Abstract
Practical relevance:
Procedural sedation and analgesia (PSA) describes the process of depressing a patient’s conscious state to perform unpleasant, minimally invasive procedures, and is part of the daily routine in feline medicine. Maintaining cardiopulmonary stability is critical while peforming PSA.
Clinical challenges:
Decision-making with respect to drug choice and dosage regimen, taking into consideration the cat’s health status, behavior, any concomitant diseases and the need for analgesia, represents an everyday challenge in feline practice. While PSA is commonly perceived to be an uneventful procedure, complications may arise, especially when cats that were meant to be sedated are actually anesthetized.
Aims:
This clinical article reviews key aspects of PSA in cats while exploring the literature and discussing complications and risk factors. Recommendations are given for patient assessment and preparation, clinical monitoring and fasting protocols, and there is discussion of how PSA protocols may change blood results and diagnostic tests. An overview of, and rationale for, building a PSA protocol, and the advantages and disadvantages of different classes of sedatives and anesthetics, is presented in a clinical context. Finally, injectable drug protocols are reported, supported by an evidence-based approach and clinical experience.
Sedation in cats: a routine challenge in feline practice
Sedation, chemical restraint and analgesia are part of the daily routine in feline practice. In some cats, especially those with fractious temperaments or showing fearful or excited behavior, sedation is required to render the patient cooperative. It is also used to facilitate diagnostics (eg, venipuncture for hematology, imaging, etc) (see box).

The ultimate goal of sedation is to provide comfort and often analgesia while reducing fear, anxiety and stress in these patients. Sedation will also prevent inadvertent injuries to personnel and promote a better hospital experience for cats undergoing minor procedures. However, the choice of drugs and dosage regimens can be challenging since there is no ‘one size fits all’; protocols should be adjusted and adapted on a case-by-case basis. The clinician must consider the cat’s health status and behavior, concomitant diseases, the need for analgesia, and the magnitude of the procedure and hence the level and duration of sedation required. Other challenges in sedation and analgesia include drug unavailability and lack of familiarity with specific medicines.
Procedural sedation and analgesia (PSA) is a term used in human medicine to describe the process of depressing a patient’s conscious state, in order to perform unpleasant, minimally invasive or objectionable procedures. 1 This clinical review explores the scientific literature to address complications and risk factors, as well as drug protocols used for PSA in cats. Recommendations are made using an evidence-based approach and the authors’ experience.
It’s not ‘just a quick sedation’: complications and risk factors
Sedation is regularly seen as an uneventful procedure with a low risk of complications. However, some protocols used for PSA may induce unconsciousness, amnesia and the loss of protective reflexes (ie, general anesthesia). There may be a mistaken belief that these patients are ‘just sedated’ when they are, in fact, anesthetized, and a more comprehensive monitoring and supportive care plan should be in place given that complications may easily arise. In addition, PSA is not always safer than general anesthesia and is often chosen for its practicality rather than representing the best option for the cat. For instance, general anesthesia offers better airway control and an easy means of ventilation, monitoring and oxygenation when cats are intubated for more extensive and invasive procedures. A study showed that profound sedation may not be suitable for all patients and can increase the risk of anesthesia-related death. 2 Therefore, the decision between PSA and general anesthesia should be taken cautiously, evaluating the advantages and disadvantages of each technique.
Documented risk factors for sedation-related morbidity and mortality are scarce in the veterinary literature.2–4 When risks have been reported, they are often presented in combination with data from anesthetized patients.2,3 This creates difficulty in distinguishing sedation- from anesthesia-specific risk factors. However, one study reported that the risks of mortality following sedation and anesthesia are not significantly different. 3 Extrapolation from previous studies evaluating sedation- and anesthesia-related risk factors can, therefore, provide useful information when determining the likelihood of complications during PSA.
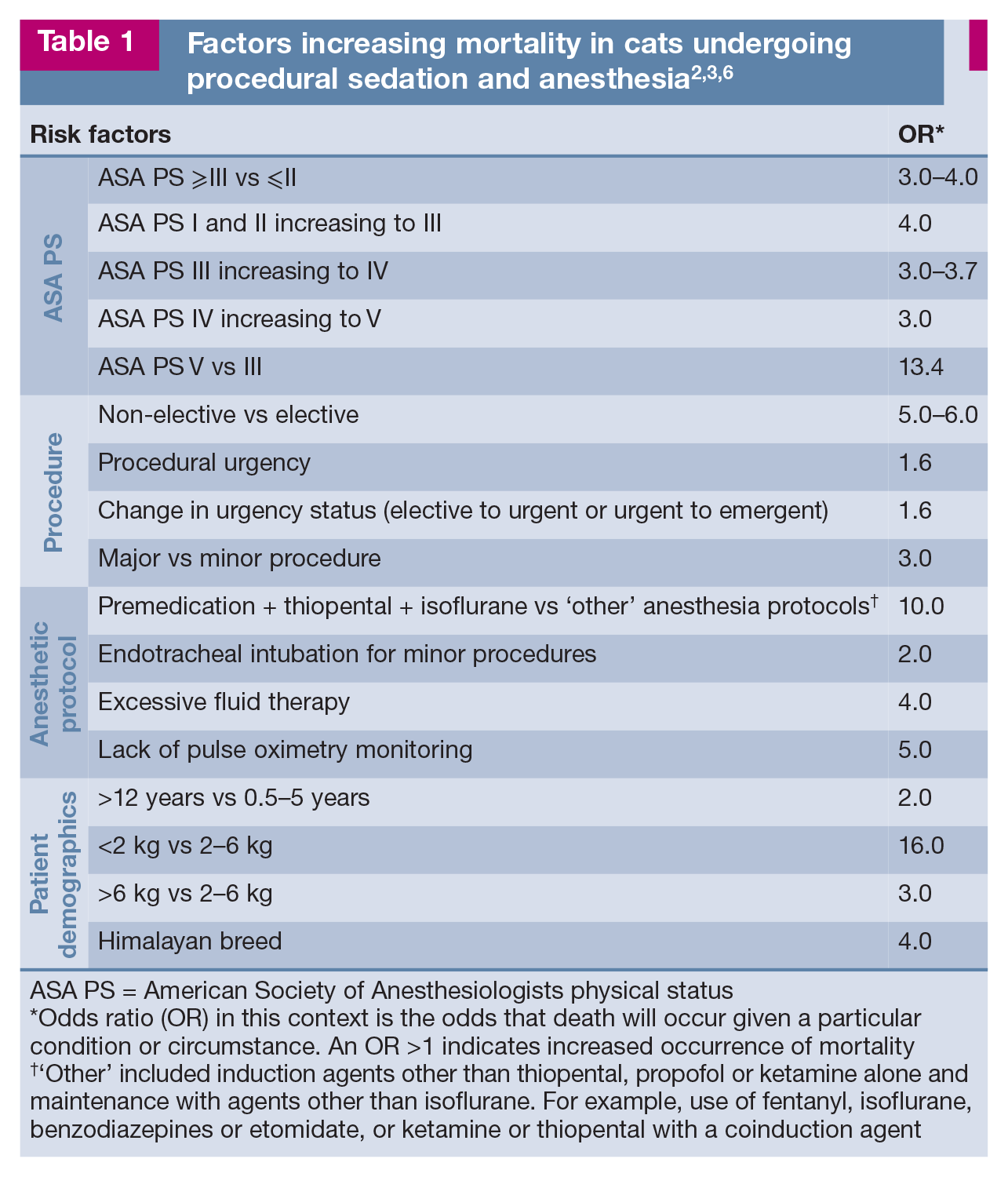
Cats have a higher risk of anesthetic death than dogs.2,3,5–7 The rate of mortality following anesthesia and/or sedation in cats can be as high as 0.24%. 3 The primary causes for anesthesia- or sedation-related mortality in cats are presented in Figure 1. An association between extremes in age and body weight and an increased risk of mortality following sedation and anesthesia has been recognised in cats.2,3 Senior and pediatric patients are more susceptible to the depressant effects of anesthetics, have prolonged recovery due to decreased hepatic blood flow and impaired thermoregulation, and have limited ability to respond to abnormal physiologic states such as hypotension.3,8 Obese patients are not only susceptible to comorbidities due to an enhanced pro-inflammatory state, but they also have reduced cardiovascular reserves and impaired ventilation and mobility due to excessive fat deposition. 3 Intravenous (IV) access, endotracheal intubation and cardio-pulmonary monitoring can be difficult in small cats and kittens. In terms of cat breeds, Himalayans have an increased risk of complications, likely due to their brachy-cephalic conformation, 9 making them prone to respiratory compromise and aspiration pneumonia during anesthesia.10,11 Other risk factors for feline sedation- and anesthetic-related death are detailed in Table 1.

Primary causes of sedation- and anesthesia-related deaths in cats. From Brodbelt et al (2008) 5
ASA PS = American Society of Anesthesiologists physical status
Odds ratio (OR) in this context is the odds that death will occur given a particular condition or circumstance. An OR >1 indicates increased occurrence of mortality
‘Other’ included induction agents other than thiopental, propofol or ketamine alone and maintenance with agents other than isoflurane. For example, use of fentanyl, isoflurane, benzodiazepines or etomidate, or ketamine or thiopental with a coinduction agent
Fluid therapy may be required during PSA. The administration of inappropriate quantities of IV fluids has been found to significantly increase the likelihood of anesthesia- and/or sedation-related deaths in cats. 3 Guidelines for maintenance fluids rate during anesthesia have been published in cats, recommending a significantly lower rate than previously suggested (3–5 vs 5–10 ml/kg/h). 12 Inappropriate use of IV fluids in euvolemic patients can result in volume overload and the development of pulmonary and peripheral edema. A thorough evaluation of the patient’s hydration status and estimated losses during the procedure should help guide the veterinarian in determining if IV fluids are required, and the type and rate of administration to compensate for any ongoing losses and dehydration.
Lastly, fasting may reduce the risk of regurgitation and aspiration pneumonia and, in turn, the sequelae of desaturation, hypoxemia, sympathetic activation and potentially death (see ‘patient assessment and preparation before PSA’). Veterinarians should thus avoid or minimize regurgitation and consider general anesthesia with intubation of the trachea in cats with severe central nervous system, respiratory, cardiovascular or gastrointestinal disorders.

Patient assessment and preparation before PSA
Preanesthetic assessment should be performed in cats before PSA as it may identify potential risk factors and avoid complications. It is not uncommon for a procedure to start under PSA and evolve into a more complex anesthetic challenge. There is a particular problem when procedures become significantly more painful and inadequate analgesia (eg, a weak opioid analgesic) has been administered as part of the PSA protocol. In these circumstances, alternative analgesic agents may be required as the weaker opioids (eg, butorphanol or buprenor-phine) may negatively impact the analgesic efficacy of pure opioid agonists (eg, morphine, methadone and hydromorphone).18,19
The patient’s identification should be confirmed and its medical history reviewed, focussing on previous procedures, and any concomitant medications and diseases. This is also the time to discuss with clients the risks associated with PSA and the planned procedure. Some cats may need fluid therapy and stabilization before PSA; fasting protocols should be in place. Current recommendations include shorter fasting times (3–4 h) when compared with dogs. 20 The administration of a small amount of wet food 3–4 h before sedation may reduce gastroesophageal reflux and acidic reflux. 20 Water should be available until the time of PSA. Specific developmental stages (neonatal, pediatric) and conditions such as gastrointestinal and central nervous system disease or diabetes, brachycephalic conformation or a history of gastroesophageal reflux and/or aspiration may require specific fasting times based on the patient’s individual needs, and further information can be found elsewhere. 21
The assignment of American Society of Anesthesiologists (ASA) physical status is important to predict complications and identify the risk of anesthetic-related death (Table 1).2,3 Feline-friendly handling techniques should always be adopted (gentle approach, patience, positive attitude, appropriate petting, etc) (Figure 2). Cats with a timid or fearful demeanor should be examined in their carriers; top- or side-opening carriers provide a safe and secure environment for these individuals (Figure 3). Indeed, the cat’s behavior is important in the decision-making process involving PSA and dosage regimens. Blood collection for additional laboratory diagnostics (ie, hematology and blood chemistry analysis), if needed, can be performed after drug administration and during venous catheterization. The authors will often use the blood extracted from the catheter hub to perform a hematocrit and total protein evaluation and avoid unnecessary jugular veni-puncture. However, venous catheterization may not always be performed as part of the PSA protocol and there is no consensus on the administration of fluid therapy in these cats. Additionally, hematocrit values in some cases may decrease by up to 30% after the administration of sedatives, especially dexmedetomidine, ketamine and acepro-mazine.22,23 (Clinicians should also be aware of the potential for increased ultrasonographic and radiographic splenic measures after the administration of acepromazine; 22 and several drugs used during PSA may change echocar-diographic results, as discussed later.)
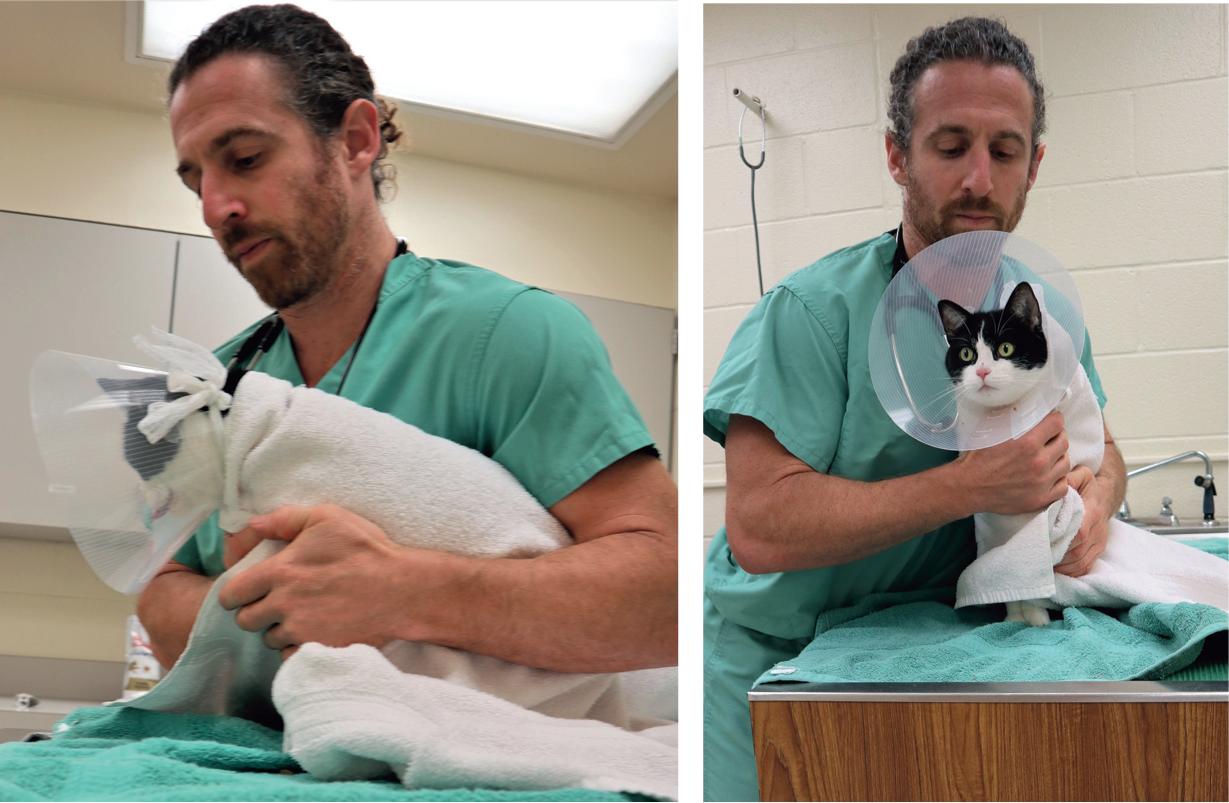
Feline-friendly handling techniques should be used throughout procedural sedation and analgesia (PSA) to minimize stress and fear and provide a good hospital experience. The mantras ‘less is more’ and ‘go slow to go fast’ are true during feline handling. Towels can be used around the neck and body for physical restraint and gentle care, and to avoid scruffing. An Elizabethan collar will provide additional protection for the handler from aggressive cats. This particular patient is safely restrained for a procedure such as intramuscular injection or intravenous catheter placement

Cats may be examined and handled in top-opening or side-opening carriers to facilitate the procedure and maximize comfort during veterinary consultations
Materials, supplies and equipment should be ready and in good order before PSA. Prevention is key to avoiding complications. Doses and volumes of emergency drugs should be calculated beforehand. It is good practice to have an anesthetic machine and means of intubation and ventilation available when sedation is profound, with a risk of regur-gitation and aspiration, and/or with patients that are at high risk of complications (ie, senior or pediatric patients, or those with extremes of body weight) (Figure 4).2–4 In some practices, an anesthetic machine may not be available. Oxygenation may still be required, and portable oxygen cylinders should be available. Oxygen supplementation may be needed to prevent or treat hypoxemia in cats undergoing PSA. 24 A manual resuscitation bag is a practical means of providing oxygenation (Figure 4).

Supplies prepared in advance of PSA, in case intubation of the cat’s trachea is required. a = cuffed endotracheal tube; b = cuff inflation syringe; c = lidocaine with a syringe and catheter for topical application on the arytenoids to prevent laryngospasm during intubation; d = topical eye lubrication; e = sterile endotracheal tube lubrication; f = roll gauze for securing the endotracheal tube in place; g = mask for oxygen supplementation; h = square gauze to help grasp the patient’s tongue during intubation; i = laryngoscope; j = stylet for placement within the endotracheal tube; the stylet should be introduced only as far as 3–5 cm past the arytenoids, and is removed once the endotracheal tube is in place, taking care not to cause pharyngeal or tracheal trauma; k = manual resuscitation bag and attachment for oxygen insufflation. Note: A Mapleson D (Bain) non-rebreathing circuit attached to an anesthetic machine may be used in lieu of a manual resuscitation bag at flow rates of 150–300 ml/kg/min
Hypothermia can be avoided by using a warming system (ie, forced air warming blanket or circulating warm water device) to prevent heat losses during PSA. Additional measures for the prevention of hypothermia include use of bubble wrap, blankets and quilts, or the Hibler’s method. The last is a low-cost technique combining an outer vapor barrier (ie, plastic wrapping) with an inner insulating layer (ie, blankets). 25 In humans, the Hibler’s method has been shown to reduce hypothermia and promote faster rewarming than use of blankets and bubble wrap. 25
Drug protocols for PSA: overview and rationale
Before focussing on specific injectable protocols for PSA, some important principles deserve particular mention. Firstly, patient assessment incorporating health status and behavior is paramount when choosing a drug protocol for PSA. So too is an individualized approach, allowing adjustments to doses and protocols as required. The ideal protocol should maintain appropriate cardiorespiratory function while unpleasant, minimally invasive procedures are performed. What constitutes ‘appropriate’ for each cat undergoing PSA is vague. For example, a healthy cat may tolerate decreases in cardiac output and heart rate induced by agonists of alpha(a)2-adrenergic receptors during hip radiography. Similar changes in cardiovascular function could, however, be severely detrimental in a senior cat with sepsis and undergoing abdominal ultrasonography.
When possible, neuroleptanalgesia is recommended during PSA and involves the combination of an opioid analgesic and a tranquillizer (ie, acepromazine) or sedative (benzodiazepine). 26 This has the potential benefits of producing a greater degree of sedation and analgesia with reduced adverse cardio-pulmonary effects when compared with either drug administered alone at similar doses. Clinical judgment is important in the decision-making process and antagonist agents (ie, atipamezole, flumazenil or naloxone) should always be available and drawn-up ready for use in critical cases. Butorphanol has been reported to preserve analgesia better than naloxone when used as a reversal agent in cats. 19
Acepromazine, benzodiazepines (ie, diazepam or midazolam) or agonists of α2-adrenergic receptors (ie, dexmedetomi-dine, medetomidine or xylazine) are used in combination with an opioid for PSA (Table 2). Local anesthetic blocks are not specifically discussed in this article, but should be incorporated to provide a smooth PSA while decreasing drug requirements.
Examples of neuroleptanalgesia protocols used for procedural sedation and analgesia (PSA) in cats. 27 If necessary, these suggested protocols can be used for premedication prior to induction of general anesthesia

If additional doses of PSA drugs are required due to decreases in depth of sedation, the authors recommend administering 25–50% of the original dose
Buprenorphine at standard concentration (0.3 mg/ml)
Other full μ-opioid receptor agonists can be used (ie, fentanyl, oxymorphone, morphine)
ASA PS = American Society of Anesthesiologists physical status; HCM = hypertrophic cardiomyopathy; IM = intramuscularly
Note that alternative methods for achieving PSA, such as chamber induction using inhalant anesthetics, can be useful to protect handlers from injury but may cause airway irritation 28 and an excitatory phase in patients, 29 in addition to the environmental concerns over waste anesthetic gases. 30 Moreover, this technique may be associated with increased risk of anesthetic-related morbidity or mortality due to the excessive inhalant anesthetic concentrations required to induce adequate sedation and avoid involuntary excitement. 20
The magnitude/level of sedation and analgesia and the quality of recovery are better with the combination than with each drug used alone;31,32 lower doses of each drug can normally be administered. Also, adverse effects may be reduced with neuroleptanalgesia. For example, in a prospective, randomized, blinded study of 30 cats, the prevalence of vomiting was 70% when dexmedetomidine was used alone, compared with 10% after dexmedetomidine-butorphanol. 33 The dose of single-agent dexmedetomidine also had to be increased two-fold to produce similar sedative effects to a combination of dexmedetomidine with butorphanol or meperidine (pethidine) for various clinical procedures. 33 However, there are some occasional cases where opioids are used alone for PSA or when the specific combination of benzodiazepine-opioid is indicated (see box on right). The specific choice of opioid will depend on the onset, duration and level of analgesia required for the procedure (see box on left; Table 3). For further information, readers are referred elsewhere.18,34–36

Opioids commonly used for procedural sedation and analgesia in cats 34

IM = intramuscular; IV = intravenous; OTM = oral transmucosal (buccal); NMDA = N-methyl D-aspartate; CRI = constant rate infusion; SC = subcutaneous
Sedation is superior when agonists of α2-adrenoreceptors are administered alone or in combination with opioids when compared with acepromazine alone or with an opioid, and also benzodiazepinebased protocols.22,23 Ketamine, tiletamine– zolazepam and alfaxalone-based protocols have also been used for neuroleptanalgesia to provide robust chemical restraint (Table 4). However, sedative effects may vary according to dosage regimens, individual patient variability in response to the drugs, disease and patient behavior, and the quality of sedation is sometimes unpredictable.
Selected drug protocols used in published studies for procedural sedation and analgesia (PSA) in cats

Additional PSA protocols are described within the text
IM = intramuscular; HR = heart rate; RR = respiratory rate; BP = blood pressure; SpO2 = saturation of peripheral oxygen; IV = intravenous; SC = subcutaneous
Note: Some PSA protocols may significantly impact diagnostic testing (eg, echocardiography and blood work analysis; Table 5) and this should be considered when interpreting the results
Effects of procedural sedation and analgesia protocols on diagnostic tests


The following section provides additional information on protocols commonly used for PSA in cats using an evidence-based approach. Detailed pharmacology of these drugs is not presented herein. Drug combinations for PSA are also routinely used for premedication, with important anestheticsparing effects.
Drug protocols for feline PSA: an evidence-based approach
The sedative, cardiorespiratory and metabolic effects of, as well as quality of recovery for, several drug combinations for feline PSA have been reported (see Table 4 for an overview). These drug combinations, together with some additional PSA protocols, are discussed in the text below.
It is important to bear in mind that comparisons are difficult between these studies. A scoring system for feline sedation assessment has not been validated. Most studies have evaluated sedation using interactive visual analog and simple descriptive scales that are dependent on the observer’s experience, leading to large inter-study variability. 56 Various multidimensional scales for sedation scores have also been used but with little validation.24,44,57 Different doses, drug combinations and routes of administration have been reported using either clinical or experimental populations. Protocols have been reported for a single procedure (ie, echocardiography) or for premedication, which cannot be generalized to other situations.
Acepromazine combinations
Acepromazine blocks dopaminergic receptors and decreases reaction to external stimuli. The drug has been used in combination with butorphanol, buprenorphine or methadone as premedication in clinical trials.58–61 Sedation was not always systematically evaluated. These combinations produce a mild calming effect with third eyelid protrusion, purring and kneading, or a state of tranquilization for 2-3 h.58,60–62 Moderate sedation following ace-promazine administration was also effective in lowering propofol dosing requirements for anesthetic induction in cats. 59
In some studies, a eutectic mixture of lidocaine-prilocaine cream was applied over the cephalic vein, and protected with occlusive dressing, after premedication with acepromazine-buprenorphine and approximately 20–30 mins before IV catheterization. This technique avoids excessive physical restraint and behavioral responses to manipulation, especially in cats responding poorly to the sedation.18,60,61 Indeed, the application of this local anesthetic cream reduced the reaction of cats to IV catheterization, as assessed by a numerical rating scale. 63 Even so, acepromazine-opioid combinations will usually produce mild sedation and experienced support staff or veterinarians are often needed for physical restraint and handling. 64
Acepromazine should not be used for PSA in cats with hypovolemia or dehydration, or in procedures with high risk of bleeding (Table 2). Hypothermia and hypotension may occur due to blockade of α1-adrenergic receptors, especially in cats undergoing general anesthesia. Acepromazine combinations should be used with caution in hypovolemic and in hypotensive patients. Acepromazine has some antihistaminic effects and should not be administered to patients undergoing intradermal skin testing.
Agonists of α2-adrenergic receptor combinations
Agonists of α2-adrenergic receptors provide sedation, muscle relaxation, analgesia and chemical restraint in a dose-dependent man-ner. 65 They can produce emesis, especially when administered alone, due to direct stimulation of the chemoreceptor trigger zone (more commonly after xylazine). 33 Vomiting becomes an issue in cats with increased intraocular and intracranial pressures, with a detrimental impact on patient health and welfare. These drugs cause peripheral vaso-constriction, hypertension with reflex brady-cardia and decreases in cardiac output. For this reason, agonists of α2-adrenergic receptors are mostly reserved for cats with stable hemodynamic function. Hypothermia can occur via depression of the hypothalamic thermoregulatory center and the lack of muscle activity during PSA.
Dexmedetomidine and medeto-midine are selective agonists of α2-adrenergic receptors and should be preferred over xylazine. Xylazine should not be administered to systemically ill, pediatric or senior cats. Dexmedetomidine is the active isomer of medeto-midine and twice as potent. Dexmedetomidine-butorphanol is a popular drug combination for PSA with a short onset of action (approximately 5 mins). 46 This combination provides superior sedative effects and a lower prevalence of vomiting than dexmedetomidine-buprenorphine during PSA for diagnostic imaging (ultrasound examination, radiography, CT), blood sampling, minor wound care and chemotherapy treatment administration. 48 However, studies have shown that the analgesia produced by dexmedetomidine-buprenorphine is superior.66,67 The thermal antinociceptive effects of dexmedetomidine were enhanced when the drug was combined with buprenorphine. 68 The addition of ketamine (3 mg/kg) to dexmedetomidine (5 ug/kg) and butorphanol (0.3 mg/kg) increased the median duration of action for blood sampling by approximately two-fold. However, sedation scores were not different when compared with dexmedetomidine–butorphanol alone. 69
Dexmedetomidine combinations usually decrease ejection fraction and fractional shortening, and increase end-diastolic and end-systolic volume in cats. This may affect the interpretation of echocardiography results. These combinations (eg, midazolam-butorphanol-dexmedetomidine or ketamine-dexmedetomidine) decrease cardiac output (approximately 50%), significantly more so than ketamine-midazolam-butorphanol (34%). 23 In contrast, medetomidine alone has been shown to increase afterload and was able to attenuate signs of dynamic left ventricular outflow tract obstruction in cats. 70 This is the reason why some veterinarians use low-dose medetomidine combinations for sedation in cats with HCM. Cardiac output is predominantly maintained via heart rate rather than contractility in the pediatric patient (cats <4 months of age). Administration of any α2 receptor agonist in pediatric cats can severely reduce cardiac output and tissue perfusion and requires caution. Similarly, these drugs should not be administered to patients with life-threatening brady-arrythmias (eg, bradycardia secondary to urinary obstruction).
The oral transmucosal (buccal) route of administration has been used with medetomidine or dexmedetomidine as an alternative to intramuscular (IM) injections in cats.57,71 The drug is injected with a 1 ml syringe into the cheek pouch to avoid oral administration (ie, drug swallowing) (Figure 5). Dexmedetomidine (40 jg/kg) in combination with buprenorphine (0.02 mg/kg) produced chemical restraint in cats when drugs were administered either buccally or IM. Sedative effects were not different between the groups. However, IM dosing produced superior sedation compared with buccal dosing when lower doses of dexmedetomidine (20 ug/kg) were used with buprenorphine. 57 Antagonists of α2-adrenergic receptors (atipamezole for dexmedetomidine and medetomidine; yohimbine for xylazine) can be administered IM to hasten recovery and antagonize potential adverse effects. Analgesia and muscle relaxation will also be antagonized. Atipamezole is typically administered IM at equal dose volume as the dexmedetomidine (0.5 mg/ml) or medetomi-dine volume. When using dexmedetomidine at 0.1 mg/ml concentration, the volume of atipamezole should be reduced to one-fifth of the dexmedetomidine dose volume. To reverse xylazine, administer 0.025–0.05 mg/kg of yohimbine IV slowly (25% of the published total dose), with the remaining 75% of the dose administered IM. This technique can help minimize yohimbine’s unwanted side effects (ie, excitement and hypotension). Note that the use of anticholinergics with agonists of α2-adrenergic receptors is normally contraindicated (see box).

Oral transmucosal (buccal) administration of dexmedetomidine and buprenorphine can provide 'hands-off' sedation, analgesia and chemical restraint. Palatability is generally acceptable. Drug absorption is by the transmucosal route, assuming the drug is not spilled or swallowed by the cat

Benzodiazepine combinations
The administration of benzodiazepines inconsistently produces sedation, and more commonly results in a transient period of excitement in adult, healthy cats. Patients may actually show signs of paradoxical aggressive and defensive behaviors.23,72 There is generally a clinical impression that benzo-diazepines cause minimal cardiovascular depression, but in one early study doses of 0.1 mg/kg of diazepam reduced systolic blood pressure and cardiac contractility, potentially because of the presence of propylene glycol in the formulation. 73 Nevertheless, midazolam and diazepam may be used for coinduction after the administration of propofol for endotracheal intubation in systemically ill cats. Benzodiazepines enhance the affinity of the neurotransmitter gamma-aminobutyric acid (GABAA) for its receptor and have a synergistic effect with barbiturates, propofol and alfaxalone. Doses as low as 0.2 and 0.3 mg/kg of midazolam and diazepam, respectively, may have a propofol-sparing effect. 59
In the authors’ experience, midazolam-opioid combinations can be used in senior or critically ill cats (see top-right box on page 1035) to produce sedation for minor diagnostic procedures. Benzodiazepines are also used in combination with ketamine (see below). Diazepam should not be administered IM because the drug is hydrophobic and contains propylene glycol, which causes pain at injection; midazolam is water soluble and recommended for injectable PSA protocols. Flumazenil (0.01–0.04 mg/kg) is the antagonist of benzodiazepines.
Ketamine-based combinations
Ketamine, an N-methyl D-aspartate (NMDA) antagonist, causes dissociation between the thalamoneocortical and limbic systems. 74 It is used as an adjunct analgesic in patients with hyperalgesia or central sensitization, 75 and can be administered IV, IM or buccally in cats.76–78 In humans, ketamine used at subanesthetic doses (<1 mg/kg) rapidly decreased the perception of severe, acute pain and provided an opioid-sparing effect.79,80 Its ability to produce similar analgesic effects at low doses is yet to be determined in cats. 81 This drug is known for causing ‘emergence phenomenon’ (dysphoria during recovery) in a variety of species, including humans, although it is not routinely seen with subanesthetic doses in cats (<0.5 mg/kg). 81 Ketamine is always administered in combination with sedatives and analgesics. The swallowing reflex is well maintained, but the risk of aspiration is still present. 82
Ketamine-based protocols are used to produce chemical restraint for PSA (Table 4). The drug must be combined with a muscle relaxant (ie, midazolam or an agonist of α2-adrenergic receptors) to prevent muscle rigidity, seizure activity and hypersalivation. The drug has an acidic pH and may cause pain during IM injection. 50 For this reason, appropriate physical restraint is required for the administration of ketamine combinations. Ketamine has the potential to increase heart rate via increases in sympathetic tone and should be avoided in patients with HCM or tachy arrhythmias.
Tiletamine-zolazepam and combinations
Tiletamine-zolazepam is available as a white powder combination (500 mg combined) and is reconstituted with 5 ml of sterile water to produce a 100 mg/ml solution. Alternatively, tiletamine-zolazepam powder can be reconstituted with 100 mg of xylazine and 400 mg of ketamine and administered IM at 3.3 mg/kg to cats for anesthesia. Following reconstitution, any unused drug should be discarded after 7 days when stored at room temperature or after 56 days when refrigerated.
This drug combination is widely used in feline practice for PSA and surgery. Sympathetic stimulation is observed after drug administration due to central nervous system stimulation, depression of baroreceptors and inhibition of norepinephrine (noradrenaline) uptake at adrenergic nerve endings. 83 Tiletamine-zolazepam can produce muscle rigidity, myoclonus, salivation, respiratory depression and prolonged recovery from anesthesia, which is a potential concern for patient welfare. 84 The combination of low doses of tiletamine-zolazepam with methadone provided superior sedation to acepromazine-methadone in healthy cats before neutering (Table 4). 37 Similar to ketamine combinations, tiletamine-zolazepam combinations should be avoided in cats with HCM.
Alfaxalone and combinations
Alfaxalone is a synthetic neurosteroid anesthetic drug that has been studied for PSA in cats.24,41,42 The sedative and cardiores-piratory effects of alfaxalone have been evaluated when the drug was administered alone or in combination with butorphanol, hydromorphone or dexmedetomidine (Table 4). These protocols are versatile with a wide margin of safety and used for various procedures in combination with opioids (Table 4). Hypoventilation is a potential adverse effect of alfaxalone use in cats. 85 Some studies have reported ataxia, excitement and hyper-reactivity in some individuals during the recovery phase when doses of 5 mg/kg were used; 43 these effects may be attenuated by the administration of acepromazine and butorphanol. 86
Alfaxalone-based protocols may involve large-volume IM injections. For example, a dose of 5–10 mg/kg may require 2–5 ml of volume administration. Cats may react to the injection even after sedation with dexmedeto-midine-butorphanol. 43 The high volume produces discomfort and violent reactions at injection when administered at 10 mg/kg. 43 Other studies have reported that IM injections are normally tolerated when low doses are used (2 mg/kg). 46 The quality of anesthetic recovery is also a concern with alfaxalone. 87 Studies have reported cats thrashing, paddling, trembling and pacing in the cage.43,87 Jurox, the manufacturer of Alfaxan Multidose, reports that repeated, supraclinical doses (25 mg/kg) of the product administered IV q48h for three doses does not result in adverse effects in cats. 88
Propofol
The phenolic compound propofol is used as an anesthetic induction agent and, in some cases, for maintenance of anesthesia (total IV anesthesia). Propofol is sometimes administered as small boluses for PSA but it can induce adverse effects, especially in systemically ill cats. The drug produces dose-dependent cardiorespiratory depression with variable changes in heart rate, decreases in cardiac contractility and respiratory rate, hypoxia, hypercapnia and vasodilation.52,89–93 Although hypotension may be observed following propofol administration, it is often mild in healthy cats. 92 Apnea may occur, especially after rapid bolus administration.89,94–96 Caution is required when using propofol for PSA in patients with respiratory compromise and oxygen supplementation should be provided.
Small doses of propofol (1 mg/kg) may induce light sedation with signs of excitement and increased muscular activity in healthy cats (ASA PS I or II) sedated with acepro-mazine-methadone. 59 In contrast, these doses have been shown to induce smooth and mild sedation in critically ill cats with urinary obstruction requiring PSA for urethral catheterization after sedation with a midazolam-opioid combination. 97 Indeed, benzodiazepines may reduce propofol requirements for endotracheal intubation by 35% in healthy cats sedated with acepromazine-methadone 59 and by 26% in unpremedicated cats. 98
The development of Heinz bodies after a 30-min infusion of propofol has been reported in cats, but only following the third day of administration; 99 after 7 consecutive days of these 30-min infusions, hemolysis was not detected. 99 The concern for Heinz body development associated with propofol administration in cats is, as yet, undetermined, as it does not appear to have clinical significance even following 24-h propofol infusions. 93
PropoFlo 28 (Zoetis) contains 20 mg/ml of benzyl alcohol, a bacteriostatic preservative that requires glucuronidation for metabolism, and is a multiuse formulation of propofol with a 28-day shelf life. Excessive quantities of benzyl alcohol (>156 mg in 6.5 h) may cause central nervous system toxicity and potentially death. 100 For a standard 3 kg cat, an extreme overdose of propofol (approximately 26 mg/kg) would be required to produce these adverse effects.
Monitoring during PSA
Appropriate monitoring prevents, detects and aids in the treatment of complications such as hypoxia, hypoventilation and apnea, hypothermia, bronchospasm, cardiovascular depression, post-sedation delirium, prolonged recovery, vomiting and aspiration.4,9,101 The American College of Veterinary Anesthesiology and Analgesia (ACVAA) has published guidelines for monitoring of patients undergoing sedation (see box).


Capnography (performed using a side-stream adapter), pulse oximetry and cardiac auscultation in a cat undergoing procedural sedation and analgesia for an abdominal ultrasound examination. Courtesy of Mary T Schacher
Recovery procedures
The quality of recovery is dependent on body temperature, duration of the procedure, drug antagonism (whether performed and/or effective), pain, and patient health status and behavior. Complications may arise during the sedation period; however, the majority of fatalities (52–60%) have been reported to occur within the first 3 h after the procedure has ended.2,5
Prolonged hypothermia and emergence, collapse and/or excitement may occur during recovery. 9 This is particularly true when anesthetics are administered for PSA. Post-procedure excitement or delirium can be controlled with dexmedetomidine (0.5–1 jg/kg IV) administered slowly for re-sedation in a quiet environment away from other animals. Management and prevention of hypothermia during PSA was discussed earlier (see section on ‘patient assessment preparation before PSA’) and the same principles apply during recovery. Reversal agents for opioids, benzodiazepines and α2-adrenergic agonists should be readily available and administered in the event of prolonged emergence from PSA. Hypotension has been reported following the reversal of dexmedetomidine with atipamezole in isoflurane-anesthetized cats; 104 however, this should not be a concern during PSA. Monitoring should continue for a minimum of 3 h following completion of the procedure and animals should not be left unattended for long periods of time until they have recovered.
Patients are considered fully recovered when they are responsive to normal stimuli, have regained protective airway reflexes and are able to maintain normal cardiopulmonary parameters. Some cats may require continuous monitoring and oxygenation (eg, oxygen tent or chamber). If intubation was necessary, extubation is recommended when a convincing swallowing reflex is present; cats should be monitored for laryngospasm and upper airway obstructions following extubation. A small amount of water and food can be offered to patients that are awake and standing with minimal to no ataxia.
Key Points
Feline patients often require PSA in veterinary practice for various procedures.
Complications may arise, especially when risk factors are present.
The choice of drug protocol depends on the procedure and the health status of the patient, including any concomitant diseases.
The need for analgesia dictates the type of opioid to be administered.
The results of clinical and experimental trials are difficult to extrapolate to every case, and an individualized approach to PSA should be taken. 1
Monitoring, fluid therapy and good practices, as discussed in this article, are paramount for successful PSA in cats.
Footnotes
Conflict of interest
Dr Paulo Steagall has provided consultancy services to Boehringer Ingelheim, Dechra Pharmaceuticals, Elanco, Procyon and Zoetis; has acted as a key opinion leader to Boehringer Ingelheim, Dechra Pharmaceuticals, Elanco, Vetoquinol and Zoetis; and has received speaker honoraria from Boehringer Ingelheim, Dechra Pharmaceuticals, Elanco and Zoetis.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
This work did not involve the use of animals and therefore ethical approval was not necessarily required.
Informed consent
This work did not involve the use of animals and therefore informed consent was not required. For any animals or humans individually identifiable within this publication, informed consent (either verbal or written) for their use in the publication was obtained from the people involved.