Abstract
Pituitary glands from 141 feline autopsy cases were reviewed histologically. Adenoma and hyperplasia were the most common lesions at 13 cases each. Pituitary adenoma was more likely than hyperplasia to be associated with clinical evidence of endocrinopathy or an intracranial mass (
Keywords
Proliferation (hyperplasia or neoplasia) is the most common pituitary lesion in domestic mammals.
25,38
However, in a review of pituitary glands from 65 feline autopsy cases,
36
only one proliferative lesion (hyperplasia) was encountered, and pituitary cysts, considered an incidental finding, were the most common lesion (8 cats). In a review of magnetic resonance (MR) images from 46 cats with histologically confirmed intracranial neoplasms, only 1 pituitary tumor was found.
47
Although pituitary neoplasia is uncommon in cats, pituitary tumors (
Tumors of the pituitary gland and sellar region account for 15% of human intracranial tumors, with pituitary adenoma being the most common skull-base tumor. 24,31,46 The 2017 World Health Organization classification of human pituitary adenomas 31 is based on immunohistochemistry (IHC) for adenohypophyseal cell lineage transcription factors (pituitary transcription factor 1 [Pit-1], pituitary-restricted transcription factor [TPit], and steroidogenic factor 1 [SF-1]) and trophic hormones. Designation of a pituitary neuroendocrine tumor as carcinoma currently requires documentation of cerebrospinal or systemic metastasis. 31 The International Pituitary Pathology Club proposed the term pituitary neuroendocrine tumor (PitNET) to avoid the histologic distinction between pituitary adenoma and carcinoma. 37 Although PitNET is not part of the World Health Organization classification, the term recognizes the potential for aggressive behavior in a minority of pituitary tumors currently classified as adenoma and the existence of other types of neuroendocrine tumor. 24,31,37,46
Much of the knowledge of feline pituitary neoplasia comes from individual case reports. 7,12,13,16,17,34 Feline pituitary neuroendocrine tumors are most commonly classified as somatotroph adenomas and less commonly as corticotroph, or at least adrenocorticotrophic hormone (ACTH)-expressing, adenomas. 15,17,35 A few melanotroph, plurihormonal, and double adenomas have also been reported. 3,6,14,20,21,43
Growth hormone production by a somatotroph adenoma is the usual cause of feline hypersomatotropism, which can lead to the phenotypic changes of acromegaly. 9,13,29,30,42 In a study of 21 acromegalic cats, 41 the pituitary lesion was either somatotroph hyperplasia or adenoma, based on expansion or disruption, respectively, of reticulin fibers. An ACTH-producing adenoma is the usual cause of feline pituitary-dependent hypercortisolism. 18,28,49 Insulin-resistant diabetes mellitus is an important and common complication of both feline hypersomatotropism and hypercortisolism. 1,4,18,35,49
Treatment options for feline pituitary adenoma include radiation, somatostatin analogs, dopaminergic agonists, and transsphenoidal hypophysectomy, 2,5,8,10,33,39,40,44,45 but depend on the endocrine functionality of the tumor. Hypophysectomy has the advantage of allowing antemortem histologic diagnosis. 18,19,22,23,34 However, comprehensive IHC panels for pituitary trophic hormones are not widely available in veterinary diagnostic laboratories.
The objectives of this study were to determine the prevalence of pituitary lesions in cats and to correlate histologic and immunohistochemical features of feline pituitary adenomas with clinical findings and case outcome. The hypotheses were that pituitary adenoma is the most common clinically relevant hypophyseal lesion and that most feline pituitary adenomas are derived from somatotrophs with fewer corticotroph, melanotroph, or plurihormonal adenomas.
Materials and Methods
Case Selection
Records for all cats in which the pituitary gland had been examined histologically were retrieved from the Indiana Animal Disease Diagnostic Laboratory (ADDL) from July 2000 through January 2019. To evaluate the prevalence of pituitary lesions, only postmortem specimens were used. Both postmortem and hypophysectomy specimens from the ADDL were used for the histochemistry and IHC of pituitary adenomas. Additional cases of pituitary adenoma were selected from the Animal Medical Center (AMC), New York City, database by searching feline pathology reports from March 2008 to July 2019 with the terms pituitary neoplasm, hyperplasia, or adenoma.
Cases were included if histologic sections of adequate sample size without severe autolysis or tissue artifact were available. Signalment, history, clinical findings, and pathologic findings were tabulated from medical records, autopsy or biopsy submission forms, and pathology reports. Hematoxylin and eosin (HE)–stained sections of the pituitary gland were classified 15,25 into one or more of the following categories: histologically normal, adenohypophyseal proliferation (hyperplasia or adenoma), cysts, or miscellaneous lesions.
Histochemistry
Adenohypophyseal proliferations were classified as hyperplasia if a nodule composed mainly of one cell type expanded an intact reticulin network, and as adenoma if the reticulin fibers were lost or disrupted. 25 Pituitary adenomas were categorized as acidophilic, basophilic, or chromophobic by tinctorial characteristics of cytoplasmic granules in HE-stained sections. Sections were also evaluated with periodic acid-Schiff (PAS) and with Gordon and Sweet’s reticulin.
Immunohistochemistry
Immunohistochemistry (see Supplemental Table S1 for antibodies, reagents, and procedures) was performed on pituitary adenomas for growth hormone (GH), prolactin, ACTH, melanocyte-stimulating hormone (MSH), follicle-stimulating hormone (FSH), luteinizing hormone (LH), and thyroid-stimulating hormone (TSH). For the AMC cases (cats 19–43), LH IHC was used only for adenomas that expressed FSH or that did not express any other trophic hormone. Normal feline pituitary gland was used to standardize each trophic hormone antibody and as the positive control tissue. For the negative reagent control, a universal negative control for mouse and rabbit antibodies (Biocare) was used instead of the primary antibody. The IHC protocol utilized the dilution, retrieval method, and detection system with the best signal-to-noise ratio. A proliferation index was calculated for hypophysectomy specimens as the percentage of neoplastic cells with Ki-67-immunopositive nuclei. At least 1000 cells were counted (count tool in Adobe Photoshop version 21.1.0) in photomicrographs taken at 400× magnification. 27
Medical Record Review
The following data were compiled for cats with pituitary adenoma: age, breed, sex, and body weight at biopsy or autopsy; maximal pituitary dimension as determined from MR images, 11 pathology reports, or histologic sections; clinical signs of hypersomatotropism/acromegaly, hypercortisolism, diabetes mellitus, hyperthyroidism, or other endocrine disease; case outcome and postdiagnosis or postsurgical survival time. Criteria for a diagnosis of hypersomatotropism were elevated serum insulin-like growth factor (IGF)-1 concentration after insulin treatment for at least 2 weeks and documentation of pituitary enlargement. Clinical features of acromegaly, such as weight gain, broad facial features, abdominal organ enlargement, prognathia inferior, enlarged paws, or heart murmur, although present in many cases, were not required to confirm the diagnosis. A diagnosis of diabetes mellitus was based on the presence of compatible clinical signs, persistent hyperglycemia and glycosuria, and/or elevated serum fructosamine concentration. A diagnosis of pituitary-dependent hypercortisolism was based on presence of clinical signs (eg, insulin-resistant or poorly controlled diabetes mellitus, endocrine alopecia, or skin fragility), compatible adrenal function test results (ACTH stimulation or dexamethasone suppression test), and documentation of pituitary enlargement. Hyperthyroidism was diagnosed on the basis of compatible clinical signs (eg, weight loss, polyphagia, polyuria, polydipsia), presence of a palpable thyroid nodule, and increased total thyroxine (TT4) serum concentration or high normal TT4 with elevated free T4 (FT4) concentration.
Data Analysis
Fisher’s exact test was used to evaluate the correlation between a histologic diagnosis of pituitary hyperplasia or adenoma and clinical evidence of endocrinopathy or mass effect. The association of sex (male vs female) with pituitary adenoma type was also evaluated by Fisher’s exact test. The survival of cats with somatotroph or melanotroph adenoma was estimated using the Kaplan-Meier method. Median survival time was reported. Cox proportional hazards regression was used to compare hazard rate between cats treated by hypophysectomy and nonsurgical cases while adjusting for age, adenoma size, and adenoma type. Significance was set at
Results
Prevalence of Pituitary Lesions
Initially, 167 feline accessions with histologic examination of the pituitary gland were retrieved from the ADDL records, but hypophysectomy cases (8) were excluded from the lesion prevalence study. In addition, 18 postmortem cases were excluded because no histologic sections were available for review. Of 141 cases that met the inclusion criteria, 105 pituitary glands were histologically normal. Lesions in the remaining 36 glands were classified as adenoma (
Hyperplastic nodules were up to 1 mm in diameter with expanded but intact reticulin fiber network, and composed of basophilic to chromophobic cells or of acidophils. Adenohypophyseal hyperplasia was not associated with clinical pituitary-dependent disease, except for one cat with somatotroph hyperplasia and a diagnosis of hypersomatotropism. Cats with pituitary adenoma were more likely (12/13) than cats with hyperplasia (1/13) to have clinical evidence of endocrinopathy or of an intracranial mass (
Histochemistry and Immunohistochemistry of Pituitary Adenomas
Twelve postmortem cases (one of which had recurred after hypophysectomy 2 years earlier) plus 8 hypophysectomy specimens of pituitary adenoma from ADDL accessions were selected for histochemistry and IHC, but 1 postmortem and 1 surgical case were excluded for lack of sufficient paraffin-embedded lesional tissue. The histochemistry and IHC panel was also applied to pituitary tumors from 26 cats (19 postmortem and 7 hypophysectomy specimens) from AMC. One of those was immunohistochemically negative for all trophic hormones, diagnosed as a histiocytic sarcoma based on immunoreactivity for Iba-1, and excluded from the study. In total, the histochemistry and IHC panel was applied to pituitary adenomas from 43 cats.
The 43 cats had 44 pituitary tumors that were classified as 20 somatotroph adenomas, 11 melanotroph adenomas, 11 gonadotroph adenomas, and 2 thyrotroph adenomas. Findings for the 44 adenomas are summarized in Table 1; individual cat data are in Supplemental Table S2. None of the adenomas expressed prolactin.
Clinical and Pathologic Features of 44 Pituitary Adenomas in 43 Cats.
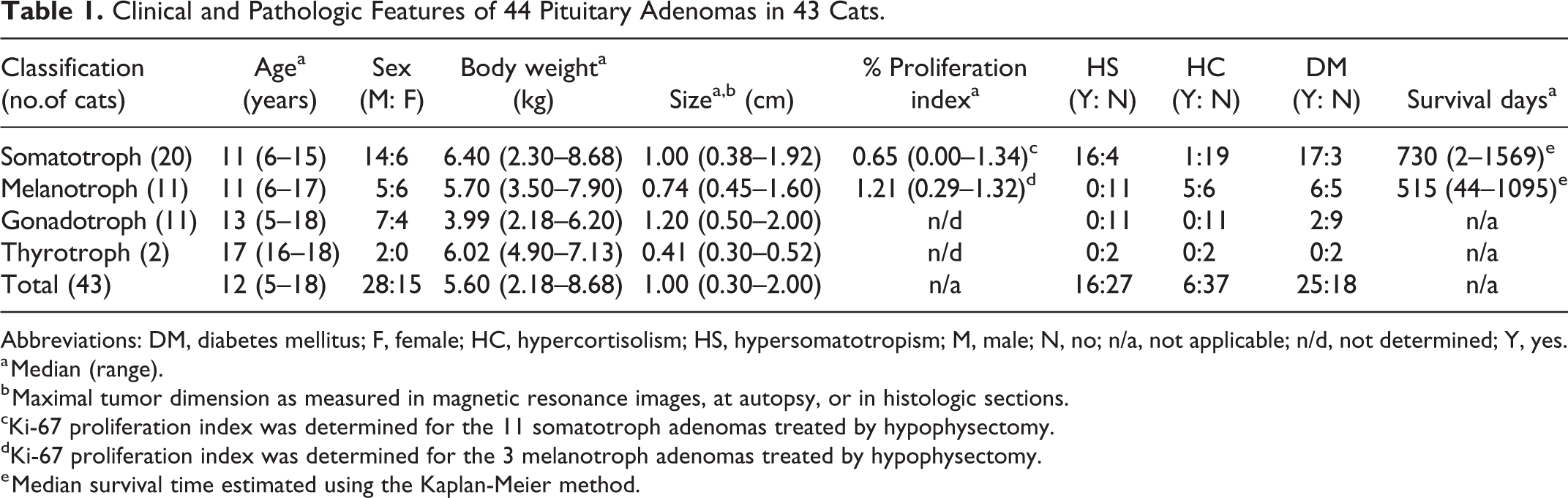
Abbreviations: DM, diabetes mellitus; F, female; HC, hypercortisolism; HS, hypersomatotropism; M, male; N, no; n/a, not applicable; n/d, not determined; Y, yes.
a Median (range).
b Maximal tumor dimension as measured in magnetic resonance images, at autopsy, or in histologic sections.
cKi-67 proliferation index was determined for the 11 somatotroph adenomas treated by hypophysectomy.
dKi-67 proliferation index was determined for the 3 melanotroph adenomas treated by hypophysectomy.
e Median survival time estimated using the Kaplan-Meier method.
Twenty somatotroph adenomas (Figs. 1–4) comprised 46% of all feline pituitary adenomas and 11/14 (79%) of hypophysectomy cases. With the exception of one sparsely granulated and pale acidophil tumor, neoplastic somatotrophs had densely granulated and brightly eosinophilic cytoplasm. The cytoplasmic granules were PAS-negative and expressed growth hormone, but none of the other trophic hormones.
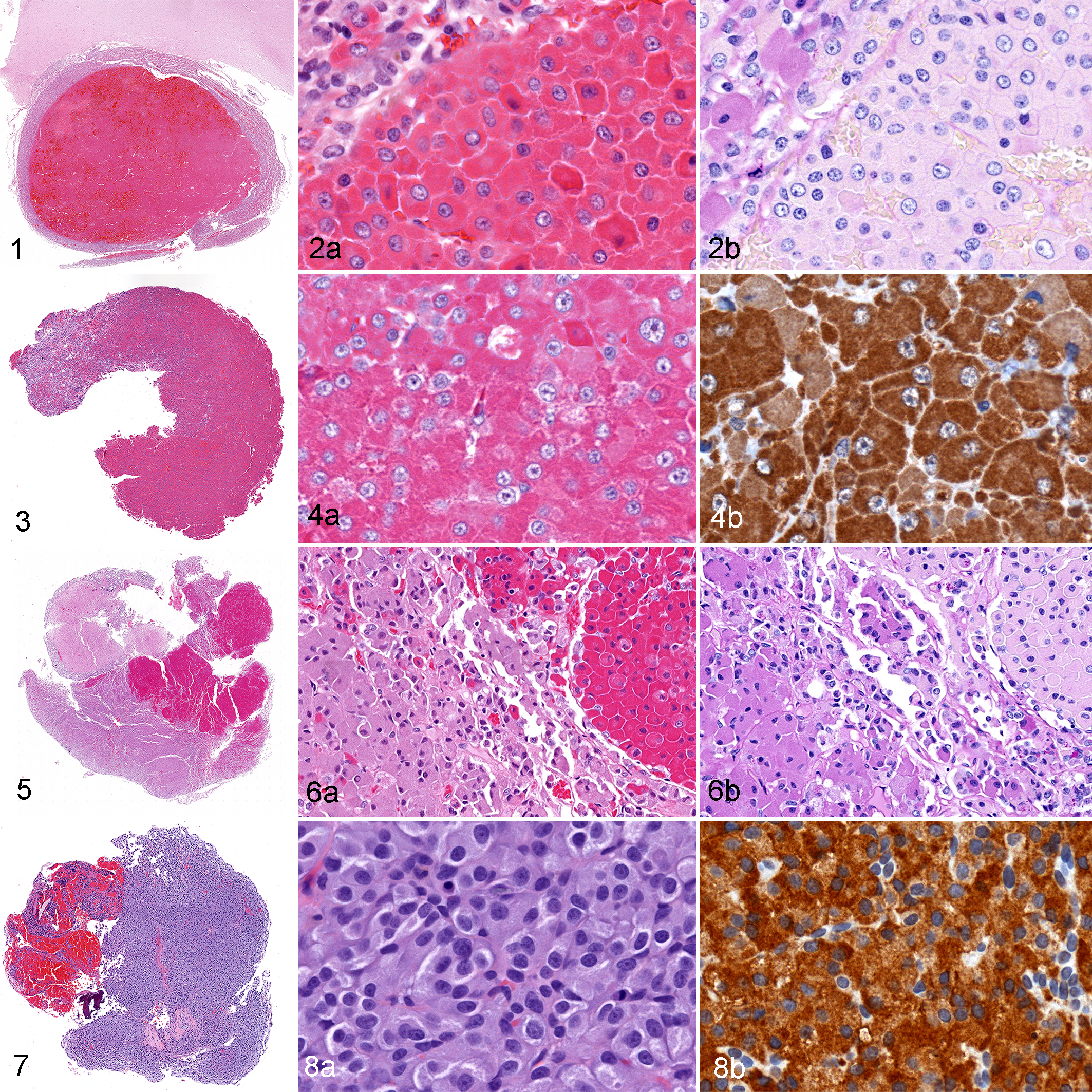
Somatotroph adenoma, pituitary gland, cat 3. The acidophil adenoma is surrounded by nonneoplastic pars distalis and compresses the overlying hypothalamus. Hematoxylin and eosin (HE).
Plurihormonal adenomas were not observed in this series, but one cat (cat 37) had a double adenoma (Figs. 5, 6). The separate somatotroph and melanotroph microadenomas were 0.38 cm and 0.45 cm in diameter, respectively. The double adenoma was discovered at autopsy; the cat had no history of hypersomatotropism, hypercortisolism, diabetes mellitus, or other endocrine disease. Another cat (cat 43) had a diagnosis of double adenoma in its medical record, with an acidophil adenoma in the pars distalis and a separate ACTH-immunoreactive chromophobic microadenoma in the pars intermedia. However, only the acidophil adenoma remained in the paraffin block when the current study commenced. It was classified as a somatotroph adenoma, and the ACTH-positive microadenoma was not included in this study because it could not be reviewed or evaluated for other trophic hormones.
Eleven melanotroph adenomas (Figs. 7, 8) comprised 25% of all pituitary adenomas and 3/14 (21%) of the hypophysectomy cases. Neoplastic melanotrophs were basophilic to chromophobic cells that were PAS-positive and expressed both MSH and ACTH, but none of the other trophic hormones.
Eleven gonadotroph adenomas (Figs. 9–11) comprised 25% of all pituitary adenomas, but none of the hypophysectomy cases. Neoplastic gonadotrophs were chromophobic, PAS-negative cells that expressed FSH and/or LH, but none of the other trophic hormones. Nine of the 11 gonadotroph adenomas expressed both FSH and LH; two expressed only LH.
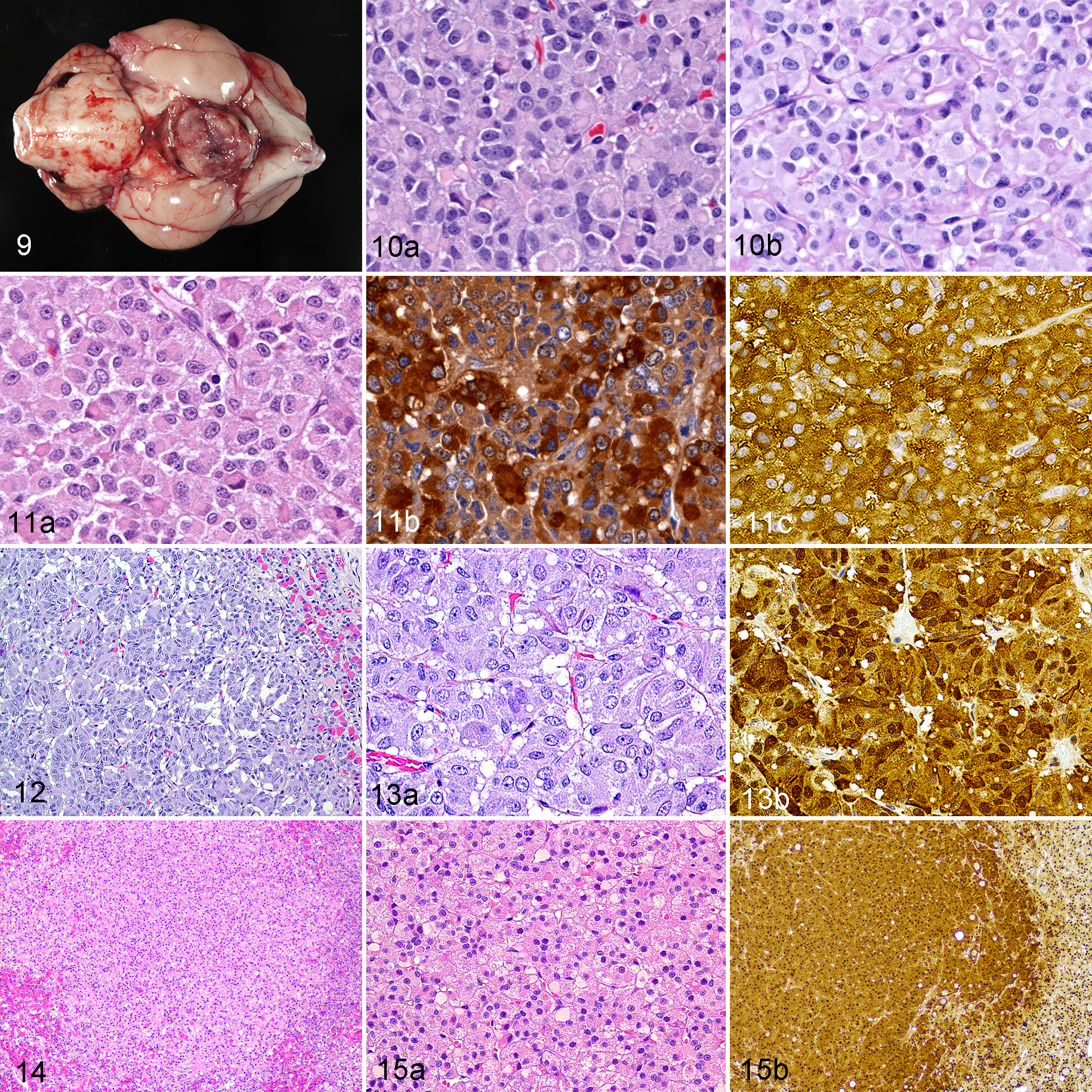
Gonadotroph adenoma, pituitary gland, cat 12. The sellar mass encroaches on cranial nerves and compresses the hypothalamus and thalamus.
The 2 thyrotroph adenomas (Figs. 12–15) accounted for 4.5% of all pituitary adenomas, but none of the hypophysectomy cases. The smaller (0.3 cm diameter) thyrotroph adenoma (Figs. 12, 13) consisted of basophilic, PAS-negative cells with moderate to strong TSH expression. The larger (0.52 cm) adenoma (Figs. 14, 15) consisted of chromophobic, PAS-negative cells with weak to moderate TSH expression.
Clinical Findings and Case Outcome in Cats With Pituitary Adenoma
Thirty-one of the 43 cats were domestic shorthairs; other breeds included American shorthair, domestic longhair, Ragdoll, Rex, and Siamese. Age, sex, body weight, tumor size, proliferation index (for surgical cases only), and clinical diagnosis of hypersomatotropism, hypercortisolism, or diabetes mellitus are summarized for each type of pituitary adenoma in Table 1. The male-to-female ratio was 1.75 for all cats with pituitary adenoma and 2.33, 0.83, 1.75, and 2.00 for cats with somatotroph, melanotroph, gonadotroph, or thyrotroph adenomas, respectively. The sex distribution among cats with the 4 different types of pituitary adenoma did not differ significantly (
Hypersomatotropism was diagnosed in 16 of the 20 cats with somatotroph adenoma and in none of the cats with other types of pituitary adenoma. Hypercortisolism was diagnosed in 5/11 cats with melanotroph adenoma. One of the cats (cat 43) with a somatotroph adenoma had clinical signs more consistent with hypercortisolism than hypersomatotropism. This cat had elevated serum endogenous ACTH and bilateral adrenocortical hyperplasia at autopsy, but the ACTH stimulation test result was normal. On review of original pathology records, the cat had, in addition to the somatotroph adenoma evaluated in the current study, a pars intermedia ACTH-positive microadenoma (immunohistochemistry by a reference laboratory) that, unfortunately, was no longer in the paraffin block, so could not be evaluated in the current study. None of the cats with gonadotroph or thyrotroph adenomas had hypercortisolism. Hypergonadotropism was not diagnosed in any cat, but 4 cats (cats 4, 11, 15, 31) with gonadotroph adenoma had a history of neurologic disease (obtunded or nonspecified neurologic clinical signs). The median tumor size (1.6 cm) was larger than for cats without a history of neurologic signs (0.9 cm). One of the 2 cats (cat 41) with a thyrotroph adenoma had bilateral thyroid follicular hyperplasia and granulomatous thyroiditis along with a history of cardiac hypertrophy that was attributed to hyperthyroidism. Cat 37 with the double somatotroph and melanotroph adenomas was the only other cat with history of hyperthyroidism.
A diagnosis of diabetes mellitus was most common (17/20) in cats with somatotroph adenoma. Six of 11 cats with a melanotroph adenoma also had diabetes mellitus. Two of the 11 cats with gonadotroph adenoma had diabetes mellitus, but insulin-resistance was not noted, and pituitary disease was not suspected antemortem.
Postdiagnosis survival was evaluated only for cats with somatotroph or melanotroph adenoma because gonadotroph or thyrotroph adenomas were not suspected antemortem. Cats with somatotroph adenoma had longer median survival than those with melanotroph adenoma (Table 1). Cats with somatotroph adenoma treated by hypophysectomy (
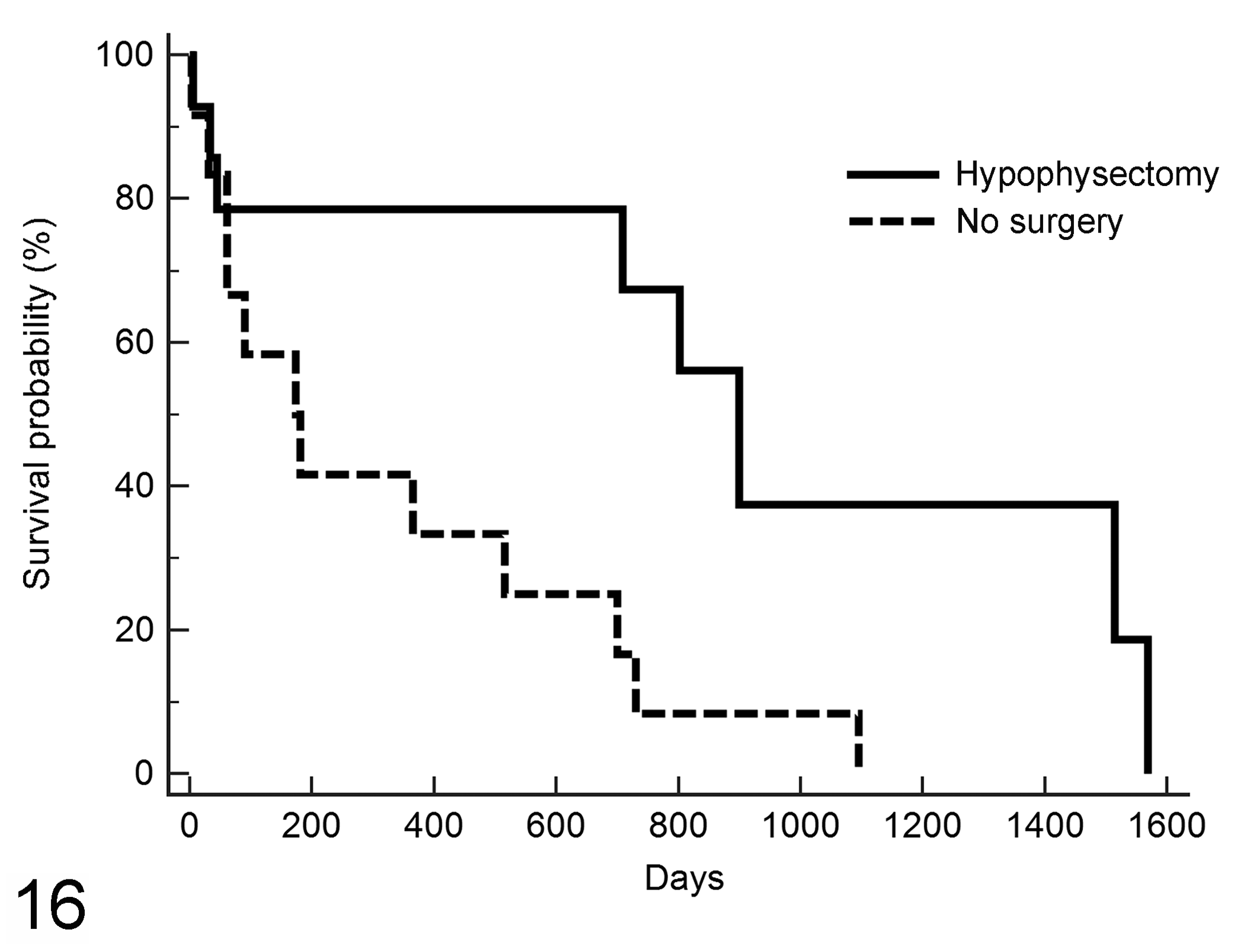
Postdiagnosis survival in 31 cats with somatotroph or melanotroph adenoma treated by hypophysectomy (
Discussion
As in other domestic mammals, 25,26 adenohypophyseal hyperplasia and adenoma were the most common diagnoses in archived feline pituitary glands. However, only 1 case of hyperplasia (and no adenoma) was found in a review of 65 feline autopsy cases. 36 Hyperplastic lesions were generally considered incidental findings in the current study because, with one exception, clinical evidence of pituitary disease was not reported. In contrast, in a study of 21 acromegalic cats, 41 the pituitary lesion in 10 of the 15 with altered reticulin structure was classified as hyperplasia; however, the authors reported inter-pathologist variation in interpretation of reticulin fiber disruption.
In the current study, the histochemical and IHC features of 44 pituitary adenomas in 43 cats were correlated with clinical findings and case outcome. Somatotroph adenomas were most common (20 cats), followed by melanotroph and gonadotroph adenomas (11 each), and 2 thyrotroph adenomas. Pituitary-dependent clinical disease was observed with most somatotroph and melanotroph adenomas, but not with gonadotroph or thyrotroph adenomas. Cats that underwent hypophysectomy for either somatotroph or melanotroph adenoma survived longer than nonsurgical cases. This study underscores the importance of immunohistochemical classification and surgical treatment of feline pituitary adenomas.
The derivation of feline pituitary adenomas from all 3 adenohypophyseal cell lineages (Pit-1, TPit, and SF-1) is like that in human PitNETs, 24,31,46 though corticotroph adenoma is the only TPit-lineage human tumor (melanotroph adenomas are not recognized among human PitNETs, potentially because the human pars intermedia regresses postnatally). Neither corticotroph adenomas nor lactotroph adenomas were diagnosed in the cats, though both are major types of human PitNET. In contrast, canine and equine pituitary adenomas are usually derived from just one lineage, TPit, 15,25,38 with dogs developing corticotroph or melanotroph adenomas, 27 and almost all equine pituitary adenomas derived from melanotrophs. 15,25
Most cats with pituitary adenoma other than melanotroph adenoma were male. However, although hypersomatotropism is reportedly more common in male cats, 35 the male-to-female ratio in cats with somatotroph adenoma in this study did not differ significantly from that for other types of pituitary adenoma. A larger sample size might have resulted in detection of a significant difference.
Pituitary adenoma was suspected or confirmed antemortem in most cats with somatotroph adenoma and in about half the cats with melanotroph adenoma because of the secondary endocrine disease. 1,29,49 Although the neutered status of these cats would have dampened expression of hypergonadotropism, all gonadotroph adenomas were clinically nonfunctional, and all cases were diagnosed postmortem. Gonadotroph adenoma is also a major human tumor, accounting for about 75% of nonfunctional PitNETs. 31 Neither of the 2 feline thyrotroph adenomas was suspected antemortem. However, one of the cats had a clinical history of hyperthyroidism with thyroid follicular hyperplasia at autopsy.
The median proliferation index, which was only determined for cats that underwent hypophysectomy, was higher (though ranges overlapped) in the 3 melanotroph adenomas than in the 11 somatotroph adenomas, and cats with melanotroph adenoma had shorter median survival time than cats with somatotroph adenoma. Cats treated by hypophysectomy had an overall longer survival compared with cats that were not treated surgically, even after adjusting for age and adenoma size and type.
As expected, 15,17,35 somatotroph adenoma was the most common feline pituitary adenoma, especially among hypophysectomy cases. Somatotroph adenomas account for 10% to 27% of resected human PitNETs. 24,46 Humans with acromegaly usually have densely granulated somatotroph adenomas with benign behavior; sparsely granulated somatotroph adenomas tend to be more aggressive and less responsive to somatostatin analogs. 24 Lactotroph adenomas, the most common functional human PitNET, 24,46 were not diagnosed in this feline series. Nor were mixed somatotroph and lactotroph (dual cell population) adenomas, mammosomatotroph (single cell population) adenomas, or Pit-1 plurihormonal adenomas diagnosed in the current study. Co-expression of prolactin in somatotroph hyperplastic or neoplastic lesions was also rare in a study of 21 cats with acromegaly. 41
The lack of corticotroph adenomas in the current study was unexpected, because ACTH-producing pituitary adenomas have long been linked to feline hypercortisolism. 18,21 –23 Corticotroph tumors accounted for 13% of resected human PitNETs in one study. 46 Corticotroph adenomas, which express ACTH but not MSH, are also common in dogs. 27 However, all ACTH-expressing adenomas in this feline study also expressed MSH, so were classified as melanotroph adenoma. Because IHC for MSH is not routine in all veterinary diagnostic laboratories, feline melanotroph adenomas may have been underdiagnosed or misclassified as corticotroph adenomas based on their ACTH expression. As in the dog, 27 hypercortisolism was diagnosed in about half the cats with a melanotroph adenoma.
The number of gonadotroph adenomas (25% of the pituitary adenomas) in this study was unexpected. Although 2 cats with gonadotroph adenoma had a history of diabetes mellitus, pituitary-associated endocrine disease was not suspected, and none of the gonadotroph adenomas was diagnosed antemortem. Four of the cats presented for neurologic signs, presumably from the mass-effect of the adenoma. Gonadotroph adenomas, other than a few plurihormonal adenomas that expressed FSH, 6,14 are seldom reported in cats, but may be underdiagnosed because routine IHC of feline pituitary adenomas seldom includes antibodies to FSH or LH. In addition, a comprehensive IHC panel may be more commonly applied to the classification of hypophysectomy specimens than to postmortem cases. Human gonadotroph adenomas are common (35%) among resected PitNETS, 46 and typically have a mass-effect rather than clinical evidence of hormone overexpression. 24 They comprise the majority of nonfunctional human PitNETs, 31 and before IHC for SF-1 became routine, were often misclassified as null cell adenomas for failure to express any trophic hormone. 24 The current diagnostic criterion for null cell adenoma, also known as immunonegative tumor, and now comprising only 2% of resected human PitNETs, 46 is the lack of IHC expression of pituitary transcription factors or trophic hormones. 32 Null cell adenomas were not diagnosed in this feline study.
Thyrotroph adenoma is rare in all species, 15,24 so it was surprising to encounter 2 cases (4.5%) in this series. Thyrotroph adenoma is the least common (2%) human PitNET, 46 but has been diagnosed in hyperthyroid, hypothyroid, and euthyroid patients. 24 Although 1 of the 2 cats with thyrotroph adenoma had a clinical diagnosis of hyperthyroidism and thyroid follicular hyperplasia at autopsy, serum TSH levels were not recorded. Blood TSH concentration is typically low in feline hyperthyroidism because of the negative feedback from excessive thyroid hormones, but would be expected to be high if the hyperthyroidism were due to a thyrotroph adenoma.
Diabetes mellitus was the most common clinical endocrine disorder in the cats with pituitary adenoma. Not surprisingly, most cats with somatotroph adenoma had insulin-resistant diabetes mellitus, attributable to GH-induced increased production of IGF-1. 30 Diabetes mellitus was also detected in about half the cats with melanotroph adenoma. The cause for diabetes in 2 cats with gonadotroph adenoma was not determined, but non-pituitary-dependent predisposing conditions include genetics, diet, pancreatitis, islet amyloidosis, and obesity. 39
The diversity of feline pituitary adenomas highlights the importance of both a standardized diagnostic assessment and a broad IHC panel for trophic hormones, particularly as transsphenoidal hypophysectomy becomes a more common treatment. Reticulin histochemistry facilitates the distinction of pituitary adenoma from adenohypophyseal hyperplasia. 15,25,41 Acidophil adenomas, such as somatotroph adenomas or lactotroph adenomas (not diagnosed in the current study), can be distinguished from basophil or chromophobe adenomas in HE-stained sections. The presence of PAS-positive cytoplasmic granules is a marker of proopiomelanocortin-producing cells and thus expected only in corticotroph (not diagnosed in the current study) or melanotroph adenomas. 15,25
Immunohistochemistry is necessary for accurate adenoma classification. Certain non-neuroendocrine skull-base tumors, for example, histiocytic sarcoma, can resemble pituitary adenoma, so generic IHC markers can be used to eliminate these mimics or to document neuroendocrine origin. A comprehensive trophic hormone IHC panel may not be necessary in every case. An algorithm (Fig. 17) is proposed for limited IHC of feline pituitary adenomas. Somatotroph origin, expected for acidophil adenomas, should be documented with IHC for growth hormone. Prolactin IHC could be reserved for growth hormone-negative acidophil adenomas or to detect mixed somatotroph/lactotroph adenoma or mammosomatotroph adenoma, all of which seem to be rare in the cat. Feline basophil or chromophobe adenomas should be evaluated with IHC for ACTH; all ACTH-positive adenomas require MSH IHC to distinguish melanotroph from corticotroph origin. The pathologist should have a high level of suspicion for gonadotroph origin in postmortem cases, so FSH and LH should be part of the IHC panel for chromophobe adenomas that are ACTH-negative. Because thyrotroph adenomas seem particularly rare in cats, IHC for TSH could be reserved for those PitNETs that are negative for all other trophic hormones. For biopsy specimens, Ki-67 IHC may provide prognostic information, but more feline pituitary adenomas should be studied to correlate the proliferation index with case outcome.
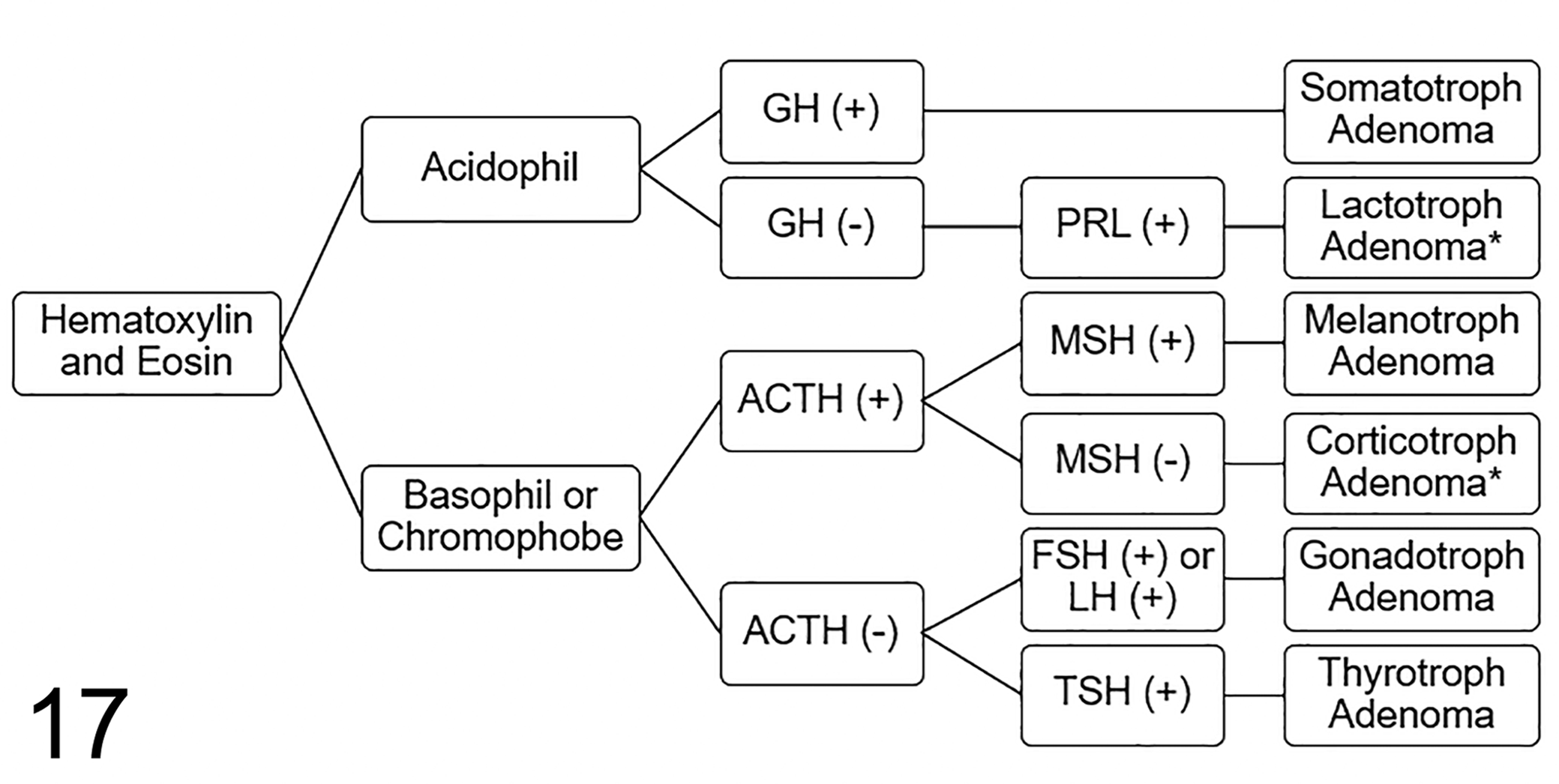
Diagnostic algorithm for trophic hormone immunohistochemistry of feline pituitary adenomas. Abbreviations: ACTH, adrenocorticotrophic hormone; FSH, follicle-stimulating hormone; GH, growth hormone; LH, luteinizing hormone; MSH, melanocyte-stimulating hormone; PRL, prolactin; TSH, thyroid-stimulating hormone. *Not diagnosed in this series.
Supplemental Material
Supplemental Material, Combined_supplemental_materials-Miller_et_al - Feline Pituitary Adenomas: Correlation of Histologic and Immunohistochemical Characteristics With Clinical Findings and Case Outcome
Supplemental Material, Combined_supplemental_materials-Miller_et_al for Feline Pituitary Adenomas: Correlation of Histologic and Immunohistochemical Characteristics With Clinical Findings and Case Outcome by Margaret A. Miller, Stacey L. Piotrowski, Taryn A. Donovan, J. Catharine Scott-Moncrieff, Tina J. Owen, John P. McCue, Deidre M. DuSold, José A. Ramos-Vara, Hsin-Yi Weng, Annie V. Chen, Linda G. Martin and David S. Bruyette in Veterinary Pathology
Footnotes
Acknowledgements
The authors thank histology technicians, pathologists, and pathology residents at the Indiana Animal Disease Diagnostic Laboratory, Washington Animal Disease Diagnostic Laboratory, and Animal Medical Center. The authors gratefully acknowledge the assistance of Lizelle Hidalgo, Purdue University; Connie Fraser, Washington State University; and Amanda Ramkissoon, Animal Medical Center, in immunohistochemistry, patient management, and data acquisition, respectively.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Author DSB is employed by Anivive Lifesciences, which provided financial support to the study. The other authors have no conflicts of interest to disclose.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported in part by Anivive Lifesciences, Long Beach, California. S. Piotrowski is a fellow in the NIH Comparative Biomedical Scientist Training Program and supported by the National Cancer Institute and the National Institute of Neurological Disorders and Stroke, in partnership with Purdue University.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
