routinely noted in cats with spontaneous disease. 29
Laboratory findings
Results of complete blood counts for cats with concurrent DM and HAC are generally within normal limits. Abnormalities on routine biochemical profiles generally reflect the diabetic state and include hyperglycemia, hypercholesterolemia and hypertriglyceridemia.12,13 As cats do not have the steroid-induced alkaline phosphatase isoenzyme seen in dogs, this parameter often remains within the reference interval. Concurrent hyperaldosteronism may result in hypokalemia and/or increased muscle enzyme activities. 18 Glycosuria ± ketonuria is expected; proteinuria and/or evidence of urinary tract infection may also be noted.
Serum thyroxine concentrations for cats with spontaneous HAC are usually within the reference interval but are occasionally (<20% of cats) subnormal. 12

Imaging
A mid-abdominal mass or area of mineralization may be noted on abdominal radiography in cats with ADH. 19 Thoracic radiography may reveal metastatic lesions in patients with malignant cortical tumors, and should always been performed prior to surgery in patients with ADH.
Transabdominal ultrasonography can provide useful supportive evidence for the diagnosis of HAC and may also differentiate between ADH and pituitary-dependent hyperadrenocorticism (PDH). 10 Bilateral adrenomegaly (defined as caudal pole height >4.5 mm) is routinely noted in cats with PDH, but is also associated with non-adrenal disease, including hypersomatotropism and hyperthyroidism.30,31 Interestingly, one study of cats with DM suggested that this condition alone does not result in adrenomegaly and found that the adrenals in poorly regulated diabetic cats were significantly smaller than those with confirmed HAC. 32 In cats with ADH, an enlarged or otherwise abnormal gland may be identified, along with atrophy of the contralateral adrenal (Figure 5).12,15,27,28 The adjacent vasculature should be assessed for possible tumor invasion or thrombus formation.
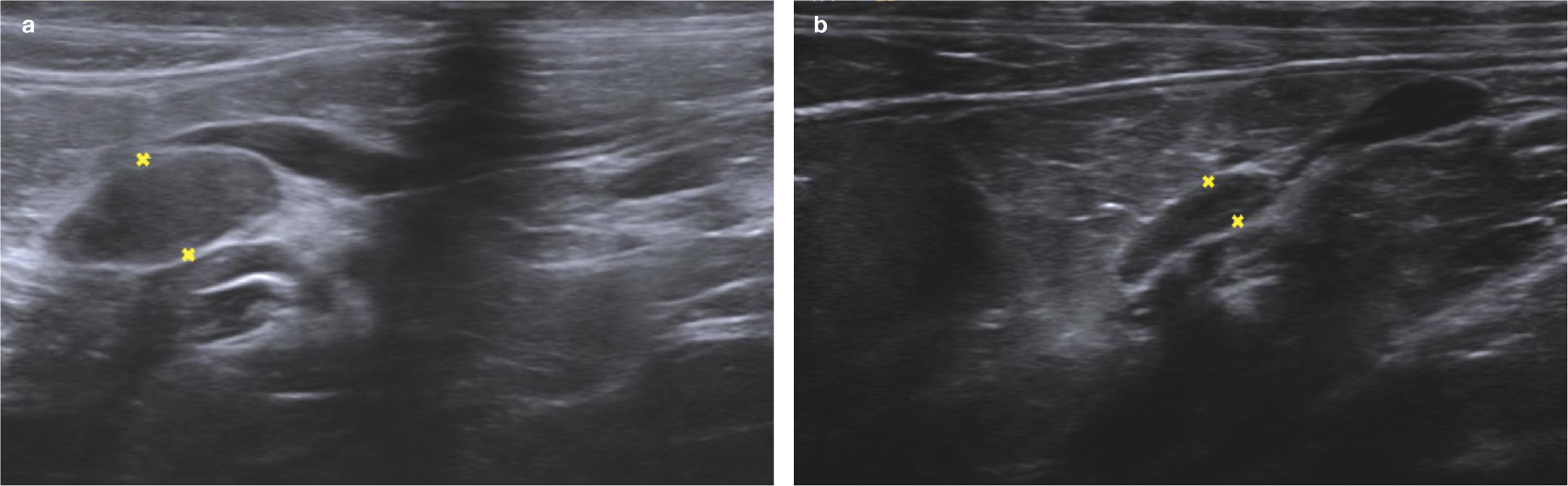
Transabdominal ultrasound images from a 14-year-old castrated male domestic longhair cat with an adrenal tumor. These show (a) an enlarged right adrenal gland (between the measuring calipers) with loss of normal architecture and (b) an atrophied left adrenal gland (between the measuring calipers). Images courtesy of the Texas A&M Veterinary Medical Teaching Hospital Imaging/Radiology Service
CT examination of the abdomen may provide additional information regarding operability in cats with ADH and should be routinely recommended prior to adrenalectomy. 33
Advanced imaging of the pituitary is indicated in cats with PDH.14,22,24,35 Direct comparisons have not been reported in this species, but studies in people with Cushing’s disease suggest that MRI is more sensitive than conventional contrast-enhanced CT. 36 Failure to identify a mass on CT does not exclude the possibility of PDH.26,35 Dynamic CT timed to generate a ‘pituitary flush’ enhances differentiation between the neurohypophysis and adeno-hypophysis, and may facilitate the identification of small tumors (Figure 6). 37 It is also important to bear in mind that a pituitary tumor in a cat with insulin resistance is substantially more likely to be secreting growth hormone than ACTH, as the prevalence of hypersomatotropism far exceeds that of PDH in this species.4,14,22

CT images of the head of a 12-year-old castrated male domestic shorthair cat with a pituitary tumor (indicated by the yellow circle); (a) transverse view and (b) sagittal view. Images courtesy of the Texas A&M Veterinary Medical Teaching Hospital Imaging/Radiology Service
Confirming the diagnosis of HAC
Various tests have been used to identify HAC in cats, including dexamethasone suppression tests (DSTs), ACTH stimulation tests and measurements of urine cortisol. Cortisol is a fairly robust hormone, and remains stable in serum or urine maintained at –20°C for several days.38,39 Samples can therefore be shipped to a reference laboratory using icepacks and an insulated container. Cortisol results from the same sample can vary widely among laboratories, and it is therefore advisable to use a facility that participates in an external quality assurance scheme. In addition, a reputable laboratory should establish its own reference intervals for feline adrenal function testing. Practitioners should preferentially rely on these guidelines, and be cautious about the strict application of published diagnostic cut-offs that may have been generated using older methodology. It is the opinion of the authors that currently available in-house cortisol assays lack adequate reliability.
Dexamethasone suppression test (DST)
Dexamethasone is a potent analog of cortisol, and intravenous administration is expected to suppress the release of corticotropin-releasing hormone from the hypothalamus and ACTH from the anterior pituitary for a prolonged period of time. Continued secretion of cortisol therefore indicates a derangement in the hypothalamic–pituitary–adrenal axis, and supports a diagnosis of HAC. Various protocols have been described, but most clinicians rely on the collection of a baseline blood sample, followed by additional samples 4 and 8 h post-dexamethasone.
Cats appear to be more resistant to the inhibitory effects of dexamethasone than other species, and the traditional ‘low dose’ of 0.01 mg/kg used in dogs has not been shown to consistently suppress cortisol production in healthy cats. 40 Consequently, most experts suggest using 0.1 mg/kg of dexa-methasone in this species, although this may reduce the sensitivity of this test when screening for HAC. The definition of suppression is somewhat variable, but several sources define suppression as cortisol concentrations ≤30 nmol/l (1.1 μg/dl) at 4 and 8 h.12,41,42 A diagnosis of HAC is supported by 8-h post-dexamethasone cortisol concentrations ≥40 nmol/l (1.5 μg/dl).
Non-adrenal disease can impact the function of the hypothalamus, and negate the normal suppressive effect of dexamethasone. However, DM per se does not appear to alter the specificity of the 0.1 mg/kg DST in cats. 32
There is limited data available to establish the sensitivity and specificity of the DST in cats. However, in one case series, 27/28 cats with a final diagnosis of HAC had abnormal DST results. 12
Adrenocorticotropic hormone (ACTH) stimulation test
Appropriate doses of exogenous ACTH stimulate cortisol production by the adrenal cortices, and can be used to determine maximal adrenal function. Cats with PDH are expected to have an exuberant response to exogenous ACTH due to hyperplasia;15,25 responses in cats with ADH may also be exaggerated, but are less predictable due to variable ACTH receptor expression/function. 12 As in other species, chronic stress secondary to non-adrenal disease may produce a physiologically appropriate hyperplasia of the zona fasciculata in cats; this can result in an exaggerated response to exogenous ACTH and a false-positive test result. It is therefore always important to consider the entire clinical picture when making a diagnosis of HAC.
Established protocols support the use of cosyntropin (tetracosactrin) at 125 μg/cat IV, although lower doses may be equally effective.43,44 The dose used may impact the timing of the cortisol peak, so practitioners should determine optimal sampling times if using lower or higher doses, or a gel formulation.44–47 A baseline sample is collected before and 60 mins after ACTH administration. Some sources advocate collecting an additional sample at 30, 90 or 120 mins post-ACTH.1,12,13,46 Transient vomition has been reported in cats receiving ACTH products.
The diagnosis of HAC is supported by a post-ACTH cortisol concentration ≥400 nmol/l (≈15 μg/dl).12,14,15 There is limited data regarding the impact of DM alone on ACTH stimulation testing, but one study reported that two cats with DM (without evidence of concurrent HAC) repeatedly displayed abnormal responses to ACTH administration. 48 A ‘flat-line’ response should prompt consideration of iatrogenic HAC, or the production of a biologically active precursor by an adrenal tumor.19,20
Overall, the ACTH stimulation test is less sensitive than the DST in cats, with one case series reporting a sensitivity of 46% when only the 60-min post-ACTH result was used, or 56% when results from all time points (30, 60 and 120 mins post-ACTH) were used. 12
Urinary cortisol:creatinine ratio (UCCR)
The UCCR reflects renal excretion of free cortisol, and is expected to be increased in patients with HAC. There is limited information regarding this test in cats, but one study using two or three pooled samples reported significantly higher UCCRs in six cats with HAC compared with healthy controls (n = 42). 26 Although at-home sample collection has some advantages, this test appears to lack specificity as results are significantly impacted by non-adrenal illness. 49
Differentiating PDH from ADH
As treatment options for HAC depend on the underlying cause, it is important to differentiate PDH from ADH. Abdominal ultrasonography is often helpful in this regard but may be unavailable or inconclusive.
Transient suppression of cortisol secretion following administration of 0.01 mg/kg dexamethasone (ie, cortisol <50% of baseline at the 4-h time point) is indicative of PDH in dogs; a similar effect has been reported in a small number of cats with PDH following administration of dexamethasone at either 0.01 mg/kg or 0.1 mg/kg.26,35 In one large case series, suppression at the 4-h mark was noted in fewer than 20% of cats with PDH; most of these DSTs used the 0.1 mg/kg dose. 12 Various authors have anecdotally described higher incidences of suppression (approximately 50% of cats with PDH) either 4 or 8 h following 1.0 mg/kg dexamethasone.
Endogenous ACTH concentrations can also be used to differentiate PDH from ADH.12,27 This hormone decays quickly following sample collection and must be handled carefully to prevent spurious results. Cortisol from an adrenal tumor will suppress the higher centers, and result in subnormal ACTH secretion. Normal or increased ACTH concentrations are therefore consistent with PDH.12,26,27,35,50 Although measurement of ACTH precursors is not routinely available, these may be predictive of pituitary tumor size. 50
Treatment of HAC
Various options are available to address feline HAC, and treatment decisions will depend on the cat’s final diagnosis (ie, PDH vs ADH), and the owner’s willingness and ability to care for its needs. Clinicians must also evaluate the cat’s overall clinical condition, paying careful attention to its neurologic, dermatologic, cardiac and renal status. A suitable plan for a cat with minimal dermatologic issues and a small left-sided adrenal tumor may be inappropriate for a cat with severe skin fragility and an invasive right-sided mass. Similarly, a treatment plan dependent on daily oral medication may be successful in an easy-going individual, but a failure in a less amenable cat.
In all instances, the owner and the practitioner need to anticipate changes in insulin needs. It is not unusual for cats with concurrent DM to require substantially less insulin or enter diabetic remission with effective management of their HAC.12,13,14,35 In either scenario, failure to quickly identify changes in glycemic control and appropriately adjust the insulin dose may result in life-threatening hypoglycemia.
Options for cats with PDH include medications to suppress adrenal function, radiation therapy, hypophysectomy and bilateral adrenalectomy. For cats with ADH, surgical removal of the affected gland is generally the most appropriate option; medical therapy may otherwise be considered.
Medical therapy: trilostane
Trilostane is a synthetic steroid analog, and inhibits 3-beta-hydroxysteroid dehydrogenase (3β-HSD), a key enzyme required for steroid hormone synthesis. Although this drug impacts the production of both cortisol and aldosterone, the zona fasciculata is preferentially affected, so aldosterone synthesis usually remains adequate. Trilostane (as Vetoryl; Dechra) is licensed for the treatment of both PDH and ADH in dogs, but has not been approved for use in cats. However, it is widely regarded as the first choice for medical management in this species as it is readily available, generally well tolerated and often beneficial.12,13,27
A starting dose of 10 mg/cat q12h PO is generally appropriate. Although q24h therapy may be adequate in non-diabetic Cushingoid cats, q12h administration ensures that each dose of insulin is ‘matched’ by the same dose of trilostane. Owners should be advised to discontinue the medication if the cat is anorexic or lethargic, as these may be signs of (relative) cortisol deficiency. Owners should be provided with a short-acting glucocorticoid and instructed to administer a dose (eg, prednisolone 0.5 mg/kg PO) if the cat seems unwell. This will rapidly reverse the signs of hypocortisolemia and protect again insulin-induced hypoglycemia.
The efficacy of treatment should be determined by careful clinical assessment (ie, appetite, thirst, attitude, glycemic control/insulin needs, physical examination) combined with a direct measurement of cortisol production/adrenal function. An ACTH stimulation test, performed 4 h posttrilostane, is the established method for monitoring dogs on trilostane, and had also been advocated for cats.13,27 However, target values for post-ACTH cortisol levels are unclear, and the less predictable timing of the maximal response to ACTH in this species further confuses the issue. It is the authors’ opinion that a 60-min post-ACTH cortisol <50 nmol/l (1.8 μg/dl) or >250 nmol/l (9.1 μg/dl) should prompt a dose adjustment. In dogs, pre-trilostane cortisol concentrations seem to correlate more reliably with owner-derived assessment of clinical control. 51 However, more information is needed before this method can be recommended as an alternative for use in cats.
Trilostane doses needed to achieve a significant amelioration of HAC in cats vary considerably, with reported means ranging from 2.7 mg/kg q24h to 10 mg/kg q12h.13,27 Outcomes are generally positive, with survival times >12 months routinely reported.12,13,27 Most cats experience a decrease in insulin requirements;12,13,27 diabetic remission appears to be uncommon, but was recently documented in one cat following 7 months of trilostane therapy. 52
Other medical options
Prior to the approval of trilostane for canine HAC, various medical options were explored for cats with this condition, including mitotane, ketoconazole, metyrapone and aminoglutethimide.12,19,48,53,54 Outcomes were often poor, and the authors do not recommend their routine use in cats with HAC.
Radiation therapy
Outcomes following pituitary irradiation have been described for cats with PDH.12,21,22 Unfortunately, many case series include cats with hypersomatotropism, which makes it difficult to parse out information specifically pertinent to those with HAC and DM. Reports suggest that some cats with PDH undergoing radiation therapy enter diabetic remission, but the majority remain insulin dependent and may need continued medical therapy to control hypercortisolemia.12,21,22 Protocols used may impact the interval to clinical improvement, with slower responses to single high doses (eg, 1500–2000 cGy; improvement in several months) compared with fractionated protocols (eg, 18 x 300 cGy; improvement in a few weeks).
Overall, pituitary irradiation appears to be well tolerated, with minimal acute side effects. Late complications have included blindness and hearing loss. 21 Again, it is hard to determine long-term outcome in cats with PDH and DM undergoing pituitary irradiation, although reported median survival times for cats with either PDH or hypersomatotropism range from 18–25 months.21,22
Hypophysectomy
Transsphenoidal hypophysectomy, in which the pituitary gland is removed via an incision in the soft palate, has the potential to be curative in animals with PDH, and excellent outcomes have been reported in cushingoid dogs. 55 In a case series describing seven cats with PDH due to an adenoma, two died within 4 weeks of surgery. 35 However, the HAC went into clinical and biochemical remission in the other five. Four cats had concurrent DM: one died in the immediate postoperative period; one was never treated with insulin; and the other two went into diabetic remission within 5 months.
Hypophysectomized cats will need transient desmopressin therapy and life-long glucocorticoid and thyroid hormone supplementation. Recurrence of HAC has been described, and may be more likely in cats with larger tumors.
Adrenalectomy
Surgical removal of the affected gland is indicated in cats with ADH, as it may be curative in those with an adenoma and palliative, and potentially curative, in those with a carcinoma. However, there is scant information regarding the outcome of unilateral adrenalectomy in cats with ADH ± concurrent DM; most of the literature on feline adrenalectomy instead describes cats with hyperaldosteronism.17,56 Although direct comparisons between these two distinct patient populations should be made with caution, these reports indicate perioperative mortality rates of 20–30% for cats undergoing unilateral adrenalectomy with a standard laparotomy.17,56 Outcomes in the limited number of cats undergoing laparoscopic adrenalectomy appear to be superior (10% mortality rate).16,33
Postoperative hypocortisolemia is expected following removal of a cortisol-secreting tumor, and should be addressed initially with injectable dexamethasone or hydrocortisone. The cat can then be transitioned to oral prednisolone or hydrocortisone, tapered over 2–3 weeks to allow for functional recovery of the contralateral gland.
Bilateral adrenalectomy has been used to manage cats with PDH.12,15,57 Although this approach effectively abrogates HAC, cats will need life-long glucocorticoid and mineralocorticoid supplementation and are still vulnerable to neurologic issues related to the underlying pituitary tumor. In one study, 2/6 cats that survived to discharge lived >9 months. 57
The impact of preoperative trilostane treatment on perioperative mortality in cats undergoing adrenalectomy is unknown, although this is now the standard approach for dogs with ADH in the authors’ hospital, and is thought to reduce the risks of thromboembolism, sepsis and delayed healing. The available reports describing cats with concurrent HAC and DM suggest a >50% likelihood of diabetic remission following adrenalectomy. 57

Management of concurrent DM
Effective mitigation of hypercortisolemia is likely to improve insulin sensitivity and may facilitate diabetic remission. The factors that determine the onset of remission are complex, and we have limited ability to predict the likelihood of pancreatic β-cell recovery. In any individual, this represents a balance between β-cell shutdown vs apoptosis, and is probably also influenced by the extent of pancreatic islet amyloidosis and the presence of other conditions causing insulin resistance. Reports in non-cushingoid diabetic cats indicate that those that are regulated within 6 months are significantly more likely to go into remission than those with delayed regulation (84% vs 35%, respectively). 58 However, 14/18 cats with hypersomatotropism and long-standing DM achieved insulin independence following hypophysectomy; 59 this suggests that factors other than duration of hyperglycemia impact β-cell recovery.
It is certainly prudent to assume that mitigation of HAC is likely to impact insulin requirements in cushingoid diabetics, and to strategize appropriately. Clients should be encouraged to monitor glycemic status at home, using capillary samples and a veterinary-validated monitor or a flash glucose monitoring system such as the FreeStyle Libre (Abbott Laboratories). The latter has been evaluated in cats, and found to correlate well with blood glucose measurements based on a hexokinase method. 60 At-home monitoring lets the owner identify hypoglycemia and decrease the insulin dose appropriately, and should be strongly encouraged. If at-home surveillance is not an option, the cat should be regularly evaluated in the clinic.

Stress hyperglycemia can markedly impact measured BG values, but low readings can reliably indicate insulin overdose. Additionally, urine can be tested once or twice daily for the presence of glucose; a negative dipstick result suggests a prolonged period of time with BG below the renal threshold (ie, <14 mmol/l [250 mg/dl]) and should prompt an insulin dose decrease. Serum fructosamine concentration provides a similarly crude reflection of glycemic events over the previous 7–10 days; a value near to or below the reference interval indicates sustained hypoglycemia.
If in doubt, the insulin dose should always be decreased, as hypoglycemia can be fatal. Clients need to be given clear and specific instructions about the signs of hypoglycemia and how to appropriately adjust insulin therapy. It is also prudent to supply owners with a short-acting glucocorticoid (eg, oral prednisolone or injectable dexamethasone) to counter hypocortisolemia and reduce the risk of insulin-induced hypoglycemia. The latter is a significant concern in cats with compromised cortisol release, as this counter-regulatory hormone is protective against a rapid drop in BG.
Key Points
HAC is an uncommon endocrinopathy in cats, but is routinely associated with DM.
Cats with concurrent HAC and DM are not necessarily insulin resistant, and clinicians should not exclude the possibility of HAC in an apparently well-controlled diabetic.
Although cats with HAC can have dramatic dermatologic issues such as skin fragility and tearing, many patients have more subtle manifestations of their cushingoid condition. A pot-bellied appearance, a scruffy haircoat or a loss of normal skin elasticity should prompt an assessment of adrenal function.
Treatment options for the diabetic with HAC are complex and imperfect, but mitigation of HAC is expected to improve patient wellbeing and may promote the onset of diabetic remission.
Footnotes
Conflict of interest
Audrey Cook has a consulting relationship with Dechra, the manufacturer of Vetoryl (trilostane).
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
This work did not involve the use of animals and therefore ethical approval was not specifically required for publication in JFMS.
Informed consent
This work did not involve the use of animals and therefore informed consent was not required. For any animals or humans individually identifiable within this publication, informed consent (either verbal or written) for their use in the publication was obtained from the people involved.
