Abstract
Objectives
The goal of this study was to document the outcomes and toxicity of a novel multimodality treatment protocol for feline gastrointestinal intermediate- or large-cell lymphoma (FGL) in which cats were treated at 21-day intervals.
Methods
This was a prospective, single-arm study. Twelve client-owned cats with cytologically diagnosed FGL were treated with a combination of abdominal cavity radiation therapy (RT; 8 Gy total dose administered in two 4 Gy fractions, 21 days apart), lomustine chemotherapy (approximately 40 mg/m2, administered orally at 21-day intervals for four treatments), prednisolone (5 mg PO q24h) and cobalamin (250 µg/week SC).
Results
Three cats were euthanized prior to the second treatment and it was difficult to discern treatment-associated toxicity from progressive disease. Four of the remaining cats developed cytopenias, resulting in 7–14-day lomustine treatment delays and/or dose reductions. Six cats had a partial response to treatment and three had stable disease based on ultrasound at day 21 (50% overall response rate). Three of these six cats completed the study and lived >240 days; one died of refractory diabetes mellitus with no clinical evidence of FGL, and the other two died as a result of FGL. The median overall survival time was 101 days (95% confidence interval [CI] 9–240). The median progression-free survival time was 77 days (95% CI 8–212). Necropsies were performed in eight cats, which revealed multifocal lymphoma throughout the gastrointestinal tract and other organs.
Conclusions and relevance
Oncological outcomes reported herein are comparable to those achieved with multiagent injectable chemotherapy (eg, CHOP). Treatment was seemingly well tolerated in most cats and was relatively cost-effective. It is therefore plausible that improved disease control may be achievable through continued optimization and intensification of the combinatorial chemoradiotherapy protocol.
Keywords
Introduction
Gastrointestinal (GI) lymphoma can be divided histopathologically into low-grade (small-cell, lymphocytic) and intermediate- or high-grade (intermediate- or large-cell) types. Large granular lymphoma is a subtype characterized by the presence of natural killer T lymphocytes that have characteristic intracytoplasmic granules. 1 Lymphocytic GI lymphoma is an indolent disease that is usually treated with oral prednisolone and chlorambucil.2,3 Feline GI intermediate- or large-cell lymphoma (FGL) is a debilitating disease that results in life-threatening maldigestion/malabsorption and often painful mass lesions that can result in GI tract perforation or ulceration. Acute onset of weight loss, hyporexia, vomiting, icterus and diarrhea is common. Ultrasound findings in cats with FGL may include mass lesions, transmural intestinal thickening, disruption or loss of normal wall layering, reduced wall echogenicity, localized hypomotility, lymphadenopathy, abdominal effusion, and diffuse echogenicity changes and/or nodular lesions in the liver, pancreas and spleen. Fine-needle aspiration and cytology of affected organs, abdominal fluid analysis and/or surgical resection of obstructing masses is used to obtain the diagnosis.
Treatment of FGL is challenging, as cats are often debilitated and have comorbidities that can impact their tolerance for therapy. Surgery for cats with obstructing GI masses is indicated when possible, though this is rarely curative as multifocal intra-abdominal disease is common. 4 Multidrug chemotherapy protocols (most commonly ‘CHOP’-based chemotherapy, utilizing cyclophosphamide, doxorubicin, vincristine and prednisolone) are used as an adjunct to, or instead of, surgery for non-resectable masses; various studies document response rates of 20–40% with reported median survival times (MSTs) of 3–7 months.4–13 Lomustine is a reasonable treatment option owing to its relatively infrequent dosing schedule (one oral dose every 3–6 weeks), similar response rate to CHOP and favorable toxicity profile.14–18 Subcutaneous cobalamin supplementation should be administered to cats with FGL even if serum concentrations are not measured, as this vitamin deficiency can contribute to inflammation and clinical signs of GI disease.2,19
Radiation therapy (RT) has been used to treat lymphoma since the early 1900s, and is a mainstay of therapy for both local and systemic lymphoma in humans. 20 In dogs, it has been used as a consolidation treatment along with chemotherapy and as a rescue therapy for dogs with refractory lymphoma. 21 In cats, RT is often used as the sole treatment for nasal cavity lymphoma22,23 but is likely an underutilized treatment modality in cats with GI lymphoma. In a study of eight cats with FGL or multicentric lymphoma confined to the abdominal cavity treated with CHOP chemotherapy followed by 10 daily 1.5 Gy fractions of abdominal cavity RT (total dose 15 Gy), 5/8 cats had long-term (>266 days) progression-free survival (PFS). 24 In another study, cats that failed chemotherapy for treatment of GI lymphoma were treated with RT as a rescue therapy. 25 Doses of 4 Gy (n = 1) or 8 Gy (n = 10; 4 Gy/day for two consecutive days) were administered, and 90% of cats had improvement in their clinical signs. In both studies, RT was well tolerated (toxicities thought to be related to RT were not reported).
In this study, multimodality therapy was prescribed to cats with FGL to determine whether this approach would improve response rates and/or survival times as compared with historical data. The protocol was designed to be cost-effective and convenient as oral chemotherapy was used and treatments were scheduled at 21-day intervals.
Materials and methods
Enrollment criteria
From 2015 to 2019, cats with a cytologic or histologic diagnosis of FGL were eligible for this prospective, single-arm clinical trial. Cats with liver enzyme (alanine transaminase, alkaline phosphatase, total bilirubin) increases of more than two times the normal upper limit, cats with resectable GI masses, and cats with significant comorbidities or an expected survival time <3 weeks without treatment were excluded. Prior to 2018, cats were not allowed to have received steroids or other chemotherapy drugs for >14 days prior to enrollment. Owing to a lack of robust case enrollment, starting in 2018, cats receiving steroids for >7 days prior to being considered for the study were allowed to enroll and continue with steroid treatment throughout the study, and cats receiving chemotherapy other than lomustine >7 days prior to being considered for the study (with no response documented to that therapy) were allowed to enroll. The study was funded by a private donor, and costs associated with RT, chemotherapy and restaging were entirely covered; after completion of the study or if the client elected to withdraw their cat from the study, cat owners could elect to continue restaging and treatment at their own expense.
Institutional approval and owner consent
Approval was granted by the Institutional Animal Care and Use Committee. Written consent from cat owners was obtained prior to enrollment into the study.
Staging tests
Baseline bloodwork (complete blood count [CBC]/chemistry panel), urinalysis and abdominal ultrasound (AUS) performed by a board-certified radiologist was obtained at the start of the study and prior to each dose of lomustine. Other staging tests, such as thyroxine, thoracic radiographs and feline leukemia virus/feline immunodeficiency virus serology were recommended to screen cats for comorbidities but were not required.
Radiation therapy
Cats were premedicated with butorphanol alone (0.2 mg/kg IM or IV) or a combination of butorphanol and alfaxalone (1–3 mg/kg IM or IV), followed by propofol (4–6 mg/kg) or alfaxalone IV induction and isoflurane maintenance. Following induction of general anesthesia and placement in lateral recumbency, cats were treated with external beam RT delivered via a linear accelerator. They were scheduled to receive 4 Gy on days 0 and 21 of the study administered to the entire abdominal cavity via a manual treatment plan using parallel opposed beams and 6 MV photons. The secondary (light-field) collimator and/or manual blocks were used to shield the spinal cord/vertebral column and distal half of the femurs (to decrease radiation dose to the bone marrow in these sites). The dose was calculated to midline using source-to-axis distance geometry and was verified by two or more radiation oncology clinicians and the institutional medical physicist. Upon recovery from anesthesia, lomustine chemotherapy was administered and cats were discharged to their owners’ care.
Chemotherapy and treatment-associated toxicity monitoring
Lomustine was scheduled to be administered orally at a dose of 40–50 mg/m2 every 21 days for a total of four doses during an outpatient hospital visit. As our institution has licensed pharmacists able to compound medications, lomustine was either dosed in 5 or 10 mg capsule sizes, or as a liquid formulation that was prepared immediately prior to administration (for cats that were difficult to pill but allowed liquid oral medication to be given). As the nadir of neutropenia and thrombocytopenia is variable and can occur 7–28 days post-administration,11–15 weekly CBCs were obtained after the first dose. Toxicities were defined by a published chemotherapy toxicity scheme; it was not possible to definitively separate chemotherapy- and radiation-associated toxicities due to concurrent administration. 26 Neutropenia of <2000/µl at 21 days after treatment triggered a 7-day treatment delay for subsequent doses. If the nadir of neutropenia after the first dose was <1000/µl, the dose was reduced by approximately 20% for subsequent doses. Platelet decreases were addressed on a case-by-case basis to decide on treatment delays or dose reductions. A serum chemistry panel was obtained prior to administration of each dose of lomustine, and abnormalities were evaluated on an individual patient basis if they arose.
Cobalamin and steroids
Prednisolone was prescribed to be given orally at a dose of 5 mg/cat/day. Cobalamin was prescribed to be given weekly at 250 µg SC.
Management of toxicities
Cats with neutrophil counts <500/µl were treated with oral or parenteral antibiotics. GI upset was managed with antinausea medications on an outpatient basis unless hospitalization for supportive care was deemed necessary.
Outcome assessment
Subjective measures assessed at each visit included owner-reported assessment of activity level and appetite. Objective outcome measures include weight (at each visit) and AUS (prior to each dose of lomustine, so at a minimum of 21-day intervals) with measurement of lesions when possible. Although no published criteria exist in cats to document response to therapy for lymphoma, a corollary publication for dogs with nodal lymphoma was used to define partial response (PR; ⩾30% reduction in the longest node diameter), stable disease (SD; <30% decrease and <20% increase in the longest node diameter) and progressive disease (PD; >20% increase in the longest node diameter). 27 Cats that had PD during the study period were dismissed and voluntary withdrawal by cat owners was allowed.
Follow-up
Upon completion of the study, owners could elect to continue lomustine (or an alternative chemotherapy protocol) at their expense. Continued follow-up (at least monthly) was achieved through patient visits to our hospital, phone calls and referring veterinarian contacts for the remainder of the patient’s life. Owners were asked to submit their pet for necropsy at the time of death, though this was not a requirement for enrollment into the study.
Statistical methods
Overall survival time (OST) was defined as the time from the first radiation fraction until death; cases were censored if alive at the time of analysis. PFS was defined as the time from the first radiation fraction until an event occurred. An event was defined as local or distant progression of lymphoma. Survival analysis was performed using the Kaplan–Meier product-limit method (GraphPad Prism version 6 [GraphPad Software] and JMP Pro 14.1.0 [SAS Institute]).
Results
A total of 12 domestic shorthair cats were enrolled; summary data, including signalment, clinical signs and disease description, are given in Table 1.
Summary table including signalment, clinical signs and disease description for 12 cats with feline gastrointestinal intermediate- or large-cell lymphoma
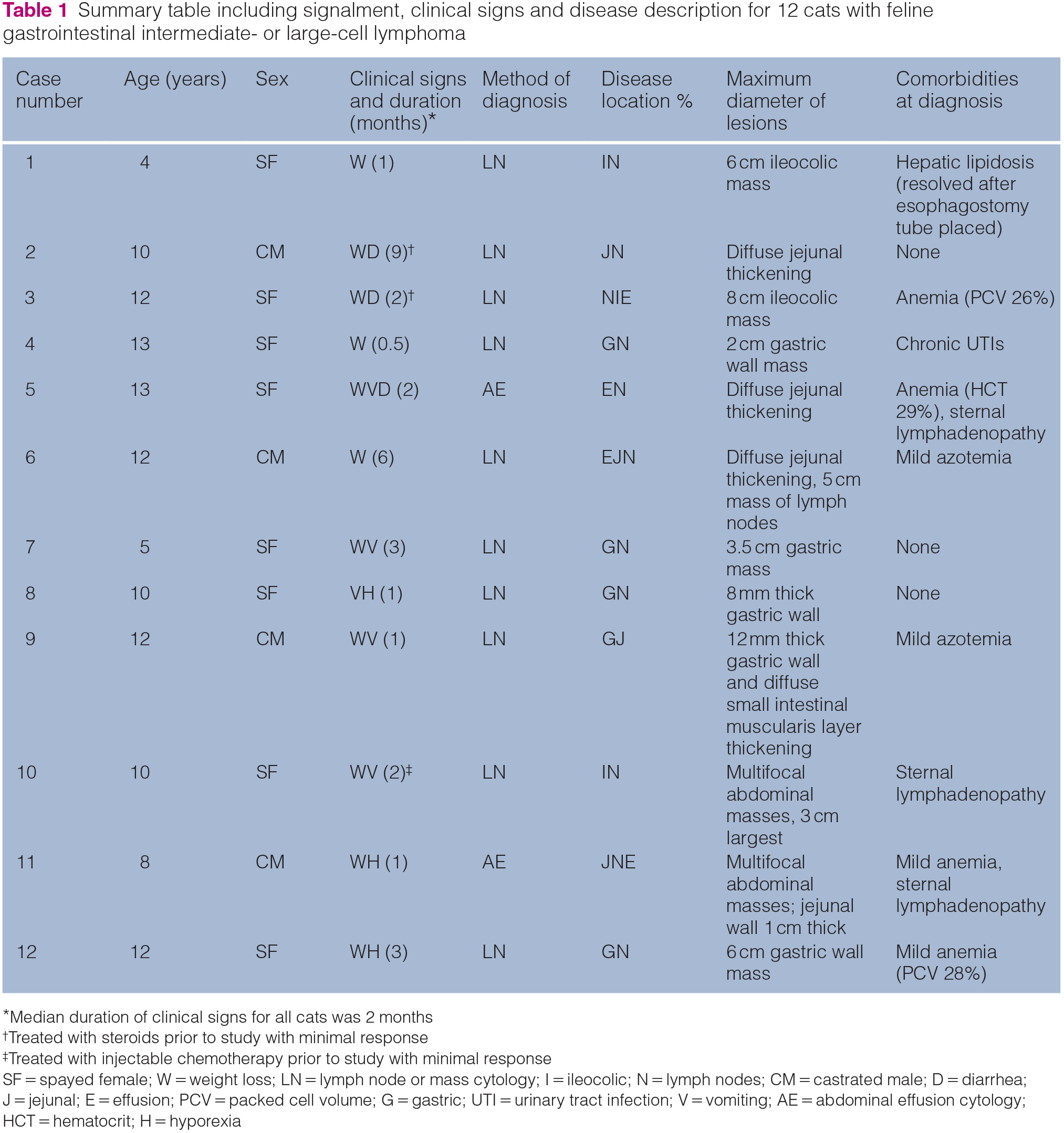
Median duration of clinical signs for all cats was 2 months
Treated with steroids prior to study with minimal response
Treated with injectable chemotherapy prior to study with minimal response
SF = spayed female; W = weight loss; LN = lymph node or mass cytology; I = ileocolic; N = lymph nodes; CM = castrated male; D = diarrhea; J = jejunal; E = effusion; PCV = packed cell volume; G = gastric; UTI = urinary tract infection; V = vomiting; AE = abdominal effusion cytology; HCT = hematocrit; H = hyporexia
Lomustine, prednisolone and cobalamin
Lomustine doses for the first treatment were 10 mg (n = 9; median 40 mg/m2 [range 33–50 mg/m2]), 9 mg (n = 1, 45 mg/m2), 8 mg (n = 1, 44 mg/m2) and 5 mg (n = 1, 25 mg/m2). In two cats, depot preparations of injectable steroids (methylprednisolone, 20 mg SC every 4 weeks) were used as owners were not able to administer daily oral medications. One of these cats developed diabetes mellitus after one dose and steroids were discontinued. All pet owners were able to administer weekly cobalamin as prescribed.
Toxicities, treatment delays and dose reductions
Table 2 gives a summary of lomustine dosing and hematologic toxicities. Three cats were euthanized prior to the second treatment, and it was difficult to discern treatment-associated toxicity from progressive disease. Nine cats survived for ⩾21 days after the initial treatment and received their second treatments of chemother-apy and RT. No cats were hospitalized for treatment-associated toxicities.
Summary table including chemotherapy dosing and graded toxicities for 12 cats treated with a chemoradiation protocol consisting of lomustine and 8 Gy abdominal cavity radiation therapy

Withdrawn from study owing to progressive disease (confirmed by ultrasound) at day 20
Euthanized 10 days after onset of treatment owing to overall declining condition
Euthanized 10 days after onset of treatment owing to increased vomiting including hematemesis
Withdrawn from study owing to progressive disease (confirmed by ultrasound) after third round of lomustine
RT = radiation therapy; NA = not available
Restaging prior to second treatment
Restaging with AUS prior to the second treatment was completed in nine cats. For six cats, a PR to treatment was seen on imaging (50% overall response rate; owners also reported improvement of clinical signs in these cats). Three cats had SD on imaging and clinically.
Outcome
Three cats completed the study (two in PR and one in CR); two of these cats received continued chemotherapy with lomustine (one received one dose and one received three doses prior to disease progression or discontinuation). Nine cats did not complete the study, including two cats that completed two cycles of treatment (including both RT and chemotherapy on days 0 and 21) and had SD before the owners elected to withdraw the cats to pursue other therapies (both cats were treated with weekly injectable chemotherapy for 2 months prior to disease progression). For cats that had PD and were dismissed from the study prior to completion, rescue therapies included RT for one cat (5 Gy single dose) and CHOP chemotherapy for one cat. Eleven cats died of FGL. The remaining cat was euthanized with no clinical evidence of FGL (though neither imaging prior to euthanasia nor necropsy was performed on that cat) 489 days after starting treatment because of insulin-refractory diabetes mellitus and urinary tract infections.
The median OST was 101 days (95% confidence interval [CI] 9–240). The median PFS was 77 days (95% CI 8–212). No cases were censored from the OST analysis as all died during the study period. Four cats were censored from the PFS analysis, including the two with SD that were withdrawn from the study by the owners (censored from the PFS analysis on the date of study withdrawal), one without evidence of disease progression at completion of the study (censored from the PFS analysis at the time of study completion) and one euthanized for a cause other than lymphoma (censored on the date of death).
Necropsy
Eight cats were necropsied. In seven cats, FGL was confirmed; in one cat, diffuse lymphocytic lymphoma with abdominal cavity effusion was present. Lymphoma was present diffusely throughout the intestinal tract in most cats, and in locations such as kidneys, omentum, gallbladder, uterine stump, bladder and sternal lymph nodes. Partial-thickness mucosal ulcerations were detected in various parts of the intestinal tract in four cats. Unexpected findings besides lymphoma included suppurative pancreatitis and bacterial cholangitis, each in one cat.
Discussion
The cats in the present study tolerated treatment and the objective and clinical response rate was 50%. The median OST was 101 days and the median PFS was 77 days. These results are similar to that of a study of 32 cats with treatment-naive intermediate-to-large-cell GI lymphoma treated with lomustine (some were also treated with L-asparaginase and most were also treated with steroids). 17 The response rate in that study (defined by the authors as a duration of clinical response for at least 28 days) was 50% but was short-lived in the majority of cases as the overall MST was only 108 days. The median number of lomustine doses was three per cat and lomustine was dosed a median of every 5 weeks (range 4–7.5 weeks) owing to non-clinical hematologic toxicity (neutropenia and/or thrombocytopenia). Prognostic factors associated with faster disease progression included a history of hyporexia, presence of anemia and a dose of lomustine lower than the median (40 mg/m2). The authors of that study suggested that using a higher dose of lomustine may improve the outcome. 17 As in the present study, no cats required antibiotics, supportive care or hospitalization for toxicity, and the median dose for the nine cats receiving the commercially available capsule size of 10 mg was 40 mg/m2; perhaps a higher lomustine dose could have improved the outcome. In the original phase 1 study of cats treated with lomustine for various tumors, the drug was dosed at 50–60 mg/m2 and the dosing interval was 6 weeks as the nadir for cytopenias ranged from 7 to 28 days after administration. 16 A practical challenge with dose adjustments for lomustine is the need for compounding if the desired dose is higher or lower than the commercially available 10 mg size, which could increase the risk of dosing error and force treatment delays if an outside pharmacy is used.
One of the challenges in comparing publications regarding feline lymphoma is that most studies evaluating response to therapy and survival time combine various stages and sites of disease (which may have varied prognoses), as well as histopathologic types (lymphocytic vs intermediate or large cell). Few studies have focused solely on GI lymphoma and even fewer on the intermediate- or large-cell subtype.11–13,17,24,28 This could be related to the fact that it can be difficult to cytologically distinguish lymphocytic vs intermediate- or large-cell lymphoma on fine-needle aspirates or fluid analyses, and histopathology may be required in some cases. In the present study, one cat had cytologically confirmed FGL via cytologic fluid analysis, but, at necropsy, diffuse small-cell lymphoma was present throughout the GI tract. Perhaps in this cat, the intermediate- or large-cell component of the disease resolved with chemoradiotherapy and the lymphocytic part remained; it is also possible that the original diagnosis of FGL was incorrect.
It is disappointing that use of RT in the cats of this study did not result in further improvement in outcome. This may be related to the total radiation dose, the interval of 21 days between doses or the lack of response of the disease process. The protocol (two doses of 4 Gy separated by an interval of 21 days, given on the same day as the planned doses of lomustine) was selected in an attempt to decrease cost and number of trips to the hospital for the cats. In regard to the total radiation dose, a higher total dose could be considered if it was tolerated by the cat’s GI tract. As the entire abdominal cavity must be irradiated (owing to the common presence of multifocal disease), normal tissue sparing is not possible and so increasing the total dose must be done gradually and with careful monitoring for toxicity. In a study of cats with nasal lymphoma, those treated with a total radiation dose of >32 Gy had longer survival times; 22 however, it is unlikely that the entire GI tract would be able to tolerate that dose, even if treatment was fractionated over a period of weeks. In the published study of eight cats treated with low-dose daily fractionation for FGL or multicentric lymphoma confined to the abdominal cavity after induction with 6 weeks of CHOP chemotherapy, the total dose was 15 Gy in 10 days (1.5 Gy/fraction on weekdays over 2 weeks), which was well tolerated. Five of the eight cats survived for >266 days, which is a relative improvement, compared with the present study in which only 3/12 cats lived for >240 days. 24 Although the numbers of cats in each study is small, the relative differences in outcomes may be due to either/or differences in total RT dose, dose/fraction or use of CHOP as compared to lomustine. A possible future study could use a combination of low-dose, daily irradiation (such as the 1.5 Gy × 10 daily fractions as used by Williams et al 21 ) with lomustine; this protocol would still result in fewer trips to the hospital and a lower cost than multidrug, weekly injectable protocols.
In a study of cats such as this one in which all have pre-existing GI disease, it can be difficult to discern treatment-associated toxicity from disease progression. In addition, a combinatorial chemoradiation protocol could further enhance either or both efficacy and toxicity. Toxicities that can be associated with irradiation of the GI tract can be acute (those starting during a course of fractionated RT treatment and continuing for as long as 2–3 months after completion of treatment, including vomiting, diarrhea, nausea, anorexia and ulceration) or late (those occurring several months/years after completion of treatment, including ulceration, stricture and fibrosis that can lead to perforation).29,30 In addition, RT-induced nausea and vomiting (RINV) is a poorly understood phenomenon occurring in humans undergoing any type/site of RT, typically occurring within a few minutes to hours after RT. When the upper abdomen is irradiated, 60–90% of patients experience RINV in the absence of prophylaxis with antiemetics (typically ondansetron and/or metoclopramide). Patient-specific factors, such as concurrent chemotherapy administration, female sex, prior history of nausea or vomiting, and increasing age, further increase the risk of RINV. 31 In the cats described here, we attributed continued GI signs after treatment to disease progression rather than to treatment-associated toxicity. Although no cats appeared to experience nausea after treatment, this could be related to either RINV or to general anesthesia if it occurred. Three of the cats in this study were presumed to have disease progression within 7–20 days following the initial treatment and were euthanized prior to restaging. It is possible that acute radiation toxicity or RINV could have contributed to their demise. It is also possible that in the remaining cats, radiation- and or chemotherapy-induced toxicity could have contributed to their ongoing clinical signs of GI disease. The partial-thickness mucosal ulcerations detected in various parts of the intestinal tract in four cats at necropsy could have been caused by RT. The other two studies of feline abdominal cavity irradiation also reported a perception of minimal acute and late toxicity associated with RT, but, again, this conclusion could be confounded by the relative difficulty in discerning toxicity vs disease progression.
One of the challenges of this prospective study was case selection. Although our enrollment criteria excluded cats with ‘expected survival times of less than 3 weeks without treatment’, this was difficult to determine objectively. Some of the cats in this study had relatively mild and prolonged clinical signs, while others were more profoundly and acutely affected. In the latter group of cats, supportive care measures such as subcutaneous fluid therapy and enteral nutrition via feeding tubes were employed, when possible, to attempt to improve quality of life. Another aspect when counseling pet owners regarding therapy for cats with FGL is the difficulty in determining an individual cat’s likelihood of response to therapy and survival times. The most consistent prognostic indicator in cats with FGL is response to therapy; several studies have documented that cats that have a ‘complete response’ to therapy have longer survival times than those that only have a ‘partial response’.9–12 Typically, defining response to therapy is difficult and subjective, and most studies have reported responses as defined by weight gain, improved quality of life, and decreased or improved vomiting/diarrhea for cats with those clinical signs at the start of therapy. Objective measurements of response to therapy in cats with FGL are challenging owing to the costs incurred to restage the disease with abdominal ultrasonography and/or repeat endoscopy. Additionally, using aggressive treatment in the first month after diagnosis is essential, as long-term survival depends on the initial response to therapy. In a study of cats treated for FGL, cats that lost >5% of their body weight at 1 month after initiation of therapy had shorter survival times than those that had gained or had stable weight. 28 In another study, cats that responded to the first (3-week) cycle of cyclophosphamide-, vincristine- and prednisone-based chemotherapy had a median PFS time of 364 days vs 31 days for cats that did not respond to the first cycle, prompting the authors to make yes/no recommendations for continued chemotherapy to pet owners after the first cycle. 5 Ideally, future studies will further elucidate prognostic factors for this disease.
Conclusions
Although this treatment protocol was well tolerated and relatively cost-effective, oncologic outcomes reported herein were not superior to those achieved with multiagent injectable chemotherapy (eg, CHOP) alone. Continued optimization and intensification of the combinatorial chemoradiotherapy protocol will be required to improve disease control in cats with FGL.
Footnotes
Acknowledgements
The authors acknowledge Emily Griffith for her contributions to the statistical methods.
Author note
The results of this study were partially presented at the 2018 Annual Meeting of the American College of Veterinary Radiology.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was generously supported by the NCSU Feline Health Research Fund.
Ethical approval
This work involved the use of non-experimental animals only (including owned or unowned animals and data from prospective or retrospective studies). Established internationally recognised high standards (‘best practice’) of individual veterinary clinical patient care were followed. Ethical approval from a committee, while not necessarily required, was nonetheless obtained, as stated in the manuscript.
Informed consent
Informed consent (either verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (either experimental or non-experimental animals) for the procedure(s) undertaken (either prospective or retrospective studies). No animals or humans are identifiable within this publication, and therefore additional informed consent for publication was not required.
