Abstract
A 13-year-old male, castrated, crossbred cat was referred for insulin-resistant diabetes mellitus. The cat had a ravenous appetite and a dull coat. Basal urinary corticoid/creatinine ratios were normal. In the low-dose dexamethasone suppression test there was no suppression of the (nonelevated) plasma cortisol concentration, whereas the (nonelevated) plasma adrenocorticotropic hormone (ACTH) concentration declined to low values. Basal plasma α-melanocyte-stimulating hormone (α-MSH) concentrations were highly elevated (> 1,500 ng/liter). Computed tomography revealed a pituitary tumor originating from the pars intermedia (PI). After microsurgical transsphenoidal hypophysectomy, the clinical signs resolved and the cat no longer required insulin administration. Microscopic examination of the surgical specimen revealed a pituitary adenoma originating from the PI with infiltration into the neural lobe. The adenoma immunostained intensely positive for α-MSH and only weakly for ACTH. It is concluded that the ACTH-independent cortisol production was probably due to the (weak) glucocorticorticotropic effects of the extremely high plasma concentration of α-MSH and related peptides.
In dogs, pituitary-dependent hyperadrenocorticism (PDH) is a common disease. The causative adrenocorticotropic hormone (ACTH)–producing tumor in the adenohypophysis may arise not only from the anterior lobe (AL) but also from the pars intermedia (PI). 16 , 23 In cats, PDH is a rare disease and is usually associated with diabetes mellitus.
In both dogs and cats the melanotroph cell is the abundant cell type of the PI. Corticotropic cells are a minority of the parenchymal cells of the canine PI, this being even more pronounced in the feline PI. 7 , 24 Functional and morphologic studies have provided evidence that in dogs PI tumors may originate from both corticotroph and melanotroph cells. 22 , 28 Also, in cats with PDH PI tumors have been found, staining positively for both ACTH and α-melanocyte–stimulating hormone (α-MSH). 15 So far there are no reports on the occurrence of distinct melanotroph adenomas in cats.
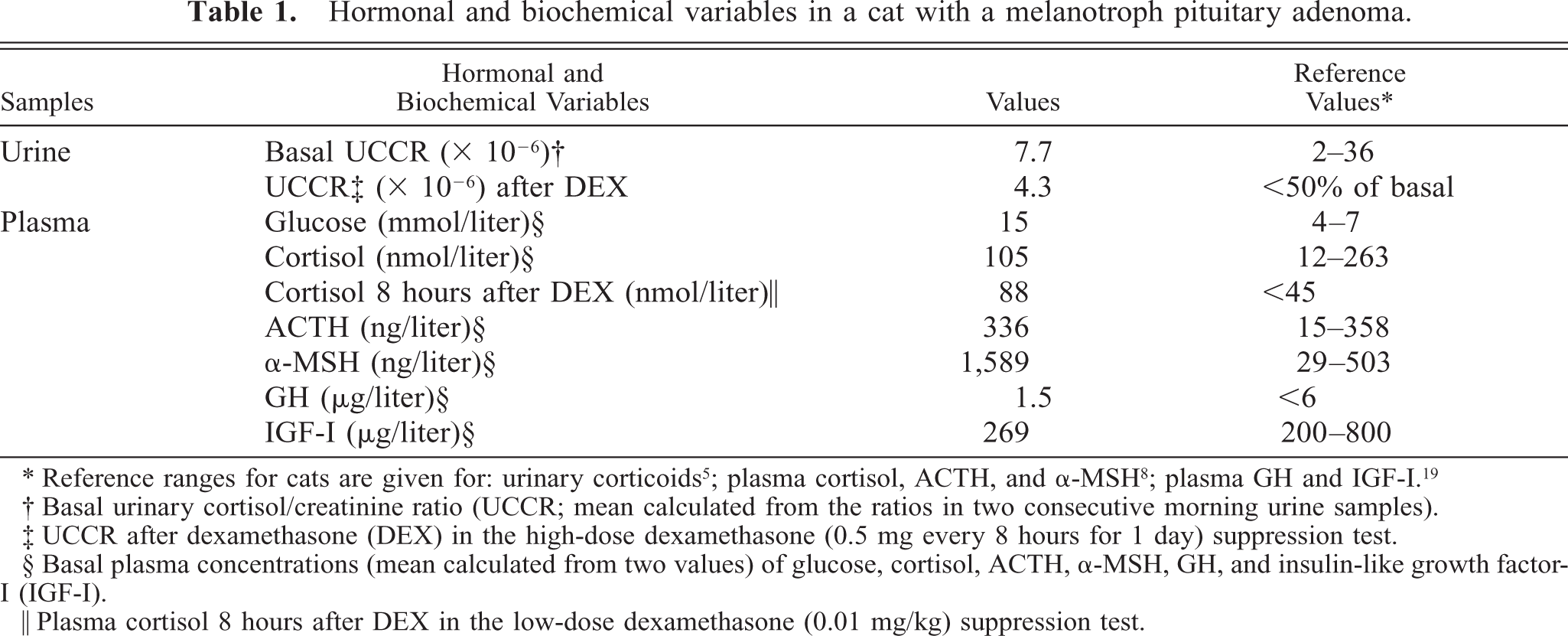
Over a period of 1 year, a 13-year-old male, castrated, crossbred cat had developed a ravenous appetite that had led to a body weight of 7.4 kg. The cat had a dull hair coat and diabetes mellitus and was injected twice daily with 11 IU of insulin (Caninsulin®, Intervet, Boxmeer, The Netherlands). Basal urinary cortisol/creatinine ratios (UCCRs), measured in two consecutive morning urine samples (11 × 10−6 and 4.3 × 10−6), were within the reference limits. Immediately after collection of the second urine sample, the cat had received three oral doses of 0.5-mg dexamethasone at 8-hour intervals. The next morning, the UCCR was 4.3 × 10−6, indicating resistance to suppression by dexamethasone. 5 Four months later UCCRs (17 × 10−6 and 14 × 10−6) were again normal. In a day curve of plasma glucose, while on insulin treatment (9 IU twice daily), the lowest plasma glucose value of 15 mmol/liter indicated insulin resistance (Table 1).
Hormonal and biochemical variables in a cat with a melanotroph pituitary adenoma.
∗ Reference ranges for cats are given for: urinary corticoids5; plasma cortisol, ACTH, and α-MSH8; plasma GH and IGF-I.19
† Basal urinary cortisol/creatinine ratio (UCCR; mean calculated from the ratios in two consecutive morning urine samples).
‡ UCCR after dexamethasone (DEX) in the high-dose dexamethasone (0.5 mg every 8 hours for 1 day) suppression test.
§ Basal plasma concentrations (mean calculated from two values) of glucose, cortisol, ACTH, α-MSH, GH, and insulin-like growth factor-I (IGF-I).
‖ Plasma cortisol 8 hours after DEX in the low-dose dexamethasone (0.01 mg/kg) suppression test.
Plasma concentrations of growth hormone (GH) 4 and insulin-like growth factor 19 were not elevated, and acromegaly was excluded as the cause of the insulin resistance (Table 1). The integrity of the hypothalamus–pituitary–adrenocortical system was further investigated by measuring plasma concentrations of cortisol, ACTH, and α-MSH in a low-dose (0.01 mg/kg body weight, intravenous administration) dexamethasone suppression test (LDDST). Plasma cortisol concentration was measured by radioimmunoassay (RIA) (Coat-A-Count® Cortisol, Diagnostic Product Corporation, Los Angeles, CA). Plasma ACTH concentration was measured using a commercially available two-site immunoradiometric assay (Nichols Institute, Wijchen, The Netherlands). The assay is highly specific for ACTH1–39 and neither cross-reacts with α-MSH nor with ACTH precursors. 2 Plasma α-MSH concentration was determined by RIA without extraction. 28
The plasma concentration of cortisol was within the reference range, and it was not suppressed by dexamethasone in the LDDST (Table 1, Fig. 1). The plasma ACTH concentration was within the reference range, but the plasma α-MSH concentration was extremely high (> 1,500 ng/liter) (Table 1, Fig. 1). Dexamethasone suppressed plasma ACTH and α-MSH concentrations by 300% and 52%, respectively, at 2 and 4 hours after the injection, but basal levels were reached again at 6 and 8 hours (Fig. 1).
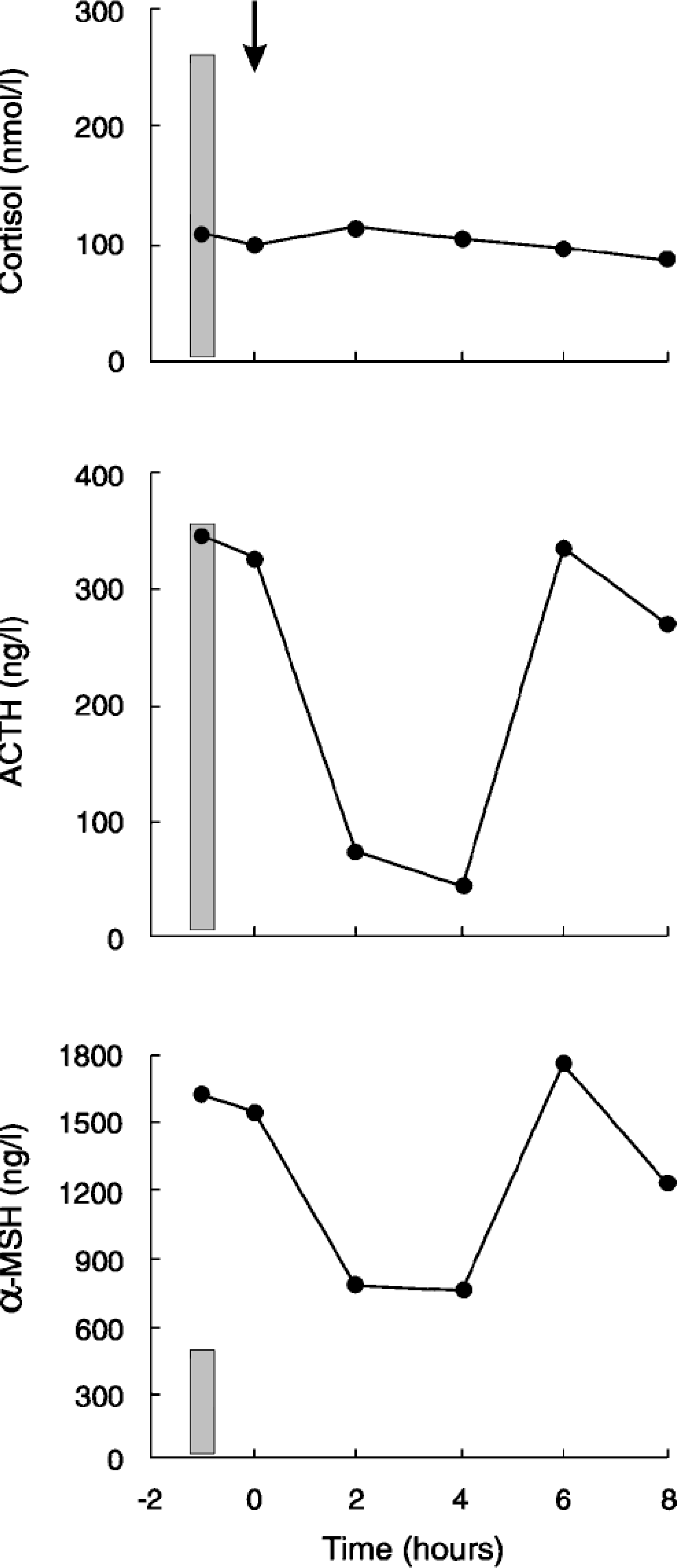
Plasma cortisol, ACTH, and α-MSH concentrations in a cat with a melanotroph pituitary adenoma, before and after the injection of low-dose (0.01 mg/kg body weight) dexamethasone (arrow). Shaded areas represent basal values for healthy cats. 8 .
Preoperative contrast-enhanced computed tomography (CT) images 15 revealed a uniformly enhanced and enlarged pituitary gland bulging dorsally (Fig. 2). Dynamic CT revealed a large strongly enhancing area in the right-central part of the pituitary gland (Fig. 3), indicating the presence of a pituitary tumor. During transsphenoidal hypophysectomy, 14 , 15 the caudal and ventral part of the pituitary was judged to be normal and collected as the unaffected specimen (specimen 1). The rostral and dorsal part of the pituitary was judged to be abnormal and was collected as the tumor specimen (specimen 2).
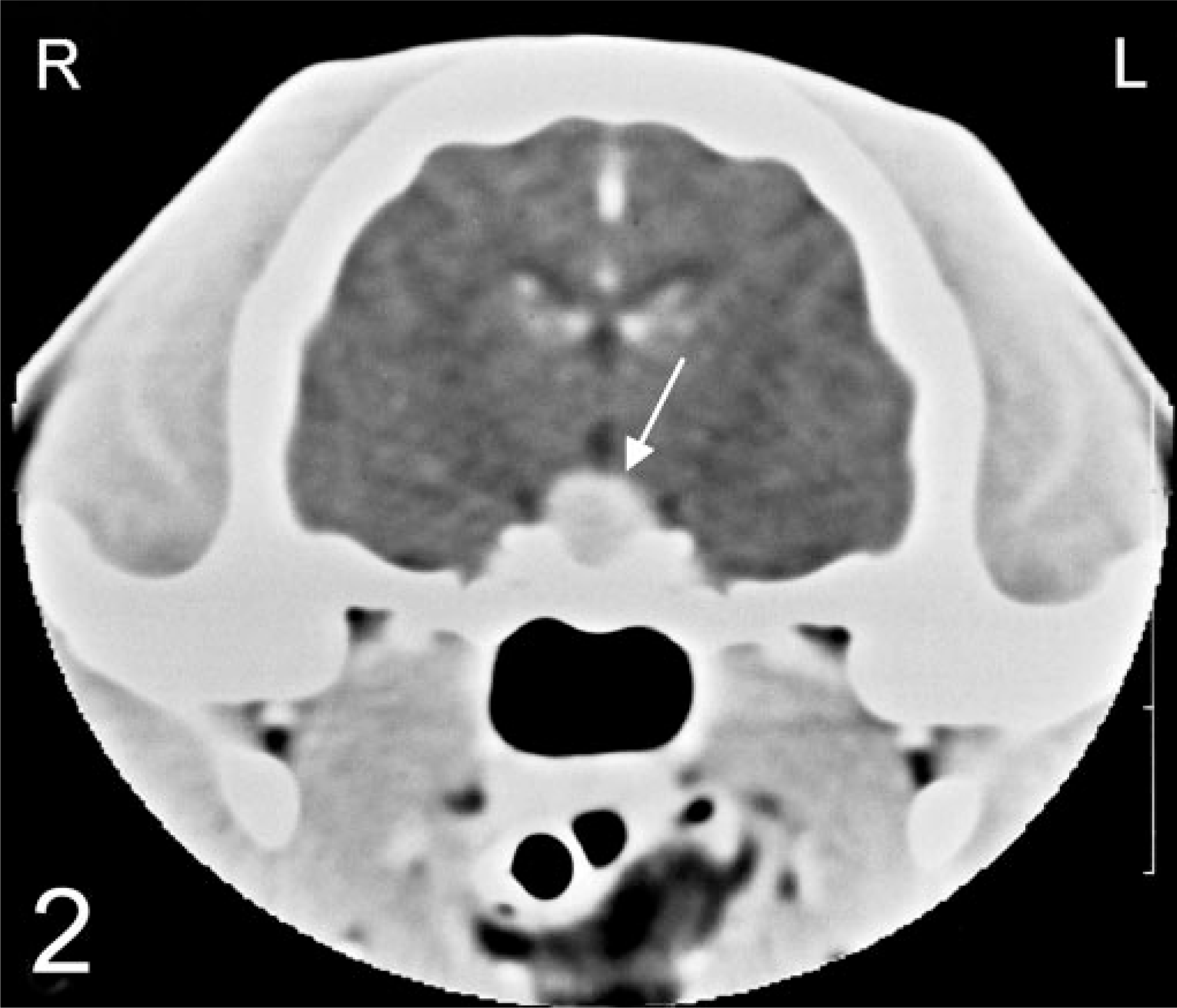
Contrast-enhanced computed tomography in a 13-year-old male, castrated, domestic shorthair cat with a melanotroph pituitary adenoma. A transverse image revealed an enlarged, uniformly enhancing pituitary gland (arrow) measuring 6.2 mm in height and 6.8 mm in width.
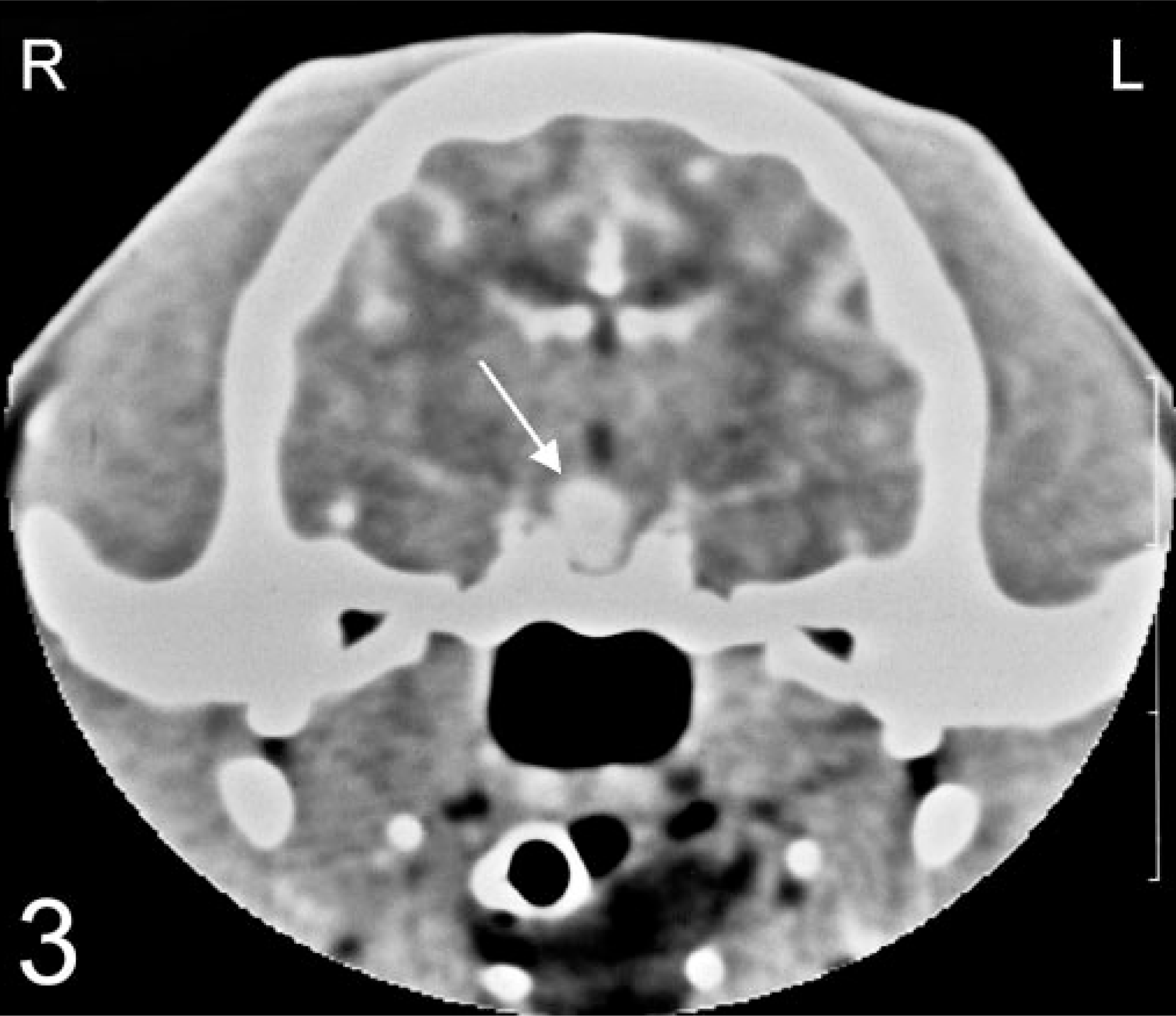
Dynamic contrast-enhanced computed tomography in a 13-year-old male, castrated, domestic shorthair cat with a melanotroph pituitary adenoma. A strongly enhancing area (arrow) in the right-central part of the pituitary indicated the presence of an adenoma.
Immunohistochemical staining was performed by the avidin–biotin–peroxidase complex (ABC) procedure. The primary antibodies were a monoclonal mouse antibody to synthetic ACTH1–24 (Department of Infectious Diseases and Immunology, Faculty of Veterinary Medicine, Utrecht University, Utrecht, The Netherlands), a polyclonal rabbit antibody to synthetic human α-MSH (PU060-UP, Biogenex Laboratories, San Remon, CA), and a polyclonal rabbit antibody to porcine GH.
25
Normal canine pituitary tissue served as control tissue. The dilution factor for the antibodies was 1 : 100 for ACTH, 1 : 5,000 for GH, and 1 : 600 for α-MSH. The deparaffinized sections were treated with 1% H2O2 for 30 minutes to neutralize endogenous peroxidases. In consecutive steps, sections were incubated with the primary antibody at 4 C for 16 hours for ACTH and α-MSH and for 90 minutes for GH; with the biotinylated secondary antibody (horse anti-mouse immunoglobulin [IgG] for ACTH, goat anti-rabbit IgG for α-MSH and GH) for 30 minutes at room temperature; and with avidin–peroxidase conjugate for 30 minutes. Finally, immunoreactivity was observed by incubation with 0.3% H2O2 and 3,3′-diaminobenzidine tetrahydrochloride (DAB) 0.5%, diluted in Tris/HCl buffer, for 10 minutes. The slides were counterstained with Mayer's hematoxylin. Consecutive sections of the tumor specimen were furthermore immunostained with three heterologous antisera in rabbits raised against
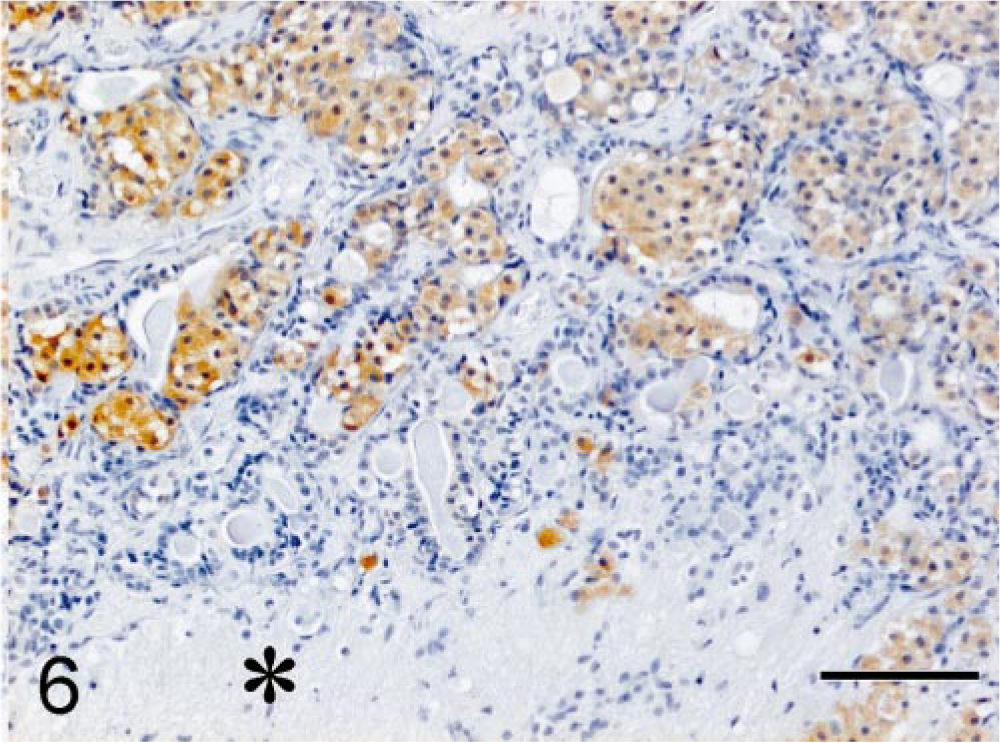
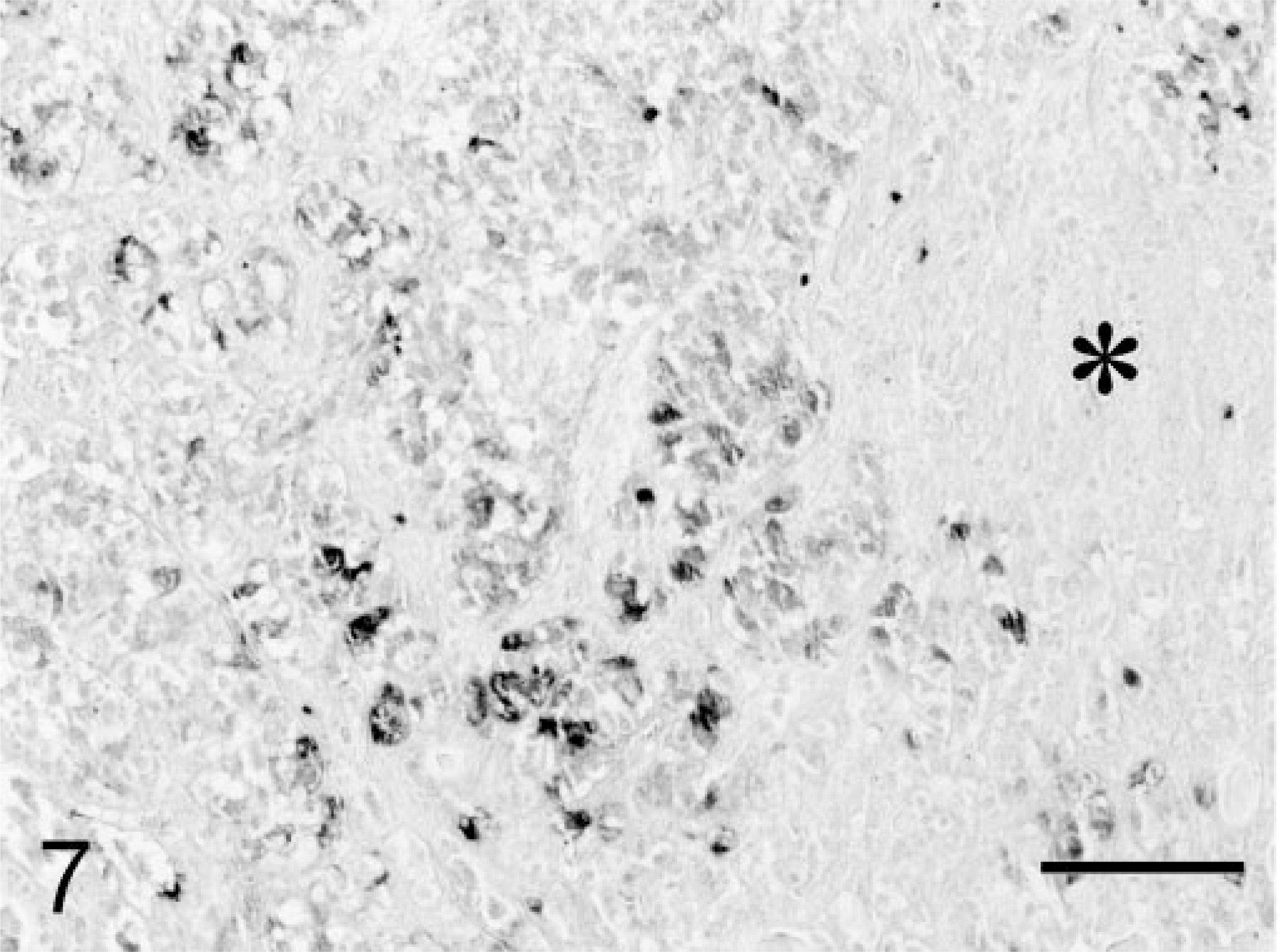
Microscopic examination of hematoxylin and eosin (HE)–stained sections of specimen 1 revealed unaffected adeno-hypophyseal tissue containing a normal mixture of acidophilic, basophilic, and chromophobic cells. Specimen 2 contained some unaffected adenohypophyseal tissue and a large pituitary tumor mass. The pituitary tumor consisted of solid fields and trabecular regions of basophilic cells with a moderate amount of cytoplasm and large round oval nuclei with a prominent nucleolus (Fig. 4). The adenoma showed infiltration of the PI and the neurohypophyseal lobe (Fig. 4). Specimen 1 stained immunohistochemically positive for GH, but there was no staining for ACTH and α-MSH. In specimen 2, the adenomatous tissue stained negative for GH and POMC, positive for ACTH1–24 (Fig. 5) and synthetic α-MSH (Fig. 6), moderately positive for carp ACTH10–23 (Fig. 7), and intensely positive for ACTH1–13 (Fig. 8).
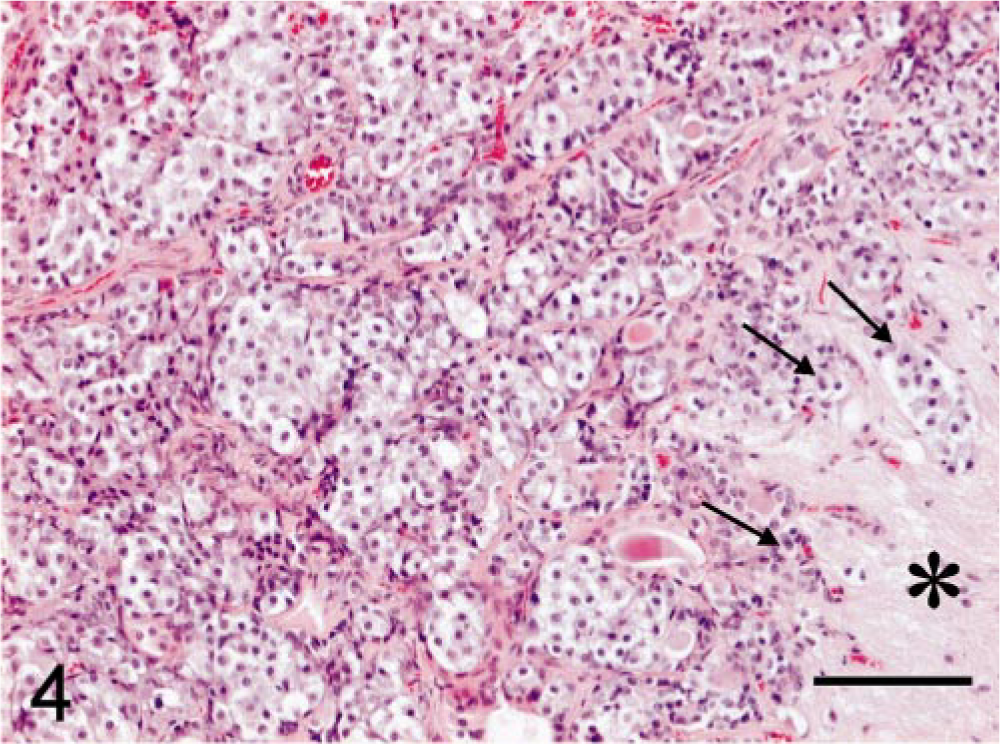
Melanotroph pituitary adenoma; cat. Basophilic cells infiltrating (arrows) the neurohypophyseal lobe (asterisk). HE. Bar = 100 µm.
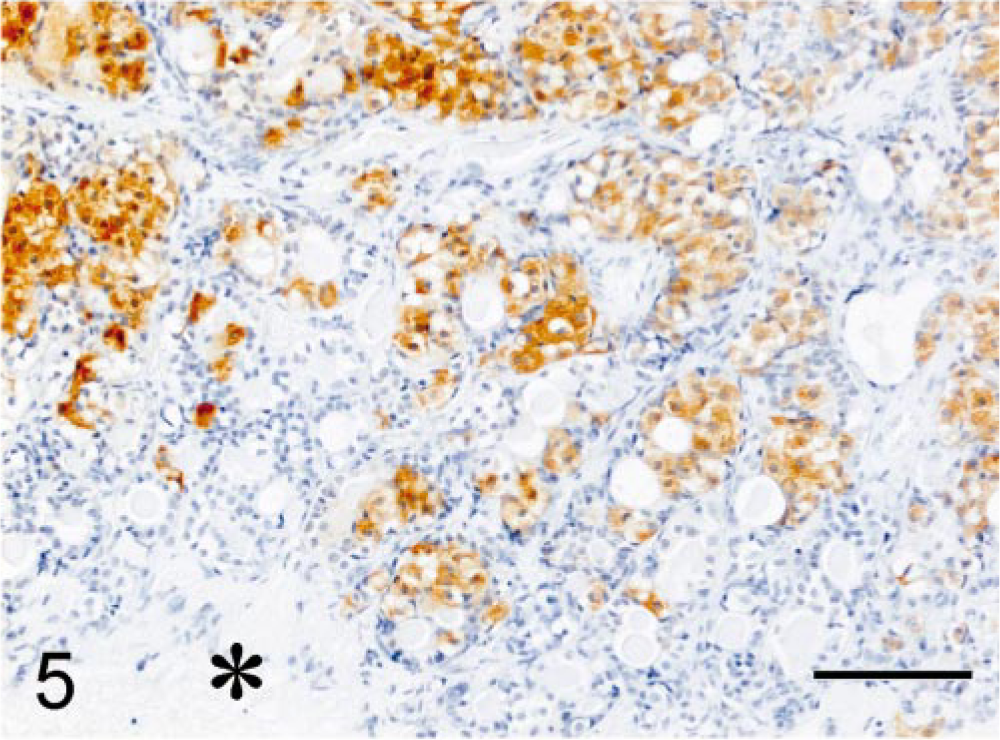
Melanotroph pituitary adenoma; cat. Positive immunohistochemical staining of the adenomatous tissue for ACTH1–24. ABC complex method, Mayer's hematoxylin counterstain. Asterisk = neurohypophyseal lobe. Bar = 100 µm.
Melanotroph pituitary adenoma; cat. Positive immunohistochemical staining of the adenomatous tissue for synthetic α-MSH. ABC complex method, Mayer's hematoxylin counterstain. Asterisk = neurohypophyseal lobe. Bar = 100 µm.
Melanotroph pituitary adenoma; cat. Moderate positive immunohistochemical staining of the adenomatous tissue for carp ACTH10–23. PAP conjugate method, DAB/Ni complex stain. Asterisk = neurohypophyseal lobe. Bar = 100 µm.
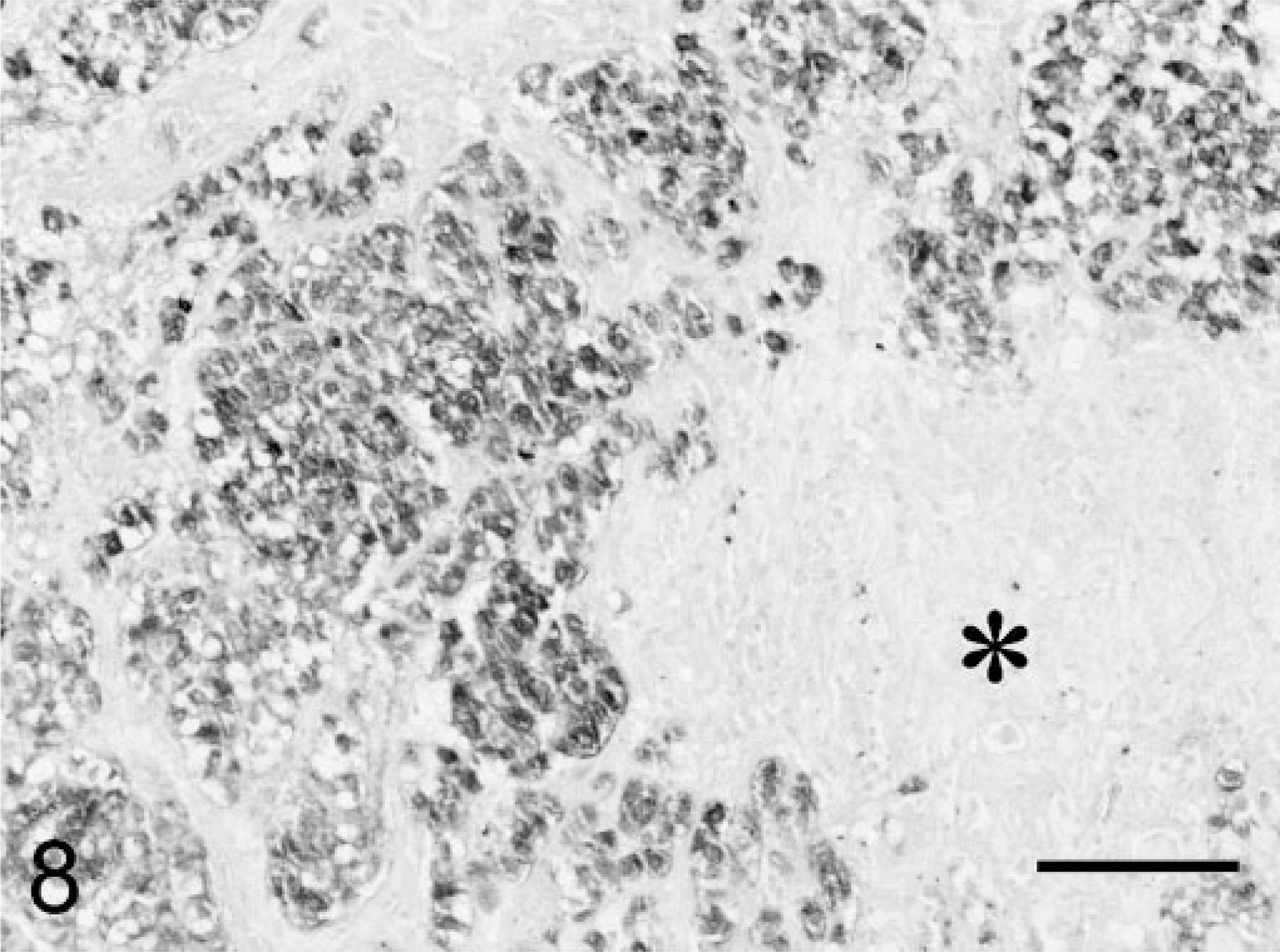
Melanotroph pituitary adenoma; cat. Intense positive immunohistochemical staining of the adenomatous tissue for ACTH1–13 (= α-MSH). PAP conjugate method, DAB/Ni complex stain. Asterisk = neurohypophyseal lobe. Bar = 100 µm.
Recovery from surgery was uncomplicated: the cat started to eat and drink the day after surgery. The insulin demand decreased steadily from 11 IU twice a day, and insulin treatment was discontinued at day 6 after surgery. The cat was discharged from the hospital on desmopressin, cortisone acetate, and thyroxine as described previously. 14 , 15 Polydipsia and polyphagia resolved, and there was renewal of the hair coat 3 months after surgery. The cat returned to its normal eating behavior. UCCR in samples collected at home were 2.8 × 10−6 and 3.4 × 10−6 (1 month after surgery) and 0.9 × 10−6 and 0.5 × 10−6 (3 months after surgery). Five months after surgery the cat died unexpectedly. No postmortem examination was performed.
The cat had presented with insulin-resistant diabetes mellitus, insatiable appetite, and a dull hair coat, a combination of features characteristic of feline hyperadrenocorticism. However, there was no evidence of hypercortisolism because urinary corticoids and plasma cortisol concentrations were within the reference range. There was resistance to suppression by dexamethasone of UCCR in the high-dose dexamethasone suppression test and plasma cortisol concentrations in the LDDST. In contrast, plasma ACTH concentrations declined strongly in the LDDST. Apparently, the cortisol production was not under direct control of pituitary ACTH. This may be explained by the extremely high plasma concentrations of α-MSH that were only partially suppressed in the LDDST. In a dog with a POMC-producing pituitary tumor not causing hyperadrenocorticism, not only ACTH but also cortisol was suppressed in the LDDST. However, in that case the plasma α-MSH concentrations were not as highly elevated as in the cat in this study. 6 POMC-derived peptides, other than ACTH (e.g., lipotropin and α-MSH), are known as weak glucocorticotropic hormones. 20 , 21 , 26 It is likely that the weak glucocorticotropic activity of α-MSH accounted for the dexamethasone resistance and possibly also for some of the clinical signs resembling those in hyperadrenocorticism.
POMC-derived peptides such as β-endorphin and α-MSH are exclusive products of melanotroph cells in the PI and play a key role in appetitive aspects of feeding. Melanocortin receptors in mice studies were involved with the development of obesity, insulin resistance, and diabetes mellitus. 18 Alpha-MSH is anorexigenic, and β-endorphin has an acute stimulatory effect on food consumption. 1 , 3 , 11 , 17 In individuals with pituitary tumors, the release of these (and other) POMC-derived peptides in supraphysiologic quantities may disturb the regulatory balance of food intake. Increased food intake will lead to obesity, which in turn causes peripheral insulin resistance and ultimately diabetes mellitus.
Pituitary imaging supported PI localization of the tumor. The neurointermediate lobe (NIL) has a direct arterial vascularization through the caudal hypophyseal artery, whereas the AL receives blood by way of the long portal vessels. Dynamic CT enables separate observation of the NIL and the AL, which is reflected by an early and strong enhancement of the NIL (‘pituitary flush’) and a delayed (less strong) enhancement of the peripheral AL. 27 The cat in this study exhibited the pituitary flush sign, which was enlarged and extended to the right side, indicating an early-enhancing pituitary tumor of the NIL.
In the surgery specimen the pituitary adenoma showed infiltration into the neural lobe, which is enveloped by the PI. This localization of the adenoma proved the PI origin of the tumor. Absence of immunostaining for POMC, positive staining for α-MSH (= ACTH1–13), and only very moderate staining for ACTH10–23 allowed the diagnosis of pituitary melanotroph adenoma.
Pituitary adenomas are considered benign tumors. However, in humans they may invade surrounding tissues such as dura mater, cavernous sinus, and the sphenoid sinus. 12 On microscopic examination, the incidence of dural invasion by pituitary adenomas has been reported to be as high as 45%. 12 The so-called giant invasive pituitary adenomas are characterized by one or more of the following features: 1) extensive invasion of dura mater and cavernous sinus, 2) extensive suprasellar extension with lateralization, and 3) extensive erosion of the skull base and invasion of sphenoid sinus. 9 These tumors look histologically benign, but because of their extension and infiltration of regional surrounding structures, they have an aggressive and rather malignant clinical course (high rate of recurrence after surgical resection). 9 The authors realize that there is a contradiction in the term ‘invasive adenoma.’ Nevertheless, for comparative reasons the authors feel that the use of this terminology in the cat in this study as well is justified.
Pituitary carcinoma is a diagnosis that is best made on both pathologic and clinical criteria. Although histologic features may show cellular pleomorphism, mitotic figures, and nuclear atypia, which are suggestive of a high proliferating potential, the clinical course may still be relative benign. For this reason, the diagnosis of pituitary carcinoma has been reserved for tumors with cerebrospinal or extracranial metastasis. 10 Invasive pituitary adenomas are not considered pituitary carcinomas. 10 In the cat in this study, the adenoma was found to infiltrate the neural lobe, and histologically the tumor was an adenoma. There were no characteristics of a pituitary carcinoma.
In cats presenting with insulin-resistant diabetes mellitus, one should consider GH excess (acromegaly) and hyperadrenocorticism. In addition, there is the possibility of a double adenoma, i.e., somatotroph and corticotroph adenoma leading to GH excess and hypercortisolism. 13 As illustrated by this case, there is also the possibility that such a cat harbors a melanotroph adenoma in the PI.
